Asia-Pacific Stool Diagnostic Market, By Test Type (Fecal Occult Blood Test, Fecal Biomarker Test, Pathogen Tests, Stool Antigen Test, Fecal Elastase Test, Microscopy Test, and Other Tests), Product Type (Instruments and Reagents), Mode (Laboratories Based Testing and Point of Care Testing), Application (Irritable Bowel Syndrome, Ulcerative Colitis, Crohn's Disease, Cancer, Diarrhea, Hemorrhoids, and Others), End User (Hospitals, Diagnostic Centers, Laboratory, Specialty Clinics, Academic Institutions and Research Centers, and Others), Distribution Channel (Direct Tender, Retail Sales, Online Sales, and Others) - Industry Trends and Forecast to 2030.
Asia-Pacific Stool Diagnostic Market Analysis and Insights
The increasing awareness about stool diagnosis has increased the demand for the market. The rising healthcare expenditure for better health services also contributes to market growth—the focus of major market players on various service launches and approvals during the crucial period. In addition, the increase in improved advancement of processes and techniques also contributes to the rising demand for stool diagnostics.
The Asia-Pacific stool diagnostic market is expected to grow in the forecast year due to the rise in market players and the availability of advanced products and services. Along with this, manufacturers are also engaged in the developmental activity for launching novel services and efficient, accurate stool diagnosis in the market.
Increasing healthcare expenditure on advancement and development is expected to give opportunities to the market. However, the high cost of tests may challenge market growth.
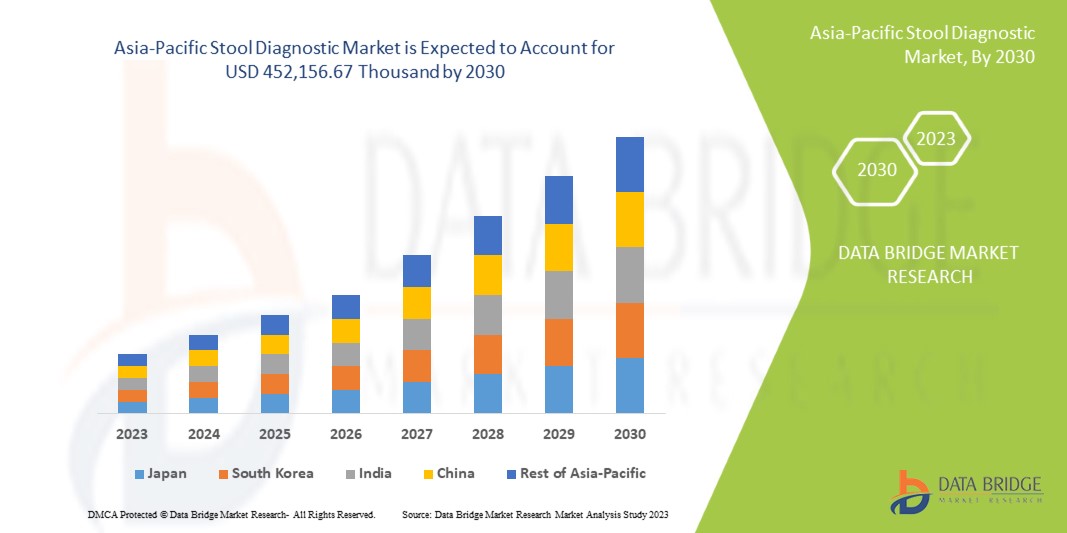
Data Bridge Market Research analyzes that the Asia-Pacific stool diagnostic market is expected to reach USD 452,156.67 thousand by 2030, at a CAGR of 7.1% during the forecast period.
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015-2020) |
|
Quantitative Units |
Revenue in USD Million, Pricing in USD |
|
Segments Covered |
By Test Type (Fecal Occult Blood Test, Fecal Biomarker Test, Pathogen Tests, Stool Antigen Test, Fecal Elastase Test, Microscopy Test, and Other Tests), Product Type (Instruments and Reagents), Mode (Laboratories Based Testing and Point of Care Testing), Application (Irritable Bowel Syndrome, Ulcerative Colitis, Crohn's Disease, Cancer, Diarrhea, Hemorrhoids, and Others), End User (Hospitals, Diagnostic Centers, Laboratory, Specialty Clinics, Academic Institutions, and Research Centers, and Others), Distribution Channel (Direct Tender, Retail Sales, Online Sales, and Others) |
|
Countries Covered |
Japan, China, South Korea, India, Singapore, Thailand, Indonesia, Malaysia, Philippines, Australia, New Zealand, Vietnam, Taiwan, Brunei, Nepal, Bangladesh, Sri Lanka, Pakistan, Hong Kong, Maldives, Mongolia, Cambodia. Laos, and Rest of Asia-Pacific |
|
Market Players Covered |
Some major companies dealing in the Asia-Pacific stool diagnostic market are Abbott, Meridian Bioscience Inc., BIOMERIEUX, Beckman Coulter, Inc. (Subsidiary of Danaher), DiaSorin S.p.A., Runmei, Molbio Diagnostics Pvt. Ltd., Quidel Corporation, Prenetics Limited, Sekisui Diagnostics (A Part of SEKISUI Chemical Co. Ltd.), BioVendor Group, LifeSign LLC, Alfa Scientific Designs, Inc., Eiken Chemical Co., Ltd., SD Biosensor, INC., Cenogenics Corporation, TransGen Biotech Co., Ltd., AdvaCare Pharma, Kibion GmbH, and Beijing Huagen Anbang Technology Co., Ltd. among others. |
Asia-Pacific Stool Diagnostic Market Definition
Stool diagnostic is a series of tests performed on a stool (feces) sample to help diagnose certain digestive tract conditions. Some of these conditions include infection (from parasites, viruses, or bacteria), poor nutrient absorption, and cancer. Microscopic examination and chemical, immunologic, and microbiologic tests are all part of a stool analysis. Leukocytes, occult blood, fat, sugars (reducing substances), pH, pancreatic enzymes, alpha-1 antitrypsin, calprotectin, and infectious causes can all be detected in stool samples with the help of stool diagnosis.
Stool tests can help determine whether or not a bacteria or other microorganism has infected the intestines. However, not all microorganisms in the gut are harmful; in fact, some are required for normal digestion. When harmful bacteria or parasites infect the intestines, they cause bloody diarrhea, and stool diagnosis aid in determining the source of this condition.
Asia-Pacific Stool Diagnostic Market Dynamics
This section deals with understanding the market drivers, advantages, opportunities, restraints, and challenges. All of this is discussed in detail below:
Driver
- INCREASE IN THE PREVALENCE OF GASTROINTESTINAL DISEASE
The increasing prevalence of gastrointestinal diseases is driving the demand for stool diagnosis. Gastrointestinal diseases can cause many symptoms, including diarrhea, constipation, abdominal pain, and nausea. Various conditions, including infections, and inflammatory bowel disease, can cause these symptoms.
In addition to the growing demand for stool diagnosis for gastrointestinal diseases, there is increasing interest in using stool tests to monitor the microbiome. The microbiome refers to the community of microorganisms that live in the gut and play an important role in digestive and overall health. Stool tests can provide information about the composition and function of the microbiome, which can be useful for identifying dysbiosis (an imbalance in the microbiome) and guiding interventions to improve gut health.
The growing use of non-invasive stool tests for screening and monitoring the microbiome can potentially improve patient outcomes and reduce the burden of gastrointestinal diseases. Thus, the increasing prevalence of gastrointestinal diseases drives the demand for stool diagnosis, particularly in Asia-Pacific.
Restraint
- STRICT GOVERNMENT REGULATIONS
The use of various testing kits across the Asia-Pacific is rapidly increasing. With the increasing prevalence of various gastrointestinal diseases and cancers, there is a need for timely diagnosis and treatment. At the same time, the manufacturers in the market must follow certain regulations to get approval from the upper authorities for launching the product. These stringent guidelines need to be followed; this is one of the most difficult tasks of all the steps.
The increased prevalence of various diseases surged the demand for treatment options amongst the population. However, the uncertainty and risks involved with these stool diagnostics result in stringent and complex regulatory landscapes. As this method of clearance of the products is rigorous and time-consuming, products find it difficult to get approved due to not complying with the regulations. Therefore, the stringent rules and regulations for product approval restrain the growth of the Asia-Pacific stool diagnostic market.
Opportunity
ADVANCEMENTS IN SAMPLE COLLECTION AND HANDLING FOR STOOL DIAGNOSTICS
Advancements in sample collection and handling have played a crucial role in improving the accuracy and reliability of stool diagnostics. Traditional stool diagnostic tests require patients to collect and handle their stool samples, which could be inconvenient and unpleasant. However, advances in sample collection and handling technologies have made stool diagnostics more patient-friendly, efficient, and accurate.
Thus, advancements have made stool diagnostics more accessible and efficient, allowing for earlier detection and treatment of gastrointestinal disorders. As technology advances, we can expect more advancements in sample collection and handling to benefit the field of stool diagnostics. Thus, advancements in sample collection and handling provide an opportunity to improve the process's accuracy, reliability, and patient friendliness.
Challenge
LACK OF SKILLED AND CERTIFIED PROFESSIONALS
The requirement for skilled and certified professionals is a big challenge for stool diagnosis. The demand for stool diagnosis is increased due to increased gastrointestinal disease and colorectal cancer cases in Asia-Pacific. Still, the fewer skilled professionals in the diagnostic center are hampering the market's growth.
A stool diagnosis is a dynamic component in today's difficult time, providing patients with essential information for diagnosing, preventing, treating, and managing their disease and cancer. The requirement for trained staff is a big issue for the Asia-Pacific stool diagnostic market. Due to the lack of skilled and certified professionals, end users cannot install advanced products for stool diagnosis; this may challenge the growth of the Asia-Pacific stool diagnostic market.
Post-Covid-19 Impact Analysis on Asia-Pacific Stool Diagnostic Market
Covid-19 made a massive dent in the diagnostics sector, with a steep fall in patient volumes since the beginning of 2020. The pandemic developed an eerie sense of fear among people, which eventually impacted hospital/clinic and lab visits. Regular checkups became cumbersome, stressful, and time-consuming. While the overall diagnostics sector seemed to have suffered a decline, the area of home diagnostics was the silver lining in the new scheme of things.
Recent Developments
- In June 2022, Prenetics Limited (Nasdaq: PRE) announced the introduction of a novel, non-invasive stool DNA at-home screening test named ColoClear by Circle (ColoClear) for detecting early detection signs of colorectal cancer. This product launch helped the company in its product portfolio expansion
- In June 2022, Meridian Bioscience, Inc. announced the launch of two new sample-specific master mixes, Lyo-Ready Direct DNA qPCR Stool Mix and Lyo-Ready Direct RNA/DNA qPCR Stool Mix. These innovative master mixes improve DNA and RNA molecular detection from crude fecal specimens while allowing room temperature stabilization of diagnostic assays. This product launch helped the company in its product portfolio expansion
Asia-Pacific Stool Diagnostic Market Scope
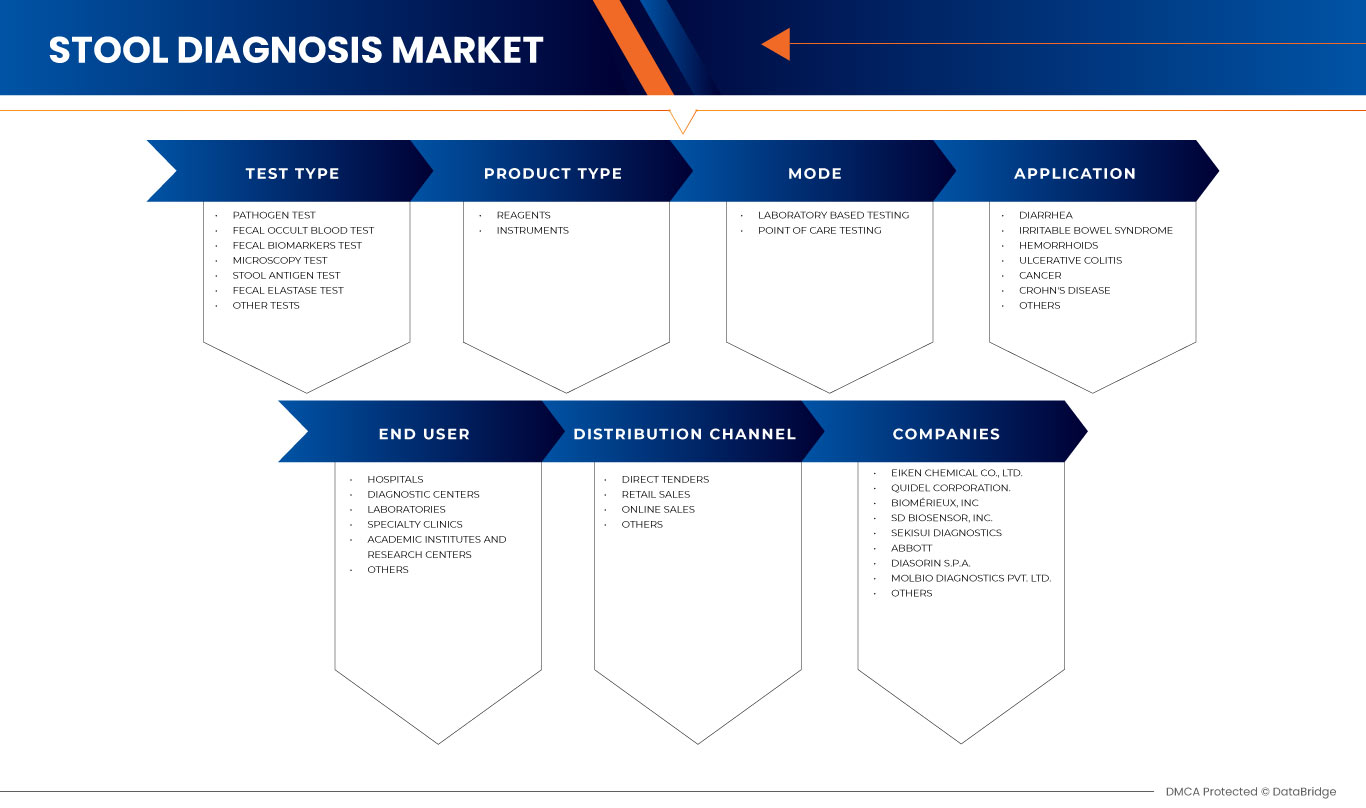
Asia-Pacific stool diagnostic market is segmented into six notable segments such as test type, product type, mode, application, end user, and distribution channel. The growth among segments helps you analyze niche pockets of growth and strategies to approach the market and determine your core application areas and the difference in your target markets.
ASIA-PACIFIC STOOL DIAGNOSTIC MARKET, BY TEST TYPE
- Fecal Occult Blood Test
- Fecal Biomarkers Test
- Pathogen Tests
- Stool Antigen Test
- Fecal Elastase Test
- Microscopy Test
- Other Tests
On the basis of test type, the Asia-Pacific stool diagnostic market is segmented into fecal occult blood test, fecal biomarker test, pathogen tests, stool antigen test, fecal elastase test, microscopy test, and other tests.
ASIA-PACIFIC STOOL DIAGNOSTIC MARKET, BY PRODUCT TYPE
- Instruments
- Reagents
On the basis of product type, the Asia-Pacific stool diagnostic market is segmented into instruments and reagents.
ASIA-PACIFIC STOOL DIAGNOSTIC MARKET, BY MODE
- Laboratory Based Testing
- Point Of Care Testing
On the basis of mode, the Asia-Pacific stool diagnostic market is segmented into laboratories based testing and point of care testing.
ASIA-PACIFIC STOOL DIAGNOSTIC MARKET, BY APPLICATION
- Irritable Bowel Syndrome
- Ulcerative Colitis
- Crohn's Disease
- Cancer
- Diarrhea
- Hemorrhoids
- Others
On the basis of application, the Asia-Pacific stool diagnostic market is segmented into irritable bowel syndrome, ulcerative colitis, crohn's disease, cancer, diarrhea, hemorrhoids, and others.
ASIA-PACIFIC STOOL DIAGNOSTIC MARKET, BY END USER
- Hospitals
- Diagnostic Centres
- Laboratory
- Specialty Clinics
- Academic Institutions And Research Centres
- Others
On the basis of end user, the Asia-Pacific stool diagnostic market is segmented into hospitals, diagnostic centers, laboratory, specialty clinics, academic institutions and research centers, and others.
ASIA-PACIFIC STOOL DIAGNOSTIC MARKET, BY DISTRIBUTION CHANNEL
- Direct Tenders
- Retail Sales
- Online Sales
- Others
On the basis of distribution channel, the Asia-Pacific stool diagnostic market is segmented into direct tenders, retail sales, online sales, and others.
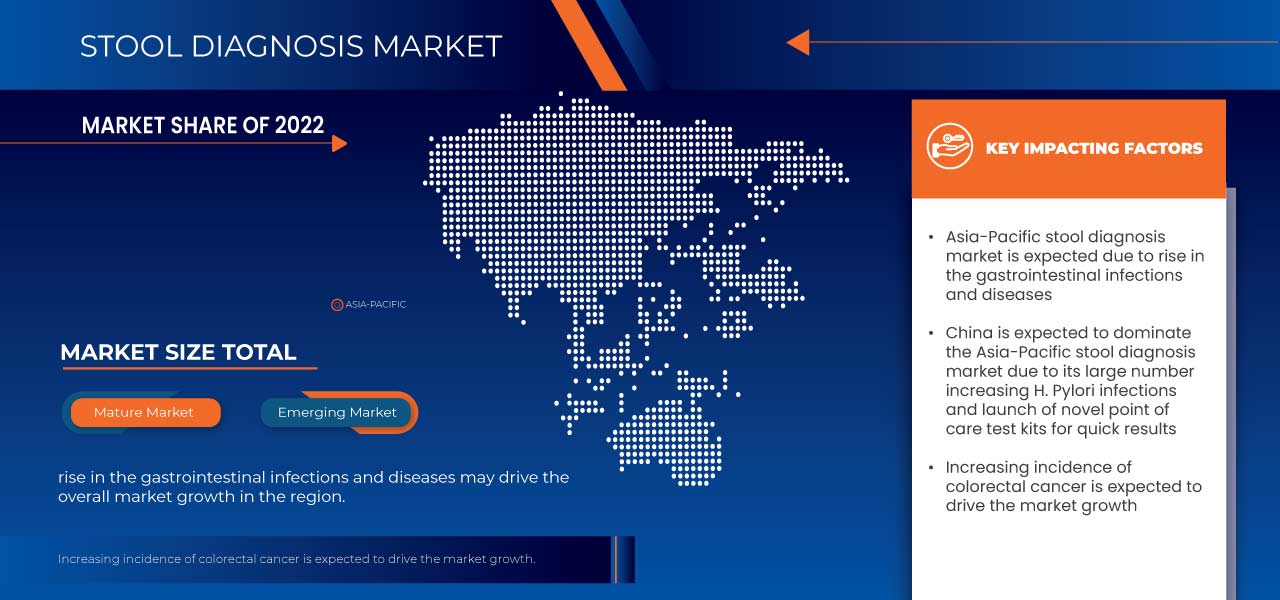
Asia-Pacific Stool Diagnostic Market Regional Analysis/Insights
Asia-Pacific stool diagnostic market is categorized into six notable segments: test type, product type, mode, application, end user, and distribution channel.
The countries covered in this market report are Japan, China, South Korea, India, Singapore, Thailand, Indonesia, Malaysia, Philippines, Australia, New Zealand, Vietnam, Taiwan, Brunei, Nepal, Bangladesh, Sri Lanka, Pakistan, Hong Kong, Maldives, Mongolia, Cambodia, Laos, and rest of Asia-Pacific.
China is expected to dominate Asia-Pacific due to increasing customer inclinations towards advanced technological processes.
The country section of the report also provides individual market-impacting factors and domestic regulation changes that impact the current and future trends of the market. Data points such as new sales, replacement sales, country demographics, regulatory acts, and import-export tariffs are some of the major pointers used to forecast the market scenario for individual countries. Also, the presence and availability of Asia-Pacific brands and their challenges due to large or scarce competition from local and domestic brands and the impact of sales channels are considered while providing forecast analysis of the country data.
Competitive Landscape and Asia-Pacific Stool Diagnostic Market Share Analysis
Asia-Pacific stool diagnostic market competitive landscape provides details by a competitor. Details included are company overview, company financials, revenue generated, market potential, investment in R&D, new market initiatives, production sites and facilities, company strengths and weaknesses, product launch, product approvals, product width and breadth, application dominance, and product type lifeline curve. The above data points only relate to the company's focus on the Asia-Pacific stool diagnostic market.
Some major companies dealing in the Asia-Pacific stool diagnostic market are Abbott, Meridian Bioscience Inc., BIOMERIEUX, Beckman Coulter, Inc. (Subsidiary of Danaher), DiaSorin S.p.A., Runmei, Molbio Diagnostics Pvt. Ltd., Quidel Corporation, Prenetics Limited, Sekisui Diagnostics (A Part of SEKISUI Chemical Co. Ltd.), BioVendor Group, LifeSign LLC, Alfa Scientific Designs, Inc., Eiken Chemical Co., Ltd., SD Biosensor, INC., Cenogenics Corporation, TransGen Biotech Co., Ltd., AdvaCare Pharma, Kibion GmbH, and Beijing Huagen Anbang Technology Co., Ltd. among others.
SKU-
Obtenha acesso online ao relatório sobre a primeira nuvem de inteligência de mercado do mundo
- Painel interativo de análise de dados
- Painel de análise da empresa para oportunidades de elevado potencial de crescimento
- Acesso de analista de pesquisa para personalização e customização. consultas
- Análise da concorrência com painel interativo
- Últimas notícias, atualizações e atualizações Análise de tendências
- Aproveite o poder da análise de benchmark para um rastreio abrangente da concorrência
Metodologia de Investigação
A recolha de dados e a análise do ano base são feitas através de módulos de recolha de dados com amostras grandes. A etapa inclui a obtenção de informações de mercado ou dados relacionados através de diversas fontes e estratégias. Inclui examinar e planear antecipadamente todos os dados adquiridos no passado. Da mesma forma, envolve o exame de inconsistências de informação observadas em diferentes fontes de informação. Os dados de mercado são analisados e estimados utilizando modelos estatísticos e coerentes de mercado. Além disso, a análise da quota de mercado e a análise das principais tendências são os principais fatores de sucesso no relatório de mercado. Para saber mais, solicite uma chamada de analista ou abra a sua consulta.
A principal metodologia de investigação utilizada pela equipa de investigação do DBMR é a triangulação de dados que envolve a mineração de dados, a análise do impacto das variáveis de dados no mercado e a validação primária (especialista do setor). Os modelos de dados incluem grelha de posicionamento de fornecedores, análise da linha de tempo do mercado, visão geral e guia de mercado, grelha de posicionamento da empresa, análise de patentes, análise de preços, análise da quota de mercado da empresa, normas de medição, análise global versus regional e de participação dos fornecedores. Para saber mais sobre a metodologia de investigação, faça uma consulta para falar com os nossos especialistas do setor.
Personalização disponível
A Data Bridge Market Research é líder em investigação formativa avançada. Orgulhamo-nos de servir os nossos clientes novos e existentes com dados e análises que correspondem e atendem aos seus objetivos. O relatório pode ser personalizado para incluir análise de tendências de preços de marcas-alvo, compreensão do mercado para países adicionais (solicite a lista de países), dados de resultados de ensaios clínicos, revisão de literatura, mercado remodelado e análise de base de produtos . A análise de mercado dos concorrentes-alvo pode ser analisada desde análises baseadas em tecnologia até estratégias de carteira de mercado. Podemos adicionar quantos concorrentes necessitar de dados no formato e estilo de dados que procura. A nossa equipa de analistas também pode fornecer dados em tabelas dinâmicas de ficheiros Excel em bruto (livro de factos) ou pode ajudá-lo a criar apresentações a partir dos conjuntos de dados disponíveis no relatório.