Global Crispr Lipid Nanoparticle Delivery Therapeutics Market
Tamanho do mercado em biliões de dólares
CAGR :
% 
 USD
1.28 Billion
USD
5.76 Billion
2025
2033
USD
1.28 Billion
USD
5.76 Billion
2025
2033
| 2026 –2033 | |
| USD 1.28 Billion | |
| USD 5.76 Billion | |
|
|
|
|
CRISPR Global Lipid Nanoparticle Delivery Therapeutics Market Segmentation, By Payload Type (mRNA, CRISPR/Cas9 Components, siRNA/saRNA, Plasmid DNA, and Others), By Lipid Nanoparticle Type (Solid Lipid Nanoparticles, Nanostructured Lipid Carriers, Liposomes, and Others), By Application (Therapeutics, Research), By Therapeutic Area (Oncoology, Rare Genetic Disorders, Cardiovascular Disorders, Neurological Disorders, Infectious Diseases, and Others), By End-User (Pharmaceutical and Biotechnology Companies, Acadêmicos and Research Institutes, and Others)- Indústria Tendências and Forecast to 2033
CRISPR Lipid Nanoparticle Delivery TerapeuticsTamanho do Mercado
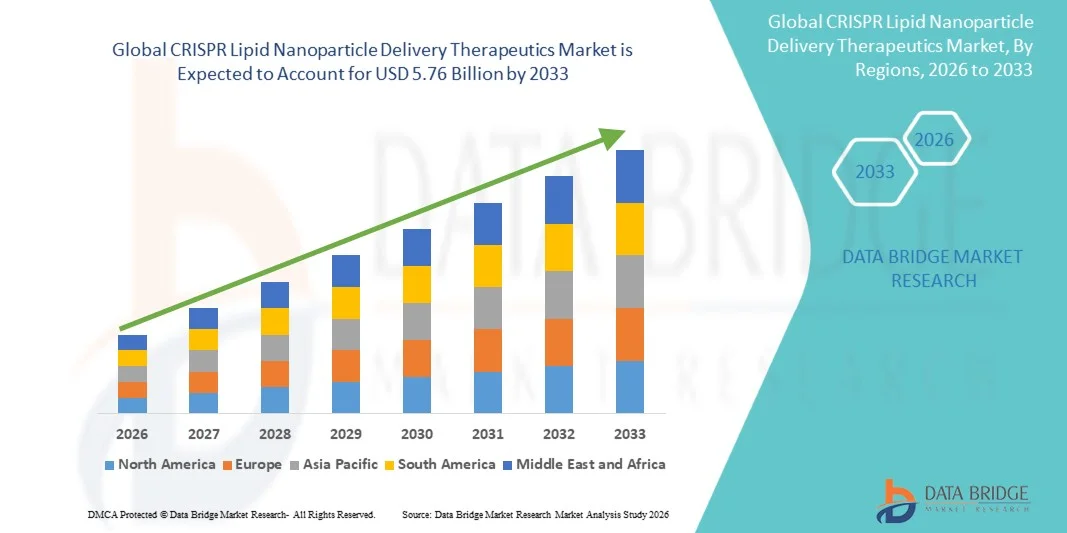
- O CRISPR global Lipid Nanoparticle Delivery Therapeutics tamanho do mercado foi avaliado em1,28 mil milhões de dólares em 2025e espera-se alcançarUSD 5,76 mil milhões até 2033, em umaCAGR de 20,70%durante o período de previsão
- O crescimento do mercado é amplamente impulsionado pelo rápido avanço de programas clínicos de edição de genes in vivo CRISPR utilizando plataformas de entrega de LNP, o desenvolvimento acelerado de terapias de mRNA e siRNA alavancando encapsulamento de nanopartículas lipídicas, e crescente reconhecimento de LNPs como uma alternativa mais segura e escalável aos vetores virais para o fornecimento de ácido nucleico.
- Além disso, o aumento do investimento farmacêutico em plataformas de tecnologia LNP-CRISPR para o tratamento de doenças genéticas, oncológicas e doenças cardiovasculares está estabelecendo a terapia com nanopartículas lipídicas CRISPR como a pedra angular da medicina de precisão de próxima geração. Esses fatores convergentes estão acelerando a absorção de soluções de entrega LNP-CRISPR, aumentando significativamente o crescimento da indústria
CRISPR Lipid Nanoparticle Delivery TerapeuticsAnálise de mercado
- A terapia de entrega de nanopartículas lipídicas CRISPR, oferecendo uma plataforma não viral biocompatível para o fornecimento intracelular eficiente de componentes CRISPR/Cas9, mRNA, siRNA e DNA plasmídeo, são componentes cada vez mais vitais da moderna paisagem de medicina de precisão devido à sua menor imunogenicidade, alta eficiência de encapsulamento e escalabilidade superior em comparação aos sistemas de liberação convencionais baseados em vetores virais
- A crescente demanda pela terapêutica com CRISPR LNP é principalmente alimentada pela expansão do pipeline clínico de programas de edição de genes in vivo, o sucesso comercial das plataformas vacinais de LNP-mRNA validadas por meio de campanhas globais de imunização COVID-19, e o crescente investimento em P&D farmacêutico em formulações lipídicas ionizáveis otimizadas para entrega direcionada de CRISPR para tecidos hepáticos e extra-hepáticos.
- A América do Norte dominou o mercado de terapias de entrega de nanopartículas lipídicas CRISPR com a maior parcela de receita de 41,20% em 2025, caracterizada pela presença de desenvolvedores pioneiros de LNP-CRISPR, incluindo Intellia Therapeutics, CRISPR Therapeutics, Verve Therapeutics e Alnylam Pharmaceuticals, juntamente com robustas vias regulatórias da FDA que apoiam o desenvolvimento clínico acelerado de programas de edição de genes in vivo
- Asia-Pacific é esperado para ser a região de crescimento mais rápido no mercado de nanopartículas lipídicas CRISPR entrega terapêutica durante o período de previsão devido à rápida expansão ecossistemas biotecnológicos, iniciativas de medicina genômica apoiada pelo governo, e crescente atividade de pesquisa clínica na China, Japão e Coreia do Sul
- O segmento de pesquisa foi responsável pela maior parcela de receita de mercado de 57,3% em 2025, impulsionada pelo amplo uso global de sistemas CRISPR-LNP em estudos pré-clínicos, análise de função gênica e gasodutos de desenvolvimento terapêutico
Âmbito do relatório eCRISPR Lipid Nanoparticle Delivery Therapeutics Market Segmentação
|
Atributos |
CRISPR Lipid Nanoparticle Delivery Therapeutics chavePerspectivas de mercado |
|
Segmentos Cobertos |
|
|
Países abrangidos |
América do Norte
Europa
Ásia- Pacífico
Médio Oriente e África
América do Sul
|
|
Jogadores do mercado chave |
|
|
Oportunidades de Mercado |
|
|
Informações sobre o Valor Adicionado |
Além dos insights sobre cenários de mercado, como valor de mercado, taxa de crescimento, segmentação, cobertura geográfica e grandes atores, os relatórios de mercado curados pela Data Bridge Market Research também incluem análise de especialistas em profundidade, epidemiologia de pacientes, análise de pipelines, análise de preços e marco regulatório. |
CRISPR Lipid Nanoparticle Delivery Therapeutics Mercado Tendências
“Formulações lipídicas ionizáveis otimizadas por IA que permitem CRISPR extra-hepático Entrega“
- Uma tendência significativa e acelerada no mercado global de terapias de entrega de nanopartículas lipídicas CRISPR é o aprofundamento da integração da inteligência artificial (IA) e do aprendizado de máquina com a ciência da formulação LNP, permitindo a rápida identificação de novas composições lipídicas ionizáveis que podem fornecer componentes de edição de genes CRISPR para além do fígado, incluindo o pulmão, músculo e sistema nervoso central
- Por exemplo, em outubro de 2023, Mana.bio revelou sua plataforma de entrega de LNP alimentado por IA especificamente projetado para otimizar formulações de lipídios para CRISPR e RNA de entrega terapêutica tecidular. Esta plataforma alavanca a triagem de alto rendimento combinada com modelagem computacional preditiva para identificar candidatos a lipídios ionizáveis capazes de alcançar eficiência de edição de 40 a 60 por cento em modelos de tecido extra-hepático pré-clínico
- A otimização de LNP orientada por IA permite características como controle preciso do tamanho das partículas, distribuição de carga e eficiência de encapsulamento, que são parâmetros críticos que regem a eficácia e segurança da entrega de CRISPR. Além disso, lipídios ionizáveis projetados incorporando headgroups sensíveis ao pH estão permitindo a otimização da fuga endossômica, o que melhora diretamente a liberação intracelular de complexos de cas9 mRNA ou ribonucleoproteína em tecidos alvo com efeitos mínimos fora do alvo.
- A integração perfeita de plataformas LNP otimizadas por IA com sistemas modulares de entrega de CRISPR está facilitando o desenvolvimento centralizado de formulação através de múltiplas indicações terapêuticas de uma única arquitetura de plataforma. Através de fluxos de trabalho computacionais e experimentais unificados, os desenvolvedores podem adaptar o mesmo andaime lipídico ionizável para fornecer diversas cargas úteis de edição de genes em alvos de oncologia, cardiovascular e doenças raras
- Essa tendência para sistemas de liberação de LNP mais inteligentes, direcionados ao tecido e programáveis está fundamentalmente reformulando a paisagem competitiva do desenvolvimento terapêutico do CRISPR. Conseqüentemente, empresas como ReCode Therapeutics e Nutcracker Therapeutics estão desenvolvendo plataformas de LNP seletivas de órgãos que expandem a endereçamento da entrega de CRISPR para além dos alvos hepáticos para os tecidos pulmonar, SNC e gastrointestinal.
- A demanda por terapia CRISPR LNP otimizada por IA com capacidade de entrega extra-hepática ampliada está crescendo rapidamente em ambientes clínicos e de pesquisa, uma vez que os desenvolvedores farmacêuticos priorizam cada vez mais soluções escaláveis e não virais capazes de possibilitar a edição de genes curativos em uma ampla gama de indicações de doenças genéticas.
CRISPR Lipid Nanoparticle Delivery Therapeutics Market Dynamics
Controlador
“Validação Clínica dos Programas da Vivo LNP-CRISPR Conduzindo Confiança Comercial”
- O avanço bem-sucedido de múltiplos programas de CRISPR in vivo utilizando a entrega de LNP em ensaios clínicos em fase tardia, combinado com resultados clínicos de referência que demonstram uma edição gênica profunda e durável em pacientes humanos, é um motor primário para acelerar o desenvolvimento comercial do mercado de nanopartículas de lipídios CRISPR.
- Por exemplo, em novembro de 2024, resultados publicados no New England Journal of Medicine da Intellia Therapeutics NTLA-2001 O ensaio de fase II confirmou uma redução média de aproximadamente 90% da proteína TTR relacionada com a doença em doentes com amiloidose hereditária transtirretina após uma dose IV única de CRISPR/Cas9 administrada via LNP, estabelecendo um marco clínico que valide a segurança e eficácia da abordagem de administração de LNP-CRISPR em seres humanos. Tais resultados de empresas-chave deverão impulsionar o crescimento da indústria terapêutica CRISPR LNP significativamente no período de previsão
- Como médicos e pagadores reconhecem cada vez mais o potencial da edição do gene CRISPR entregue por LNP para fornecer resultados duradouros e potencialmente curativos para pacientes com doenças genéticas graves, espera-se que a demanda por produtos CRISPR LNP em fase clínica e comercialmente aprovados cresça substancialmente, incentivando ainda mais o investimento em I&D em todo o setor.
- Além disso, o crescente portfólio de programas de CRISPR in vivo entregues por plataformas proprietárias de LNP, incluindo CTX310 e CTX320 para doenças cardiovasculares e VERVE-101 para hipercolesterolemia familiar, está demonstrando a versatilidade da entrega de LNP em múltiplos sistemas de órgãos e indicações genéticas além do foco hepático inicial.
- A crescente disponibilidade de matérias-primas lipídicas ionizáveis de grau GMP, o avanço das tecnologias de fabricação microfluídica que permitem a produção consistente de lote de LNP e o crescente ecossistema de CDMO especializado no desenvolvimento de formulações de LNP estão reduzindo as barreiras de comercialização e apoiando a ampliação de programas de LNP-CRISPR desde as fases clínicas até comerciais de fabricação
Restrição/Desafio
“Limitações de entrega extra-hepáticas e complexidade de fabricação“
- A administração sistêmica de LNP tem uma tendência natural de se acumular predominantemente em hepatócitos, criando um desafio significativo para programas terapêuticos CRISPR visando tecidos como pulmão, músculo, cérebro e tumores onde a biodistribuição de LNP é inerentemente menos eficiente. Esta limitação biológica restringe o espaço de doença endereçável das plataformas atuais de LNP-CRISPR e requer engenharia de formulação adicional substancial para alcançar edição adequada de genes extra-hepáticos
- Por exemplo, enquanto o ensaio NTLA-2001 demonstrou uma eficiência de edição hepática excepcional, traduzindo o desempenho comparável do LNP-CRISPR em alvos extra-hepáticos, exigiu o desenvolvimento de andaimes lipídicos ionizáveis totalmente novos, combinações excipientes seletivas de órgãos e vias de entrega alternativas, incluindo administração intratecal e intramuscular, todas as quais aumentam substancialmente a complexidade e os custos do desenvolvimento.
- A fabricação da terapêutica CRISPR baseada em LNP envolve processos altamente complexos, incluindo homogeneização de alta pressão, mistura microfluídica e criopreservação precisa, que requerem equipamentos especializados e rigorosos protocolos de controle de qualidade para manter a consistência batelada em tamanho de partículas, índice de polidispersão e eficiência de encapsulamento. Mesmo desvios menores nesses parâmetros podem impactar significativamente no desempenho de edição vivo e perfis de segurança do paciente
- Empresas como Acuitas Therapeutics e Genevant Sciences estão investindo substancialmente em plataformas avançadas de fabricação de LNP, bibliotecas de lipídios ionizáveis proprietárias e capacidades analíticas de caracterização para enfrentar esses desafios e apoiar a produção comercial escalável de terapias CRISPR LNP de próxima geração
- Superar esses desafios através da inovação contínua na engenharia de LNP orientada para órgãos, otimização do processo de fabricação e o desenvolvimento de ferramentas analíticas avançadas para caracterização de LNP será vital para sustentar o impulso de crescimento do mercado de nanopartículas de entrega de lipídios CRISPR através de indicações de doença não hepática
CRISPR Lipid Nanoparticle Delivery Therapeutics Market Scope
O mercado é segmentado com base no tipo de carga útil, tipo de nanopartícula lipídica, aplicação, área terapêutica e usuário final.
• Por tipo de carga útil
Com base no tipo de carga útil, o mercado terapêutico de entrega de nanopartículas lipídicas CRISPR é segmentado em componentes de mRNA, CRISPR/Cas9, siRNA/saRNA, DNA plasmídeo e outros. O segmento CRISPR/Cas9 Components dominou a maior parcela de receita de mercado de 38,6% em 2025, impulsionada pela rápida expansão da edição de genomas pesquisa e desenvolvimento terapêutico voltado para transtornos genéticos. Estes componentes são amplamente utilizados devido à sua precisão, eficiência e capacidade para permitir o knockout e correção de genes. O aumento dos ensaios clínicos com foco em terapias baseadas em CRISPR está aumentando significativamente a demanda. O crescente investimento em empresas de biotecnologia de edição de genes apoia ainda mais a liderança de segmentos. A forte adopção em investigação em oncologia e doenças raras aumenta a utilização. Os sistemas de nanopartículas lipídicas (LNP) melhoram a eficiência de entrega e reduzem a degradação dos componentes CRISPR. A ampliação das aprovações regulatórias para estudos de edição de genes está acelerando o crescimento do mercado. As colaborações crescentes entre empresas farmacêuticas e institutos de investigação estão a reforçar a inovação. O sucesso crescente em estudos pré-clínicos está incentivando a pesquisa translacional. Estes factores asseguram colectivamente a dominância dos componentes CRISPR/Cas9.
Espera-se que o segmento de mRNA testemunhe a taxa de crescimento mais rápida de 18,9% CAGR de 2026 a 2033, impulsionada pelo aumento do uso de terapias e vacinas baseadas em mRNA entregues através de sistemas de nanopartículas lipídicas. Aumentar o sucesso das plataformas de RNAm em aplicações de doenças infecciosas e oncologia está alimentando a expansão. A crescente procura de tecnologias de desenvolvimento de drogas rápidas e escaláveis apoia a adopção. A inovação contínua em sistemas de entrega de LNP-mRNA aumenta a estabilidade e a eficácia. Expandir a tubulação de terapias médicas personalizadas impulsiona ainda mais o crescimento. O aumento do financiamento na pesquisa terapêutica do RNA fortalece a penetração do mercado. As empresas farmacêuticas estão investindo fortemente em medicamentos para a próxima geração. Ensaios clínicos crescentes em doenças genéticas e metabólicas raras suportam a demanda. Melhor eficiência de entrega através da tecnologia LNP melhora os resultados terapêuticos. Esses fatores posicionam o mRNA como o segmento de carga útil de crescimento mais rápido.
• Por Tipo de Nanopartícula Lipídica
Com base no tipo de nanopartículas lipídicas, o mercado é segmentado em nanopartículas lipídicas sólidas, porta-lipídeos nanoestruturados, lipossomas e outros. O segmento Lipossomos teve a maior parcela de receita de mercado de 44,1% em 2025, impulsionada pelo seu uso de longa data na entrega de drogas e comprovada capacidade de encapsular ácidos nucleicos e cargas de CRISPR de forma eficaz. Os lipossomas oferecem alta biocompatibilidade, baixa toxicidade e maior captação celular. Sua versatilidade os torna amplamente utilizados na terapia genética e no desenvolvimento vacinal. O aumento da aplicação na oncologia e na terapêutica das doenças raras apoia a procura. Um forte foco de pesquisa na melhoria da eficiência de entrega baseada em lipídios está impulsionando a adoção. As empresas farmacêuticas preferem lipossomas devido à familiaridade regulatória estabelecida. A expansão dos ensaios clínicos envolvendo sistemas de entrega de CRISPR baseados em lipídios reforça ainda mais a quota de mercado. O aumento do investimento na nanomedicina está a acelerar a inovação. Maior estabilidade e capacidade de direcionamento aumentam a eficiência terapêutica. Esses fatores garantem que os lipossomos mantenham a dominância no segmento.
Espera-se que o segmento Nanoestruturado Lipid Carriers (NLCs) testemunhe a taxa de crescimento mais rápida de 17,4% CAGR de 2026 a 2033, impulsionada pela sua capacidade superior de carga de medicamentos e melhoria da estabilidade em relação aos sistemas lipídicos tradicionais. Os CNPs permitem o encapsulamento eficiente de materiais genéticos complexos utilizados em terapias CRISPR. O aumento da pesquisa em nanocarregadores de próxima geração está apoiando a adoção. A crescente demanda por terapias de genes alvo e de liberação sustentada está alimentando a expansão. As empresas farmacêuticas estão focadas em melhorar a precisão de entrega para ferramentas de edição genética. A crescente utilização no tratamento da oncologia e da doença neurológica aumenta ainda mais a procura. Melhoria da escalabilidade e vantagens de fabricação suportam a comercialização. A expansão da pesquisa acadêmica em sistemas de entrega baseados em nanotecnologia está acelerando a inovação. Perfis de segurança melhorados estão aumentando a aceitação clínica. Esses fatores posicionam os CPNs como o tipo de nanopartícula lipídica de crescimento mais rápido.
• Por Aplicação
Com base na aplicação, o mercado é segmentado em Terapêutica e Pesquisa. O segmento Pesquisa foi responsável pela maior parcela de receita de mercado de 57,3% em 2025, impulsionada pelo amplo uso global de sistemas CRISPR-LNP em estudos pré-clínicos, análise de função gênica e oleodutos de desenvolvimento terapêutico. As instituições académicas e as empresas de biotecnologia dependem fortemente destes sistemas para a investigação da edição do genoma. Aumentar o financiamento da engenharia genética e da medicina de precisão está a apoiar o crescimento. A expansão das colaborações entre universidades e empresas farmacêuticas está acelerando a inovação. O aumento do número de estudos experimentais baseados em CRISPR aumenta a procura. O forte foco na modelagem de doenças e identificação de alvos fortalece ainda mais o uso. A disponibilidade de plataformas LNP avançadas aumenta a eficiência da pesquisa. Avanços tecnológicos contínuos em sistemas de liberação de genes estão impulsionando a adoção. As iniciativas governamentais e de financiamento privado apoiam a investigação genómica a nível mundial. Esses fatores asseguram que a pesquisa continue sendo o segmento de aplicação dominante.
Espera-se que o segmento Terapêutico testemunhe a taxa de crescimento mais rápida de 21,6% CAGR de 2026 a 2033, impulsionada pela rápida transição das tecnologias CRISPR-LNP de pesquisas para aplicações clínicas. Aumentar as aprovações para terapias baseadas em edição de genes está alimentando a comercialização. A crescente prevalência de doenças genéticas e cancros está a aumentar a procura de tratamentos específicos. As empresas farmacêuticas investem fortemente em gasodutos de medicamentos baseados em CRISPR. Avanços na eficiência de entrega utilizando nanopartículas lipídicas estão melhorando as taxas de sucesso clínico. Aumentar a consciência do paciente de opções de terapia genética apoia a adoção. A expansão de ensaios clínicos em oncologia, doenças raras e doenças infecciosas impulsiona ainda mais o crescimento. O progresso regulamentar nas terapias de edição de genes está acelerando a entrada no mercado. O aumento da demanda por medicamentos personalizados é outro fator chave. Esses motoristas posicionam a Terapêutica como o segmento de aplicação de crescimento mais rápido.
• Por Área Terapêutica
Com base na área terapêutica, o mercado de Terapêuticas para Entrega de Nanopartículas Lipidas CRISPR é segmentado em Oncologia, Transtornos Genéticos Raros, Doenças Cardiovasculares, Transtornos Neurológicos, Doenças Infecciosas e Outros. O segmento de Oncologia dominou a maior parcela de receita de mercado de 41,8% em 2025, impulsionada pela crescente aplicação de terapias baseadas em CRISPR para edição de gene alvo de câncer e supressão tumoral. O aumento da incidência global do cancro está a aumentar significativamente a procura. Os sistemas CRISPR-LNP são amplamente utilizados para nocaute de genes e modulação imunológica em células cancerosas. Forte investimento no desenvolvimento de drogas oncológicas apoia a liderança do segmento. A expansão de ensaios clínicos para tumores sólidos e neoplasias hematológicas está acelerando a adoção. Os avanços na oncologia de precisão estão melhorando os resultados do tratamento. As empresas farmacêuticas estão priorizando terapias genéticas focadas no câncer. O uso crescente de terapias combinadas aumenta a eficácia. O aumento do financiamento das organizações públicas e privadas reforça a investigação. Esses fatores garantem que a Oncologia continue sendo a área terapêutica dominante.
Espera-se que o segmento de Transtornos Genéticos Raros testemunhe a taxa de crescimento mais rápida de 22,3% CAGR de 2026 a 2033, impulsionada pelo aumento do desenvolvimento de terapias curativas de edição genética para doenças hereditárias. A tecnologia CRISPR-LNP oferece alta precisão na correção de mutações genéticas. A sensibilização e o diagnóstico das doenças raras estão a aumentar a procura de tratamento. Os crescentes incentivos aos medicamentos órfãos incentivam o investimento farmacêutico. Expandir ensaios clínicos visando distúrbios monogênicos ainda mais apoiar o crescimento. Sistemas de entrega melhorados estão aumentando a segurança e eficiência terapêutica. Os grupos de defesa do paciente estão acelerando o financiamento e a conscientização. Os avanços na medicina personalizada estão impulsionando a inovação. Aumentar as aprovações regulatórias para terapias genéticas está fortalecendo a expansão do mercado. Esses fatores posicionam os Transtornos Genéticos Raros como a área terapêutica de crescimento mais rápido.
• Por Usuário Final
Com base no usuário final, o mercado está segmentado em empresas farmacêuticas e biotecnológicas, institutos acadêmicos e de pesquisa e outros. O segmento Farmacêutico e Empresas de Biotecnologia representou a maior parcela de receita de mercado de 62,7% em 2025, impulsionada por amplo investimento em gasodutos de desenvolvimento e comercialização de medicamentos baseados em CRISPR. Essas empresas estão liderando a inovação em plataformas de entrega de nanopartículas lipídicas. O foco crescente na comercialização da terapia genética suporta a dominância do segmento. Fortes gastos em P&D em medicina de precisão reforçam a adoção. Parcerias com startups de biotecnologia estão acelerando os avanços tecnológicos. O número crescente de ensaios clínicos conduzidos por empresas farmacêuticas aumenta ainda mais a procura. A expansão dos portfólios de edição de genes fortalece a liderança do mercado. Aumentar as aprovações regulatórias para terapias genéticas apoiar o crescimento da indústria. Capacidades avançadas de fabricação melhoram a escalabilidade da terapêutica baseada em LNP. Esses fatores garantem o domínio das empresas farmacêuticas e de biotecnologia.
Espera-se que o segmento Academic & Research Institutes testemunhe a taxa de crescimento mais rápida de 16,8% CAGR de 2026 a 2033, impulsionada pelo aumento do financiamento global para pesquisas genômicas e estudos de edição de genes. Universidades e centros de pesquisa são os principais contribuintes para a inovação CRISPR em fase inicial. Aumentar a colaboração com empresas farmacêuticas está impulsionando a transferência de tecnologia. A crescente disponibilidade de bolsas de pesquisa apoia a adoção de sistemas de entrega baseados em LNP. Expansão da educação em biotecnologia programa novos combustíveis demanda. O crescente foco na pesquisa translacional acelera as aplicações clínicas. A infraestrutura de laboratório avançada aumenta as capacidades experimentais. Aumentar o interesse em pesquisas personalizadas de medicina fortalece o uso. As iniciativas governamentais que promovem a investigação genómica também contribuem para o crescimento. Esses fatores fazem dos Institutos Acadêmicos & de Pesquisa o segmento de usuário final de crescimento mais rápido.
CRISPR Lipid Nanoparticle Delivery Terapeutics Market Analysis Regional
- A América do Norte dominou o mercado de terapias de entrega de nanopartículas lipídicas CRISPR com a maior parcela de receita de 41,20% em 2025, impulsionada pela presença das principais empresas mundiais de LNP-CRISPR, um ecossistema CDMO bem desenvolvido especializado em fabricação de LNP, financiamento público robusto através de BARDA e NIH para o desenvolvimento de tecnologia LNP, e a maior concentração de ensaios clínicos ativos in vivo CRISPR globalmente
- Os consumidores e os prestadores de cuidados de saúde na região beneficiam-se de vias regulamentares avançadas da FDA que apoiam o desenvolvimento acelerado do LNP-CRISPR, incluindo a designação do RMAT, o estado da terapia inovadora e a designação rápida de terapêuticas CRISPR destinadas a doenças genéticas raras e com risco de vida
- Esta adoção generalizada é ainda apoiada por altos níveis de investimento em I&D biofarmacêutico público e privado, uma infraestrutura clínica sofisticada que apoia a condução de ensaios LNP-CRISPR, e crescente colaboração entre desenvolvedores de tecnologia LNP e grandes empresas farmacêuticas, estabelecendo a América do Norte como o mercado dominante e mais avançado comercialmente LNP-CRISPR globalmente
US CRISPR Lipid Nanoparticle Delivery Therapeutics Market Insight
O mercado de terapias de entrega de nanopartículas lipídicas CRISPR dos EUA capturou a maior participação de receita na América do Norte em 2025, alimentada pela rápida expansão comercial dos programas LNP-CRISPR de empresas pioneiras, incluindo Intellia Therapeutics, CRISPR Therapeutics, Verve Therapeutics e Beam Therapeutics. O mercado dos EUA deverá atingir um valor estimado de US$ 1,84 bilhões em 2032, impulsionado pelo engajamento regulatório favorável da FDA, aumentando o reconhecimento do pagador do valor transformador de tratamentos curativos únicos LNP-CRISPR e ampliando a infraestrutura de fabricação. Além disso, o estudo inovador de 2025 em um único paciente no Hospital Infantil de Filadélfia e Universidade da Pensilvânia usando LNPs Acuitas para entregar a primeira terapia CRISPR personalizada do mundo está contribuindo significativamente para a expansão do mercado.
Europa CRISPR Lipid Nanoparticle Delivery Therapeutics Market Insight
O mercado europeu de terapias de entrega de nanopartículas lipídicas CRISPR é projetado para se expandir em um CAGR substancial ao longo do período de previsão, impulsionado principalmente por forte apoio regulatório EMA para medicamentos de terapia avançada que incorporam a entrega de LNP, crescente investimento em infraestrutura de fabricação de LNP em toda a Alemanha, França e Reino Unido, e a expansão do cenário de ensaios clínicos europeus para programas de edição de genes LNP-CRISPR. Empresas europeias, incluindo a BioNTEch SE na Alemanha e a Acuitas Therapeutics, estão desempenhando papéis centrais no avanço da tecnologia de LNP lipídica ionizável que sustenta o oleoduto clínico de entrega CRISPR globalmente. Além disso, o investimento contínuo do programa EU Horizon em pesquisa de terapia genética baseada em LNP está apoiando a expansão das capacidades de desenvolvimento LNP-CRISPR acadêmicas e comerciais europeias.
U.K. CRISPR Lipid Nanoparticle Delivery Therapeutics Market Insight
Prevê-se que o mercado de terapias de entrega de nanopartículas lipídicas do Reino Unido CRISPR cresça em um notável CAGR durante o período de previsão, impulsionado pelo quadro regulatório progressivo da MHRA para a terapêutica avançada baseada em LNP, o investimento robusto em medicina genômica em NHS e a presença de centros acadêmicos líderes, incluindo o Instituto Wellcome Sanger e o Instituto Francis Crick, conduzindo pesquisas pioneiras de entrega de LNP-CRISPR. O programa Genomics England do Reino Unido e o crescente investimento em medicina de precisão estão criando um ecossistema de suporte para a tradução clínica de programas LNP-CRISPR.
Alemanha CRISPR Lipid Nanoparticle Delivery Therapeutics Market Insight
Espera-se que o mercado de terapias de entrega de nanopartículas lipídicas CRISPR da Alemanha se expanda em um considerável CAGR durante o período de previsão, alimentado pela presença da BioNTech SE, que alavanca sua infraestrutura de fabricação mRNA-LNP validada globalmente para o avanço de programas terapêuticos LNP-CRISPR, além de um forte investimento governamental em inovação em biotecnologia e um ecossistema de fabricação farmacêutica bem desenvolvido. A ênfase da Alemanha na medicina de precisão e forte colaboração acadêmico-indústria em pesquisa genômica promove o desenvolvimento de formulações avançadas de LNP-CRISPR, particularmente em aplicações de oncologia e doenças raras.
Asia-Pacific CRISPR Lipid Nanoparticle Delivery Therapeutics Market Insight
O mercado de terapias de entrega de nanopartículas lipídicas CRISPR Ásia-Pacífico está preparado para crescer no CAGR mais rápido de 24,0% durante o período de previsão de 2026 a 2033, impulsionado por investimentos em I&D biofarmacêuticos em rápida expansão, crescente apoio governamental para a inovação em medicina genômica e nanotecnologia e aumento da atividade de ensaios clínicos na China, Japão, Coreia do Sul e Índia. Por exemplo, em março de 2025, pesquisadores da East China Normal University e da YolTech Therapeutics em Xangai desenvolveram LNPs para entregar CRISPR-Cas9 para o tratamento da hiperoxaluria primária tipo 1, demonstrando a crescente capacidade da região no desenvolvimento terapêutico LNP-CRISPR. Além disso, iniciativas como o plano "Made in China 2025" da China e o BIRAC da Índia estão promovendo a I&D de nanotecnologia LNP, acelerando ainda mais o crescimento do mercado regional.
Japão CRISPR Lipid Nanoparticle Delivery Therapeutics Market Insight
O mercado de terapias de entrega de nanopartículas lipídicas CRISPR no Japão está ganhando ímpeto devido à forte tradição do país de pesquisas acadêmicas em ciência da entrega de drogas LNP, aumento do investimento industrial em terapias de RNA e crescente apoio regulatório para terapia genética avançada e desenvolvimento de medicamentos nucleicos ácidos. A iniciativa nacional de medicina de precisão do Japão e a crescente colaboração clínica entre universidades japonesas e desenvolvedores globais de LNP-CRISPR estão acelerando a tradução da inovação de entrega de LNP para aplicações clínicas.
China CRISPR Lipid Nanoparticle Delivery Therapeutics Market Insight
O mercado terapêutico de nanopartículas lipídicas CRISPR da China representou a maior quota de receita de mercado na Ásia-Pacífico em 2025, atribuída ao maior número de ensaios clínicos CRISPR do país globalmente, expandindo rapidamente o setor nacional de biotecnologia, e forte apoio do governo para terapia genética e inovação nanomedicina. Empresas domésticas, incluindo a YolTech Therapeutics e grupos de pesquisa acadêmica em universidades chinesas líderes, estão desenvolvendo formulações proprietárias de LNP para a entrega de CRISPR, enquanto parcerias internacionais estão facilitando a transferência de tecnologia e o avanço clínico dos programas LNP-CRISPR no mercado de saúde chinês.
CRISPR Lipid Nanoparticle Delivery Therapeutics Market Share
A indústria de terapia para entrega de nanopartículas lipídicas CRISPR é liderada principalmente por empresas bem estabelecidas, incluindo:
- Intellia Therapeutics (EUA)
- CRISPR Therapeutics AG (Suíça)
- Verve Therapeutics (EUA)
- Alnylam Pharmaceuticals (EUA)
- Moderna Inc. (EUA)
- BioNTech SE (Alemanha)
- Biociências de Precisão (EUA)
- Acuitas Therapeutics (Canadá)
- Ciências de Genebra (EUA)
- Arcturus Therapeutics (EUA)
- Merck KGaA (Alemanha)
- Pfizer Inc. (EUA)
- AstraZeneca (U.K.)
- Beam Therapeutics (EUA)
- Prime Medicine (EUA)
- Tessera Therapeutics (EUA)
- Tome Biosciences (EUA)
- Mana.bio (EUA)
- Recode Therapeutics (EUA)
- Terapêutica do Quebra-Nozes (EUA)
Últimos desenvolvimentos no mercado global de terapias de entrega de nanopartículas lipídicas CRISPR
- Em janeiro de 2024, a Tome Biosciences and Genevant Sciences entrou em uma colaboração estratégica avaliada em aproximadamente 114,3 milhões de dólares, combinando a experiência programável de integração genômica da Tome com a tecnologia proprietária de LNP da Genevant para desenvolver soluções de edição de genes in vivo para raras condições hepáticas. Essa parceria de referência ressaltou a importância estratégica do licenciamento de tecnologia de entrega de LNP no avanço de programas terapêuticos in vivo de próxima geração.
- Em outubro de 2023, Mana.bio desvelou sua plataforma de entrega de nanopartículas lipídicas alimentadas por IA, projetada para acelerar a identificação de novas formulações lipídicas ionizáveis para o fornecimento terapêutico de CRISPR e RNA seletivos de órgãos. A plataforma orientada por IA integra triagem experimental de alto rendimento com previsão baseada em aprendizado de máquina para otimizar composições de LNP para direcionamento de tecido extra-hepático, ampliando o espaço de doença endereçável para terapias de LNP-CRISPR.
- Em novembro de 2024, resultados publicados no New England Journal of Medicine da Intellia Therapeutics NTLA-2001 O ensaio de fase II demonstrou uma redução de aproximadamente 90% nos níveis de proteína TTR em doentes com amiloidose transtiretina hereditária após uma dose única IV de CRISPR/Cas9, com entrega de LNP, confirmando uma edição profunda e durável do gene in vivo em seres humanos. Um programa acompanhante NTLA-2002 demonstrou ainda uma redução de 95% na frequência de episódios de angioedema hereditário, validando coletivamente o LNP-CRISPR como uma plataforma clínica transformadora
- Em março de 2025, pesquisadores da East China Normal University and YolTech Therapeutics, em Xangai, publicaram dados pré-clínicos demonstrando que uma administração única de um sistema LNP-CRISPR-Cas9 obteve efeitos terapêuticos sustentados em um modelo de rato de hiperoxaluria primária tipo 1, avançando o caso de desenvolvimento clínico para a entrega não viral de LNP de terapias CRISPR para doenças hepáticas metabólicas raras para além dos mercados ocidentais
- Em junho de 2025, Eli Lilly completou a aquisição da Verve Therapeutics, integrando sua plataforma proprietária de edição LNP-base visando edição de genes cardiovasculares no portfólio de medicina de precisão da Lilly. Esta aquisição, representando o investimento direto de uma grande empresa farmacêutica na tecnologia de entrega LNP-CRISPR, sinalizou forte validação comercial do potencial terapêutico da edição de genes LNP de última geração para doenças cardiovasculares
SKU-
Obtenha acesso online ao relatório sobre a primeira nuvem de inteligência de mercado do mundo
- Painel interativo de análise de dados
- Painel de análise da empresa para oportunidades de elevado potencial de crescimento
- Acesso de analista de pesquisa para personalização e customização. consultas
- Análise da concorrência com painel interativo
- Últimas notícias, atualizações e atualizações Análise de tendências
- Aproveite o poder da análise de benchmark para um rastreio abrangente da concorrência
Metodologia de Investigação
A recolha de dados e a análise do ano base são feitas através de módulos de recolha de dados com amostras grandes. A etapa inclui a obtenção de informações de mercado ou dados relacionados através de diversas fontes e estratégias. Inclui examinar e planear antecipadamente todos os dados adquiridos no passado. Da mesma forma, envolve o exame de inconsistências de informação observadas em diferentes fontes de informação. Os dados de mercado são analisados e estimados utilizando modelos estatísticos e coerentes de mercado. Além disso, a análise da quota de mercado e a análise das principais tendências são os principais fatores de sucesso no relatório de mercado. Para saber mais, solicite uma chamada de analista ou abra a sua consulta.
A principal metodologia de investigação utilizada pela equipa de investigação do DBMR é a triangulação de dados que envolve a mineração de dados, a análise do impacto das variáveis de dados no mercado e a validação primária (especialista do setor). Os modelos de dados incluem grelha de posicionamento de fornecedores, análise da linha de tempo do mercado, visão geral e guia de mercado, grelha de posicionamento da empresa, análise de patentes, análise de preços, análise da quota de mercado da empresa, normas de medição, análise global versus regional e de participação dos fornecedores. Para saber mais sobre a metodologia de investigação, faça uma consulta para falar com os nossos especialistas do setor.
Personalização disponível
A Data Bridge Market Research é líder em investigação formativa avançada. Orgulhamo-nos de servir os nossos clientes novos e existentes com dados e análises que correspondem e atendem aos seus objetivos. O relatório pode ser personalizado para incluir análise de tendências de preços de marcas-alvo, compreensão do mercado para países adicionais (solicite a lista de países), dados de resultados de ensaios clínicos, revisão de literatura, mercado remodelado e análise de base de produtos . A análise de mercado dos concorrentes-alvo pode ser analisada desde análises baseadas em tecnologia até estratégias de carteira de mercado. Podemos adicionar quantos concorrentes necessitar de dados no formato e estilo de dados que procura. A nossa equipa de analistas também pode fornecer dados em tabelas dinâmicas de ficheiros Excel em bruto (livro de factos) ou pode ajudá-lo a criar apresentações a partir dos conjuntos de dados disponíveis no relatório.