Global Microbiome Engineering Therapeutics Market
Tamanho do mercado em biliões de dólares
CAGR :
% 
 USD
1.74 Billion
USD
7.18 Billion
2025
2033
USD
1.74 Billion
USD
7.18 Billion
2025
2033
| 2026 –2033 | |
| USD 1.74 Billion | |
| USD 7.18 Billion | |
|
|
|
|
Segmentação do Mercado de Engenharia Terapêutica Global de Microbiomes (Transplante de Microbiota Fecal, Produtos Bioterapêuticos Vivos, Consórcios Microbiais Probióticos, Terapêutica Baseada em Probióticos, Terapêutica Baseada em Prebióticos, Doenças Pós-Bióticas, Transplantes Sinbióticos e Transtornos Dermatológicos Microbiológicos), Aplicação (Transtornos Gastrointestinais, Transtornos Metabólicos, Oncologia, Doenças Infecciosas, Doenças Autoimunes e Inflamações, Transtornos Neurológicos e Transtornos Dermatológicos), Tecnologia (Plataformas de Biologia Sintésica, Engenharia Genética de Microbes, Editação Microbiana Baseada em CRISPR, Metagenômica e Microbiome Sequenciamento, Bioinformática & Microbiome, Análise Microbiome Promotiva, Culturomics & Técnicas de Isolamento Microbial e Sistemas de Engenharia Baseada em Fermentação), Usuário Final (Hospitais, Especialidades Clínicas Especializadas, Instituto de Pesquisas de Pesquisas, Empresas Farmacêutica, Empresas Farmacêutica Tendências e Previsão da Indústria para 2033
Microbiome Engenharia TerapêuticaTamanho do Mercado
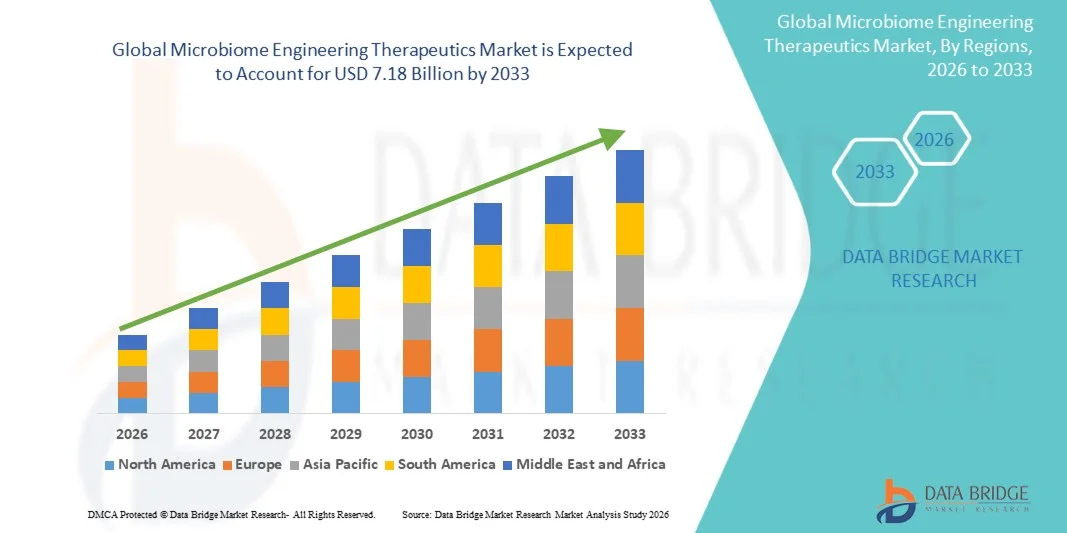
- O tamanho global do mercado de engenharia de microbiomas foi avaliado em1,74 mil milhões de USD em 2025e espera-se alcançar7,18 mil milhões de USD até 2033, emCAGR de 19,40 %durante o período de previsão
- O crescimento do mercado é impulsionado principalmente por avanços crescentes na biologia sintética, sequenciamento de microbiomas e desenvolvimento de produtos bioterapêuticos vivos, juntamente com a crescente validação clínica de intervenções baseadas em microbiomas em várias áreas da doença
- Além disso, a crescente demanda por abordagens médicas de precisão, a expansão de aplicações em doenças gastrointestinais, oncologia e doenças metabólicas e o aumento do investimento de empresas farmacêuticas e de biotecnologia estão acelerando a adoção de terapias de engenharia de microbiomas, aumentando significativamente o crescimento da indústria.
Microbiome Engenharia TerapêuticaAnálise de mercado
- A terapia de engenharia de microbiomas, que envolve a modificação ou modulação deliberada da microbiota humana usando micróbios projetados, bioterapêuticos vivos e abordagens de biologia sintética, estão emergindo como um componente transformador da medicina de precisão de próxima geração devido à sua capacidade de direcionar mecanismos de doença no nível microbiano e metabólico através de distúrbios gastrointestinais, metabólicos, oncológicos e imunes.
- A crescente demanda por terapias de engenharia de microbiomas é impulsionada principalmente por avanços rápidos na biologia sintética, aumento da validação clínica de produtos bioterapêuticos vivos e crescente reconhecimento do papel do microbioma intestinal na saúde humana, além do aumento do investimento de empresas farmacêuticas e de biotecnologia em gasodutos de desenvolvimento de medicamentos baseados em microbiomas.
- A América do Norte dominou o mercado de terapias de engenharia de microbiomas com a maior parcela de receita de 41,8% em 2025, apoiada por forte infraestrutura de biotecnologia, adoção precoce de terapias avançadas, financiamento significativo de P&D e presença de principais atores da indústria e instituições de pesquisa clínica, com os EUA liderando em ensaios clínicos e aprovações regulatórias para produtos bioterapêuticos vivos e terapias microbianas projetadas.
- Asia-Pacific é esperado para ser a região de crescimento mais rápido no mercado de engenharia de microbiomas terapêuticos durante o período de previsão devido à expansão dos investimentos em biotecnologia, aumento da inovação em saúde, aumento da prevalência de doenças crônicas e adoção crescente de abordagens de medicina de precisão em países como China, Japão e Índia
- O segmento de Produtos Bioterapêuticos Vivos dominou o mercado de engenharia de microbiomas terapêuticos com a maior participação de mercado de 38,6% em 2025, impulsionado pelo forte progresso clínico no tratamento de infecções difficile Clostridioides recorrentes e gasodutos de desenvolvimento em expansão visando distúrbios gastrointestinais e metabólicos
Âmbito do relatório eMicrobiome Engenharia Terapêutica Segmentação de Mercado
|
Atributos |
Chave Terapêutica de Engenharia de MicrobiomePerspectivas de mercado |
|
Segmentos Cobertos |
|
|
Países abrangidos |
América do Norte · U.S. · Canadá · México Europa · Alemanha · França · U.K. · Países Baixos · Suíça · Bélgica · Rússia · Itália · Espanha · Turquia · Resto da Europa Ásia- Pacífico · China · Japão · Índia · Coreia do Sul · Singapura · Malásia · Austrália · Tailândia · Indonésia · Filipinas · Resto da Ásia-Pacífico Médio Oriente e África · Arábia Saudita · U.A.E. · África do Sul · Egito · Israel · Resto do Oriente Médio e África América do Sul · Brasil · Argentina · Resto da América do Sul |
|
Jogadores do mercado chave |
|
|
Oportunidades de Mercado |
· Expansão de consórcios microbianos projetados para fornecer moléculas terapêuticas direcionadas · Oportunidade crescente para terapias adjuvantes baseadas em microbiomas em oncologia e imunoterapia |
|
Informações sobre o Valor Adicionado |
Além dos insights sobre cenários de mercado, como valor de mercado, taxa de crescimento, segmentação, cobertura geográfica e principais atores, os relatórios de mercado curados pela Data Bridge Market Research também incluem análise de especialistas em profundidade, epidemiologia do paciente, análise de pipelines, análise de preços e marco regulatório |
Tendências do mercado da terapia da engenharia do microbiome
“Avançamento da Terapia Microbial Projetada e Modulação do Microbiome de Precisão”
- Uma tendência significativa e acelerada no mercado global de terapias de engenharia de microbiomas é o rápido desenvolvimento de terapia microbiana e produtos bioterapêuticos vivos projetados para modular precisamente o microbioma humano para tratamento de doenças direcionadas em doenças gastrointestinais, metabólicas e relacionadas com a imunidade
- Por exemplo, a terapia SER-109 da Seres Therapeutics para infecção recorrente por C. difficile demonstra como os candidatos a medicamentos baseados em microbiomas estão avançando através da validação clínica tardia para se tornarem opções terapêuticas padronizadas
- Biologia sintética e ferramentas de edição de genes estão sendo cada vez mais usadas para projetar micróbios com maior funcionalidade, permitindo-lhes entregar proteínas terapêuticas, regular vias metabólicas e restaurar o equilíbrio microbiano em condições de doença
- A integração de tecnologias multi-omics tais como metagenomics, transcriptomics, e metabolomics com a análise orientada por AI está permitindo uma compreensão mais profunda das interações do microbiome-hospedeiro e melhorar a precisão da descoberta da droga
- Expansão de aplicações oncológicas baseadas em microbiomas, incluindo bactérias projetadas usadas para melhorar a resposta inibidora do controle imunológico, está emergindo como uma área de inovação de alto valor no desenvolvimento do tratamento do câncer
- Esta tendência para terapias de microbiomas altamente orientadas, projetadas e orientadas por dados está fundamentalmente reformulando paradigmas de tratamento em doenças crônicas e complexas, onde os medicamentos convencionais têm eficácia limitada.
- A demanda por plataformas de engenharia de microbiomas escaláveis, regulamentadas e clinicamente validadas está crescendo rapidamente nos setores farmacêutico e biotecnológico, à medida que a adoção de medicamentos personalizados continua se expandindo.
Microbiome Engenharia Terapêutica Mercado Dinâmica
Controlador
“Demanda crescente de medicina de precisão e tratamento de doenças baseada em microbiomas”
- A crescente demanda por medicina de precisão e terapias direcionadas ao microbioma, aliada à crescente evidência clínica que liga o desequilíbrio da microbiota a múltiplas doenças crônicas, é um fator importante para a expansão da terapêutica da engenharia do microbioma.
- Por exemplo, a aprovação da Ferring Pharmaceuticals REBYOTA para prevenção de infecção recorrente por C. difficile destaca aceitação regulatória de terapias baseadas em microbiomas em sistemas de saúde tradicionais
- O aumento da prevalência de distúrbios gastrointestinais, doenças metabólicas e câncer está acelerando a necessidade de novas abordagens de tratamento que alavancam a modulação do microbioma para melhores resultados clínicos
- Além disso, os investimentos crescentes de empresas farmacêuticas e de empresas de biotecnologia em gasodutos de I&D de microbiome estão apoiando a inovação rápida e a comercialização de terapias microbianas projetadas.
- O aumento da adoção de diagnósticos de acompanhantes baseados em microbiomas está ajudando a identificar assinaturas microbianas específicas do paciente, melhorando o direcionamento do tratamento e taxas de sucesso terapêutico
- Expansão de dutos de ensaios clínicos focados em bioterapêuticas vivas projetadas está fortalecendo ainda mais a confiança entre reguladores e prestadores de cuidados de saúde
- A adoção crescente de tecnologias avançadas de sequenciamento e plataformas de biologia sintética está aumentando ainda mais a eficiência do desenvolvimento de drogas e ampliando as aplicações terapêuticas
- A mudança para soluções personalizadas de saúde está impulsionando significativamente a adoção de terapias de engenharia de microbiomas em sistemas de saúde globais
Restrição/Desafio
“Caminhos Regulatórios Complexos e Normalização Limitada de Produtos Baseados em Microbiomas”
- As preocupações com as complexas vias de aprovação regulatória e a falta de estruturas padronizadas de fabricação e caracterização de terapias baseadas em microbiomas representam um desafio significativo para o crescimento do mercado.
- Por exemplo, a variabilidade nas diretrizes regulatórias entre as regiões para produtos bioterapêuticos vivos gera atrasos na aprovação clínica e comercialização de terapias de microbioma projetadas
- A dificuldade em garantir composição microbiana consistente, estabilidade e reprodutibilidade de terapias projetadas adiciona complexidade aos processos de fabricação e controle de qualidade em larga escala
- Além disso, os dados clínicos a longo prazo limitados sobre a segurança, eficácia e interações microbiome-hospedeiro restringem a adoção mais ampla destas terapêuticas pelo médico e pelo paciente.
- Alta dependência de armazenamento especializado em cadeia fria e requisitos de manuseio sensíveis aumenta a complexidade logística e os custos operacionais para produtos baseados em microbiomas
- Desafios de propriedade intelectual relacionados com estirpes microbianas de origem natural também criam barreiras para garantir e proteger gasodutos de inovação comercial
- O alto custo e a complexidade técnica associados ao desenvolvimento de consórcios microbianos e de terapias baseadas em biologia sintética dificultam ainda mais a comercialização em mercados emergentes.
- Superar estes desafios através de quadros regulamentares harmonizados, de melhores normas de fabrico e de validação clínica a longo prazo será fundamental para um crescimento sustentado do mercado
Microbiome Engenharia Terapêutica Âmbito de Mercado
O mercado é segmentado com base no tipo de terapia, aplicação, tecnologia e usuário final.
- Por tipo de terapia
Com base no tipo de terapia, o mercado de terapias de engenharia de microbiomas é segmentado em transplante de microbiota fecal (FMT), produtos bioterapêuticos vivos (LBPs), consórcios microbianos projetados, terapias baseadas em probióticos, terapêutica baseada em prebióticos, pós-bióticos, sinbióticos e terapias baseadas em metabólitos microbianos. O segmento de Produtos Bioterapêuticos Vivos (LBPs) dominou o mercado com a maior parcela de receita de mercado de 38,6% em 2025, impulsionada pelo aumento da validação clínica e avanço regulatório da terapêutica baseada em microbiomas. As LBPs são amplamente utilizadas no tratamento da infecção difficile recorrente por Clostridioides e estão se expandindo rapidamente para doenças inflamatórias intestinais, distúrbios metabólicos e aplicações oncológicas. Sua capacidade de fornecer modulação direcionada do microbioma intestinal com formulações padronizadas torna-os comercialmente mais viáveis em comparação com terapias convencionais de microbioma. Empresas farmacêuticas e de biotecnologia estão cada vez mais investindo em gasodutos de desenvolvimento de LBP devido a resultados clínicos favoráveis e crescente aceitação do médico
Prevê-se que o segmento de consórcios microbianos projetados testemunhe a taxa de crescimento mais rápida de 22,4% de 2026 a 2033, alimentado por avanços nas tecnologias de engenharia de microbiomas de precisão e biologia sintética. Os consórcios microbianos projetados envolvem o uso de múltiplas cepas microbianas projetadas que podem desempenhar funções terapêuticas especializadas dentro do corpo humano. Essas terapias estão ganhando atenção para o seu potencial em oncologia, doenças metabólicas e doenças autoimunes devido à sua maior funcionalidade e capacidade de direcionamento de precisão. O aumento da pesquisa em micróbios programáveis capazes de produzir compostos terapêuticos está acelerando a inovação neste segmento. Empresas de biotecnologia estão colaborando ativamente com institutos acadêmicos para desenvolver plataformas de consórcios microbianos de última geração para aplicações personalizadas de medicina.
- Por Aplicação
Com base na aplicação, o mercado é segmentado em distúrbios gastrointestinais, distúrbios metabólicos, oncologia, doenças infecciosas, doenças autoimunes e inflamatórias, distúrbios neurológicos e distúrbios dermatológicos. O segmento de distúrbios gastrointestinais dominou o mercado com a maior parcela de receita de mercado de 45,1% em 2025, impulsionada pelo amplo papel do microbioma intestinal na saúde digestiva e regulação imunológica. Alta prevalência de infecção recorrente por C. difficile, doença inflamatória intestinal, doença de Crohn e colite ulcerativa tem aumentado significativamente a demanda por terapias baseadas em microbiomas. As evidências clínicas que apoiam a eficácia da modulação do microbioma na restauração da flora intestinal saudável fortaleceram a confiança do médico e a adoção do paciente. As aprovações regulatórias para a terapêutica do microbioma visando transtornos gastrointestinais também aceleraram as atividades de comercialização nesse segmento.
Espera-se que o segmento oncológico testemunhe a taxa de crescimento mais rápida de 24,7% de 2026 a 2033, impulsionada pelo aumento da pesquisa sobre interações do sistema microbioma-imune e seu impacto na eficácia da terapia oncológica. Cientistas estão explorando terapias de microbioma projetadas para melhorar a resposta do paciente a imunoterapias, como inibidores de controle imunológico. A capacidade de certas cepas microbianas para melhorar a atividade imune antitumoral está criando oportunidades significativas na oncologia de precisão. O aumento da incidência global do cancro e a crescente necessidade de abordagens de tratamento adjuvante estão a acelerar o investimento em terapêuticas oncológicas baseadas em microbiomas. Empresas farmacêuticas e institutos de pesquisa sobre câncer estão cada vez mais realizando ensaios clínicos focados em terapias microbianas projetadas para tumores sólidos e cânceres gastrointestinais.
- Por Tecnologia
Com base na tecnologia, o mercado é segmentado em plataformas de biologia sintética, engenharia genética de micróbios, edição microbiana baseada em CRISPR, metagenômica e sequenciamento de microbiomas, análise de microbiomas baseada em bioinformática e IA, técnicas de cultivo e isolamento microbiano e sistemas de engenharia baseados em fermentação. O segmento metagenômica e sequenciamento de microbiomas dominou o mercado com a maior parcela de receita de mercado de 33,5% em 2025, impulsionada pelo seu papel crítico na identificação da composição microbiana, diversidade e assinaturas microbianas relacionadas à doença. Tecnologias de sequenciamento são amplamente utilizadas na descoberta de microbiomas, identificação de biomarcadores e processos de desenvolvimento terapêutico. O declínio do custo das tecnologias de sequenciamento de próxima geração aumentou significativamente a acessibilidade entre institutos de pesquisa e empresas de biotecnologia. A crescente demanda por medicina de precisão e terapêutica personalizada está incentivando ainda mais a adoção de plataformas avançadas de sequenciamento de microbiomas. As empresas farmacêuticas dependem fortemente da análise metagenômica para entender as interações microbiome-hospedeiro e otimizar o direcionamento terapêutico.
O segmento de edição microbiana baseado em CRISPR é esperado para testemunhar a taxa de crescimento mais rápida de 25,9% de 2026 a 2033, alimentado pelo crescente interesse na engenharia de precisão de cepas microbianas para aplicações terapêuticas. A tecnologia CRISPR permite a modificação altamente direcionada dos genomas microbianos, permitindo que os pesquisadores melhorem as funções microbianas benéficas e suprimem as vias associadas à doença. Essa tecnologia vem ganhando força substancial na oncologia, no tratamento de doenças metabólicas e no manejo de distúrbios inflamatórios devido à sua precisão e escalabilidade. Aumentar os investimentos em tecnologias de edição de genes e startups de biologia sintética estão apoiando esforços rápidos de pesquisa e comercialização. Institutos acadêmicos e empresas de biotecnologia estão ativamente explorando probióticos de engenharia CRISPR capazes de fornecer moléculas terapêuticas diretamente dentro do corpo humano
- Por Usuário Final
Com base no usuário final, o mercado é segmentado em hospitais, clínicas especializadas, institutos de pesquisa, empresas farmacêuticas, empresas de biotecnologia e laboratórios acadêmicos e clínicos. O segmento de empresas de biotecnologia dominou o mercado com a maior quota de receita de mercado de 29,8% em 2025, impulsionada pelo seu papel de liderança na descoberta de drogas microbiológicas, desenvolvimento de terapia microbiana e execução de ensaios clínicos. As empresas de biotecnologia são altamente ativas no desenvolvimento de produtos bioterapêuticos vivos, terapias microbianas sintéticas e plataformas de modulação de microbiomas de precisão. Fortes financiamentos de capital de risco e colaborações estratégicas com empresas farmacêuticas estão permitindo uma rápida expansão de gasodutos focados em microbiomas. Essas empresas estão cada vez mais investindo em tecnologias avançadas de sequenciamento, biologia sintética e ferramentas de análise de microbiomas guiados por IA para acelerar a inovação terapêutica. A crescente procura de medicamentos personalizados e de terapias biológicas específicas reforçou ainda mais a posição das empresas de biotecnologia neste mercado
Espera-se que o segmento de institutos de pesquisa testemunhe a taxa de crescimento mais rápida de 23,6% de 2026 a 2033, alimentada pelo crescente interesse científico em interações microbiome-hospedeiro e mecanismos microbianos específicos da doença. Governos e organizações privadas estão aumentando significativamente o financiamento para pesquisas acadêmicas e estudos translacionais relacionados ao microbioma. Os institutos de pesquisa desempenham um papel crucial na descoberta de novas cepas microbianas, biomarcadores terapêuticos e assinaturas de microbiomas associados à doença. A colaboração entre instituições acadêmicas, hospitais e empresas de biotecnologia está acelerando a transição das descobertas de microbiomas da pesquisa laboratorial para a aplicação clínica. Expandir o acesso a tecnologias de sequenciamento de alto desempenho e plataformas avançadas de análise computacional está melhorando ainda mais a eficiência da pesquisa e a qualidade da saída.
Microbiome Engenharia Terapêutica Mercado Análise Regional
- A América do Norte dominou o mercado de terapias de engenharia de microbiomas com a maior parcela de receita de 41,8% em 2025, apoiada por forte infraestrutura de biotecnologia, adoção precoce de terapias avançadas, financiamento significativo de P&D e presença de principais atores do setor e instituições de pesquisa clínica.
- Os profissionais de saúde e as instituições de pesquisa da região priorizam altamente a terapêutica avançada do microbioma para aplicações gastrintestinais, metabólicas e oncológicas devido à crescente validação clínica, suporte regulatório favorável e sensibilização para o manejo da doença associada ao microbioma.
- Esta ampla liderança de mercado é ainda apoiada pela presença de empresas líderes em biotecnologia, alto gasto com saúde, sequenciamento avançado e capacidades de biologia sintética, e aumento da atividade de ensaios clínicos, estabelecendo a terapia de engenharia de microbiomas como um segmento rapidamente emergente na indústria biofarmacêutico norte-americana
U.S. Microbiome Engenharia Terapêutica Mercado Insight
O mercado de engenharia terapêutica de microbiomas dos EUA capturou a maior parcela de receita de 79% em 2025 na América do Norte, alimentada por forte inovação em biotecnologia, aumento de ensaios clínicos e ampliação da adoção de abordagens de medicina de precisão. Os profissionais de saúde e as empresas de biotecnologia estão cada vez mais focados em terapias baseadas em microbiomas para distúrbios gastrointestinais, oncologia e doenças metabólicas. A presença crescente de tecnologias avançadas de sequenciamento, plataformas de biologia sintética e suporte da FDA para produtos bioterapêuticos vivos impulsiona ainda mais a indústria de engenharia de microbiomas. Além disso, o aumento dos investimentos das empresas farmacêuticas e o aumento das colaborações entre instituições acadêmicas e empresas de biotecnologia contribuem significativamente para a expansão do mercado.
Europa Microbiome Engenharia Terapêutica Mercado Insight
Prevê-se que o mercado europeu da engenharia terapêutica de microbiomas se expanda num CAGR substancial ao longo do período de previsão, impulsionado principalmente pelo aumento das actividades de investigação relacionadas com microbiomas e pelo crescente enfoque nas soluções de cuidados de saúde de precisão. A crescente prevalência de doenças gastrointestinais e autoimunes crônicas, aliada ao forte apoio governamental à inovação em biotecnologia, está promovendo a adoção de terapias com microbiomas. Os sistemas de saúde europeus também enfatizam terapias biológicas avançadas e abordagens médicas personalizadas para melhorar os resultados do tratamento. A região está experimentando um crescimento significativo na pesquisa clínica e no desenvolvimento de medicamentos baseados em microbiomas, com empresas farmacêuticas investindo cada vez mais em produtos bioterapêuticos vivos e terapias microbianas projetadas.
U.K. Microbiome Engenharia Terapêutica Introdução ao Mercado
O mercado de terapias de engenharia de microbiomas do Reino Unido está previsto para crescer em um CAGR notável durante o período de previsão, impulsionado pelo aumento dos investimentos em pesquisa de microbiomas e crescente demanda por terapias biológicas direcionadas. Além disso, a crescente conscientização sobre a microbiota intestinal e sua associação com doenças crônicas está incentivando os profissionais de saúde e institutos de pesquisa a adotar abordagens de tratamento baseadas em microbiomas. Espera-se que o forte ecossistema biotecnológico do Reino Unido, juntamente com sua infraestrutura de pesquisa clínica em expansão, continue estimulando o crescimento do mercado. Além disso, parcerias entre universidades, hospitais e empresas de biotecnologia estão acelerando a inovação em engenharia terapêutica microbiana e aplicações de medicina de precisão.
Alemanha Microbiome Engenharia Terapêutica Introdução ao Mercado
Espera-se que o mercado de terapias de engenharia de microbiomas da Alemanha se expanda em um considerável CAGR durante o período de previsão, alimentado pela adoção crescente de soluções avançadas de biotecnologia e foco crescente na medicina de precisão orientada por microbiomas. A bem estabelecida infraestrutura farmacêutica e de pesquisa da Alemanha, aliada à ênfase na inovação científica e na modernização da saúde, promove o desenvolvimento de terapias baseadas em microbiomas. A crescente integração da biologia sintética, análise de microbiomas orientada por IA e tecnologias de sequenciamento metagenômico também está apoiando a expansão do mercado. Além disso, o aumento dos investimentos em terapias oncológicas e de microbiomas gastrintestinais está se alinhando com o forte foco do país em pesquisas clínicas avançadas e soluções personalizadas de saúde.
Asia-Pacific Microbiome Engenharia Terapêutica Introdução ao Mercado
O mercado de engenharia terapêutica de microbiomas Ásia-Pacífico está preparado para crescer no CAGR mais rápido de 24,3% durante o período de previsão de 2026 a 2033, impulsionado pelo aumento dos investimentos em saúde, ampliação das capacidades de biotecnologia e aumento da prevalência de doenças crônicas em países como China, Japão e Índia. O crescente foco da região na medicina de precisão e na biologia avançada, apoiado por iniciativas governamentais que promovem a inovação em biotecnologia, está impulsionando a adoção de terapias de engenharia de microbiomas. Além disso, como o APAC surge como um dos principais centros para a fabricação farmacêutica e pesquisa clínica, a acessibilidade às terapias baseadas em microbiomas está se expandindo em uma população de pacientes mais ampla.
Japão Microbiome Engenharia Terapêutica Introdução ao Mercado
O mercado de engenharia terapêutica de microbiomas do Japão está ganhando impulso devido à infraestrutura avançada de saúde do país, fortes capacidades de pesquisa em biotecnologia e crescente demanda por soluções terapêuticas inovadoras. O mercado japonês coloca ênfase significativa na medicina de precisão e cuidados de saúde preventivos, e a adoção da terapêutica do microbioma é impulsionada pelo aumento da pesquisa sobre distúrbios gastrointestinais e metabólicos. A integração de tecnologias de sequenciamento de microbiomas, análises baseadas em IA e plataformas de biologia sintética está alimentando o crescimento do mercado. Além disso, o envelhecimento da população do Japão provavelmente estimulará a demanda por terapias avançadas baseadas em microbiomas com o objetivo de melhorar a saúde digestiva, a imunidade e o manejo de doenças crônicas em ambientes hospitalares e de cuidados especializados.
Índia Microbiome Engenharia Terapêutica Introdução ao Mercado
O mercado de engenharia terapêutica de microbiomas da Índia representou a maior participação de receita de mercado na Ásia Pacífico em 2025, atribuída ao setor de biotecnologia em expansão do país, aumento da conscientização sobre saúde e rápido crescimento na adoção de medicina de precisão. A Índia é um dos mercados emergentes de pesquisa e desenvolvimento biológico de microbiomas, com o aumento da tração no tratamento de doenças gastrointestinais e metabólicas. O impulso para a inovação da biotecnologia, juntamente com investimentos crescentes de empresas farmacêuticas nacionais e internacionais, está apoiando a expansão do mercado na Índia. Além disso, o aumento das atividades de pesquisa clínica e a crescente disponibilidade de tecnologias avançadas de sequenciamento são fatores fundamentais para impulsionar o mercado terapêutico de engenharia de microbiomas no país.
Microbiome Engenharia Terapêutica Market Share
A indústria de Microbiome Engineering Therapeutics é liderada principalmente por empresas bem estabelecidas, incluindo:
- Seres Therapeutics, Inc. (EUA)
- MaaT Pharma (França)
- Vedanta Biosciences, Inc. (EUA)
- Finch Therapeutics Group, Inc. (EUA)
- EnteroBiotix Limited (U.K.)
- Microbiotica Ltd (U.K.)
- Siolta Therapeutics, Inc. (EUA)
- Segundo Genoma, Inc. (EUA)
- Axial Therapeutics, Inc. (EUA)
- Pendulum Therapeutics, Inc. (EUA)
- Assembly Biosciences, Inc. (EUA)
- SNIPR Biome ApS (Dinamarca)
- BiomX Inc. (Israel)
- Synlogic Operating Company, Inc. (EUA)
- AOBIIOME LLC (EUA)
- Evelo Biosciences, Inc. (EUA)
- Locus Biosciences, Inc. (EUA)
- A Akkermansia Company NV (Bélgica)
- 4D pharma plc (U.K.)
- Seed Health, Inc. (EUA)
Quais são os recentes desenvolvimentos no mercado global de terapias de engenharia de microbiomas
- Em Junho de 2025, a MaaT Pharma anunciou a apresentação de um pedido de autorização de introdução no mercado (MAA) à Agência Europeia de Medicamentos (EMA) para o Xervyteg (MaaT013), uma terapêutica baseada em microbiomas para doença aguda do enxerto contra o hospedeiro (aGvHD). A terapia demonstrou fortes resultados de eficácia de Fase 3 e poderia se tornar a primeira terapêutica aprovada com microbiota em hemato-oncologia na Europa. Este desenvolvimento destaca a crescente maturidade regulatória e comercial da engenharia terapêutica do microbioma em aplicações oncológicas
- Em janeiro de 2025, MaaT Pharma relatou resultados positivos dos ensaios clínicos de Fase 3 de MaaT013 em doença gastrointestinal aguda do enxerto contra hospedeiro, atingindo uma taxa de resposta global gastrointestinal de 62% no Dia 28. Os achados reforçaram o potencial terapêutico das terapias ecossistêmicas de microbiomas em distúrbios imunomediados e fortaleceram a confiança em plataformas de medicina de precisão baseadas em microbiomas
- Em maio de 2025, a Ferring Pharmaceuticals apresentou achados iniciais do estudo de Fase 3b CDI-SCOPE avaliando REBIOTA administrado por colonoscopia para infecção difficile Clostridioides recorrente. O estudo teve como foco a segurança e a efetividade do mundo real, apoiando a adoção clínica mais ampla de produtos bioterapêuticos vivos baseados em microbiomas no manejo de doenças gastrointestinais
- Em setembro de 2024, Nexilico colaborou com a Siolta Therapeutics para desenvolver produtos bioterapêuticos vivos de precisão com microbioma visando enterocolite necrosante (NEC). A parceria combinou a análise de microbiomas guiados por IA com terapia microbiana projetada para avançar estratégias de tratamento direcionadas para as condições de saúde infantil
- Em junho de 2024, a Nestlé Health Science adquiriu direitos globais ao VOWST da Seres Therapeutics fortalecendo sua posição na indústria de microbiomas terapêuticos. A aquisição permitiu à Nestlé Health Science ampliar a capacidade de comercialização e fabricação de terapias baseadas em microbiomas visando infecção recorrente por C. difficile e distúrbios gastrointestinais relacionados.
SKU-
Obtenha acesso online ao relatório sobre a primeira nuvem de inteligência de mercado do mundo
- Painel interativo de análise de dados
- Painel de análise da empresa para oportunidades de elevado potencial de crescimento
- Acesso de analista de pesquisa para personalização e customização. consultas
- Análise da concorrência com painel interativo
- Últimas notícias, atualizações e atualizações Análise de tendências
- Aproveite o poder da análise de benchmark para um rastreio abrangente da concorrência
Metodologia de Investigação
A recolha de dados e a análise do ano base são feitas através de módulos de recolha de dados com amostras grandes. A etapa inclui a obtenção de informações de mercado ou dados relacionados através de diversas fontes e estratégias. Inclui examinar e planear antecipadamente todos os dados adquiridos no passado. Da mesma forma, envolve o exame de inconsistências de informação observadas em diferentes fontes de informação. Os dados de mercado são analisados e estimados utilizando modelos estatísticos e coerentes de mercado. Além disso, a análise da quota de mercado e a análise das principais tendências são os principais fatores de sucesso no relatório de mercado. Para saber mais, solicite uma chamada de analista ou abra a sua consulta.
A principal metodologia de investigação utilizada pela equipa de investigação do DBMR é a triangulação de dados que envolve a mineração de dados, a análise do impacto das variáveis de dados no mercado e a validação primária (especialista do setor). Os modelos de dados incluem grelha de posicionamento de fornecedores, análise da linha de tempo do mercado, visão geral e guia de mercado, grelha de posicionamento da empresa, análise de patentes, análise de preços, análise da quota de mercado da empresa, normas de medição, análise global versus regional e de participação dos fornecedores. Para saber mais sobre a metodologia de investigação, faça uma consulta para falar com os nossos especialistas do setor.
Personalização disponível
A Data Bridge Market Research é líder em investigação formativa avançada. Orgulhamo-nos de servir os nossos clientes novos e existentes com dados e análises que correspondem e atendem aos seus objetivos. O relatório pode ser personalizado para incluir análise de tendências de preços de marcas-alvo, compreensão do mercado para países adicionais (solicite a lista de países), dados de resultados de ensaios clínicos, revisão de literatura, mercado remodelado e análise de base de produtos . A análise de mercado dos concorrentes-alvo pode ser analisada desde análises baseadas em tecnologia até estratégias de carteira de mercado. Podemos adicionar quantos concorrentes necessitar de dados no formato e estilo de dados que procura. A nossa equipa de analistas também pode fornecer dados em tabelas dinâmicas de ficheiros Excel em bruto (livro de factos) ou pode ajudá-lo a criar apresentações a partir dos conjuntos de dados disponíveis no relatório.