North America Liver Cancer Diagnostics Market
Tamanho do mercado em biliões de dólares
CAGR :
% 
 USD
4,031.47 Million
USD
6,707.57 Million
2022
2030
USD
4,031.47 Million
USD
6,707.57 Million
2022
2030
| 2023 –2030 | |
| USD 4,031.47 Million | |
| USD 6,707.57 Million | |
|
|
|
North America Liver Cancer Diagnostics Market, By Test Type (Imaging Test, Biopsy, Genomic Test, and Others), Cancer Stages (Stage 0, Stage I , Stage II, Stage III, and Stage IV), Cancer Type (Primary Liver Cancer and Secondary Liver Cancer), Product (Platform-Based Products, Instrument Based Products, Kits and Reagents, and Other Consumables), Technology (Fluorescent In Situ Hybridization, Next Generation Sequencing, Fluorimmunoassay, Comparative Genomic Hybridization, Immunohistochemical, and Others), Application (Screening, Diagnostic and Predictive, Prognostic, and Research), Gender (Female and Male), End-User (Hospitals, Diagnostic Centers, Cancer Research Centers, Academic Institutes, Ambulatory Surgical Centers, and Others), Distribution Channel (Direct Tender, Retail Sales, and Others) - Industry Trends and Forecast to 2030.
North America Liver Cancer Diagnostics Market Analysis and Insights
North America liver cancer diagnostics market is expected to grow in the forecast year due to the rise in market players and the availability of advanced services. Along with this, manufacturers are engaged in R&D activity for launching novel services in the market. The growing demand for better quality healthcare for liver cancer and rising preference for preventative health checkups is expected to boost the market’s growth.
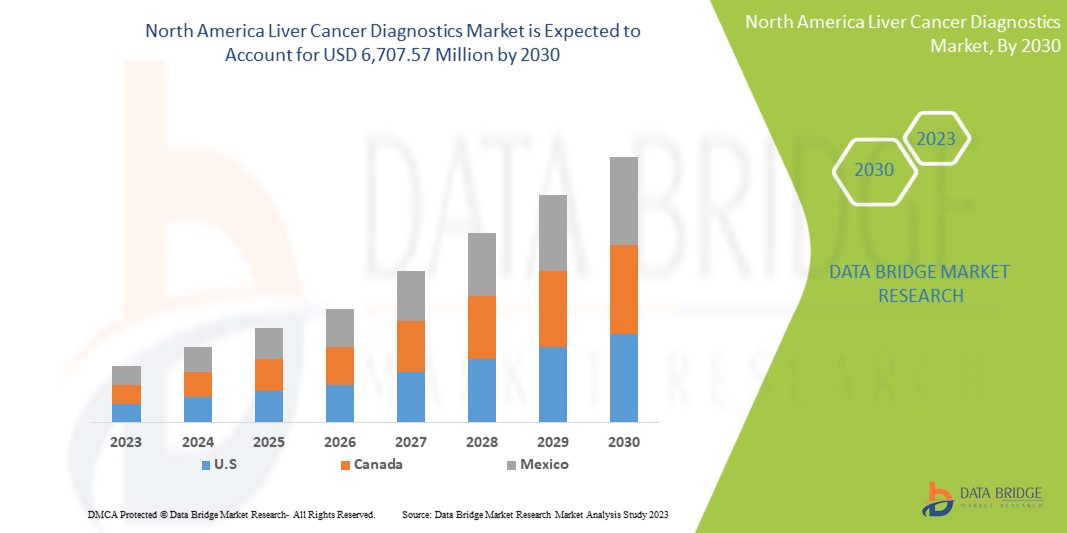
North America liver cancer diagnostics market is expected to gain market growth in the forecast period of 2023 to 2030. Data Bridge Market Research analyses that the market is growing with a CAGR of 6.6% in the forecast period of 2023 to 2030 and is expected to reach USD 6,707.57 Million by 2030 from USD 4,031.47 Million in 2022.
North America liver cancer diagnostics market report provides details of market share, new developments, and product pipeline analysis, the impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, product approvals, strategic decisions, product launches, geographic expansions, and technological innovations in the market. To understand the analysis and the market scenario, contact us for an analyst brief. Our team will help you create a revenue impact solution to achieve your desired goal. The scalability and business expansion of the retail units in the developing countries of various regions and partnership with suppliers for the safe distribution of machine and drug products are the major drivers which propelled the demand of the market in the forecast period.
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015-2020) |
|
Quantitative Units |
Revenue in USD Million |
|
Segments Covered |
By Test Type (Imaging, Biopsy, Genomic Test, and Others), Cancer Stages (Stage 0, Stage I, Stage II, Stage III, and Stage IV), Cancer Type(Primary Liver Cancer and Secondary Liver Cancer), Product (Platform-Based Products, Instrument based products, Kits and Reagents, and Other Consumables), Technology (Fluorescent In Situ Hybridization, Next Generation Sequencing, Fluorimmunoassay, Comparative Genomic Hybridization, Immunohistochemical, and Others), Application (Screening, Diagnostic and Predictive, Prognostic, and Research), Gender (Female and Male), End User (Hospitals, Diagnostic Centers, Cancer Research Centers, Academic Institutes, Ambulatory Surgical Centers, and Others), Distribution Channel (Direct Tender, Retail Sales, and Others) |
|
Countries Covered |
U.S., Canada, Mexico |
|
Market Players Covered |
Siemens Healthcare GmbH (Germany), Koninklijke Philips N.V.(Netherland), Agilent Technologies, Inc. (U.S.), Illumina, Inc. (U.S.), Epigenomics AG (Germany), Thermo Fisher Scientific Inc. (U.S.), QIAGEN(U.S.), F. Hoffmann-La Roche Ltd(Switzerland), Diagnostic Biosystems Inc. (U.S.), FUJIFILM Corporation (Japan), BD(U.S.), MOLGEN (Netherlands), BIOCEPT, INC. (U.S.), Sysmex Corporation (Japan), Elabscience Biotechnology Inc. (U.S.), Hipro Biotechnology Co., Ltd. (China), Altogen Biosystems (U.S.), ABK Biomedical Inc.(Canada), Diazyme Laboratories, Inc. (U.S.), and AB Sciex Pte Ltd. (Subsidiary of Danaher) (U.S.) among others |
Market Definition
Liver cancer diagnostics refers to the various methods and techniques used to detect and diagnose liver cancer. This may include laboratory tests, imaging tests, and physical examinations.
The liver cancer diagnostic market includes a wide range of products and services from various companies, including diagnostic laboratories, medical device manufacturers, and pharmaceutical companies. The market is driven by the increasing incidence of liver cancer and the growing demand for accurate and reliable diagnostic tests.
North America Liver Cancer Diagnostics Market Dynamics
Drivers
-
Unmet need for non-invasive, accurate, and reliable diagnostic tests for earlier cancer detection
Diagnostics is also becoming an increasingly important tool for converting the most recent developments in basic research into better clinical outcomes for patients. The development of novel, quick, sensitive, less invasive, and more accurate molecular diagnostic tests is being accelerated by some of the most exciting scientific developments of the time, including genomics, proteomics, and other "omics" technologies. This significantly impacts the ability to identify and treat different cancers earlier and more precisely. By customizing medicines based on each patient's distinct molecular profile, diagnostics help doctors make better-educated treatment decisions.
Thus, the unmet need for non-invasive, accurate, and reliable diagnostic tests for earlier cancer detection demands is expected to drive the market's growth.
-
Increasing early diagnosis of liver cancer
Machine learning could revolutionize early cancer diagnosis, which trains computers to see patterns in complex data. Tools include assessments of common health data, medical imaging, biopsy samples, and blood tests to help in early diagnosis and risk stratification. In many tumor types, the likelihood of undergoing successful therapy increases with an early cancer diagnosis. One important strategy is to assess patients at risk who do not exhibit symptoms and respond quickly and appropriately to those who do.
Thus, earlier detection improves treatment options, patient outcomes, and survival and is expected to drive the growth of the North America liver cancer diagnostics market.
Restraint
-
Barriers to liver cancer diagnosis and poor prognosis
Cancer is the leading cause of death in developed and developing countries. Cancer mortality is expected to rise to an estimated 13.1 million deaths annually by 2030. However, certain types of cancer have a high chance of being cured if they are detected at an early stage and adequately treated. The delays in cancer diagnosis may occur throughout the diagnostic pathway: patient, primary care, and secondary care. Patient delays may occur when the patient fails to recognize and act on suspicious cancer symptoms. Poor public awareness of early cancer symptoms is considered the predominant reason for delayed presentation, particularly if symptoms are atypical in nature.
The late diagnosis often results from these factors, which result in poor prognosis. Thus, these are expected to restrain the growth of the North America liver cancer diagnostics market.
Opportunity
-
Increasing awareness towards liver cancer
Liver cancer awareness is a chance to increase knowledge about these diseases and to put the spotlight on research into their causes, prevention, diagnosis, treatment, and survival. The objective is to assist persons who have been impacted by the liver and to promote healthy habits. Primary liver cancer is the name for cancer that develops in the liver. Hepatocellular carcinoma is the most prevalent type of primary liver cancer in adults (HCC). The third most common cause of cancer-related fatalities worldwide is this particular form of liver cancer. About 25,000 men and 11,000 women are diagnosed with liver cancer each year in the US, and the illness claims the lives of 19,000 men and 9,000 women.
Thus, government initiatives toward liver cancer diagnostics is expected to create an opportunity for the growth of the market.
Challenge
- Strict regulations for the approval of liver cancer diagnostic products
The strict guidelines for product approval and market commercialization are proving to be a major obstacle for manufacturers of cancer diagnostic products worldwide. Every nation has its own regulations and employs a different regulating body.
The players involved in the production and marketing of medical devices must adjust as a result of the strict regulations. This would affect all stakeholders globally, which is projected to challenge the market throughout the forecast period. Thus, it is expected to act as a challenge for the North America liver cancer diagnostics market to hamper its growth.
Recent Development
- In December 2022, FUJIFILM Holdings America Corporation announced the company has a purchase agreement asset with Inspirata, Inc. to acquire a digital pathology business to expand its robust enterprise imaging offering. This results in enabling the integration of pathology images and data into a healthcare organization’s electronic health record system to streamline care delivery for oncology patients
North America Liver Cancer Diagnostics Market Scope
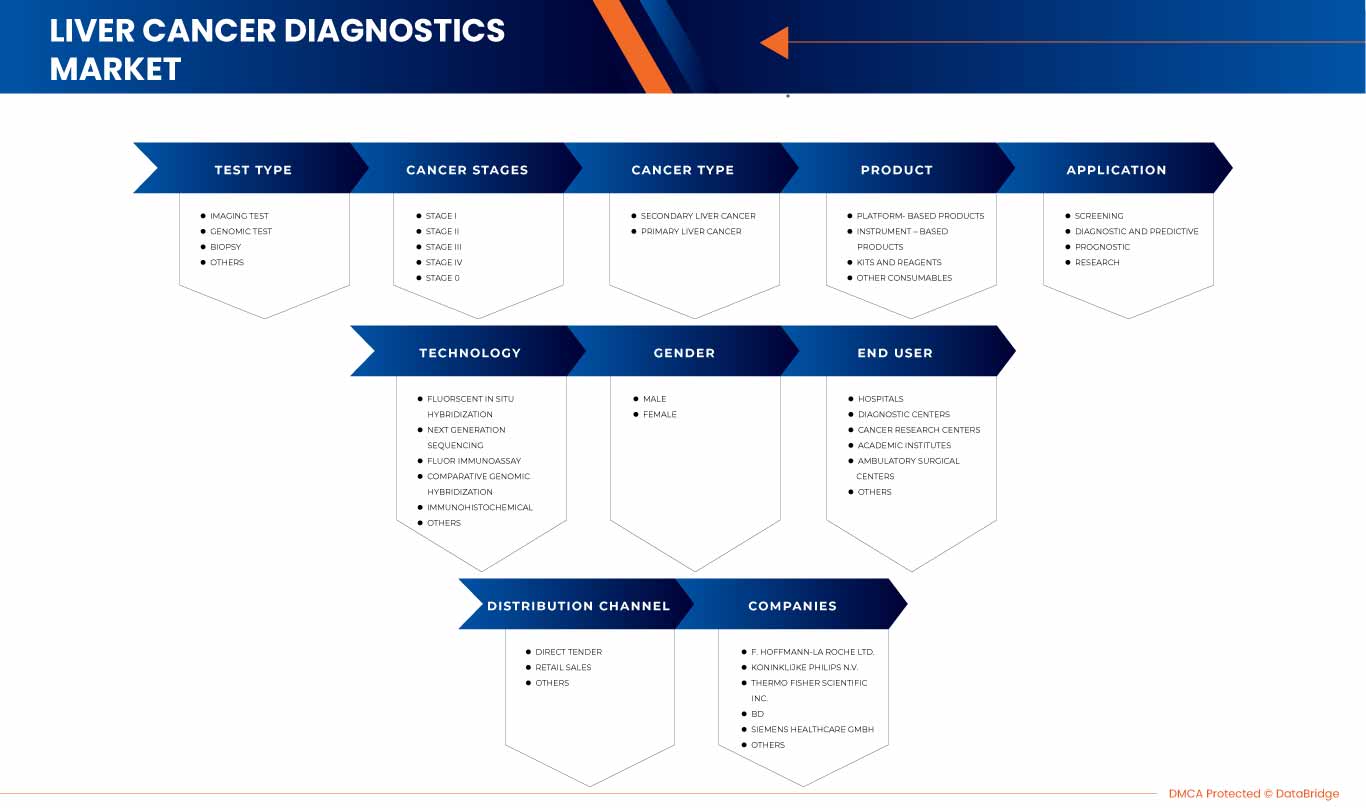
North America liver cancer diagnostics market is categorized into nine notable segments, which are test type, cancer type, cancer stages, product, application, technology, gender, end-user, and distribution channels.
By Test Type
- Imaging Test
- Biopsy
- Genomic Test
- Others
By Cancer Stages
- Stage 0
- Stage I
- Stage II
- Stage III
- Stage IV
By Cancer Type
- Primary Liver Cancer
- Secondary Liver Cancer
By Product
- Platform-Based Products
- Instrument-Based Products
- Kits and Reagents
- Other Consumables
By Technology
- Fluorescent in Situ Hybridization
- Next Generation Sequencing
- Fluoroimmunoassay
- Comparative Genomic Hybridization
- Immunohistochemical
- Others
By Application
- Screening
- Diagnostic and Predictive
- Prognostic
- Research
By Gender
- Female
- Male
By End-User
- Hospitals
- Cancer Research Centers
- Academic Institutes
- Diagnostic Centers
- Ambulatory Surgical Centers
- Others
By Distribution Channel
- Direct Tenders
- Retail Sales
- Others
North America Liver Cancer Diagnostics Market Regional Analysis/Insights
North America liver cancer diagnostics market is segmented into nine notable segments based on test type, cancer stages, cancer type, product, application, technology, gender, end-user, and distribution channel.
The countries covered in this market report are the U.S., Canada, and Mexico.
The U.S. is dominating due to the presence of key market players in the largest consumer market with high GDP.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impact the current and future trends of the market. Data points such as new sales, replacement sales, country demographics, regulatory acts, and import-export tariffs are some of the major pointers used to forecast the market scenario for individual countries. Also, the presence and availability of North America brands and their challenges faced due to large or scarce competition from local and domestic brands, and impact of sales channels are considered while providing forecast analysis of the country data.
Competitive Landscape and North America Liver Cancer Diagnostics Market Share Analysis
North America liver cancer diagnostics market competitive landscape provides details by the competitor. Details included are company overview, company financials, revenue generated, market potential, investment in R&D, new market initiatives, production sites and facilities, company strengths and weaknesses, product launch, product trials pipelines, product approvals, product width and breath, application dominance, technology lifeline curve. The above data points provided are only related to the company’s focus on the North America Liver cancer diagnostics market.
Some of the major players operating in the North America liver cancer diagnostics market are Siemens Healthcare GmbH (Germany), Koninklijke Philips N.V.(Netherland), Agilent Technologies, Inc. (U.S.), Illumina, Inc. (U.S.), Epigenomics AG (Germany), Thermo Fisher Scientific Inc. (U.S.), QIAGEN(U.S.), F. Hoffmann-La Roche Ltd(Switzerland), Diagnostic Biosystems Inc. (U.S.), FUJIFILM Corporation (Japan), BD(U.S.), MOLGEN (Netherlands), BIOCEPT, INC. (U.S.), Sysmex Corporation (Japan), Elabscience Biotechnology Inc. (U.S.), Hipro Biotechnology Co., Ltd. (China), Altogen Biosystems (U.S.), ABK Biomedical Inc.(Canada), Diazyme Laboratories, Inc. (U.S.), AB Sciex Pte Ltd. (Subsidary of Danaher) (U.S.) among others.
SKU-
Obtenha acesso online ao relatório sobre a primeira nuvem de inteligência de mercado do mundo
- Painel interativo de análise de dados
- Painel de análise da empresa para oportunidades de elevado potencial de crescimento
- Acesso de analista de pesquisa para personalização e customização. consultas
- Análise da concorrência com painel interativo
- Últimas notícias, atualizações e atualizações Análise de tendências
- Aproveite o poder da análise de benchmark para um rastreio abrangente da concorrência
Metodologia de Investigação
A recolha de dados e a análise do ano base são feitas através de módulos de recolha de dados com amostras grandes. A etapa inclui a obtenção de informações de mercado ou dados relacionados através de diversas fontes e estratégias. Inclui examinar e planear antecipadamente todos os dados adquiridos no passado. Da mesma forma, envolve o exame de inconsistências de informação observadas em diferentes fontes de informação. Os dados de mercado são analisados e estimados utilizando modelos estatísticos e coerentes de mercado. Além disso, a análise da quota de mercado e a análise das principais tendências são os principais fatores de sucesso no relatório de mercado. Para saber mais, solicite uma chamada de analista ou abra a sua consulta.
A principal metodologia de investigação utilizada pela equipa de investigação do DBMR é a triangulação de dados que envolve a mineração de dados, a análise do impacto das variáveis de dados no mercado e a validação primária (especialista do setor). Os modelos de dados incluem grelha de posicionamento de fornecedores, análise da linha de tempo do mercado, visão geral e guia de mercado, grelha de posicionamento da empresa, análise de patentes, análise de preços, análise da quota de mercado da empresa, normas de medição, análise global versus regional e de participação dos fornecedores. Para saber mais sobre a metodologia de investigação, faça uma consulta para falar com os nossos especialistas do setor.
Personalização disponível
A Data Bridge Market Research é líder em investigação formativa avançada. Orgulhamo-nos de servir os nossos clientes novos e existentes com dados e análises que correspondem e atendem aos seus objetivos. O relatório pode ser personalizado para incluir análise de tendências de preços de marcas-alvo, compreensão do mercado para países adicionais (solicite a lista de países), dados de resultados de ensaios clínicos, revisão de literatura, mercado remodelado e análise de base de produtos . A análise de mercado dos concorrentes-alvo pode ser analisada desde análises baseadas em tecnologia até estratégias de carteira de mercado. Podemos adicionar quantos concorrentes necessitar de dados no formato e estilo de dados que procura. A nossa equipa de analistas também pode fornecer dados em tabelas dinâmicas de ficheiros Excel em bruto (livro de factos) ou pode ajudá-lo a criar apresentações a partir dos conjuntos de dados disponíveis no relatório.