Global Lysosomal Function Enhancing Drug Market
Tamanho do mercado em biliões de dólares
CAGR :
% 
 USD
964.00 Million
USD
4,285.22 Million
2025
2033
USD
964.00 Million
USD
4,285.22 Million
2025
2033
| 2026 –2033 | |
| USD 964.00 Million | |
| USD 4,285.22 Million | |
|
|
|
|
Função Lisossômica Global Reforçando a Segmentação do Mercado de Drogas, Por Classe de Medicamentos (Terapêutica de Substituição de Enzimas (ERT), Chaperonas Farmacológicas, Terapia de Redução de Substratos, Terapia Geneica e Outros), Aplicação (Doença de Gaucher, Doença de Fabry, Doença de Pompe, Doença de Niemann-Pick e Outros Distúrbios Lisossômicos de Armazenamento) - Tendências e Previsão da Indústria para 2033
Função lisossômica Melhorando a DrogaTamanho do Mercado
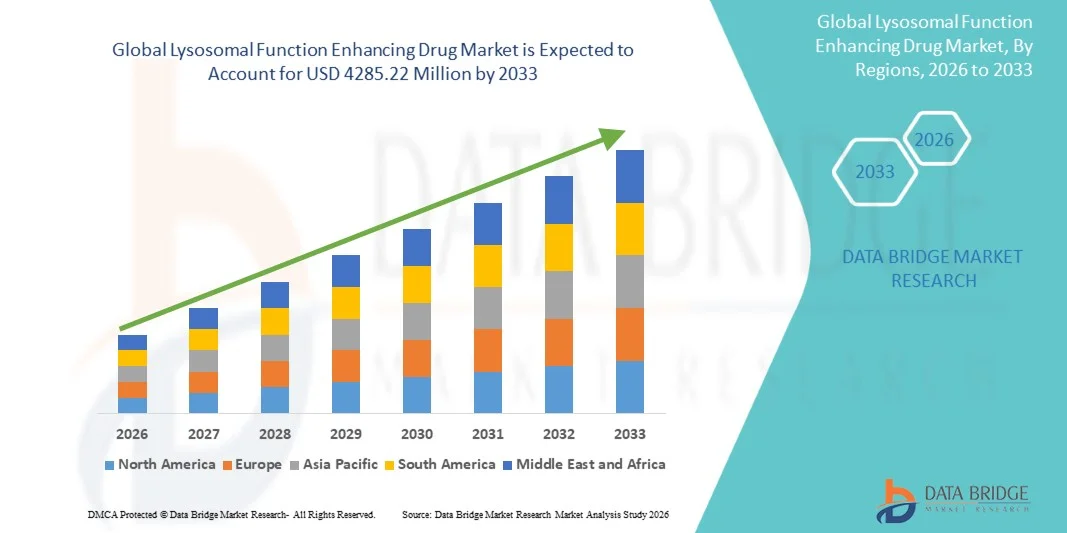
- O aumento global da função lisossómica no mercado de drogas foi avaliado emUSD 964,00 Milhões em 2025e espera-se alcançarUSD 4285,22 Milhões por 2033, emCAGR de 20,50%durante o período de previsão
- O crescimento do mercado é amplamente alimentado pela crescente prevalência de distúrbios de armazenamento lisossomal, tais como doença de Gaucher, doença de Fabry e doença de Pompe, juntamente com avanços em biotecnologia e pesquisa genética, levando a uma melhor compreensão das deficiências enzimáticas e mecanismos de doença
- Além disso, o aumento da adoção de terapia de substituição enzimática, acompanhantes farmacológicos e abordagens de terapia genética emergentes, juntamente com o crescente investimento no desenvolvimento de drogas de doenças raras e aprovações de medicamentos órfãs, estão estabelecendo a função lisossomal aumentando as soluções de medicamentos como componentes críticos da medicina de precisão moderna. Estes factores convergentes estão a acelerar a absorção da função lisossómica aumentando as terapias medicamentosas, aumentando assim significativamente o crescimento da indústria.
Função lisossômica Melhorando a DrogaAnálise de mercado
- As drogas que melhoram a função lisossômica, incluindo as terapias de substituição enzimática (ERT), as acompanhantenas farmacológicas e as terapias gênicas emergentes, são cada vez mais vitais no tratamento de distúrbios raros de armazenamento lisossómico, através do tratamento de deficiências enzimáticas subjacentes e da melhoria dos mecanismos de processamento de resíduos celulares.
- A crescente demanda por funções lisossômicas que aumentam as terapias medicamentosas é principalmente alimentada pela crescente prevalência de doenças genéticas raras, avanços no diagnóstico molecular, aumento das aprovações de medicamentos órfãos e crescente investimento em pesquisas de medicina de precisão e biotecnologia
- A América do Norte dominou a função lisossômica aumentando o mercado de medicamentos com a maior parcela de receita de 39,6% em 2025, caracterizada por forte infraestrutura de I&D biofarmacêutico, alta adoção de biológicos avançados, quadros regulatórios de suporte para medicamentos órfãos e aumento da atividade de ensaios clínicos em tratamentos de doenças raras
- Espera-se que a Ásia-Pacífico seja a região de crescimento mais rápido na função lisossomal que aumenta o mercado de medicamentos durante o período de previsão, devido à melhoria da infraestrutura de cuidados de saúde, ao aumento da conscientização de doenças genéticas raras, ao aumento do apoio do governo ao tratamento de doenças raras e ao aumento do acesso a terapias biológicas avançadas
- O segmento de terapia de substituição enzimática dominou a maior parcela de receita de mercado de 44,9% em 2025, impulsionada por sua forte eficácia clínica e uso generalizado como abordagem padrão de tratamento para múltiplos distúrbios de armazenamento lisossomal.
Âmbito do relatório eFunção lisossómica Melhorando a Segmentação do Mercado de Drogas
|
Atributos |
Função lisossômica Melhorando a Chave das DrogasPerspectivas de mercado |
|
Segmentos Cobertos |
|
|
Países abrangidos |
América do Norte · U.S. · Canadá · México Europa · Alemanha · França · U.K. · Países Baixos · Suíça · Bélgica · Rússia · Itália · Espanha · Turquia · Resto da Europa Ásia- Pacífico · China · Japão · Índia · Coreia do Sul · Singapura · Malásia · Austrália · Tailândia · Indonésia · Filipinas · Resto da Ásia-Pacífico Médio Oriente e África · Arábia Saudita · U.A.E. · África do Sul · Egito · Israel · Resto do Oriente Médio e África América do Sul · Brasil · Argentina · Resto da América do Sul |
|
Jogadores do mercado chave |
•F. Hoffmann- La Roche Ltd.. (Suíça) |
|
Oportunidades de Mercado |
· Avanço na terapia genética e na próxima geração biológica · Aumento da demanda em mercados emergentes |
|
Informações sobre o Valor Adicionado |
Além dos insights sobre cenários de mercado, como valor de mercado, taxa de crescimento, segmentação, cobertura geográfica e grandes atores, os relatórios de mercado curados pela Data Bridge Market Research também incluem análise de especialistas em profundidade, epidemiologia de pacientes, análise de pipelines, análise de preços e marco regulatório. |
Função lisossómica Melhorando as tendências do mercado de drogas
“Avanços na Direção de Caminho Celular e Terapêutica de Precisão“
- Uma tendência significativa e acelerada no mercado global de drogas com função lisossômica é o foco crescente em terapias de precisão visando vias de processamento de resíduos celulares. Estes medicamentos são projetados para melhorar a eficiência lisossomal, melhorar a autofagia e restaurar a homeostase celular em doenças ligadas à disfunção lisossômica
- Por exemplo, terapias emergentes de pequenas moléculas e compostos moduladores de enzimas estão sendo desenvolvidas para melhorar a atividade enzimática lisossomal em distúrbios como doenças de armazenamento lisossomal e condições neurodegenerativas. Estes avanços apoiam abordagens de tratamento mais orientadas e baseadas em mecanismos
- O uso de desenvolvimento de drogas orientadas por biomarcadores está permitindo que pesquisadores identifiquem subgrupos de pacientes com perfis específicos de disfunção lisossomal, melhorando as taxas de sucesso de ensaios clínicos e precisão terapêutica
- Além disso, os avanços na terapia genética e estratégias de substituição enzimática estão expandindo a paisagem do tratamento, abordando defeitos genéticos subjacentes responsáveis pelo comprometimento lisossomal
- A crescente investigação sobre a modulação da autofagia está a reforçar ainda mais a inovação neste domínio, uma vez que a restauração dos mecanismos de reciclagem celular é cada vez mais reconhecida como uma via terapêutica chave em múltiplas doenças crónicas e raras.
Função lisossômica Melhorando a Dinâmica do Mercado de Drogas
Controlador
“Prevalência crescente de Distúrbios de Armazenamento Lisossômico e Expansão de Pesquisa em Doenças Neurodegenerativas”
- A crescente prevalência de distúrbios de armazenamento lisossomal (DLS), juntamente com o crescente reconhecimento da disfunção lisossômica em doenças neurodegenerativas, é um dos principais motores do crescimento do mercado
- Por exemplo, condições como a doença de Gaucher, a doença de Fabry e a doença de Niemann-Pick estão conduzindo a procura de terapias de substituição enzimática e terapias de redução de substrato destinadas a restaurar a função lisossomal
- A expansão da investigação sobre a doença de Alzheimer, a doença de Parkinson e a doença de Huntington está a realçar ainda mais o papel da deficiência lisossómica na neurodegeneração, abrindo novas oportunidades terapêuticas
- O aumento dos programas de triagem de recém-nascidos e as iniciativas de testes genéticos estão melhorando as taxas de diagnóstico precoce de distúrbios lisossomais, possibilitando intervenção terapêutica oportuna
- O aumento do investimento de empresas farmacêuticas e de biotecnologia no desenvolvimento de drogas raras está a acelerar a inovação e a expandir o gasoduto de terapias orientadas para os lisossomos
Restrição/Desafio
“Altos custos de desenvolvimento, biologia complexa e acessibilidade limitada ao tratamento“
- Um dos principais desafios na função lisossómica que aumenta o mercado de drogas é o alto custo e a complexidade associados ao desenvolvimento de drogas, especialmente para doenças raras e geneticamente diversas
- Por exemplo, terapias de substituição enzimática requerem extensa pesquisa, infraestrutura de fabricação biológica e avaliação clínica de longo prazo, tornando-as caras e demoradas para desenvolver
- A complexidade biológica das vias lisossómicas também torna a droga alvo desafiador, uma vez que múltiplas enzimas e processos celulares estão frequentemente envolvidos na progressão da doença
- Populações limitadas de pacientes para muitos distúrbios de armazenamento lisossomal criam dificuldades na realização de ensaios clínicos em larga escala, retardando aprovações regulatórias e comercialização
- Além disso, custos elevados de tratamento e quadros de reembolso limitados em várias regiões restringem o acesso dos doentes a terapias avançadas, especialmente em países de baixa e média renda
- Superar esses desafios exigirá avanços na terapia gênica, melhoria da escalabilidade de fabricação e programas de acesso à saúde global mais fortes para garantir maior disponibilidade de tratamentos de transtorno lisossomal
Função lisossómica Melhorando o escopo do mercado de drogas
O mercado é segmentado com base na classe e aplicação de drogas.
- Por Classe de Drogas
Com base na classe de drogas, a função lisossômica aumentar o mercado de drogas é segmentado em terapia de substituição enzimática (ERT), acompanhantes farmacológicas, terapia de redução de substrato, terapia genética, entre outros. O segmento de terapia de substituição enzimática dominou a maior parcela de receita de mercado de 44,9% em 2025, impulsionada pela sua forte eficácia clínica e uso generalizado como abordagem padrão de tratamento para múltiplos distúrbios lisossomais de armazenamento. A TRE continua sendo a modalidade mais estabelecida devido à sua capacidade de substituir diretamente enzimas deficientes ou ausentes, melhorando os resultados do manejo da doença. O aumento da prevalência de doenças genéticas raras apoia ainda mais a demanda. Forte infraestrutura de administração hospitalar aumenta a entrega de tratamento. A ampliação da cobertura de reembolsos em regiões desenvolvidas aumenta a acessibilidade. Melhorias contínuas na produção de enzimas recombinantes melhoram a segurança e a eficácia. Aumentar a conscientização dos clínicos quanto à intervenção precoce fortalece a adoção. A validação clínica contínua em múltiplas indicações reforça sua dominância.
Espera-se que o segmento de terapia genética testemunhe o CAGR mais rápido de 23,8% de 2026 a 2033, impulsionado por rápidos avanços em tecnologias de engenharia genética e sistemas de entrega direcionados. Terapias genéticas oferecem o potencial para resultados a longo prazo ou até mesmo curativos em distúrbios de armazenamento lisossomal, reduzindo significativamente a carga de tratamento ao longo da vida. O aumento do investimento na investigação sobre doenças raras acelera o desenvolvimento de gasodutos. A expansão do uso de plataformas vetoriais virais como AAV aumenta a precisão terapêutica. O aumento das taxas de sucesso em ensaios clínicos suporta o potencial de comercialização. Fortes financiamentos das empresas de biotecnologia e iniciativas públicas impulsionam a inovação. O aumento do apoio regulamentar ao desenvolvimento de medicamentos órfãos acelera ainda mais as aprovações. O aumento da demanda por tratamentos duradouros e únicos impulsiona a adoção do mercado.
- Por Aplicação
Com base na aplicação, o mercado de drogas com função lisossômica é segmentado em doença de Gaucher, doença de Fabry, doença de Pompe, doença de Niemann-Pick e outros distúrbios de armazenamento lisossômico. O segmento da doença de Gaucher representou a maior parcela de receita de mercado de 38,4% em 2025, impulsionada por sua prevalência relativamente maior e vias de tratamento bem estabelecidas. A disponibilidade de terapias de substituição enzimática aprovadas suporta significativamente sua dominância. O diagnóstico precoce através de programas de triagem genética e neonatal melhora as taxas de início do tratamento. A forte conscientização dos profissionais de saúde aumenta a identificação do paciente. Políticas de reembolso do governo ainda melhor acesso à terapia. O aumento da gestão hospitalar das doenças raras apoia a procura sustentada. Melhorias contínuas na acurácia diagnóstica fortalecem os resultados do tratamento. A expansão da atividade de pesquisa clínica contribui para avanços terapêuticos.
Espera-se que o segmento da doença de Pompe testemunhe o CAGR mais rápido de 22,6% de 2026 a 2033, impulsionado pela adoção crescente de programas de triagem precoce e pelo aumento da conscientização de distúrbios lisossomais neuromusculares. Avanços na substituição enzimática e nas opções de terapia genética estão melhorando a sobrevida e os resultados de qualidade de vida. O aumento das iniciativas de rastreamento neonatal possibilita a detecção e início precoce do tratamento. Aumentar a disponibilidade de biológicos específicos aumenta a eficácia do tratamento. O aumento dos investimentos em pesquisas em doenças metabólicas raras acelera o desenvolvimento de drogas. O aumento dos registros dos pacientes melhora o rastreamento e as estratégias de manejo da doença. Forte atividade do gasoduto apoia o crescimento futuro. A melhoria das infra-estruturas de saúde nas regiões emergentes reforça ainda mais o acesso ao tratamento.
Função lisossómica Melhorando a Análise Regional do Mercado de Drogas
- A América do Norte dominou a função lisossômica aumentando o mercado de medicamentos com a maior parcela de receita de 39,6% em 2025, caracterizada por forte infraestrutura de I&D biofarmacêutico, alta adoção de biológicos avançados, quadros regulatórios de suporte para medicamentos órfãos e aumento da atividade de ensaios clínicos em tratamentos de doenças raras
- A região beneficia de um gasoduto bem estabelecido de terapias de substituição enzimática (ERTs), terapias baseadas em genes e terapias de acompanhamento de pequenas moléculas visando distúrbios de armazenamento lisossomal, apoiados por uma forte colaboração entre academias e empresas farmacêuticas
- Políticas de reembolso favoráveis, programas de diagnóstico precoce de doenças e crescente inscrição de pacientes em registros de doenças raras reforçam ainda mais o crescimento do mercado na América do Norte
U.S. Função lisossômica Melhorando a Perspectiva do Mercado de Drogas
A função lisossômica dos EUA, o aumento do mercado de drogas, captou a maior participação na receita da América do Norte em 2025, impulsionada por avanços contínuos no desenvolvimento de drogas raras e forte presença de empresas biofarmacêuticos líderes. Aumentar as aprovações de medicamentos órfãos visando transtornos de armazenamento lisossomal, juntamente com a expansão de ensaios clínicos para terapias de genes de próxima geração, estão aumentando significativamente a expansão do mercado. Além disso, grupos fortes de defesa de pacientes e melhores capacidades diagnósticas estão permitindo a detecção e início precoce do tratamento, melhorando os resultados a longo prazo.
Europa Função lisossómica Melhorar a Perspectiva do Mercado de Drogas
Prevê-se que a função lisossómica europeia que aumenta o mercado de medicamentos se expanda num CAGR estável durante o período previsto, apoiada por legislação robusta em matéria de medicamentos órfãos, aumento do financiamento da investigação para doenças raras e adopção crescente de biológicos. Os fortes incentivos regulamentares da região, incluindo a exclusividade do mercado e as aprovações rápidas, encorajam a inovação farmacêutica. Além disso, a crescente colaboração entre instituições de pesquisa e empresas de biotecnologia está acelerando o desenvolvimento da terapia para distúrbios de armazenamento lisossomal.
Função lisossômica do Reino Unido Melhorando a Perspectiva do Mercado de Drogas
Prevê-se que a função lisossomal do Reino Unido que aumenta o mercado de medicamentos cresça de forma constante, impulsionada pelo forte apoio da NHS ao tratamento de doenças raras e pela crescente participação em programas de investigação clínica. Aumentar o acesso a terapias de substituição enzimática e aumentar o conhecimento de distúrbios de armazenamento lisossomal estão melhorando as taxas de diagnóstico e tratamento. Além disso, o forte ecossistema de pesquisa acadêmica do Reino Unido continua a contribuir para a inovação em terapias baseadas em genes e proteínas.
Alemanha Função lisossómica Melhorando a Perspectiva do Mercado de Drogas
Prevê-se que a função lisossómica alemã que aumenta o mercado de medicamentos se expanda a um ritmo considerável, apoiada por infra-estruturas de cuidados de saúde avançadas e por uma forte ênfase na medicina de precisão. O aumento do uso de biológicos, a melhoria dos programas de rastreio genético e o aumento do investimento em investigação sobre doenças raras estão a impulsionar o crescimento do mercado. A forte base farmacêutica da Alemanha também apoia a produção e distribuição de agentes terapêuticos avançados.
Função lisossomal Ásia-Pacífico Melhorando a Perspectiva do Mercado de Drogas
Espera-se que a função lisossomal Ásia-Pacífico que aumenta o mercado de medicamentos cresça o mais rápido possível durante o período de previsão, devido à melhoria da infraestrutura de saúde, ao aumento da conscientização de doenças genéticas raras, ao aumento do apoio do governo ao tratamento de doenças raras e ao aumento do acesso a terapias biológicas avançadas. Os crescentes investimentos na modernização da saúde e na expansão das capacidades de diagnóstico estão acelerando ainda mais o desenvolvimento do mercado em toda a região.
Japão Função lisossômica Melhorando a Perspectiva do Mercado de Drogas
A função lisossômica do Japão que aumenta o mercado de medicamentos é impulsionada por capacidades avançadas de pesquisa médica e forte foco na gestão de doenças raras. A adoção crescente de terapias de substituição enzimática e a crescente integração da medicina genômica na prática clínica estão melhorando os resultados do tratamento. O envelhecimento da população do Japão também suporta maiores taxas de diagnóstico e demanda sustentada por cuidados terapêuticos de longo prazo.
China Função lisossômica Melhorando a Perspectiva do Mercado de Drogas
A função lisossômica da China, o aumento do mercado de drogas, representou uma parte significativa na Ásia-Pacífico em 2025, apoiada pela expansão da infraestrutura de saúde, aumento do investimento em pesquisa em biotecnologia e crescentes iniciativas governamentais para o gerenciamento de doenças raras. A adoção crescente de biologics avançados e a melhoria do acesso aos serviços especializados de diagnóstico estão impulsionando o crescimento do mercado em todo o país.
Função lisossómica Melhorando a participação no mercado de drogas
A indústria de drogas é liderada principalmente por empresas bem estabelecidas, incluindo:
• F. Hoffmann- La Roche Ltd. (Suíça)
• Sanofi S. A. (França)
• Pfizer Inc. (EUA)
• Novartis AG (Suíça)
• Johnson & Johnson (EUA)
• Takeda Pharmaceutical Company Limited (Japão)
• Amgen Inc. (EUA)
• Bristol- Myers Squibb Company (EUA)
• Eli Lilly e Companhia (EUA)
• AbbVie Inc. (EUA)
• BioMarin Pharmaceutical Inc. (EUA)
• Vertex Pharmaceuticals Incorporated (EUA)
• Ipsen S.A. (França)
• Chiesi Farmaceutici S.p.A. (Itália)
• Ultragenyx Pharmaceutical Inc. (EUA)
• Sobi (Orfão Sueco Biovitrum AB) (Suécia)
• Teva Pharmaceutical Industries Ltd. (Israel)
• Sun Pharmaceutical Industries Ltd. (Índia)
Mais recentes desenvolvimentos na função lisossômica global Melhorando o mercado de drogas
- Em fevereiro de 2021, Genethon iniciou um programa de terapia genética clínica para a doença de Pompe, avançando o tratamento da desordem lisossomal de armazenamento, visando a deficiência enzimática através da entrega de genes baseados em AAV, marcando um passo importante para a restauração da função lisossômica a longo prazo em doenças genéticas raras
- Em julho de 2022, pesquisas publicadas na literatura biomédica destacaram o crescente desenvolvimento de terapias de substituição enzimática (ETRs) e terapias de redução de substratos (TRSs) com o objetivo de restaurar a atividade enzimática lisossomal, reforçando a função lisossomal como um alvo terapêutico chave em distúrbios metabólicos herdados
- Em março de 2023, os avanços científicos no design de drogas enfatizaram o desenvolvimento de sistemas de liberação de drogas com foco em lisossomas e nanocarregadores responsivos ao pH, melhorando a liberação intracelular de drogas e aumentando a modulação da função lisossomal na terapia do câncer e doença genética
- Em setembro de 2023, pesquisadores relataram o uso crescente de terapias visando proteínas de membrana lisossomal, com foco na correção da disfunção lisossômica e melhoria dos mecanismos de degradação de resíduos celulares, ampliando o escopo terapêutico do desenvolvimento de drogas baseadas em lisossomos
- Em junho de 2024, os avanços na medicina molecular destacaram o surgimento de estratégias terapêuticas lisossômicas, como ativação da biogênese lisossómica e modulação da autofagia, com o objetivo de tratar doenças neurodegenerativas e metabólicas, restaurando vias de depuração celular.
- Em maio de 2025, revisões científicas relataram progresso no transplante lisossomal e na entrega de drogas mediadas pela vesícula extracelular (VE), representando uma abordagem de próxima geração para restaurar a função lisossômica em doenças genéticas e neurodegenerativas através de estratégias de substituição de organelas
SKU-
Obtenha acesso online ao relatório sobre a primeira nuvem de inteligência de mercado do mundo
- Painel interativo de análise de dados
- Painel de análise da empresa para oportunidades de elevado potencial de crescimento
- Acesso de analista de pesquisa para personalização e customização. consultas
- Análise da concorrência com painel interativo
- Últimas notícias, atualizações e atualizações Análise de tendências
- Aproveite o poder da análise de benchmark para um rastreio abrangente da concorrência
Metodologia de Investigação
A recolha de dados e a análise do ano base são feitas através de módulos de recolha de dados com amostras grandes. A etapa inclui a obtenção de informações de mercado ou dados relacionados através de diversas fontes e estratégias. Inclui examinar e planear antecipadamente todos os dados adquiridos no passado. Da mesma forma, envolve o exame de inconsistências de informação observadas em diferentes fontes de informação. Os dados de mercado são analisados e estimados utilizando modelos estatísticos e coerentes de mercado. Além disso, a análise da quota de mercado e a análise das principais tendências são os principais fatores de sucesso no relatório de mercado. Para saber mais, solicite uma chamada de analista ou abra a sua consulta.
A principal metodologia de investigação utilizada pela equipa de investigação do DBMR é a triangulação de dados que envolve a mineração de dados, a análise do impacto das variáveis de dados no mercado e a validação primária (especialista do setor). Os modelos de dados incluem grelha de posicionamento de fornecedores, análise da linha de tempo do mercado, visão geral e guia de mercado, grelha de posicionamento da empresa, análise de patentes, análise de preços, análise da quota de mercado da empresa, normas de medição, análise global versus regional e de participação dos fornecedores. Para saber mais sobre a metodologia de investigação, faça uma consulta para falar com os nossos especialistas do setor.
Personalização disponível
A Data Bridge Market Research é líder em investigação formativa avançada. Orgulhamo-nos de servir os nossos clientes novos e existentes com dados e análises que correspondem e atendem aos seus objetivos. O relatório pode ser personalizado para incluir análise de tendências de preços de marcas-alvo, compreensão do mercado para países adicionais (solicite a lista de países), dados de resultados de ensaios clínicos, revisão de literatura, mercado remodelado e análise de base de produtos . A análise de mercado dos concorrentes-alvo pode ser analisada desde análises baseadas em tecnologia até estratégias de carteira de mercado. Podemos adicionar quantos concorrentes necessitar de dados no formato e estilo de dados que procura. A nossa equipa de analistas também pode fornecer dados em tabelas dinâmicas de ficheiros Excel em bruto (livro de factos) ou pode ajudá-lo a criar apresentações a partir dos conjuntos de dados disponíveis no relatório.