デュシェンヌ型筋ジストロフィー治療の世界市場、治療タイプ別(分子ベースの治療、ステロイド治療、その他)、治療方法別(エクソン スキッピング アプローチ、変異抑制、ジストロフィン標的治療)、投与経路別(経口、非経口、その他)、エンド ユーザー別(病院、専門クリニック、在宅ケア、その他)、流通チャネル別(病院薬局、小売薬局、オンライン薬局) - 2030 年までの業界動向と予測。
デュシェンヌ型筋ジストロフィー治療市場の分析と規模
市場の成長を牽引すると予想される主な要因は、DMD 治療の認知度の高まりと、DMD 障害に対する新しい治療法の導入です。さらに、DMD 障害の有病率と影響の高まりも、市場の成長を牽引すると予想されるもう 1 つの重要な要因です。臨床試験数の増加も最近の傾向であり、市場の成長を牽引すると予想されています。ただし、高度な技術による DMD 治療の利用可能性の制限と、DMD 障害の治療費の高さが、市場の成長を抑制すると予想されます。
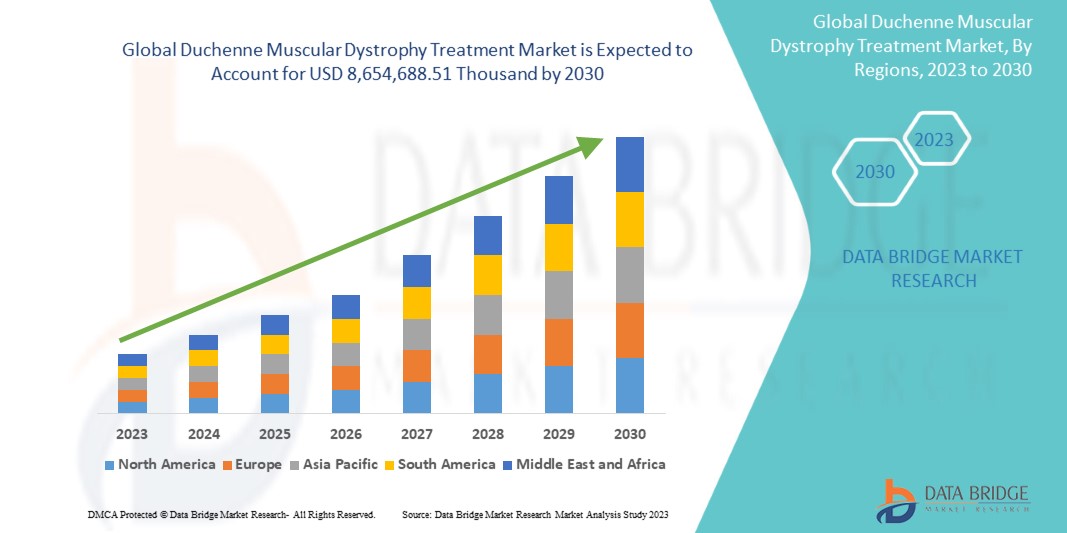
Data Bridge Market Research は、世界のデュシェンヌ型筋ジストロフィー治療市場は、2023 年から 2030 年の予測期間中に 16.8% の CAGR で成長し、2030 年までに 8,654,688.51 千米ドルに達すると予測しています。この市場レポートでは、価格分析と技術の進歩についても詳細に取り上げています。
|
レポートメトリック |
詳細 |
|
予測期間 |
2023年から2030年 |
|
基準年 |
2022 |
|
歴史的な年 |
2021 (2015 - 2020 にカスタマイズ可能) |
|
定量単位 |
収益(千米ドル) |
|
対象セグメント |
治療の種類(分子ベースの治療、ステロイド療法など)、治療法(エクソン スキッピング アプローチ、変異抑制、ジストロフィン標的療法)、投与経路(経口、非経口、その他)、エンド ユーザー(病院、専門クリニック、在宅ケアなど)、流通チャネル(病院薬局、小売薬局、オンライン薬局) |
|
対象国 |
米国、カナダ、メキシコ、ドイツ、フランス、英国、イタリア、スペイン、ロシア、トルコ、ベルギー、オランダ、スイス、その他のヨーロッパ諸国、日本、中国、韓国、インド、オーストラリア、シンガポール、タイ、マレーシア、インドネシア、フィリピン、その他のアジア太平洋諸国、ブラジル、アルゼンチン、その他の南米諸国、南アフリカ、サウジアラビア、UAE、エジプト、イスラエル、その他の中東およびアフリカ諸国 |
|
対象となる市場プレーヤー |
Sarepta Therapeutics, Inc.、GSK plc.、Capricor Therapeutics, Inc.、Dyne Therapeutics、Solid Biosciences Inc.、BioMarin、Stealth BioTherapeutics Inc.、Avidity Biosciences、ReveraGen BioPharma, Inc.、PTC Therapeutics.、NS Pharma, Inc、ITALFARMACO SpA、FibroGen, Inc、SANTHERA PHARMACEUTICALS、Pfizer Inc.、F. Hoffmann-La Roche Ltd、Akashi RX、TAIHO PHARMACEUTICAL CO., LTD など |
市場の定義
デュシェンヌ型筋ジストロフィー (DMD) は、進行性の筋肉の変性と衰弱を特徴とするまれな遺伝性疾患です。DMD の主な治療には、プレドニゾンやデフラザコートなどのコルチコステロイドの使用が含まれます。これらの薬剤は炎症を軽減し、筋肉の変性を遅らせ、最終的に歩行能力を延長し、筋肉機能を維持します。コルチコステロイド治療は、DMD 患者の筋力、呼吸機能、および全体的な生活の質を改善することが示されています。
デュシェンヌ型筋ジストロフィー治療の世界市場の動向
このセクションでは、市場の推進要因、利点、制約、課題について理解します。これらについては、以下で詳しく説明します。
ドライバー
- デュシェンヌ型筋ジストロフィー(DMD)の罹患率と影響の増加
デュシェンヌ型筋ジストロフィー (DMD) は、進行性の筋力低下と機能喪失を特徴とする、まれで衰弱性の遺伝性疾患です。主に男性に発症し、症状は典型的には幼少期に現れます。DMD と診断される人の数も増加しており、世界人口が引き続き増加しているため、有病率の上昇により効果的な治療法と療法の必要性が生じています。
DMDの罹患率の上昇により、患者と医療専門家による支援活動も増加しています。これらのグループは、DMDに関する認識を高め、研究資金を擁護し、潜在的な治療法の承認を早める上で重要な役割を果たしています。DMDコミュニティは成長しているため、この医療分野における技術の進歩と投資も増加しています。
- DMD障害に対する新しい治療法の導入
革新的な治療法の出現により、DMD 治療が増加し、市場の拡大に大きく貢献しています。従来の治療法は、主に症状の管理と支持療法に重点を置いていました。しかし、DMD の原因となる根本的な遺伝子変異に対処することを目指す新しい治療法の導入は、パラダイムシフトを表しています。
さらに、エクソン スキッピング薬が、DMD の有望な治療法のもう 1 つの種類として登場しました。これらの薬は、ジストロフィン遺伝子の特定のエクソンを「スキップ」するように設計されており、短縮されているものの部分的に機能するジストロフィン タンパク質の生成を可能にします。これらの薬は、病気の進行を大幅に遅らせ、DMD 患者の生活の質を向上させることができます。これらの治療法の開発と承認は、DMD 患者に新たな期待をもたらし、市場の成長をさらに促進すると期待されています。
機会
- 医療インフラの整備と投資の増加
DMD 患者は、設備の整った病院や診断施設など、より強力な医療インフラがあれば、より早く診断を受けることができます。早期診断により、タイムリーな介入と治療開始が可能になり、病気の進行を遅らせ、患者の転帰を改善できる可能性があります。先進的な医療システムには、DMD などの希少疾患に特化した専門センターやクリニックが数多くあります。これらのセンターでは、専門の医療専門家へのアクセス、理学療法、支援サービスなど、DMD 患者の生活の質を高める総合的なケアを提供しています。
発達した医療制度では、理学療法、作業療法、補助器具、カウンセリングなど、DMD 患者の生活の質を大幅に向上できる幅広い支援サービスが提供されていることがよくあります。政府、民間投資家、慈善団体は、医療インフラが十分に確立されている場合、DMD などの希少疾患の医薬品開発に投資する可能性が高くなります。この投資は、研究、臨床試験、革新的な治療法の開発を支援することができます。
抑制/挑戦
- 先進技術の欠如によりDMD治療の利用可能性が限られている
DMD の治療選択肢は限られており、患者とその家族には病気の進行を遅らせたり全体的な健康状態を改善したりするための選択肢がほとんどありません。
技術の進歩により、遺伝子治療や個別化医療アプローチなど、DMD に対する高度に専門化された革新的な治療法が開発されることが多くなっています。これらの最先端の治療法は、開発、製造、投与が複雑です。特に遺伝子治療の開発には、複雑なプロセスと特殊な製造技術が関係しています。これらの治療法では、DMD の根本的な原因に対処するために欠陥のある遺伝子の修正または置換が必要であり、治療費が高額になります。さらに、遺伝子治療を製造できる製造施設の数が限られているため、その利用がさらに制限されています。
高度な DMD 治療は複雑であるため、製造と投与が困難であり、入手が困難になることがあります。製造には専門の施設と専門知識が必要になる場合があり、これらの治療の投与には専門の医療専門家が必要になる可能性がありますが、特定の地域では医療専門家が不足している可能性があります。
最近の動向
- 2023年3月、Dyne Therapeuticsは、エクソン51スキップが可能なデュシェンヌ型筋ジストロフィー(DMD)変異の治験薬であるDYNE-251が、米国FDAの希少疾病用医薬品および小児希少疾患指定を受けたと発表しました。DYNE-251は、フェーズ1/2のDELIVER臨床試験で評価されています。これは、組織が製品カテゴリーと全体的な収益を増やすのに役立ちます。
- 2022年8月、FibroGen社は、全身性コルチコステロイドを背景としたDMDの外来患者の治療を目的としたパムレブルマブの第3相LELANTOS-2試験のトップラインデータを発表しました。これにより、同社はパイプラインポートフォリオを強化することができました。
デュシェンヌ型筋ジストロフィー治療の世界市場規模
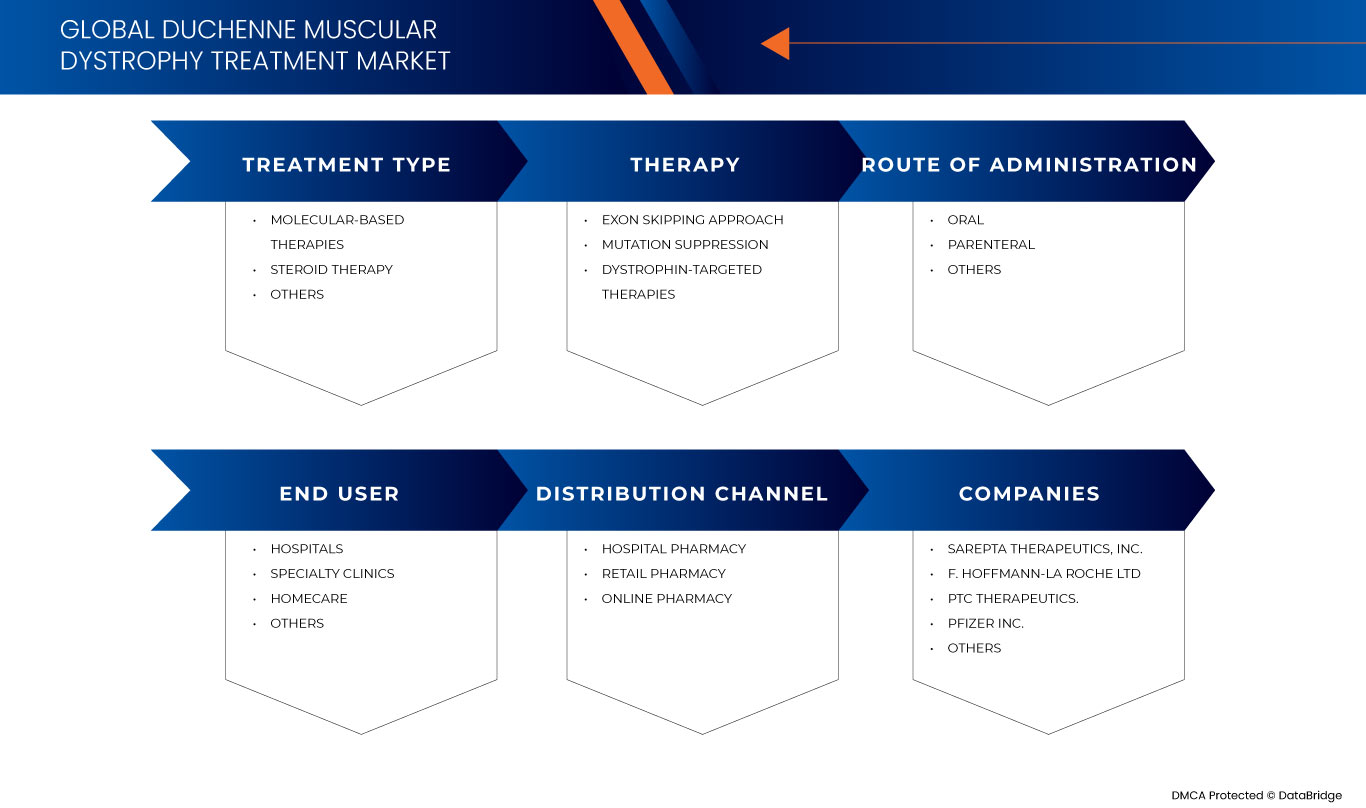
デュシェンヌ型筋ジストロフィー治療の世界市場は、治療の種類、療法、投与経路、エンドユーザー、流通チャネルに基づいて 5 つの主要なセグメントに分かれています。これらのセグメントの成長は、業界のわずかな成長セグメントの分析に役立ち、ユーザーに貴重な市場概要と市場洞察を提供して、コア市場アプリケーションを特定するための戦略的決定を下すのに役立ちます。
治療の種類
- 分子ベースの治療法
- ステロイド療法
- その他
治療の種類に基づいて、市場は分子ベースの治療、ステロイド治療、その他に分類されます。
治療
- エクソンスキッピングアプローチ
- 突然変異の抑制
- ジストロフィン標的療法
治療法に基づいて、市場はエクソンスキッピングアプローチ、変異抑制、およびジストロフィン標的療法に分類されます。
投与経路
- オーラル
- 非経口
- その他
投与経路に基づいて、市場は経口、非経口、その他に分類されます。
エンドユーザー
- 病院
- 在宅医療
- 専門クリニック
- その他
エンドユーザーに基づいて、市場は病院、在宅医療、専門クリニック、その他に分類されます。
流通チャネル
- 病院薬局
- オンライン薬局
- 小売薬局
流通チャネルに基づいて、市場は病院薬局、オンライン薬局、小売薬局に分類されます。
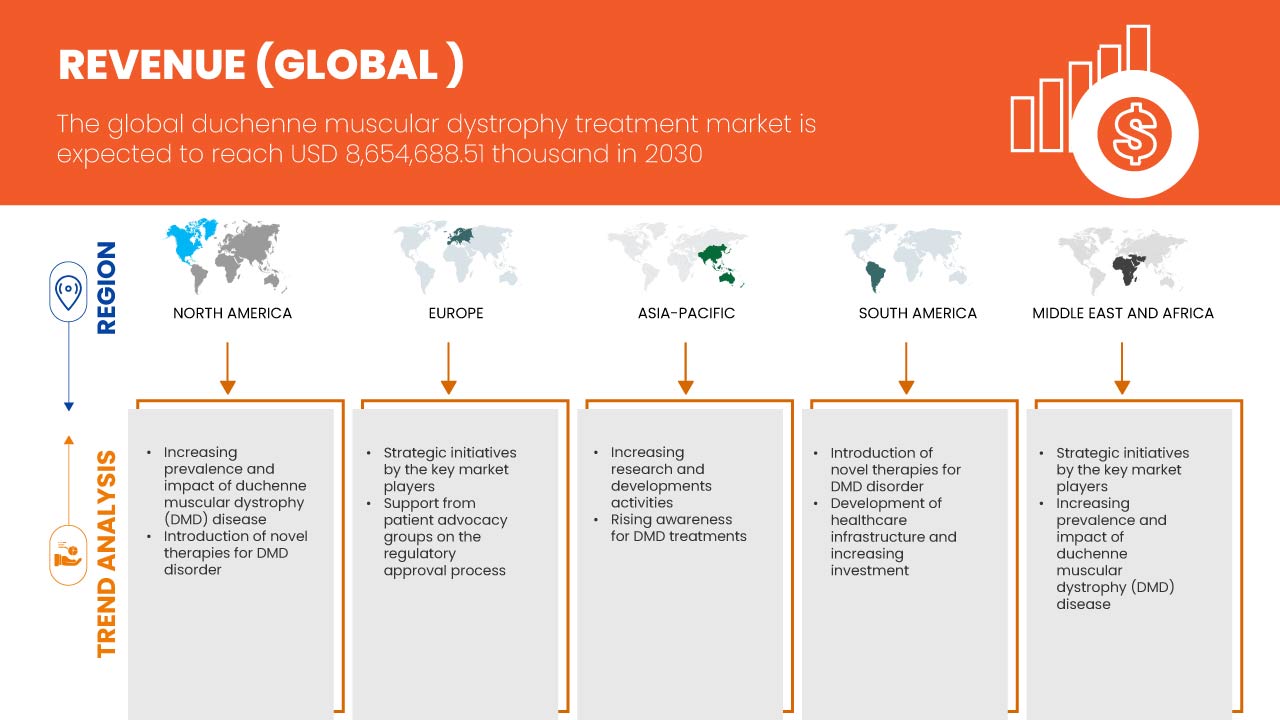
デュシェンヌ型筋ジストロフィー治療市場の世界地域分析/洞察
世界のデュシェンヌ型筋ジストロフィー治療市場は、治療の種類、療法、投与経路、エンドユーザー、流通チャネルに基づいて 5 つの主要なセグメントに分類されています。
この世界的なデュシェンヌ型筋ジストロフィー治療市場レポートで取り上げられている国は、米国、カナダ、メキシコ、ドイツ、フランス、英国、イタリア、スペイン、ロシア、トルコ、ベルギー、オランダ、スイス、その他のヨーロッパ諸国、日本、中国、韓国、インド、オーストラリア、シンガポール、タイ、マレーシア、インドネシア、フィリピン、その他のアジア太平洋諸国、ブラジル、アルゼンチン、その他の南米諸国、南アフリカ、サウジアラビア、UAE、エジプト、イスラエル、その他の中東およびアフリカ諸国です。
米国は、DMD の認知度とスクリーニングの高まりにより、北米で優位に立つと予想されています。ドイツは、筋ジストロフィーの罹患率の上昇と新しい技術進歩の浸透により、ヨーロッパで優位に立つと予想されています。中国は、主要な市場プレーヤーによる戦略的イニシアチブが大幅に拡大しているため、アジア太平洋で優位に立つと予想されています。
レポートの国別セクションでは、市場の現在および将来の傾向に影響を与える個別の市場影響要因と市場規制の変更も提供しています。下流および上流のバリュー チェーン分析、技術動向、ポーターの 5 つの力の分析、ケース スタディなどのデータ ポイントは、個々の国の市場シナリオを予測するために使用される指標の一部です。また、国別データの予測分析を提供する際には、グローバル ブランドの存在と可用性、および地元および国内ブランドとの競争が激しいか少ないために直面する課題、国内関税の影響、貿易ルートも考慮されます。
競争環境と世界デュシェンヌ型筋ジストロフィー治療市場シェア分析
デュシェンヌ型筋ジストロフィー治療の世界市場の競争状況は、競合他社の詳細を提供します。含まれる詳細には、会社概要、会社の財務状況、生み出される収益、市場の可能性、新しい市場への取り組み、世界的なプレゼンス、生産拠点と施設、生産能力、会社の強みと弱み、製品の発売、製品の幅と広さ、アプリケーションの優位性などがあります。提供されている上記のデータ ポイントは、市場に関連する会社の焦点にのみ関連しています。
世界のデュシェンヌ型筋ジストロフィー治療市場で活動している主要な市場プレーヤーには、Sarepta Therapeutics, Inc.、GSK plc.、Capricor Therapeutics, Inc.、Dyne Therapeutics、Solid Biosciences Inc.、BioMarin、Stealth BioTherapeutics Inc.、Avidity Biosciences、ReveraGen BioPharma, Inc.、PTC Therapeutics.、NS Pharma, Inc、ITALFARMACO SpA、FibroGen, Inc、SANTHERA PHARMACEUTICALS、Pfizer Inc.、F. Hoffmann-La Roche Ltd、Akashi RX、TAIHO PHARMACEUTICAL CO., LTD などがあります。
SKU-
世界初のマーケットインテリジェンスクラウドに関するレポートにオンラインでアクセスする
- インタラクティブなデータ分析ダッシュボード
- 成長の可能性が高い機会のための企業分析ダッシュボード
- カスタマイズとクエリのためのリサーチアナリストアクセス
- インタラクティブなダッシュボードによる競合分析
- 最新ニュース、更新情報、トレンド分析
- 包括的な競合追跡のためのベンチマーク分析のパワーを活用
目次
1 はじめに
1.1 研究の目的
1.2 市場の定義
1.3 デュシェンヌ型筋ジストロフィー治療薬の世界市場の概要
1.4 通貨と価格
1.5 制限
1.6 対象市場
2 市場セグメンテーション
2.1 対象市場
2.2 地理的範囲
研究期間2.3年
2.4 DBMR TRIPODデータ検証モデル
2.5 主要なオピニオンリーダーとの一次インタビュー
2.6 多変量モデリング
2.7 治療タイプセグメントライフライン曲線
2.8 市場エンドユーザーカバレッジグリッド
2.9 DBMR市場ポジショングリッド
2.1 ベンダーシェア分析
2.11 二次資料
2.12 仮定
3 概要
4つのプレミアムインサイト
4.1 PESTELのモデル
4.2 ポーターの5つの力モデル
4.3 価格分析
5 デュシェンヌ型筋ジストロフィー治療薬の世界市場:規制
5.1 米国における規制
5.2 欧州における規制
5.3 オーストラリアの規制
5.4 南アフリカの規制
5.5 ブラジルの規制
6 市場概要
6.1 ドライバー
6.1.1 デュシェンヌ型筋ジストロフィー(DMD)の罹患率と影響の増加
6.1.2 DMD障害に対する新しい治療法の導入
6.1.3 DMD治療に対する意識の高まり
6.1.4 臨床試験数の増加は最近の傾向である
6.2 拘束
6.2.1 先進技術の欠如によるDMD治療の制限
6.2.2 DMD障害の高額な治療費
6.3 機会
6.3.1 医療インフラの整備と投資の増加
6.3.2 個別化された効果的な治療に対する患者の関心の高まり
6.3.3 主要市場プレーヤーによる戦略的取り組み
6.3.4 規制承認プロセスに関する患者擁護団体からの支援
6.4 課題
6.4.1 DMD診断における標準化の欠如
6.4.2 患者の遺伝子コードの永久的な変更に関する倫理的考慮
7 治療タイプ別デュシェンヌ型筋ジストロフィー治療の世界市場
7.1 概要
7.2 分子ベースの治療法
7.2.1 アンチセンスオリゴヌクレオチド療法
7.2.1.1 エクソンディス51
7.2.1.2 アモンディス45
7.2.1.3 ヴィヨンディス53
7.3 ナンセンス変異
7.3.1.1 翻訳
7.4 ステロイド療法
7.4.1 プレドニゾン
7.4.2 デフラザコート
7.5 その他
8 デュシェンヌ型筋ジストロフィー治療薬の世界市場(治療法別)
8.1 概要
8.2 エクソンスキッピングアプローチ
8.2.1 マルチエクソンスキッピングアプローチ
8.2.2 単一エクソンスキップアプローチ
8.3 突然変異抑制
8.4 ジストロフィン標的療法
8.4.1 遺伝子治療
8.4.2 細胞療法
8.4.2.1 遺伝子編集
8.4.2.2 遺伝子の追加
9 投与経路別デュシェンヌ型筋ジストロフィー治療薬の世界市場
9.1 概要
9.2 非経口投与
9.3 口頭
9.4 その他
10 エンドユーザー別デュシェンヌ型筋ジストロフィー治療の世界市場
10.1 概要
10.2 病院
10.3 専門クリニック
10.4 ホームケア
10.5 その他
11 流通チャネル別デュシェンヌ型筋ジストロフィー治療の世界市場
11.1 概要
11.2 病院薬局
11.3 小売薬局
11.4 オンライン薬局
12 地域別デュシェンヌ型筋ジストロフィー治療の世界市場
12.1 概要
12.2 北米
12.2.1 米国
12.2.2 カナダ
12.2.3 メキシコ
12.3 ヨーロッパ
12.3.1 ドイツ
12.3.2 フランス
12.3.3 英国
12.3.4 イタリア
12.3.5 スペイン
12.3.6 ロシア
12.3.7 トルコ
12.3.8 ベルギー
12.3.9 オランダ
12.3.10 スイス
12.3.11 その他のヨーロッパ
12.4 アジア太平洋
12.4.1 中国
12.4.2 日本
12.4.3 インド
12.4.4 韓国
12.4.5 オーストラリア
12.4.6 シンガポール
12.4.7 タイ
12.4.8 マレーシア
12.4.9 インドネシア
12.4.10 フィリピン
12.4.11 その他のアジア太平洋地域
12.5 南アメリカ
12.5.1 ブラジル
12.5.2 アルゼンチン
12.5.3 南米のその他の地域
12.6 中東とアフリカ
12.6.1 南アフリカ
12.6.2 サウジアラビア
12.6.3 アラブ首長国連邦
12.6.4 エジプト
12.6.5 イスラエル
12.6.6 その他の中東およびアフリカ
13 世界のデュシェンヌ型筋ジストロフィー治療市場、企業概要
13.1 企業シェア分析: 世界
14 北米デュシェンヌ型筋ジストロフィー治療市場、企業概要
14.1 企業シェア分析: 北米
15 欧州デュシェンヌ型筋ジストロフィー治療市場、企業概要
15.1 企業シェア分析: ヨーロッパ
16 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場、企業概要
16.1 企業シェア分析: アジア太平洋地域
17 SWOT分析
18 企業プロフィール
18.1 サレプタセラピューティクス社
18.1.1 会社概要
18.1.1 収益分析
18.1.2 企業株式分析
18.1.3 製品ポートフォリオ
18.1.4 パイプラインポートフォリオ
18.1.5 最近の動向
18.2 F.ホフマン・ラ・ロッシュ株式会社
18.2.1 会社のスナップショット
18.2.2 収益分析
18.2.3 企業株式分析
18.2.4 製品ポートフォリオ
18.2.5 パイプラインポートフォリオ
18.2.6 最近の動向
18.3 PTCセラピューティクス。
18.3.1 会社のスナップショット
18.3.2 収益分析
18.3.3 企業株式分析
18.3.4 製品ポートフォリオ
18.3.5 最近の開発
18.4 ファイザー株式会社
18.4.1 会社のスナップショット
18.4.2 収益分析
18.4.3 企業株式分析
18.4.4 パイプラインポートフォリオ
18.4.5 製品ポートフォリオ
18.4.6 最近の開発
18.5 明石RX
18.5.1 会社のスナップショット
18.5.2 パイプラインポートフォリオ
18.5.3 最近の動向
18.6 アビディティバイオサイエンス
18.6.1 会社のスナップショット
18.6.2 収益分析
18.6.3 パイプラインポートフォリオ
18.6.4 最近の動向
18.7 バイオマリン
18.7.1 会社のスナップショット
18.7.2 収益分析
18.7.3 パイプラインポートフォリオ
18.7.4 最近の動向
18.8 カプリコールセラピューティクス社
18.8.1 会社のスナップショット
18.8.2 パイプラインポートフォリオ
18.8.3 最近の開発
18.9 ダイン・セラピューティクス
18.9.1 会社概要
18.9.2 パイプラインポートフォリオ
18.9.3 最近の動向
18.1 ファイブロゲン株式会社
18.10.1 会社概要
18.10.2 収益分析
18.10.3 パイプラインポートフォリオ
18.10.4 最近の開発
18.11 イタルファルマコ スパ
18.11.1 会社概要
18.11.2 パイプラインポートフォリオ
18.11.3 最近の開発
18.12 NSファーマ株式会社
18.12.1 会社概要
18.12.2 パイプラインポートフォリオ
18.12.3 最近の動向
18.13 リベラジェンバイオファーマ株式会社
18.13.1 会社概要
18.13.2 パイプラインポートフォリオ
18.13.3 最近の開発
18.14 サンセラ製薬
18.14.1 会社概要
18.14.2 収益分析
18.14.3 パイプラインポートフォリオ
18.14.4 最近の開発
18.15 大鵬薬品工業株式会社
18.15.1 会社概要
18.15.2 パイプラインポートフォリオ
18.15.3 最近の動向
18.16 ソリッドバイオサイエンス株式会社
18.16.1 会社概要
18.16.2 パイプラインポートフォリオ
18.16.3 最近の開発
18.17 ステルスバイオセラピューティクス社
18.17.1 会社概要
18.17.2 パイプラインポートフォリオ
18.17.3 最近の開発
19 アンケート
関連レポート20件
表のリスト
表1 世界のデュシェンヌ型筋ジストロフィー治療薬市場における承認薬の価格一覧
表2 デュシェンヌ型筋ジストロフィー治療の世界市場(治療タイプ別、2021年~2030年、1000米ドル)
表3 デュシェンヌ型筋ジストロフィー治療市場における分子ベース療法の世界市場、地域別、2021年~2030年(千米ドル)
表4 デュシェンヌ型筋ジストロフィー治療市場における分子ベースの治療、治療タイプ別、2021年~2030年(千米ドル)
表5 デュシェンヌ型筋ジストロフィー治療におけるアンチセンスオリゴヌクレオチド療法の世界市場、治療タイプ別、2021年~2030年(千米ドル)
表6 デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場(治療タイプ別)、2021年~2030年(千米ドル)
表7 デュシェンヌ型筋ジストロフィー治療におけるステロイド療法の世界市場、地域別、2021年~2030年(千米ドル)
表8 デュシェンヌ型筋ジストロフィー治療におけるステロイド療法の世界市場、治療タイプ別、2021年~2030年(千米ドル)
表9 デュシェンヌ型筋ジストロフィー治療市場における世界のその他の市場、地域別、2021年~2030年(千米ドル)
表10 デュシェンヌ型筋ジストロフィー治療の世界市場、治療法別、2021年~2030年(千米ドル)
表11 デュシェンヌ型筋ジストロフィー治療市場におけるエクソンスキッピングアプローチの世界市場、地域別、2021年~2030年(千米ドル)
表12 デュシェンヌ型筋ジストロフィー治療市場におけるエクソンスキッピングアプローチ、治療法別、2021年~2030年(千米ドル)
表13 デュシェンヌ型筋ジストロフィー治療における変異抑制の世界市場、地域別、2021年~2030年(千米ドル)
表14 デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、地域別、2021年~2030年(千米ドル)
表15 デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、治療別、2021年~2030年(千米ドル)
表16 デュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021年~2030年(千米ドル)
表17 デュシェンヌ型筋ジストロフィー治療の世界市場、投与経路別、2021年~2030年(千米ドル)
表18 デュシェンヌ型筋ジストロフィー治療における世界の非経口市場、地域別、2021年~2030年(千米ドル)
表19 デュシェンヌ型筋ジストロフィー治療における世界の経口薬市場、地域別、2021年~2030年(千米ドル)
表20 デュシェンヌ型筋ジストロフィー治療市場における世界のその他の企業、地域別、2021年~2030年(千米ドル)
表21 世界のデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021年~2030年(千米ドル)
表22 デュシェンヌ型筋ジストロフィー治療市場における世界の病院数、地域別、2021年~2030年(千米ドル)
表23 デュシェンヌ型筋ジストロフィー治療市場における世界の専門クリニック、地域別、2021年~2030年(千米ドル)
表24 デュシェンヌ型筋ジストロフィー治療における世界の在宅ケア市場、地域別、2021年~2030年(千米ドル)
表25 デュシェンヌ型筋ジストロフィー治療市場における世界のその他の企業、地域別、2021年~2030年(千米ドル)
表26 デュシェンヌ型筋ジストロフィー治療の世界市場、流通チャネル別、2021年~2030年(千米ドル)
表27 デュシェンヌ型筋ジストロフィー治療市場における世界の病院薬局、地域別、2021年~2030年(千米ドル)
表28 デュシェンヌ型筋ジストロフィー治療市場における世界の小売薬局、地域別、2021年~2030年(千米ドル)
表29 デュシェンヌ型筋ジストロフィー治療市場における世界のオンライン薬局、地域別、2021年~2030年(千米ドル)
表30 デュシェンヌ型筋ジストロフィー治療の世界市場、地域別、2021年~2030年(千米ドル)
表31 北米デュシェンヌ型筋ジストロフィー治療市場、国別、2021年~2030年(千米ドル) 国
表32 北米デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表33 北米の分子ベース療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表34 北米のデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表35 北米のデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(数量)
表36 北米におけるデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表37 北米におけるデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(数量)
表38 北米のデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021年~2030年(千米ドル)
表39 北米のデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021年~2030年(数量)
表40 北米デュシェンヌ型筋ジストロフィー治療市場、治療法別、2021年~2030年(千米ドル)
表41 北米のデュシェンヌ型筋ジストロフィー治療市場におけるエクソンスキッピングアプローチ、治療法別、2021年~2030年(千米ドル)
表42 北米のデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021年~2030年(千米ドル)
表43 北米デュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021年~2030年(千米ドル)
表44 北米デュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表45 北米デュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021年~2030年(千米ドル)
表46 北米デュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021年~2030年(千米ドル)
表47 米国デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表48 デュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021年~2030年(千米ドル)
表49 デュシェンヌ型筋ジストロフィー治療における米国のアンチセンスオリゴヌクレオチド療法市場、治療タイプ別、2021年~2030年(千米ドル)
表50 デュシェンヌ型筋ジストロフィー治療市場における米国のアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(数量)
表51 デュシェンヌ型筋ジストロフィー治療市場における米国のアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(ASP)
表52 米国デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表53 米国デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(数量)
表54 米国デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(ASP)
表55 デュシェンヌ型筋ジストロフィー治療における米国のステロイド療法市場、治療タイプ別、2021年~2030年(千米ドル)
表56 デュシェンヌ型筋ジストロフィー治療における米国のステロイド療法市場、治療タイプ別、2021年~2030年(数量)
表 57 米国デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表58 米国デュシェンヌ型筋ジストロフィー治療市場、治療法別、2021年~2030年(千米ドル)
表59 米国デュシェンヌ型筋ジストロフィー治療市場におけるエクソンスキッピングアプローチ、治療法別、2021年~2030年(千米ドル)
表60 米国のデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021年~2030年(千米ドル)
表61 デュシェンヌ型筋ジストロフィー治療市場における米国細胞療法、療法別、2021年~2030年(千米ドル)
表62 米国デュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表63 米国デュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021年~2030年(千米ドル)
表64 米国デュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021年~2030年(千米ドル)
表65 カナダのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表66 カナダの分子ベースの治療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表67 カナダのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表68 カナダのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(数量)
表 69 カナダのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表70 カナダのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 71 カナダのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 72 カナダのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表73 カナダのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(千米ドル)
表 74 カナダのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 75 カナダのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030 年 (ASP)
表 76 カナダのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 77 カナダのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 78 カナダ デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 79 カナダのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表80 カナダのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表81 カナダのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021年~2030年(千米ドル)
表82 カナダのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021年~2030年(千米ドル)
表83 メキシコのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表84 メキシコのデュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021年~2030年(千米ドル)
表85 メキシコのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表86 メキシコのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(数量)
表 87 メキシコのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表88 メキシコ デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表89 メキシコ デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(数量)
表90 メキシコ デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(ASP)
表91 メキシコのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(千米ドル)
表92 メキシコのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(数量)
表93 メキシコのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(ASP)
表94 メキシコのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021年~2030年(千米ドル)
表95 メキシコのデュシェンヌ型筋ジストロフィー治療市場におけるエクソンスキッピングアプローチ、治療法別、2021年~2030年(千米ドル)
表96 メキシコのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021年~2030年(千米ドル)
表97 メキシコのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021年~2030年(千米ドル)
表98 メキシコのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表99 メキシコのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021年~2030年(千米ドル)
表 100 メキシコのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030 年 (千米ドル)
表101 ヨーロッパのデュシェンヌ型筋ジストロフィー治療市場、国別、2021年~2030年(千米ドル)
表 102 ヨーロッパのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 103 ヨーロッパの分子ベースの治療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表104 ヨーロッパのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表105 ヨーロッパのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(数量)
表106 ヨーロッパのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 107 ヨーロッパのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(数量)
表108 ヨーロッパのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(千米ドル)
表 109 ヨーロッパのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 110 ヨーロッパのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 111 ヨーロッパのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 112 ヨーロッパのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 113 ヨーロッパのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表114 ヨーロッパのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表 115 欧州デュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表116 欧州デュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021年~2030年(千米ドル)
表117 ドイツデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表118 ドイツのデュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021年~2030年(千米ドル)
表119 ドイツにおけるデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表 120 ドイツ デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 121 ドイツ デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表122 ドイツのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表123 ドイツのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(数量)
表 124 ドイツのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表125 ドイツ デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(千米ドル)
表 126 ドイツ デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 127 ドイツ デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 128 ドイツのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 129 ドイツのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 130 ドイツのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 131 ドイツのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表132 ドイツのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表 133 ドイツのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030 年 (千米ドル)
表134 ドイツのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021年~2030年(千米ドル)
表135 フランスデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 136 フランス デュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021-2030年 (千米ドル)
表 137 フランス デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年 (千米ドル)
表 138 フランス デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 139 フランス デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表140 フランス デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 141 フランス デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 142 フランス デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 143 フランス デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (千米ドル)
表 144 フランス デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (数量)
表 145 フランス デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 146 フランス デュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030 年 (千米ドル)
表 147 フランスのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 148 フランス デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 149 フランス デュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030 年 (千米ドル)
表 150 フランスのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 151 フランスのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030 年 (千米ドル)
表 152 フランス デュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030 年 (千米ドル)
表153 英国デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 154 英国のデュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021-2030年 (千米ドル)
表155 デュシェンヌ型筋ジストロフィー治療市場における英国のアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表 156 英国デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 157 英国デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表158 英国デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 159 英国のナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(数量)
表 160 英国のデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表161 英国デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(千米ドル)
表 162 英国デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 163 英国デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 164 英国デュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年(千米ドル)
表 165 英国のデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 166 英国のデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 167 英国のデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表168 英国デュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表 169 英国のデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030 年 (千米ドル)
表 170 英国デュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 171 イタリアのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 172 イタリアのデュシェンヌ型筋ジストロフィー治療市場における分子ベースの治療法、治療タイプ別、2021-2030年 (千米ドル)
表 173 イタリアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年 (千米ドル)
表 174 イタリアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 175 イタリアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表 176 イタリアのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 177 イタリア デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (数量)
表 178 イタリア デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 179 イタリアのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年 (千米ドル)
表 180 イタリアのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (数量)
表 181 イタリアのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 182 イタリアデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030 年 (千米ドル)
表 183 イタリアのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 184 イタリアのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 185 イタリアのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 186 イタリアのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 187 イタリアのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030 年 (千米ドル)
表 188 イタリアのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 189 スペインのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 190 スペインのデュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021-2030年 (千米ドル)
表 191 スペインのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 192 スペインのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 193 スペインのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表 194 スペイン デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 195 スペイン デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 196 スペイン デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 197 スペインのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(千米ドル)
表 198 スペインのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 199 スペインのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 200 スペインのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 201 スペインのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 202 スペインのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 203 スペインのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年(千米ドル)
表 204 スペインのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 205 スペインのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表 206 スペインのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 207 ロシアのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 208 ロシアのデュシェンヌ型筋ジストロフィー治療市場における分子ベースの治療法、治療タイプ別、2021-2030年 (千米ドル)
表 209 ロシアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 210 ロシアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 211 ロシアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表212 ロシアのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 213 ロシアのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(数量)
表 214 ロシアのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030 年 (ASP)
表 215 ロシアのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年 (千米ドル)
表 216 ロシアのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 217 ロシアのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 218 ロシアのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 219 ロシアのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 220 ロシアのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 221 ロシアのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 222 ロシアのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 223 ロシアのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表 224 ロシアのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 225 トルコのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 226 トルコのデュシェンヌ型筋ジストロフィー治療市場における分子ベースの治療法、治療タイプ別、2021-2030年(千米ドル)
表 227 トルコのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 228 トルコのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 229 トルコのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年 (ASP)
表 230 トルコ デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 231 トルコ デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 232 トルコ デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 233 トルコのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(千米ドル)
表 234 トルコのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 235 トルコのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 236 トルコのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年(千米ドル)
表 237 トルコのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 238 トルコ デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 239 トルコのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 240 トルコのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 241 トルコのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表 242 トルコのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 243 ベルギーのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 244 ベルギーのデュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021-2030年(千米ドル)
表 245 ベルギーのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 246 ベルギーのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 247 ベルギーのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表 248 ベルギーのデュシェンヌ型筋ジストロフィー治療市場(治療タイプ別)、2021年~2030年(千米ドル)
表 249 ベルギーのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 250 ベルギーのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 251 ベルギーのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(千米ドル)
表 252 ベルギーのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 253 ベルギーのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 254 ベルギーのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 255 ベルギーのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 256 ベルギーのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 257 ベルギーのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 258 ベルギーのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 259 ベルギーのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表 260 ベルギーのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 261 オランダのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 262 オランダの分子ベースの治療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 263 オランダのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 264 オランダのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 265 オランダのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表 266 オランダのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 267 オランダのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(数量)
表 268 オランダのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (ASP)
表 269 オランダのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(千米ドル)
表 270 オランダのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 271 オランダのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 272 オランダのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 273 オランダのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 274 オランダのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 275 オランダのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 276 オランダのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 277 オランダのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表 278 オランダのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 279 スイスのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 280 スイスのデュシェンヌ型筋ジストロフィー治療市場における分子ベースの治療法、治療タイプ別、2021年~2030年(千米ドル)
表 281 スイスのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 282 スイスのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 283 スイスのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年 (ASP)
表 284 スイスのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 285 スイスのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 286 スイスのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年 (ASP)
表 287 スイスのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(千米ドル)
表 288 スイスのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 289 スイスのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年 (ASP)
表 290 スイスのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 291 スイスのデュシェンヌ型筋ジストロフィー治療市場におけるエクソンスキッピングアプローチ、治療法別、2021-2030年(千米ドル)
表 292 スイスのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 293 スイスのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年(千米ドル)
表 294 スイスのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表 295 スイスのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表 296 スイスのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021年~2030年(千米ドル)
表 297 ヨーロッパのその他の地域におけるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 298 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場、国別、2021年~2030年(千米ドル)
表 299 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 300 アジア太平洋地域の分子ベース療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 301 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 302 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表303 アジア太平洋地域のデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 304 アジア太平洋地域のデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 305 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030年(千米ドル)
表 306 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030年(数量)
表 307 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年(千米ドル)
表 308 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場におけるエクソンスキッピングアプローチ、治療法別、2021-2030年(千米ドル)
表 309 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 310 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 311 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 312 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表313 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021年~2030年(千米ドル)
表314 中国デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 315 デュシェンヌ型筋ジストロフィー治療市場における中国の分子ベース療法、治療タイプ別、2021-2030年(千米ドル)
表316 デュシェンヌ型筋ジストロフィー治療市場における中国のアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表 317 デュシェンヌ型筋ジストロフィー治療市場における中国のアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 318 デュシェンヌ型筋ジストロフィー治療市場における中国のアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年 (ASP)
表319 中国のデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 320 中国におけるデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 321 中国におけるデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 322 中国のデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(千米ドル)
表 323 中国のデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 324 中国デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 325 中国デュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年(千米ドル)
表 326 中国のデュシェンヌ型筋ジストロフィー治療市場におけるエクソンスキッピングアプローチ、治療法別、2021-2030年(千米ドル)
表 327 中国デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 328 デュシェンヌ型筋ジストロフィー治療市場における中国の細胞療法、療法別、2021-2030年(千米ドル)
表 329 中国デュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 330 中国デュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表331 中国デュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021年~2030年(千米ドル)
表332 日本デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表333 日本デュシェンヌ型筋ジストロフィー治療市場における分子療法、治療タイプ別、2021年~2030年(千米ドル)
表334 デュシェンヌ型筋ジストロフィー治療における日本アンチセンスオリゴヌクレオチド療法市場、治療タイプ別、2021年~2030年(千米ドル)
表335 デュシェンヌ型筋ジストロフィー治療における日本アンチセンスオリゴヌクレオチド療法市場、治療タイプ別、2021年~2030年(数量)
表 336 デュシェンヌ型筋ジストロフィー治療における日本アンチセンスオリゴヌクレオチド療法市場、治療タイプ別、2021-2030年 (ASP)
表337 日本デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表338 日本におけるデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(数量)
表 339 日本 デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年 (ASP)
表340 デュシェンヌ型筋ジストロフィー治療におけるステロイド療法の日本市場、治療タイプ別、2021年~2030年(千米ドル)
表341 デュシェンヌ型筋ジストロフィー治療におけるステロイド療法の日本市場、治療タイプ別、2021年~2030年(数量)
表 342 デュシェンヌ型筋ジストロフィー治療におけるステロイド療法の日本市場、治療タイプ別、2021-2030年 (ASP)
表 343 日本デュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年(千米ドル)
表 344 日本におけるエクソンスキッピングアプローチによるデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年(千米ドル)
表 345 日本デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 346 デュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年(千米ドル)
表 347 日本デュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 348 日本デュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年(千米ドル)
表 349 日本デュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 350 インド デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030 年 (千米ドル)
表 351 インド デュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021-2030年 (千米ドル)
表 352 インド デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (千米ドル)
表 353 インド デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (数量)
表 354 インド デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表 355 インド デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 356 インド デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 357 インド デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 358 インド デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (千米ドル)
表 359 インド デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (数量)
表 360 インド デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 361 インドのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 362 インド デュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 363 インド デュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030 年 (千米ドル)
表 364 インド デュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030 年 (千米ドル)
表 365 インド デュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030 年 (千米ドル)
表 366 インド デュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030 年 (千米ドル)
表 367 韓国デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 368 韓国の分子ベース療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 369 韓国のデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 370 韓国のデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 371 韓国のデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表 372 韓国のナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 373 韓国のデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 374 韓国のデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 375 韓国のデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(千米ドル)
表 376 韓国のデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 377 韓国のデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 378 韓国の筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 379 韓国のデュシェンヌ型筋ジストロフィー治療市場におけるエクソンスキッピングアプローチ、治療法別、2021-2030年(千米ドル)
表 380 韓国のデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 381 韓国のデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年(千米ドル)
表 382 韓国デュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 383 韓国デュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年(千米ドル)
表 384 韓国デュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 385 オーストラリアのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 386 オーストラリア デュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021-2030年 (千米ドル)
表 387 オーストラリアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 388 オーストラリアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 389 オーストラリアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表 390 オーストラリア デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 391 オーストラリア デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 392 オーストラリア デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 393 オーストラリア デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年 (千米ドル)
表 394 オーストラリア デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 395 オーストラリアのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030 年 (ASP)
表 396 オーストラリアのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年(千米ドル)
表 397 オーストラリア デュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 398 オーストラリア デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 399 オーストラリアのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表400 オーストラリアのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表 401 オーストラリアのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030 年 (千米ドル)
表402 オーストラリアのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021年~2030年(千米ドル)
表403 シンガポールのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表404 シンガポールの分子ベース療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表405 シンガポールのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表406 シンガポールのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(数量)
表 407 シンガポールのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表408 シンガポールのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表409 シンガポールのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(数量)
表 410 シンガポールのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表411 シンガポールのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(千米ドル)
表412 シンガポールのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(数量)
表 413 シンガポールのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表414 シンガポールのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021年~2030年(千米ドル)
表 415 シンガポールのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 416 シンガポールのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 417 シンガポールのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表418 シンガポールのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表419 シンガポールのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021年~2030年(千米ドル)
表420 シンガポールのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021年~2030年(千米ドル)
表421 タイのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 422 タイのデュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021-2030年 (千米ドル)
表423 タイのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表424 タイのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(数量)
表 425 タイのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表426 タイのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表427 タイのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(数量)
表 428 タイのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表429 タイのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(千米ドル)
表 430 タイのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 431 タイのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年 (ASP)
表 432 タイのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年(千米ドル)
表 433 タイのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 434 タイのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 435 タイのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表436 タイのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表 437 タイのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表438 タイのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021年~2030年(千米ドル)
表439 マレーシアのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 440 マレーシア デュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021-2030年 (千米ドル)
表441 マレーシアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表 442 マレーシアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 443 マレーシアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表444 マレーシアのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表445 マレーシアのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(数量)
表 446 マレーシア デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表447 マレーシアのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(千米ドル)
表 448 マレーシアのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 449 マレーシアのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030 年 (ASP)
表 450 マレーシアのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 451 マレーシアのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 452 マレーシア デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 453 マレーシアのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表454 マレーシアのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表 455 マレーシアのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表 456 マレーシアのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 457 インドネシアのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 458 インドネシア デュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021-2030年 (千米ドル)
表 459 インドネシアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 460 インドネシアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 461 インドネシアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年 (ASP)
表462 インドネシア デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表463 インドネシアのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(数量)
表 464 インドネシア デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 465 インドネシアのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030年(千米ドル)
表 466 インドネシアのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030年(数量)
表 467 インドネシアのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030 年 (ASP)
表 468 インドネシアのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年(千米ドル)
表 469 インドネシアのエクソン スキッピング アプローチによるデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021 年 - 2030 年 (千米ドル)
表 470 インドネシア デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 471 インドネシア デュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 472 インドネシアのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 473 インドネシアのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表 474 インドネシアのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 475 フィリピンのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 476 フィリピンの分子ベースの治療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 477 フィリピンのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 478 フィリピンのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 479 フィリピンのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表 480 フィリピンのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 481 フィリピンのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(数量)
表 482 フィリピンのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 483 フィリピンのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(千米ドル)
表 484 フィリピンのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 485 フィリピンのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 486 フィリピンデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年(千米ドル)
表 487 フィリピンのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 488 フィリピンのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 489 フィリピンのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 490 フィリピンのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 491 フィリピンのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表 492 フィリピンのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 493 アジア太平洋地域のその他のデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 494 南米デュシェンヌ型筋ジストロフィー治療市場、国別、2021-2030年(千米ドル)
表 495 南米デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 496 南米デュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021-2030年(千米ドル)
表 497 南米デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 498 南米デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 499 南米のナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 500 南米のナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030 年 (数量)
表501 南米のデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(千米ドル)
表 502 南米のデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 503 南米デュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年(千米ドル)
表 504 南米のデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 505 南米のデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 506 南米デュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年(千米ドル)
表 507 南米デュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 508 南米デュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030 年 (千米ドル)
表 509 南米デュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表510 ブラジルのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表511 ブラジルのデュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021年~2030年(千米ドル)
表512 ブラジルのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表513 ブラジルのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(数量)
表 514 ブラジルのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表515 ブラジルのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表516 ブラジルのデュシェンヌ型筋ジストロフィー治療市場におけるナンセンス変異、治療タイプ別、2021年~2030年(数量)
表 517 ブラジルのデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表518 ブラジルのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(千米ドル)
表519 ブラジルのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021年~2030年(数量)
表 520 ブラジルのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表521 ブラジルのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021年~2030年(千米ドル)
表 522 ブラジルのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 523 ブラジル デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 524 ブラジルのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表525 ブラジルのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021年~2030年(千米ドル)
表 526 ブラジルのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表527 ブラジルのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021年~2030年(千米ドル)
表 528 アルゼンチン デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 529 アルゼンチンの分子ベースの治療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表530 アルゼンチンのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表 531 アルゼンチンのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 532 アルゼンチンのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表533 アルゼンチン デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 534 アルゼンチン デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 535 アルゼンチン デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 536 アルゼンチンのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(千米ドル)
表 537 アルゼンチンのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 538 アルゼンチンのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 539 アルゼンチンのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 540 アルゼンチン デュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 541 アルゼンチン デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 542 アルゼンチンのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 543 アルゼンチンのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 544 アルゼンチン デュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030 年 (千米ドル)
表 545 アルゼンチンのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030 年 (千米ドル)
表 546 南米のその他の地域におけるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 547 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィー治療市場、国別、2021-2030年 (千米ドル)
表 548 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表549 中東およびアフリカの分子ベースの治療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表550 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表551 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(数量)
表552 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(千米ドル)
表553 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021年~2030年(数量)
表554 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021年~2030年(千米ドル)
表 555 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 556 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 557 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 558 中東およびアフリカのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 559 中東およびアフリカのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 560 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030 年 (千米ドル)
表 561 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030 年 (千米ドル)
表 562 中東およびアフリカにおけるデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030 年 (千米ドル)
表 563 南アフリカのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 564 南アフリカの分子ベースの治療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 565 南アフリカのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 566 南アフリカのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 567 南アフリカのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表 568 南アフリカのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 569 南アフリカのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(数量)
表 570 南アフリカのナンセンス変異デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030 年 (ASP)
表 571 南アフリカのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030年 (千米ドル)
表 572 南アフリカのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030年(数量)
表 573 南アフリカのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030 年 (ASP)
表 574 南アフリカのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 575 南アフリカのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 576 南アフリカのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 577 南アフリカのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 578 南アフリカのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 579 南アフリカのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030 年 (千米ドル)
表 580 南アフリカのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 581 サウジアラビアのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 582 サウジアラビア デュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021-2030年 (千米ドル)
表 583 サウジアラビアにおけるデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年 (千米ドル)
表 584 サウジアラビアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 585 サウジアラビアのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表 586 サウジアラビア デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 587 サウジアラビア デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 588 サウジアラビア デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 589 サウジアラビアのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030年 (千米ドル)
表 590 サウジアラビアのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 591 サウジアラビアのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030 年 (ASP)
表 592 サウジアラビアのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 593 サウジアラビア デュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 594 サウジアラビア デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年 (千米ドル)
表 595 サウジアラビアのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 596 サウジアラビアのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年 (千米ドル)
表 597 サウジアラビアのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表 598 サウジアラビアのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年 (千米ドル)
表 599 UAE デュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030 年 (千米ドル)
表 600 UAE デュシェンヌ型筋ジストロフィー治療市場における分子ベース療法、治療タイプ別、2021-2030 年 (千米ドル)
表 601 UAE デュシェンヌ型筋ジストロフィー治療市場における抗センスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (千米ドル)
表 602 UAE デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (数量)
表 603 UAE デュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表 604 UAE デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 605 UAE デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 606 UAE デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 607 UAE デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (千米ドル)
表 608 UAE デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (数量)
表 609 UAE デュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 610 UAE デュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030 年 (千米ドル)
表 611 UAE デュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 612 UAE デュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030 年 (千米ドル)
表 613 UAE デュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030 年 (千米ドル)
表 614 UAE デュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030 年 (千米ドル)
表 615 UAE デュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030 年 (千米ドル)
表 616 UAE デュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030 年 (千米ドル)
表617 エジプトのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021年~2030年(千米ドル)
表 618 エジプトの分子ベースの治療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表619 エジプトのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(千米ドル)
表620 エジプトのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021年~2030年(数量)
表 621 エジプトのデュシェンヌ型筋ジストロフィー治療市場におけるアンチセンスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表622 エジプト デュシェンヌ型筋ジストロフィー治療市場におけるナンセンス変異、治療タイプ別、2021年~2030年(千米ドル)
表 623 エジプト デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 624 エジプト デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030 年 (ASP)
表 625 エジプトのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(千米ドル)
表 626 エジプトのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030年(数量)
表 627 エジプトのデュシェンヌ型筋ジストロフィー治療におけるステロイド療法市場、治療タイプ別、2021-2030 年 (ASP)
表 628 エジプトのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 629 エジプトのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 630 エジプトのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 631 エジプトのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 632 エジプトのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 633 エジプトのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表 634 エジプトのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 635 イスラエルのデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 636 イスラエルの分子ベースの治療法によるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年(千米ドル)
表 637 イスラエルのデュシェンヌ型筋ジストロフィー治療市場における抗センスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(千米ドル)
表 638 イスラエルのデュシェンヌ型筋ジストロフィー治療市場における抗センスオリゴヌクレオチド療法、治療タイプ別、2021-2030年(数量)
表 639 イスラエルのデュシェンヌ型筋ジストロフィー治療市場における抗センスオリゴヌクレオチド療法、治療タイプ別、2021-2030 年 (ASP)
表 640 イスラエル デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年 (千米ドル)
表 641 イスラエル デュシェンヌ型筋ジストロフィーのナンセンス変異治療市場、治療タイプ別、2021-2030年(数量)
表 642 イスラエル デュシェンヌ型筋ジストロフィー治療市場におけるナンセンス変異、治療タイプ別、2021-2030 年 (ASP)
表 643 イスラエルのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(千米ドル)
表 644 イスラエルのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030年(数量)
表 645 イスラエルのデュシェンヌ型筋ジストロフィー治療市場におけるステロイド療法、治療タイプ別、2021-2030 年 (ASP)
表 646 イスラエルのデュシェンヌ型筋ジストロフィー治療市場、治療法別、2021-2030年 (千米ドル)
表 647 イスラエルのデュシェンヌ型筋ジストロフィー治療市場におけるエクソン スキッピング アプローチ、治療法別、2021 年 - 2030 年 (千米ドル)
表 648 イスラエルのデュシェンヌ型筋ジストロフィー治療市場におけるジストロフィン標的療法、療法別、2021-2030年(千米ドル)
表 649 イスラエルのデュシェンヌ型筋ジストロフィー治療市場における細胞療法、療法別、2021-2030年 (千米ドル)
表 650 イスラエルのデュシェンヌ型筋ジストロフィー治療市場、投与経路別、2021-2030年(千米ドル)
表 651 イスラエルのデュシェンヌ型筋ジストロフィー治療市場、エンドユーザー別、2021-2030年 (千米ドル)
表 652 イスラエルのデュシェンヌ型筋ジストロフィー治療市場、流通チャネル別、2021-2030年(千米ドル)
表 653 中東およびアフリカのその他の地域におけるデュシェンヌ型筋ジストロフィー治療市場、治療タイプ別、2021-2030年 (千米ドル)
図表一覧
図1 世界のデュシェンヌ型筋ジストロフィー治療市場:セグメンテーション
図2 デュシェンヌ型筋ジストロフィー治療薬の世界市場:データの三角測量
図3 世界のデュシェンヌ型筋ジストロフィー治療市場: DROC分析
図4 デュシェンヌ型筋ジストロフィー治療薬の世界市場:世界市場と地域市場の比較分析
図5 世界のデュシェンヌ型筋ジストロフィー治療市場:企業調査分析
図6 デュシェンヌ型筋ジストロフィー治療の世界市場:インタビュー人口統計
図 7 世界のデュシェンヌ型筋ジストロフィー治療市場: 市場エンドユーザーカバレッジグリッド
図 8 世界のデュシェンヌ型筋ジストロフィー治療市場: DBMR 市場ポジション グリッド
図9 デュシェンヌ型筋ジストロフィー治療薬の世界市場:ベンダーシェア分析
図10 デュシェンヌ型筋ジストロフィー治療の世界市場:セグメンテーション
図11 デュシェンヌ型筋ジストロフィー(DMD)の有病率と影響の上昇、およびDMD障害に対する新しい治療法の導入により、2023年から2030年にかけて世界のデュシェンヌ型筋ジストロフィー治療市場の成長が促進されると予想されています。
図12 分子ベースの治療セグメントは、2023年と2030年に世界のデュシェンヌ型筋ジストロフィー治療市場で最大のシェアを占めると予想されています。
図13 世界のデュシェンヌ型筋ジストロフィー治療市場の推進要因、制約要因、機会、課題
図14 デュシェンヌ型筋ジストロフィー治療の世界市場:治療タイプ別、2022年
図15 デュシェンヌ型筋ジストロフィー治療の世界市場:治療タイプ別、2023年~2030年(千米ドル)
図16 世界のデュシェンヌ型筋ジストロフィー治療市場:治療タイプ別、CAGR(2023-2030年)
図17 デュシェンヌ型筋ジストロフィー治療の世界市場:治療タイプ別、ライフライン曲線
図18 デュシェンヌ型筋ジストロフィー治療の世界市場:治療法別、2022年
図19 デュシェンヌ型筋ジストロフィー治療の世界市場:治療法別、2023年~2030年(千米ドル)
図20 世界のデュシェンヌ型筋ジストロフィー治療市場:治療法別、CAGR(2023-2030年)
図21 デュシェンヌ型筋ジストロフィー治療の世界市場:治療法別、ライフライン曲線
図22 デュシェンヌ型筋ジストロフィー治療薬の世界市場:投与経路別、2022年
図23 デュシェンヌ型筋ジストロフィー治療の世界市場:投与経路別、2023年~2030年(千米ドル)
図24 デュシェンヌ型筋ジストロフィー治療の世界市場:投与経路別、CAGR(2023-2030年)
図25 デュシェンヌ型筋ジストロフィー治療薬の世界市場:投与経路別、ライフライン曲線
図26 世界のデュシェンヌ型筋ジストロフィー治療市場:エンドユーザー別、2022年
図 27 世界のデュシェンヌ型筋ジストロフィー治療市場: エンドユーザー別、2023-2030 年 (千米ドル)
図28 世界のデュシェンヌ型筋ジストロフィー治療市場:エンドユーザー別、CAGR(2023-2030年)
図29 世界のデュシェンヌ型筋ジストロフィー治療市場:エンドユーザー別、ライフライン曲線
図30 世界のデュシェンヌ型筋ジストロフィー治療市場:流通チャネル別、2022年
図31 世界のデュシェンヌ型筋ジストロフィー治療市場:流通チャネル別、2023年~2030年(千米ドル)
図32 世界のデュシェンヌ型筋ジストロフィー治療市場:流通チャネル別、CAGR(2023-2030年)
図33 デュシェンヌ型筋ジストロフィー治療薬の世界市場:流通チャネル別、ライフライン曲線
図34 デュシェンヌ型筋ジストロフィー治療の世界市場スナップショット
図35 デュシェンヌ型筋ジストロフィー治療の世界市場:企業シェア2022(%)
図36 北米デュシェンヌ型筋ジストロフィー治療市場:企業シェア2022(%)
図37 ヨーロッパのデュシェンヌ型筋ジストロフィー治療市場:企業シェア2022(%)
図38 アジア太平洋地域のデュシェンヌ型筋ジストロフィー治療市場:企業シェア2022(%)
調査方法
データ収集と基準年分析は、大規模なサンプル サイズのデータ収集モジュールを使用して行われます。この段階では、さまざまなソースと戦略を通じて市場情報または関連データを取得します。過去に取得したすべてのデータを事前に調査および計画することも含まれます。また、さまざまな情報ソース間で見られる情報の不一致の調査も含まれます。市場データは、市場統計モデルと一貫性モデルを使用して分析および推定されます。また、市場シェア分析と主要トレンド分析は、市場レポートの主要な成功要因です。詳細については、アナリストへの電話をリクエストするか、お問い合わせをドロップダウンしてください。
DBMR 調査チームが使用する主要な調査方法は、データ マイニング、データ変数が市場に与える影響の分析、および一次 (業界の専門家) 検証を含むデータ三角測量です。データ モデルには、ベンダー ポジショニング グリッド、市場タイムライン分析、市場概要とガイド、企業ポジショニング グリッド、特許分析、価格分析、企業市場シェア分析、測定基準、グローバルと地域、ベンダー シェア分析が含まれます。調査方法について詳しくは、お問い合わせフォームから当社の業界専門家にご相談ください。
カスタマイズ可能
Data Bridge Market Research は、高度な形成的調査のリーダーです。当社は、既存および新規のお客様に、お客様の目標に合致し、それに適したデータと分析を提供することに誇りを持っています。レポートは、対象ブランドの価格動向分析、追加国の市場理解 (国のリストをお問い合わせください)、臨床試験結果データ、文献レビュー、リファービッシュ市場および製品ベース分析を含めるようにカスタマイズできます。対象競合他社の市場分析は、技術ベースの分析から市場ポートフォリオ戦略まで分析できます。必要な競合他社のデータを、必要な形式とデータ スタイルでいくつでも追加できます。当社のアナリスト チームは、粗い生の Excel ファイル ピボット テーブル (ファクト ブック) でデータを提供したり、レポートで利用可能なデータ セットからプレゼンテーションを作成するお手伝いをしたりすることもできます。