Europe Mycoplasma Testing In Clinical Market
市场规模(十亿美元)
CAGR :
% 
 USD
52.52 million
USD
106.75 million
2022
2030
USD
52.52 million
USD
106.75 million
2022
2030
| 2023 –2030 | |
| USD 52.52 million | |
| USD 106.75 million | |
|
|
|
Europe Mycoplasma Testing in Clinical Market, By Products (Kits and Reagents, Instruments, Services), Technique (Microbial Culture Techniques/Direct Assay, Polymerase Chain Reaction, ELISA, DNA Staining/Indirect Assay, Enzymatic Methods), Application (Cell Line Testing, Virus Testing, End of Production Cells Testing, Others), Disease Area (Respiratory, Urogenital, Gastrointestinal, Musculoskeletal, Cardiovascular, Others), End User (Diagnostic Laboratories, Hospitals) – Industry Trends and Forecast to 2030.
Europe Mycoplasma Testing in Clinical Market Analysis and Size
A study released in May 2022 found a connection between the quantity of mycoplasma tests and COVID-19-related non-pharmaceutical interventions (NPIs). The incidence of M. pneumonia following the adoption of COVID-19 NPIs was examined in this study. This study revealed a decline in M. pneumoniae detection throughout the epidemic. Globally, this decrease was noticed between March 2020 and March 2021. This decrease is mostly attributable to the limited spread of M. pneumonia, which has led to a sharp decline in M. pneumoniae infections worldwide.
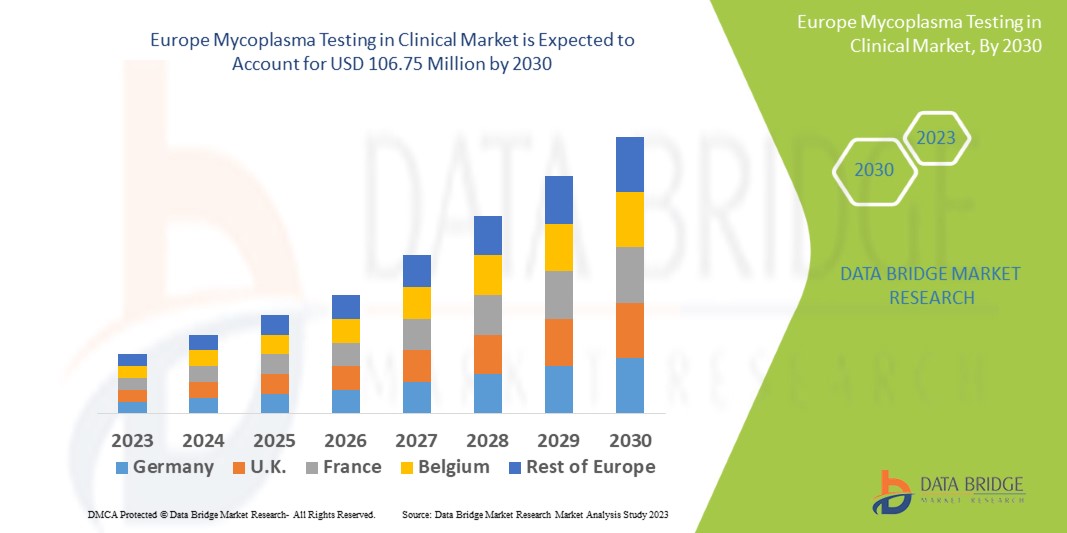
Data Bridge Market Research analyses that the mycoplasma testing in clinical market which was USD 52.52 million in 2022, is expected to reach USD 106.75 million by 2030, at a CAGR of 8.2% during the forecast period 2023 to 2030. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
Europe Mycoplasma Testing in Clinical Market Scope and Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015 - 2020) |
|
Quantitative Units |
Revenue in USD Million, Volumes in Units, Pricing in USD |
|
Segments Covered |
Products (Kits and Reagents, Instruments, Services), Technique (Microbial Culture Techniques/Direct Assay, Polymerase Chain Reaction, ELISA, DNA Staining/Indirect Assay, Enzymatic Methods), Application (Cell Line Testing, Virus Testing, End of Production Cells Testing, Others), Disease Area (Respiratory, Urogenital, Gastrointestinal, Musculoskeletal, Cardiovascular, Others), End User (Diagnostic Laboratories, Hospitals) |
|
Countries Covered |
Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe |
|
Market Players Covered |
AB ANALITICA s.r.l. (Italy), BIOMÉRIEUX (France), ELITechGroup (France), Liofilchem S.r.l. (Italy), Agilent Technologies, Inc. (U.S.), PromoCell GmbH (Germany), F. Hoffmann-La Roche Ltd (Switzerland), OSANG Healthcare (South Korea), Sacace Biotechnologies Srl (Italy), Lonza (Switzerland), Merck KGaA (Germany), Seegene Inc. (South Korea), Clongen Laboratories, LLC (U.S.), Bio-Rad Laboratories, Inc. (U.S.), Charles River Laboratories (U.S.), Bionique Testing Laboratories LLC (U.S.) and ZEAKON Diagnostics (India) |
|
Market Opportunities |
|
Market Definition
Mycoplasma is a form of bacterial infection that can be found through the use of a mycoplasma test. There are various mycoplasma infections, testing can be carried out in a number of methods depending on which one is suspected. M. pneumoniae, which causes upper respiratory infections and can be detected with a blood sample, is the most prevalent Mycoplasma. A vaginal swab, joint fluid, bodily fluids, tissue samples and other sample types may need to be collected for further mycoplasma testing such as sputum.
Europe Mycoplasma Testing in Clinical Market Dynamics
Drivers
- Innovative approaches and substantial investments
The mycoplasma testing in clinical market is receiving more attention to cutting-edge strategies and large investments from active competitors. For instance, seven of the top ten best-selling medications in the first half of 2020 were biologics. This exemplifies the rise of biologics on the international market. A variety of biologic products, including monoclonal antibodies, viral vaccines and cytokines, which are utilized for medicinal or therapeutic purposes, are produced using cell lines. Cell line metabolism, physiology, and gene expression are significantly impacted by Mycoplasma strains. Therefore, it's important to monitor mycoplasma contamination in the manufacturing of biopharmaceuticals.
- Growing R&D investments
The adoption of highly effective mycoplasma testing technologies will certainly be encouraged by rising research expenditures and rising R&D investments by major corporations. For instance, Bristol-Myers Squibb started spending money on creating experimental drugs to help clinical trials. Furthermore, it is essential to carry out safety testing to make sure that a product meets the requirements for efficacy, safety and general public health. This is because of the emergence of biosimilars and the lucrative R&D investment that followed. In December 2020, Merck KGaA increased the size of its American manufacturing footprint by investing more than USD 45.6 million (EUR 40 million) in its Massachusetts and New Hampshire production sites. The facilities were created to increase the company's capability for production while producing a range of biopharmaceutical manufacturing products. These are the factors which further propel the market growth.
Opportunities
- Globalization of clinical trials
ELISA technology is predicted to achieve a sizable market share throughout the forecast period because it allows for simple detection using labelled probes or antibodies for mycoplasma detection. In some cases, PCR and ELISA tests are combined to create PCR-ELISA, a photometric enzyme-based immunoassay that makes it easier to find mycoplasma DNA that has been amplified by PCR in samples. The segment growth will be fuelled by factors such as functional advantages, cost effectiveness, and the capacity to identify a wide variety of strains during the forecast period.
Restraints/Challenges
- High cost of instruments
The high cost of instruments, as well as the lengthy and laborious detection process, are expected to limit the growth of the mycoplasma testing in clinical market during the forecast period. One of the most frequent causes of both paediatric and adult community-acquired pneumonia is Mycoplasma pneumoniae.
This mycoplasma testing in clinical market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the mycoplasma testing in clinical market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
Recent Developments
• In 2020, Liofilchem S.r.l. introduced the enhanced AF Genital System, VTM, Lockable contact plates with microbiological testing. The company's revenue increased because to this product upgrade.
• In 2020, BioMérieux SA announced the introduction of the biofire mycoplasma test for the detection of mycoplasma in biopharmaceutical products. This strategy helped the business increase the market shares of its product in the biotherapeutics sector.
COVID-19 Impact on the Mycoplasma Testing in Clinical Market
The COVID-19 outbreak has had a detrimental impact on all sectors of the economy and is still having a negative effect. When compared to other industries, the biopharmaceutical industry and adjacent markets have not been considerably affected. A study released in May 2022 found a connection between the quantity of mycoplasma tests and COVID-19-related non-pharmaceutical interventions (NPIs). The incidence of M. pneumonia following the adoption of COVID-19 NPIs was examined in this study. This study revealed a decline in M. pneumoniae detection throughout the epidemic. Globally, this decrease was noticed between March 2020 and March 2021. This decrease is mostly attributable to the limited spread of M. pneumonia, which has led to a sharp decline in M. pneumoniae infections worldwide.
Europe Mycoplasma Testing in Clinical Market Scope
The mycoplasma testing in clinical market is segmented on the basis of products, technique, application, disease area and end user. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Products
- Kits and Reagents
- PCR Assays
- Nucleic Acid Detection Kits
- Stains
- Elimination Kits
- Standards and Controls
- Others
- Instruments
- Services
Technique
- Microbial Culture Techniques/Direct Assay
- Polymerase Chain Reaction
- ELISA
- DNA Staining/Indirect Assay
- Enzymatic Methods
Application
- Cell Line Testing
- Virus Testing
- End of Production Cells Testing
- Others
Disease Area
- Respiratory
- Urogenital
- Gastrointestinal
- Musculoskeletal
- Cardiovascular
- Others
End User
- Diagnostic Laboratories
- Hospitals
Mycoplasma Testing in Clinical Market Regional Analysis/Insights
The mycoplasma testing in clinical market is analyzed and market size insights and trends are provided by country, products, technique, application disease area and end user as referenced above.
The countries covered in the mycoplasma testing in clinical market report are Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe.
Germany is dominating the mycoplasma testing in clinical market owing to the rising introduction of novel technologies by key players, growing demand of effective testing.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Data points like down-stream and upstream value chain analysis, technical trends and porter's five forces analysis, case studies are some of the pointers used to forecast the market scenario for individual countries. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Healthcare Infrastructure Growth Installed base and New Technology Penetration
The mycoplasma testing in clinical market also provides you with detailed market analysis for every country growth in healthcare expenditure for capital equipment, installed base of different kind of products for mycoplasma testing in clinical market, impact of technology using life line curves and changes in healthcare regulatory scenarios and their impact on the mycoplasma testing in clinical market. The data is available for historic period 2011-2021.
Competitive Landscape and Mycoplasma Testing in Clinical Market Share Analysis
The mycoplasma testing in clinical market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to mycoplasma testing in clinical market.
Some of the major players operating in the mycoplasma testing in clinical market are:
- AB ANALITICA s.r.l. (Italy)
- BIOMÉRIEUX (France)
- ELITechGroup (France)
- Liofilchem S.r.l. (Italy)
- Agilent Technologies, Inc. (U.S.)
- PromoCell GmbH (Germany)
- F. Hoffmann-La Roche Ltd (Switzerland)
- OSANG Healthcare (South Korea)
- Sacace Biotechnologies Srl (Italy)
- Lonza (Switzerland)
- Merck KGaA (Germany)
- Seegene Inc. (South Korea)
- Clongen Laboratories, LLC (U.S.)
- Bio-Rad Laboratories, Inc. (U.S.)
- Charles River Laboratories (U.S.)
- Bionique Testing Laboratories LLC (U.S.)
- ZEAKON Diagnostics (India)
SKU-
研究方法
数据收集和基准年分析是使用具有大样本量的数据收集模块完成的。该阶段包括通过各种来源和策略获取市场信息或相关数据。它包括提前检查和规划从过去获得的所有数据。它同样包括检查不同信息源中出现的信息不一致。使用市场统计和连贯模型分析和估计市场数据。此外,市场份额分析和关键趋势分析是市场报告中的主要成功因素。要了解更多信息,请请求分析师致电或下拉您的询问。
DBMR 研究团队使用的关键研究方法是数据三角测量,其中包括数据挖掘、数据变量对市场影响的分析和主要(行业专家)验证。数据模型包括供应商定位网格、市场时间线分析、市场概览和指南、公司定位网格、专利分析、定价分析、公司市场份额分析、测量标准、全球与区域和供应商份额分析。要了解有关研究方法的更多信息,请向我们的行业专家咨询。
可定制
Data Bridge Market Research 是高级形成性研究领域的领导者。我们为向现有和新客户提供符合其目标的数据和分析而感到自豪。报告可定制,包括目标品牌的价格趋势分析、了解其他国家的市场(索取国家列表)、临床试验结果数据、文献综述、翻新市场和产品基础分析。目标竞争对手的市场分析可以从基于技术的分析到市场组合策略进行分析。我们可以按照您所需的格式和数据样式添加您需要的任意数量的竞争对手数据。我们的分析师团队还可以为您提供原始 Excel 文件数据透视表(事实手册)中的数据,或者可以帮助您根据报告中的数据集创建演示文稿。