Europe Prophylaxis Of Organ Rejection Market
Taille du marché en milliards USD
TCAC :
% 
 USD
959.37 Million
USD
1,253.57 Million
2025
2033
USD
959.37 Million
USD
1,253.57 Million
2025
2033
| 2026 –2033 | |
| USD 959.37 Million | |
| USD 1,253.57 Million | |
|
|
|
Marché européen de la prophylaxie du rejet d'organes, par cause (exposition épidémiologique, prophylaxie antibactérienne, prophylaxie contre d'autres agents pathogènes et autres), traitement (immunosuppresseur ambulatoire, immunosuppresseur hospitalier et autres), voie d'administration (orale, intraveineuse), organe (rein, foie, cœur, poumon, autres), type de patient (pédiatrique, adulte), utilisateur final (hôpitaux, cliniques de soins à domicile et autres), canal de distribution (appel d'offres direct, pharmacies et autres), pays (Royaume-Uni, Allemagne, France, Espagne, Italie, Pays-Bas, Suisse, Russie, Belgique, Turquie et reste de l'Europe), tendances de l'industrie et prévisions jusqu'en 2029.
Analyse et perspectives du marché : Marché européen de la prophylaxie du rejet d'organes
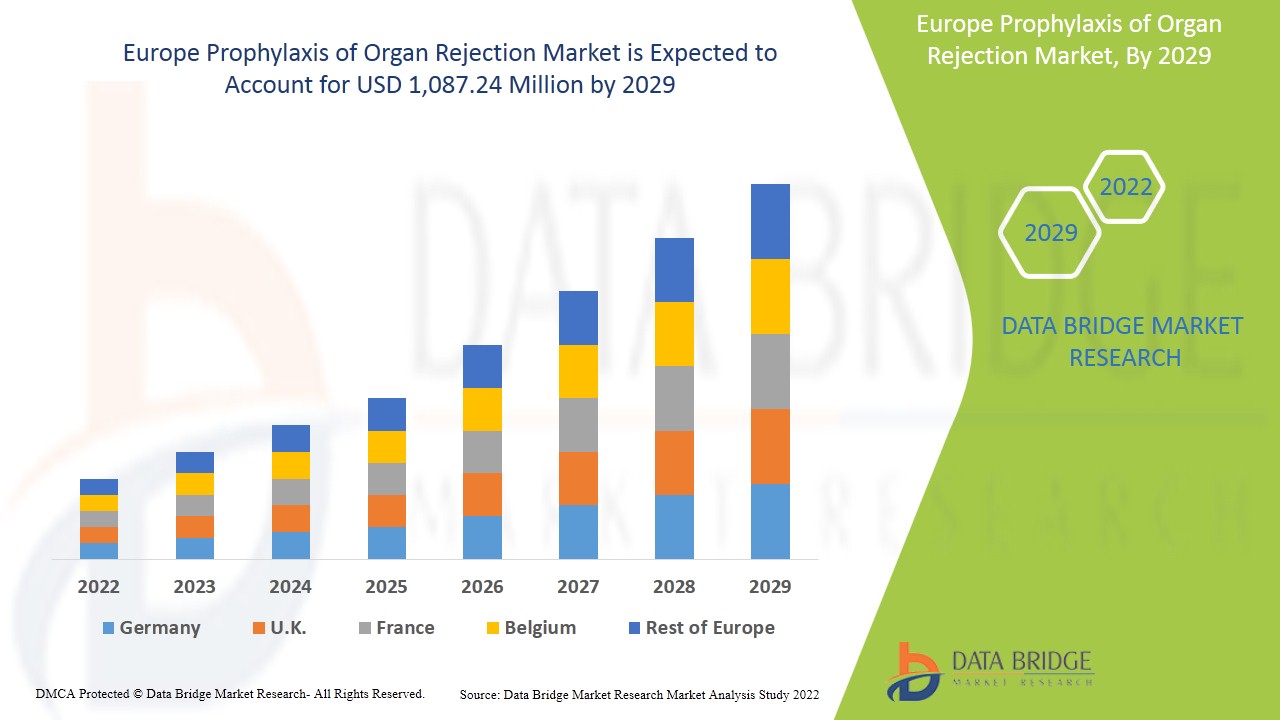
La prophylaxie européenne du rejet d'organes devrait connaître une croissance du marché au cours de la période de prévision de 2021 à 2029. Data Bridge Market Research analyse que le marché européen croît avec un TCAC de 3,4 % au cours de la période de prévision de 2022 à 2029 et devrait atteindre 1 087,24 millions USD d'ici 2029 contre 839,28 millions USD en 2021. La prévalence croissante de la transplantation d'organes et l'utilisation accrue d'immunosuppresseurs seront probablement les principaux moteurs de la demande du marché au cours de la période de prévision.
Le terme prophylaxie désigne un traitement administré ou une action entreprise pour prévenir une maladie. La prophylaxie du rejet d'organe fait référence à la prévention du rejet d'organe par l'utilisation de médicaments. Les patients qui subissent une transplantation doivent être maintenus sous un régime d'immunosuppression pour la prophylaxie du rejet afin de garantir la survie du greffon. Le rejet de greffe est un processus dans lequel l'immunité du receveur de greffe est affaiblie par l'utilisation d' inhibiteurs de la calcineurine , d'inhibiteurs de mTOR, d'agents antimétabolites et de corticostéroïdes. Les agents de traitement améliorent les conséquences à court terme de la transplantation d'organe, mais certaines améliorations sont possibles. Les patients receveurs, qui ont subi une transplantation d'organe solide, doivent prendre des médicaments anti-rejet. En effet, le système immunitaire détruirait l'organe transplanté.
Les facteurs responsables de la croissance du marché nord-américain de la prophylaxie du rejet d'organes sont la prévalence accrue de la transplantation d'organes , l'augmentation des interventions chirurgicales, l'augmentation de la population de receveurs et le lancement de nouveaux produits. Cependant, les facteurs susceptibles de freiner le marché sont l'augmentation du coût de la procédure de transplantation, le manque de sensibilisation à la transplantation d'organes et les risques encourus par le patient lors de la réception de l'organe transplanté.
- D'autre part, les initiatives stratégiques des acteurs du marché, l'augmentation de la recherche et du développement et l'utilisation d'immunosuppresseurs peuvent constituer une opportunité pour la croissance du marché nord-américain de la prophylaxie du rejet d'organes. Le besoin d'expertise qualifiée et l'approbation réglementaire peuvent créer des défis pour le marché nord-américain de la prophylaxie du rejet d'organes. Des développements récents liés au marché nord-américain de la prophylaxie du rejet d'organes ont été observés.
Le rapport sur le marché européen de la prophylaxie du rejet d'organes fournit des détails sur la part de marché, les nouveaux développements et l'analyse du pipeline de produits, l'impact des acteurs du marché national et local, analyse les opportunités en termes de poches de revenus émergentes, les changements dans la réglementation du marché, les approbations de produits, les décisions stratégiques, les lancements de produits, les expansions géographiques et les innovations technologiques sur le marché. Pour comprendre l'analyse et le scénario du marché, contactez-nous pour un briefing d'analyste, notre équipe vous aidera à créer une solution d'impact sur les revenus pour atteindre votre objectif souhaité.
Portée et taille du marché de la prophylaxie du rejet d'organes en Europe
Le marché européen de la prophylaxie du rejet d'organes est analysé et des informations sur la taille du marché sont fournies par cause, traitement, voie d'administration, organe, type de patient, utilisateur final et canal de distribution. La croissance entre les segments vous aide à analyser les niches de croissance et les stratégies pour aborder le marché et déterminer vos principaux domaines d'application et la différence entre vos marchés cibles.
- Sur la base de la cause, le marché nord-américain de la prophylaxie du rejet d'organes est segmenté en exposition épidémiologique, prophylaxie antibactérienne, prophylaxie contre d'autres agents pathogènes et autres. En 2022, le segment de l'exposition épidémiologique devrait dominer le marché nord-américain de la prophylaxie du rejet d'organes en raison de la disponibilité d'informations directes et de la présence d'agents pathogènes liés à la prophylaxie du rejet d'organes aux États-Unis
- En fonction du traitement, le marché nord-américain de la prophylaxie du rejet d'organes est segmenté en immunosuppresseurs ambulatoires, immunosuppresseurs hospitaliers et autres. En 2022, le segment des immunosuppresseurs ambulatoires devrait dominer le marché nord-américain de la prophylaxie du rejet d'organes, car il permet d'obtenir une réponse immunitaire soutenue et spécifique contre les cellules endommagées et la disponibilité de la cyclosporine aux États-Unis et au Canada.
- En Amérique du Nord, le marché de la prophylaxie du rejet d'organes est divisé en deux catégories : orale et intraveineuse, en fonction de la voie d'administration. En 2022, le segment oral devrait dominer le marché de la prophylaxie du rejet d'organes en Amérique du Nord en raison de la disponibilité et de la consommation accrues de comprimés et de gélules oraux.
- Sur la base des organes, le marché nord-américain de la prophylaxie du rejet d'organes est segmenté en rein, foie, cœur, poumon et autres. En 2022, le segment rénal devrait dominer le marché nord-américain de la prophylaxie du rejet d'organes en raison de l'incidence accrue de l'insuffisance rénale et de la présence de politiques de remboursement telles que Medicare, pour l'assurance du receveur.
- En fonction du type de patient, le marché nord-américain de la prophylaxie du rejet d'organes est segmenté en pédiatrie et en adultes. En 2022, le segment des adultes devrait dominer le marché nord-américain de la prophylaxie du rejet d'organes, en raison de la prévalence accrue des maladies chroniques et des conditions génétiques des patients adultes, de la faiblesse du système immunitaire chez les adultes et de la liste d'attente des receveurs adultes supérieure à la liste d'attente des enfants et des femmes au Nouveau-Mexique et aux États-Unis
- En fonction de l'utilisateur final, le marché nord-américain de la prophylaxie du rejet d'organes est segmenté en hôpitaux, cliniques, soins à domicile et autres. En 2022, le segment des hôpitaux devrait dominer le marché nord-américain de la prophylaxie du rejet d'organes en raison de l'augmentation des soins médicaux et de la collaboration avec d'autres hôpitaux en Amérique du Nord pour la livraison des organes des donneurs aux patients receveurs.
- En fonction du canal de distribution, le marché nord-américain de la prophylaxie du rejet d'organes est segmenté en appels d'offres directs, pharmacies et autres. En 2022, le segment des appels d'offres directs devrait dominer le marché nord-américain de la prophylaxie du rejet d'organes en raison de la forte demande d'immunosuppresseurs ambulatoires par les sociétés pharmaceutiques et du paiement garanti, qui devraient dominer le marché.
Analyse du marché de la prophylaxie du rejet d'organes en Europe
Le marché européen de la prophylaxie du rejet d’organes est analysé et des informations sur la taille du marché sont fournies par cause, traitement, voie d’administration, organe, type de patient, utilisateur final et canal de distribution.
Les pays couverts par le rapport sur le marché européen de la prophylaxie du rejet d’organes sont le Royaume-Uni, l’Allemagne, la France, l’Espagne, l’Italie, les Pays-Bas, la Suisse, la Russie, la Belgique, la Turquie et le reste de l’Europe.
L'Europe devrait croître avec le TCAC au cours des périodes prévues, comme dans le cas des activités croissantes de R&D dans le secteur de la pneumologie. L'Allemagne devrait dominer le marché européen. L'Allemagne est l'un des principaux pays en raison de la présence d'acteurs majeurs du marché et des avancées technologiques accrues de la prophylaxie européenne du rejet d'organes dans la région.
La section pays du rapport fournit également des facteurs d'impact sur les marchés individuels et des changements de réglementation sur le marché national qui ont un impact sur les tendances actuelles et futures du marché. Des points de données tels que les nouvelles ventes, les ventes de remplacement, la démographie des pays, les actes réglementaires et les tarifs d'importation et d'exportation sont quelques-uns des principaux indicateurs utilisés pour prévoir le scénario de marché pour les différents pays. En outre, la présence et la disponibilité des marques européennes et les défis auxquels elles sont confrontées en raison de la concurrence importante ou rare des marques locales et nationales, l'impact des canaux de vente sont pris en compte lors de l'analyse prévisionnelle des données nationales.
Les initiatives stratégiques des acteurs du marché pour la prophylaxie du rejet d’organes créent de nouvelles opportunités sur le marché européen de la prophylaxie du rejet d’organes.
Le marché européen de la prophylaxie du rejet d'organes vous fournit également une analyse de marché détaillée pour la croissance de chaque pays dans un secteur particulier avec les ventes de prophylaxie du rejet d'organes en Europe, l'impact des progrès dans la prophylaxie du rejet d'organes en Europe et les changements dans les scénarios réglementaires avec leur soutien au marché européen des particules de prophylaxie du rejet d'organes. Les données sont disponibles pour la période historique de 2020 à 2021.
Analyse du paysage concurrentiel et des parts de marché de la prophylaxie du rejet d'organes en Europe
Le paysage concurrentiel du marché européen de la prophylaxie du rejet d'organes fournit des détails par concurrent. Les détails inclus sont la présentation de l'entreprise, les finances de l'entreprise, les revenus générés, le potentiel du marché, les investissements dans la recherche et le développement, les nouvelles initiatives du marché, les sites et installations de production, les forces et les faiblesses de l'entreprise, le lancement de produits, les pipelines d'essais de produits, les approbations de produits, les brevets, la largeur et l'ampleur du produit, la domination des applications, la courbe de survie technologique. Les points de données ci-dessus fournis ne concernent que l'orientation de l'entreprise par rapport au marché européen de la prophylaxie du rejet d'organes.
Les principales sociétés fournissant la prophylaxie du rejet d'organe en Europe sont Hansa Biopharma, Concord Biotech, Panacea Biotec, WOCKHARDT, SEBELA PHARMACEUTICALS, Accord-UK Ltd, Veloxis Pharmaceuticals, Inc, Astellas Pharma Inc, Novartis AG, Bristol-Myers Squibb Company, Dr Reddy's Laboratories, Ltd, Viatris Inc, Strides Pharma Science Limited, Glenmark, Biocon, Pfizer Inc, Hikma Pharmaceuticals PLC, AbbVie Inc, Amneal Pharmaceuticals LLC, Zydus Pharmaceuticals, Inc., CSL Behring entre autres.
Les initiatives stratégiques des acteurs du marché ainsi que les nouvelles avancées technologiques pour le marché européen de la prophylaxie du rejet d’organes comblent le fossé des médicaments.
Par exemple,
- En novembre 2020, Concord Biotech et Lupin ont reçu l'approbation de la FDA américaine pour les capsules d'immunosuppresseurs génériques. L'approbation reçue augmenterait la vente des capsules d'immunosuppresseurs utilisées dans le rejet d'organes. L'approbation reçue a assuré l'approbation post-commercialisation des capsules d'immunosuppresseurs génériques et leur mise à disposition dans les pharmacies et les magasins de détail.
SKU-
Accédez en ligne au rapport sur le premier cloud mondial de veille économique
- Tableau de bord d'analyse de données interactif
- Tableau de bord d'analyse d'entreprise pour les opportunités à fort potentiel de croissance
- Accès d'analyste de recherche pour la personnalisation et les requêtes
- Analyse de la concurrence avec tableau de bord interactif
- Dernières actualités, mises à jour et analyse des tendances
- Exploitez la puissance de l'analyse comparative pour un suivi complet de la concurrence
Table des matières
1 INTRODUCTION
1.1 OBJECTIFS DE L'ÉTUDE
1.2 DÉFINITION DU MARCHÉ
1.3 APERÇU DU MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES
1.4 MONNAIE ET TARIFS
1.5 LIMITATIONS
1.6 MARCHÉS COUVERTS
2 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : SEGMENTATION
2.1 MARCHÉS COUVERTS
2.2 PORTÉE GÉOGRAPHIQUE
2,3 ANS CONSIDÉRÉS POUR L'ÉTUDE
2.4 MODÈLE DE VALIDATION DES DONNÉES DU TRÉPIED DBMR
2.5 ENTRETIENS PRIMAIRES AVEC DES LEADERS D'OPINION CLÉS
2.6 MODÉLISATION MULTIVARIÉE
2.7 COURBE DE LIGNE DE VIE PAR SEGMENT DE CAUSE
2.8 GRILLE DE POSITIONNEMENT DU MARCHÉ DBMR
2.9 ANALYSE DES PARTS DES FOURNISSEURS
2.1 GRILLE DE COUVERTURE DES UTILISATEURS FINAUX DU MARCHÉ
2.11 SOURCES SECONDAIRES
3 RÉSUMÉ EXÉCUTIF
4 EUROPE PROPHYLAXIE DU REJET D'ORGANE : APERÇU ÉPIDÉMIOLOGIE
5 APERÇU DU MARCHÉ
5.1 PILOTES
5.1.1 AUGMENTATION DE LA PRÉVALENCE DES TRANSPLANTATIONS D'ORGANES
5.1.2 AUGMENTATION DES MALADIES CHRONIQUES
5.1.3 AUGMENTATION DES ESSAIS CLINIQUES
5.1.4 AUGMENTATION DU NOMBRE DE PATIENTS RECEVEURS
5.1.5 APPROBATIONS DE PRODUITS
5.2 RESTRICTIONS
5.2.1 AUGMENTATION DU COÛT DE LA PROCÉDURE DE TRANSPLANTATION
5.2.2 MANQUE DE SENSIBILISATION À LA TRANSPLANTATION D'ORGANES
5.2.3 RISQUES INCLUS LORSQUE LE PATIENT REÇOIT UN ORGANE TRANSPLANTÉ
5.2.4 QUESTIONS ÉTHIQUES LIÉES À LA TRANSPLANTATION D'ORGANES
5.3 OPPORTUNITÉS
5.3.1 AUGMENTATION DE LA RECHERCHE ET DES DÉVELOPPEMENTS
5.3.2 INITIATIVES STRATÉGIQUES DES ACTEURS DU MARCHÉ
5.3.3 AUGMENTATION DE L'UTILISATION DES IMMUNOSUPPRESSEURS
5.4 DÉFIS
5.4.1 PÉNURIE DE PROFESSIONNELS QUALIFIÉS REQUIS POUR L'UTILISATION DE LA PROPHYLAXIE DU REJET D'ORGANES
5.4.2 RÉGLEMENTATIONS STRICTES
6 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR CAUSE
6.1 APERÇU
6.2 EXPOSITION ÉPIDÉMIOLOGIQUE
6.2.1 EXPOSITION À L'HÔPITAL
6.2.2 EXPOSITION COMMUNAUTAIRE
6.3 PROPHYLAXIE ANTIBACTÉRIENNE
6.3.1 PROPHYLAXIE POST-TRANSPLANTE
6.3.2 PROPHYLAXIE ANTIBACTÉRIENNE PÉRIPHÉRIQUE
6.4 PROPHYLAXIE CONTRE D'AUTRES AGENTS PATHOGÈNES
6.5 AUTRES
7 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR TRAITEMENT
7.1 APERÇU
7.2 IMMUNOSUPPRESSEUR EN CONSULTATION EXTERNE
7.2.1 INHIBITEURS DE LA CALCINEURINE
7.2.1.1 CYCLOSPORINE
7.2.1.2 TACROLIMUS
7.2.1.2.1 DIFFUSION IMMÉDIATE
7.2.1.2.2 VERSION PROLONGÉE
7.2.2 CORTICOSTÉROÏDES
7.2.2.1 MÉTHYLPREDNISOLONE
7.2.2.2 PREDNISONE
7.2.2.3 AUTRES
7.2.3 ACIDES MYCOPHÉNOLIQUES
7.2.3.1 MYCOPHÉNOLATE MOFÉTIL
7.2.3.2 MYCOPHÉNOLATE DE SODIUM
7.2.4 INHIBITEURS DE MTOR
7.2.4.1 SIROLIMUS
7.2.4.2 ÉVÉROLIMUS
7.2.5 AZATHIOPRINE
7.2.6 AUTRES
7.3 IMMUNOSUPPRESSEUR POUR PERSONNES HOSPITALISÉES
7.3.1 BELATACEPT
7.3.2 BASILIXIMAB
7.3.3 AUTRES
7.4 AUTRES
7.4.1 AGENTS STIMULANT L'ÉRYTHROPOÏÈSE
7.4.1.1 ÉRYTHROPOÏÉTINE
7.4.1.2 DARBEPOETINE
7.4.2 AGENTS ANTIVIRAUX
7.4.2.1 VALGANCICLOVIR
7.4.2.2 LAMIVUDINE
7.4.2.3 ADÉFOVIR
7.4.2.4 AUTRES
7.4.3 AUTRES
8 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR VOIE D'ADMINISTRATION
8.1 APERÇU
8.2 ORALE
8.2.1 COMPRIMÉ
8.2.1.1 DIFFUSION IMMÉDIATE
8.2.1.2 VERSION ÉTENDUE
8.2.2 SOLUTION BUVABLE
8.2.3 AUTRES
8.3 INTRAVEINEUSE
9 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR ORGANE
9.1 APERÇU
9.2 REIN
9.3 FOIE
9.4 COEUR
9,5 POUMON
9.6 AUTRES
10 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR TYPE DE PATIENT
10.1 APERÇU
10.2 ADULTES
10.3 PÉDIATRIQUE
11 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR UTILISATEUR FINAL
11.1 APERÇU
11.2 HÔPITAUX
11.3 CLINIQUES
11.4 SOINS DE SANTÉ À DOMICILE
11,5 AUTRES
12 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION
12.1 APERÇU
12.2 APPELS D'OFFRES DIRECTS
12.3 PHARMACIES
12.3.1 PHARMACIE DE L'HÔPITAL
12.3.2 PHARMACIE DE DÉTAIL
12.3.3 PHARMACIE EN LIGNE
12.4 AUTRES
13 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION
13.1 EUROPE
13.1.1 FRANCE
13.1.2 ESPAGNE
13.1.3 Royaume-Uni
13.1.4 ITALIE
13.1.5 ALLEMAGNE
13.1.6 POLOGNE
13.1.7 PAYS-BAS
13.1.8 SUÈDE
13.1.9 AUTRICHE
13.1.10 SUISSE
13.1.11 NORVÈGE
13.1.12 HONGRIE
13.1.13 DANEMARK
13.1.14 IRLANDE
13.1.15 GRÈCE
13.1.16 LITUANIE
13.1.17 RESTE DE L'EUROPE
14 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAYSAGE DES ENTREPRISES
14.1 ANALYSE DES ACTIONS DE L'ENTREPRISE : EUROPE
15 QUESTIONNAIRE
16 RAPPORTS CONNEXES
Liste des tableaux
TABLEAU 1 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 2 EXPOSITION ÉPIDÉMIOLOGIQUE EN EUROPE SUR LE MARCHÉ DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 3 EXPOSITION ÉPIDÉMIOLOGIQUE EUROPÉENNE SUR LE MARCHÉ DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 4 PROPHYLAXIE ANTIBACTÉRIENNE EN EUROPE SUR LE MARCHÉ DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 5 PROPHYLAXIE ANTIBACTÉRIENNE EN EUROPE SUR LE MARCHÉ DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 6 PROPHYLAXIE CONTRE D'AUTRES AGENTS PATHOGÈNES EN EUROPE SUR LE MARCHÉ DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 7 AUTRES MARCHÉS EUROPÉENS DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 8 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 9 MARCHÉ EUROPÉEN DES IMMUNOSUPPRESSEURS EN CONSULTATION AMBULATOIRE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 10 MARCHÉ EUROPÉEN DES IMMUNOSUPPRESSEURS EN PROPHYLAXIE DU REJET D'ORGANE EN CONSULTATION AMBULATOIRE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 11 MARCHÉ EUROPÉEN DES INHIBITEURS DE LA CALCINEURINE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 12 MARCHÉ EUROPÉEN DU TACROLIMUS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 13 MARCHÉ EUROPÉEN DES CORTICOSTÉROÏDES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 14 MARCHÉ EUROPÉEN DES ACIDES MYCOPHÉNOLIQUES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 15 MARCHÉ EUROPÉEN DES INHIBITEURS DE MTOR DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 16 MARCHÉ EUROPÉEN DES IMMUNOSUPPRESSEURS POUR LA PROPHYLAXIE DU REJET D'ORGANE EN CAS D'HOSPITALISATION, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 17 MARCHÉ EUROPÉEN DES IMMUNOSUPPRESSEURS POUR LA PROPHYLAXIE DU REJET D'ORGANE EN CAS D'HOSPITALISATION, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 18 AUTRES MARCHÉS EUROPÉENS DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 19 AUTRES MARCHÉS EUROPÉENS DANS LA PROPHYLAXIE DU REJET D'ORGANES, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 20 MARCHÉ EUROPÉEN DES AGENTS STIMULANT L'ÉRYTHROPOÏÏSE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 21 MARCHÉ EUROPÉEN DES AGENTS ANTIVIRAUX DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 22 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 23 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE ORALE DU REJET D'ORGANE, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 24 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE ORALE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 25 MARCHÉ EUROPÉEN DES COMPRIMÉS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 26 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES PAR VOIE INTRAVEINEUSE, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 27 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR ORGANE, 2020-2029 (EN MILLIONS USD)
TABLEAU 28 MARCHÉ EUROPÉEN DU REIN DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 29 MARCHÉ EUROPÉEN DU FOIE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 30 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 31 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES DANS LES POUMONS, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 32 AUTRES MARCHÉS EUROPÉENS DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 33 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 34 Marché européen des adultes bénéficiant de la prophylaxie du rejet d'organes, par région, 2020-2029 (en millions USD)
TABLEAU 35 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE PÉDIATRIQUE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 36 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
TABLEAU 37 HÔPITAUX EUROPÉENS SUR LE MARCHÉ DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 38 CLINIQUES EUROPÉENNES SUR LE MARCHÉ DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 39 MARCHÉ EUROPÉEN DES SOINS DE SANTÉ À DOMICILE DANS LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 40 AUTRES MARCHÉS EUROPÉENS DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 41 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 42 APPELS D'OFFRES DIRECTS EN EUROPE SUR LE MARCHÉ DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 43 PHARMACIES EUROPÉENNES SUR LE MARCHÉ DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 44 PHARMACIES EUROPÉENNES SUR LE MARCHÉ DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 45 AUTRES MARCHÉS EUROPÉENS DE LA PROPHYLAXIE DU REJET D'ORGANES, PAR RÉGION, 2020-2029 (EN MILLIONS USD)
TABLEAU 46 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DES TRANSPLANTATIONS D'ORGANES, PAR PAYS, 2020-2029 (EN MILLIONS USD)
TABLEAU 47 PROPHYLAXIE DE LA TRANSPLANTATION D'ORGANES EN EUROPE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 48 EXPOSITION ÉPIDÉMIOLOGIQUE EN EUROPE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 49 PROPHYLAXIE ANTIBACTÉRIENNE EN EUROPE DANS LA PRÉVENTION DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 50 PROPHYLAXIE DU REJET D'ORGANE EN EUROPE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 51 EUROPE : IMMUNOSUPPRESSEURS EN CONSULTATION AMBULATOIRE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 52 INHIBITEURS DE LA CALCINEURINE EN EUROPE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 53 EUROPE TACROLIMUS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 54 ACIDES MYCOPHÉNOLIQUES EUROPE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 55 INHIBITEURS DE MTOR EN EUROPE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 56 CORTICOSTÉROÏDES EN EUROPE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 57 IMMUNOSUPPRESSEUR EN EUROPE POUR LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 58 EUROPE AUTRES EN MATIÈRE DE PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 59 AGENTS STIMULANTS DE L'ÉRYTHROPOÏÈSE EN EUROPE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 60 AGENTS ANTIVIRAUX EUROPEENS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 61 PROPHYLAXIE DU REJET D'ORGANE EN EUROPE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 62 EUROPE VOIE ORALE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 63 EUROPE COMPRIMÉ DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 64 PROPHYLAXIE DU REJET D'ORGANE EN EUROPE, PAR ORGANE, 2020-2029 (EN MILLIONS USD)
TABLEAU 65 PROPHYLAXIE DU REJET D'ORGANE EN EUROPE, PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 66 PROPHYLAXIE DU REJET D'ORGANE EN EUROPE, PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
TABLEAU 67 PROPHYLAXIE DU REJET D'ORGANE EN EUROPE, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 68 PHARMACIES EUROPÉENNES UTILISANT LA PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 69 PROPHYLAXIE DE LA TRANSPLANTATION D'ORGANES EN FRANCE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 70 FRANCE EXPOSITION ÉPIDÉMIOLOGIQUE EN PRÉVENTION DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 71 FRANCE PROPHYLAXIE ANTIBACTÉRIENNE DANS LA PRÉVENTION DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 72 FRANCE PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 73 FRANCE IMMUNOSUPPRESSEURS EN CONSULTATION AMBULATOIRE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 74 FRANCE INHIBITEURS DE LA CALCINEURINE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 75 FRANCE TACROLIMUS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 76 ACIDES MYCOPHÉNOLIQUES EN FRANCE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 77 INHIBITEURS DE MTOR EN FRANCE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 78 CORTICOSTÉROÏDES EN FRANCE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 79 FRANCE IMMUNOSUPPRESSEUR EN PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 80 FRANCE AUTRES EN PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 81 FRANCE AGENTS STIMULANTS DE L'ÉRYTHROPOÏÏSE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 82 AGENTS ANTIVIRAUX EN FRANCE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 83 FRANCE PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 84 FRANCE VOIE ORALE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 85 FRANCE COMPRIMÉ DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 86 FRANCE PROPHYLAXIE DU REJET D'ORGANE, PAR ORGANE, 2020-2029 (EN MILLIONS USD)
TABLEAU 87 FRANCE PROPHYLAXIE DU REJET D'ORGANE, PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 88 FRANCE PROPHYLAXIE DU REJET D'ORGANE, PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
TABLEAU 89 PROPHYLAXIE DU REJET D'ORGANE EN FRANCE, PAR CIRCUIT DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 90 PHARMACIES FRANÇAISES UTILISÉES POUR LA PROPHYLAXIE DU REJET D'ORGANES, PAR CIRCUIT DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 91 ESPAGNE PROPHYLAXIE DE LA TRANSPLANTATION D'ORGANES, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 92 ESPAGNE EXPOSITION ÉPIDÉMIOLOGIQUE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 93 ESPAGNE PROPHYLAXIE ANTIBACTÉRIENNE DANS LA PRÉVENTION DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 94 ESPAGNE PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 95 ESPAGNE IMMUNOSUPPRESSEUR EN CONSULTATION AMBULATOIRE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 96 ESPAGNE INHIBITEURS DE LA CALCINEURINE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 97 ESPAGNE TACROLIMUS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 98 ESPAGNE ACIDES MYCOPHÉNOLIQUES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 99 ESPAGNE INHIBITEURS DE MTOR DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 100 CORTICOSTÉRÔDES EN ESPAGNE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 101 ESPAGNE IMMUNOSUPPRESSEUR EN PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 102 ESPAGNE AUTRES EN PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 103 ESPAGNE AGENTS STIMULANTS DE L'ÉRYTHROPOÏÈSE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 104 AGENTS ANTIVIRAUX ESPAGNOLS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 105 ESPAGNE PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 106 ESPAGNE VOIE ORALE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 107 ESPAGNE COMPRIMÉ DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 108 ESPAGNE PROPHYLAXIE DU REJET D'ORGANE, PAR ORGANE, 2020-2029 (EN MILLIONS USD)
TABLEAU 109 ESPAGNE PROPHYLAXIE DU REJET D'ORGANE, PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 110 ESPAGNE PROPHYLAXIE DU REJET D'ORGANE, PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
TABLEAU 111 ESPAGNE PROPHYLAXIE DU REJET D'ORGANE, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 112 PHARMACIES ESPAGNOLES UTILISÉES POUR LA PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 113 PROPHYLAXIE DES TRANSPLANTATIONS D'ORGANES AU ROYAUME-UNI, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 114 EXPOSITION ÉPIDÉMIOLOGIQUE AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 115 PROPHYLAXIE ANTIBACTÉRIENNE AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 116 PROPHYLAXIE DU REJET D'ORGANE AU ROYAUME-UNI, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 117 IMMUNOSUPPRESSEURS ADMINISTRÉS EN CONSULTATION EXTERNE AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 118 INHIBITEURS DE LA CALCINEURINE AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 119 TACROLIMUS AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 120 ACIDES MYCOPHÉNOLIQUES AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 121 INHIBITEURS DE MTOR AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 122 CORTICOSTÉRÔDES AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 123 IMMUNOSUPPRESSEUR POUR LES HÔPITAUX HOSPITALISÉS AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 124 AUTRES PERSONNES AU ROYAUME-UNI EN PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 125 AGENTS STIMULANTS DE L'ÉRYTHROPOÏÈSE AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 126 AGENTS ANTIVIRAUX AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 127 PROPHYLAXIE DU REJET D'ORGANE AU ROYAUME-UNI, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 128 UTILISATION ORALE AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 129 COMPRIMÉS AU ROYAUME-UNI DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 130 PROPHYLAXIE DU REJET D'ORGANE AU ROYAUME-UNI, PAR ORGANE, 2020-2029 (EN MILLIONS USD)
TABLEAU 131 PROPHYLAXIE DU REJET D'ORGANE AU ROYAUME-UNI, PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 132 PROPHYLAXIE DU REJET D'ORGANE AU ROYAUME-UNI, PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
TABLEAU 133 PROPHYLAXIE DU REJET D'ORGANE AU ROYAUME-UNI, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 134 PHARMACIES BRITANNIQUES UTILISANT LA PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 135 ITALIE PROPHYLAXIE DE LA TRANSPLANTATION D'ORGANES, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 136 EXPOSITION ÉPIDÉMIOLOGIQUE EN ITALIE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 137 ITALIE PROPHYLAXIE ANTIBACTÉRIENNE DANS LA PRÉVENTION DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 138 ITALIE PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 139 ITALIE IMMUNOSUPPRESSEUR EN CONSULTATION AMBULATOIRE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 140 ITALIE INHIBITEURS DE LA CALCINEURINE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 141 ITALIE TACROLIMUS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 142 ITALIE ACIDES MYCOPHÉNOLIQUES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 143 ITALIE INHIBITEURS DE MTOR DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 144 ITALIE CORTICOSTÉROÏDES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 145 ITALIE IMMUNOSUPPRESSEUR POUR LES HÔPITAUX HOSPITALISÉS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 146 ITALIE AUTRES EN PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 147 ITALIE AGENTS STIMULANTS DE L'ÉRYTHROPOÏÈSE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 148 ITALIE AGENTS ANTIVIRAUX DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 149 ITALIE PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 150 ITALIE VOIE ORALE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 151 ITALIE COMPRIMÉ DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 152 ITALIE PROPHYLAXIE DU REJET D'ORGANE, PAR ORGANE, 2020-2029 (EN MILLIONS USD)
TABLEAU 153 ITALIE PROPHYLAXIE DU REJET D'ORGANE, PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 154 ITALIE PROPHYLAXIE DU REJET D'ORGANE, PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
TABLEAU 155 ITALIE PROPHYLAXIE DU REJET D'ORGANE, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 156 PHARMACIES ITALIENNES UTILISANT LA PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 157 ALLEMAGNE PROPHYLAXIE DE LA TRANSPLANTATION D'ORGANES, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 158 ALLEMAGNE EXPOSITION ÉPIDÉMIOLOGIQUE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 159 ALLEMAGNE PROPHYLAXIE ANTIBACTÉRIENNE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 160 ALLEMAGNE PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 161 ALLEMAGNE IMMUNOSUPPRESSEUR EN CONSULTATION AMBULATOIRE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 162 ALLEMAGNE INHIBITEURS DE LA CALCINEURINE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 163 ALLEMAGNE TACROLIMUS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 164 ALLEMAGNE ACIDES MYCOPHÉNOLIQUES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 165 ALLEMAGNE INHIBITEURS DE MTOR DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 166 ALLEMAGNE CORTICOSTÉROÏDES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 167 ALLEMAGNE IMMUNOSUPPRESSEUR POUR LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 168 ALLEMAGNE AUTRES EN PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 169 ALLEMAGNE AGENTS STIMULANTS DE L'ÉRYTHROPOÏÈSE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 170 AGENTS ANTIVIRAUX EN ALLEMAGNE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 171 ALLEMAGNE PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 172 ALLEMAGNE VOIE ORALE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 173 ALLEMAGNE COMPRIMÉ DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 174 ALLEMAGNE PROPHYLAXIE DU REJET D'ORGANE, PAR ORGANE, 2020-2029 (EN MILLIONS USD)
TABLEAU 175 ALLEMAGNE PROPHYLAXIE DU REJET D'ORGANE, PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 176 ALLEMAGNE PROPHYLAXIE DU REJET D'ORGANE, PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
TABLEAU 177 ALLEMAGNE PROPHYLAXIE DU REJET D'ORGANE, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 178 PHARMACIES ALLEMANDES UTILISANT LA PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 179 POLOGNE PROPHYLAXIE DE LA TRANSPLANTATION D'ORGANES, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 180 POLOGNE EXPOSITION ÉPIDÉMIOLOGIQUE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 181 POLOGNE PROPHYLAXIE ANTIBACTÉRIENNE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 182 POLOGNE PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 183 POLOGNE IMMUNOSUPPRESSEUR EN CONSULTATION AMBULATOIRE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 184 POLOGNE INHIBITEURS DE LA CALCINEURINE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 185 POLOGNE TACROLIMUS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 186 POLOGNE ACIDES MYCOPHÉNOLIQUES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 187 POLOGNE INHIBITEURS DE MTOR DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 188 POLOGNE CORTICOSTÉROÏDES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 189 POLOGNE IMMUNOSUPPRESSEUR POUR LES HÔTES HOSPITALISÉS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 190 POLOGNE AUTRES EN PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 191 POLOGNE AGENTS STIMULANTS DE L'ÉRYTHROPOÏÏSE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 192 POLOGNE AGENTS ANTIVIRAUX DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 193 POLOGNE PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 194 POLOGNE VOIE ORALE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 195 POLOGNE COMPRIMÉ DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 196 POLOGNE PROPHYLAXIE DU REJET D'ORGANE, PAR ORGANE, 2020-2029 (EN MILLIONS USD)
TABLEAU 197 POLOGNE PROPHYLAXIE DU REJET D'ORGANE, PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 198 POLOGNE PROPHYLAXIE DU REJET D'ORGANE, PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
TABLEAU 199 POLOGNE PROPHYLAXIE DU REJET D'ORGANE, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 200 PHARMACIES POLOGNES UTILISÉES POUR LA PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 201 PAYS-BAS PROPHYLAXIE DE LA TRANSPLANTATION D'ORGANES, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 202 PAYS-BAS EXPOSITION ÉPIDÉMIOLOGIQUE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 203 PAYS-BAS PROPHYLAXIE ANTIBACTÉRIENNE DANS LA PRÉVENTION DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 204 PAYS-BAS PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 205 PAYS-BAS : IMMUNOSUPPRESSEUR EN CONSULTATION AMBULATOIRE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 206 INHIBITEURS DE LA CALCINEURINE AUX PAYS-BAS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 207 PAYS-BAS TACROLIMUS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 208 PAYS-BAS ACIDES MYCOPHÉNOLIQUES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 209 PAYS-BAS INHIBITEURS DE MTOR DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 210 PAYS-BAS CORTICOSTÉROÏDES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 211 IMMUNOSUPPRESSEUR HOSPITALISÉ AUX PAYS-BAS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 212 PAYS-BAS AUTRES EN MATIÈRE DE PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 213 PAYS-BAS AGENTS STIMULANTS DE L'ÉRYTHROPOÏÈSE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 214 AGENTS ANTIVIRAUX AUX PAYS-BAS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 215 PAYS-BAS PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 216 PAYS-BAS UTILISATION ORALE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 217 PAYS-BAS COMPRIMÉ DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 218 PAYS-BAS PROPHYLAXIE DU REJET D'ORGANE, PAR ORGANE, 2020-2029 (EN MILLIONS USD)
TABLEAU 219 PAYS-BAS PROPHYLAXIE DU REJET D'ORGANE, PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 220 PAYS-BAS PROPHYLAXIE DU REJET D'ORGANE, PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
TABLEAU 221 PAYS-BAS PROPHYLAXIE DU REJET D'ORGANE, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 222 PHARMACIES DES PAYS-BAS EN MATIÈRE DE PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 223 SUÈDE PROPHYLAXIE DE LA TRANSPLANTATION D'ORGANES, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 224 SUÈDE EXPOSITION ÉPIDÉMIOLOGIQUE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 225 SUÈDE PROPHYLAXIE ANTIBACTÉRIENNE DANS LA PRÉVENTION DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 226 SUÈDE PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 227 SUÈDE IMMUNOSUPPRESSEUR EN CONSULTATION AMBULATOIRE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 228 SUÈDE INHIBITEURS DE LA CALCINEURINE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLE 229 SWEDEN TACROLIMUS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 230 SWEDEN MYCOPHENOLIC ACIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 231 SWEDEN MTOR INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 232 SWEDEN CORTICOSTEROIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 233 SWEDEN INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 234 SWEDEN OTHERS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 235 SWEDEN ERYTHROPOIESIS-STIMULATING AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 236 SWEDEN ANTI-VIRAL AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 237 SWEDEN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 238 SWEDEN ORAL IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 239 SWEDEN TABLET IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 240 SWEDEN PROPHYLAXIS OF ORGAN REJECTION, BY ORGAN, 2020-2029 (USD MILLION)
TABLE 241 SWEDEN PROPHYLAXIS OF ORGAN REJECTION, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 242 SWEDEN PROPHYLAXIS OF ORGAN REJECTION, BY END USER, 2020-2029 (USD MILLION)
TABLE 243 SWEDEN PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 244 SWEDEN PHARMACY STORES IN PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 245 AUSTRIA PROPHYLAXIS OF ORGAN TRANSPLANT, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 246 AUSTRIA EPIDEMIOLOGIC EXPOSURE IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 247 AUSTRIA ANTIBACTERIAL PROPHYLAXIS IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 248 AUSTRIA PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 249 AUSTRIA OUTPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 250 AUSTRIA CALCINEURIN INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 251 AUSTRIA TACROLIMUS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 252 AUSTRIA MYCOPHENOLIC ACIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 253 AUSTRIA MTOR INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 254 AUSTRIA CORTICOSTEROIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 255 AUSTRIA INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 256 AUSTRIA OTHERS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 257 AUSTRIA ERYTHROPOIESIS-STIMULATING AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 258 AUSTRIA ANTI-VIRAL AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 259 AUSTRIA PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 260 AUSTRIA ORAL IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 261 AUSTRIA TABLET IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 262 AUSTRIA PROPHYLAXIS OF ORGAN REJECTION, BY ORGAN, 2020-2029 (USD MILLION)
TABLE 263 AUSTRIA PROPHYLAXIS OF ORGAN REJECTION, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 264 AUSTRIA PROPHYLAXIS OF ORGAN REJECTION, BY END USER, 2020-2029 (USD MILLION)
TABLE 265 AUSTRIA PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 266 AUSTRIA PHARMACY STORES IN PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 267 SWITZERLAND PROPHYLAXIS OF ORGAN TRANSPLANT, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 268 SWITZERLAND EPIDEMIOLOGIC EXPOSURE IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 269 SWITZERLAND ANTIBACTERIAL PROPHYLAXIS IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 270 SWITZERLAND PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 271 SWITZERLAND OUTPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 272 SWITZERLAND CALCINEURIN INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 273 SWITZERLAND TACROLIMUS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 274 SWITZERLAND MYCOPHENOLIC ACIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 275 SWITZERLAND MTOR INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 276 SWITZERLAND CORTICOSTEROIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 277 SWITZERLAND INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 278 SWITZERLAND OTHERS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 279 SWITZERLAND ERYTHROPOIESIS-STIMULATING AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 280 SWITZERLAND ANTI-VIRAL AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 281 SWITZERLAND PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 282 SWITZERLAND ORAL IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 283 SWITZERLAND TABLET IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 284 SWITZERLAND PROPHYLAXIS OF ORGAN REJECTION, BY ORGAN, 2020-2029 (USD MILLION)
TABLEAU 285 SUISSE PROPHYLAXIE DU REJET D'ORGANE, PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 286 SUISSE PROPHYLAXIE DU REJET D'ORGANE, PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
TABLEAU 287 SUISSE PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 288 PHARMACIES SUISSES EN PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 289 PROPHYLAXIE DE LA TRANSPLANTATION D'ORGANES EN NORVÈGE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 290 NORVÈGE EXPOSITION ÉPIDÉMIOLOGIQUE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 291 NORVÈGE PROPHYLAXIE ANTIBACTÉRIENNE DANS LA PRÉVENTION DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 292 PROPHYLAXIE DU REJET D'ORGANE EN NORVÈGE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 293 NORVÈGE IMMUNOSUPPRESSEUR EN CONSULTATION AMBULATOIRE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 294 INHIBITEURS DE LA CALCINEURINE EN NORVÈGE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 295 NORVÈGE TACROLIMUS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 296 ACIDES MYCOPHÉNOLIQUES EN NORVÈGE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 297 INHIBITEURS DE MTOR EN NORVÈGE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 298 CORTICOSTÉRÔDES NORVÉGIENNES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 299 NORVÈGE IMMUNOSUPPRESSEUR POUR LES HÔTES HOSPITALISÉS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 300 NORVÈGE AUTRES EN MATIÈRE DE PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 301 NORVÈGE AGENTS STIMULANTS DE L'ÉRYTHROPOÏÈSE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 302 AGENTS ANTIVIRAUX NORVÉGIENNES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 303 PROPHYLAXIE DU REJET D'ORGANE EN NORVÈGE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 304 NORVÈGE VOIE ORALE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 305 COMPRIMÉ NORVÉGIEN DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 306 PROPHYLAXIE DU REJET D'ORGANE EN NORVÈGE, PAR ORGANE, 2020-2029 (EN MILLIONS USD)
TABLEAU 307 PROPHYLAXIE DU REJET D'ORGANE EN NORVÈGE, PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 308 PROPHYLAXIE DU REJET D'ORGANE EN NORVÈGE, PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
TABLEAU 309 PROPHYLAXIE DU REJET D'ORGANE EN NORVÈGE, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 310 PHARMACIES NORVÉGIENNES UTILISANT LA PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 311 HONGRIE PROPHYLAXIE DES TRANSPLANTATIONS D'ORGANES, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 312 EXPOSITION ÉPIDÉMIOLOGIQUE EN HONGRIE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 313 HONGRIE PROPHYLAXIE ANTIBACTÉRIENNE DANS LA PRÉVENTION DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 314 HONGRIE PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 315 HONGRIE : IMMUNOSUPPRESSEUR EN CONSULTATION AMBULATOIRE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 316 HONGRIE INHIBITEURS DE LA CALCINEURINE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 317 HONGRIE TACROLIMUS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 318 HONGRIE ACIDES MYCOPHÉNOLIQUES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 319 HONGRIE INHIBITEURS DE MTOR DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 320 HONGRIE CORTICOSTÉROÏDES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 321 HONGRIE IMMUNOSUPPRESSEUR POUR LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 322 HONGRIE AUTRES PERSONNES EN PRÉVENTION DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 323 HONGRIE AGENTS STIMULANTS DE L'ÉRYTHROPOÏÏSE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 324 AGENTS ANTIVIRAUX HONGROIS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 325 HONGRIE PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 326 HONGRIE UTILISATION DE L'ADMINISTRATION ORALE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 327 HONGRIE COMPRIMÉ DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR VOIE D'ADMINISTRATION, 2020-2029 (EN MILLIONS USD)
TABLEAU 328 HONGRIE PROPHYLAXIE DU REJET D'ORGANE, PAR ORGANE, 2020-2029 (EN MILLIONS USD)
TABLEAU 329 HONGRIE PROPHYLAXIE DU REJET D'ORGANE, PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 330 HONGRIE PROPHYLAXIE DU REJET D'ORGANE, PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
TABLEAU 331 HONGRIE PROPHYLAXIE DU REJET D'ORGANE, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 332 PHARMACIES HONGROISES UTILISÉES POUR LA PROPHYLAXIE DU REJET D'ORGANES, PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
TABLEAU 333 DANEMARK PROPHYLAXIE DE LA TRANSPLANTATION D'ORGANES, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 334 DANEMARK EXPOSITION ÉPIDÉMIOLOGIQUE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 335 DANEMARK PROPHYLAXIE ANTIBACTÉRIENNE DANS LA PRÉVENTION DU REJET D'ORGANE, PAR CAUSE, 2020-2029 (EN MILLIONS USD)
TABLEAU 336 DANEMARK PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 337 DANEMARK IMMUNOSUPPRESSEUR EN CONSULTATION AMBULATOIRE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 338 DANEMARK INHIBITEURS DE LA CALCINEURINE DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 339 DANEMARK TACROLIMUS DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 340 DANEMARK ACIDES MYCOPHÉNOLIQUES DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLEAU 341 DANEMARK INHIBITEURS DE MTOR DANS LA PROPHYLAXIE DU REJET D'ORGANE, PAR TRAITEMENT, 2020-2029 (EN MILLIONS USD)
TABLE 342 DENMARK CORTICOSTEROIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 343 DENMARK INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 344 DENMARK OTHERS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 345 DENMARK ERYTHROPOIESIS-STIMULATING AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 346 DENMARK ANTI-VIRAL AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 347 DENMARK PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 348 DENMARK ORAL IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 349 DENMARK TABLET IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 350 DENMARK PROPHYLAXIS OF ORGAN REJECTION, BY ORGAN, 2020-2029 (USD MILLION)
TABLE 351 DENMARK PROPHYLAXIS OF ORGAN REJECTION, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 352 DENMARK PROPHYLAXIS OF ORGAN REJECTION, BY END USER, 2020-2029 (USD MILLION)
TABLE 353 DENMARK PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 354 DENMARK PHARMACY STORES IN PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 355 IRELAND PROPHYLAXIS OF ORGAN TRANSPLANT, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 356 IRELAND EPIDEMIOLOGIC EXPOSURE IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 357 IRELAND ANTIBACTERIAL PROPHYLAXIS IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 358 IRELAND PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 359 IRELAND OUTPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 360 IRELAND CALCINEURIN INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 361 IRELAND TACROLIMUS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 362 IRELAND MYCOPHENOLIC ACIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 363 IRELAND MTOR INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 364 IRELAND CORTICOSTEROIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 365 IRELAND INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 366 IRELAND OTHERS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 367 IRELAND ERYTHROPOIESIS-STIMULATING AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 368 IRELAND ANTI-VIRAL AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 369 IRELAND PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 370 IRELAND ORAL IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 371 IRELAND TABLET IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 372 IRELAND PROPHYLAXIS OF ORGAN REJECTION, BY ORGAN, 2020-2029 (USD MILLION)
TABLE 373 IRELAND PROPHYLAXIS OF ORGAN REJECTION, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 374 IRELAND PROPHYLAXIS OF ORGAN REJECTION, BY END USER, 2020-2029 (USD MILLION)
TABLE 375 IRELAND PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 376 IRELAND PHARMACY STORES IN PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 377 GREECE PROPHYLAXIS OF ORGAN TRANSPLANT, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 378 GREECE EPIDEMIOLOGIC EXPOSURE IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 379 GREECE ANTIBACTERIAL PROPHYLAXIS IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 380 GREECE PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 381 GREECE OUTPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 382 GREECE CALCINEURIN INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 383 GREECE TACROLIMUS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 384 GREECE MYCOPHENOLIC ACIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 385 GREECE MTOR INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 386 GREECE CORTICOSTEROIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 387 GREECE INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 388 GREECE OTHERS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 389 GREECE ERYTHROPOIESIS-STIMULATING AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 390 GREECE ANTI-VIRAL AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 391 GREECE PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 392 GREECE ORAL IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 393 GREECE TABLET IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 394 GREECE PROPHYLAXIS OF ORGAN REJECTION, BY ORGAN, 2020-2029 (USD MILLION)
TABLE 395 GREECE PROPHYLAXIS OF ORGAN REJECTION, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 396 GREECE PROPHYLAXIS OF ORGAN REJECTION, BY END USER, 2020-2029 (USD MILLION)
TABLE 397 GREECE PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 398 GREECE PHARMACY STORES IN PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 399 LITHUANIA PROPHYLAXIS OF ORGAN TRANSPLANT, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 400 LITHUANIA EPIDEMIOLOGIC EXPOSURE IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 401 LITHUANIA ANTIBACTERIAL PROPHYLAXIS IN PROPHYLAXIS OF ORGAN REJECTION, BY CAUSE, 2020-2029 (USD MILLION)
TABLE 402 LITHUANIA PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 403 LITHUANIA OUTPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 404 LITHUANIA CALCINEURIN INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 405 LITHUANIA TACROLIMUS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 406 LITHUANIA MYCOPHENOLIC ACIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 407 LITHUANIA MTOR INHIBITORS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 408 LITHUANIA CORTICOSTEROIDS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 409 LITHUANIA INPATIENT IMMUNOSUPPRESSANT IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 410 LITHUANIA OTHERS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 411 LITHUANIA ERYTHROPOIESIS-STIMULATING AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 412 LITHUANIA ANTI-VIRAL AGENTS IN PROPHYLAXIS OF ORGAN REJECTION, BY TREATMENT, 2020-2029 (USD MILLION)
TABLE 413 LITHUANIA PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 414 LITHUANIA ORAL IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 415 LITHUANIA TABLET IN PROPHYLAXIS OF ORGAN REJECTION, BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
TABLE 416 LITHUANIA PROPHYLAXIS OF ORGAN REJECTION, BY ORGAN, 2020-2029 (USD MILLION)
TABLE 417 LITHUANIA PROPHYLAXIS OF ORGAN REJECTION, BY PATIENT TYPE, 2020-2029 (USD MILLION)
TABLE 418 LITHUANIA PROPHYLAXIS OF ORGAN REJECTION, BY END USER, 2020-2029 (USD MILLION)
TABLE 419 LITHUANIA PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 420 LITHUANIA PHARMACY STORES IN PROPHYLAXIS OF ORGAN REJECTION, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 421 REST OF EUROPE PROPHYLAXIS OF ORGAN TRANSPLANT, BY CAUSE, 2020-2029 (USD MILLION)
Liste des figures
FIGURE 1 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: SEGMENTATION
FIGURE 2 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: DATA TRIANGULATION
FIGURE 3 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: DROC ANALYSIS
FIGURE 4 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: EUROPE VS REGIONAL MARKET ANALYSIS
FIGURE 5 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: DBMR POSITION GRID
FIGURE 8 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: VENDOR SHARE ANALYSIS
FIGURE 9 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: END USER COVERAGE GRID
FIGURE 10 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: SEGMENTATION
FIGURE 11 NORTH AMERICA IS EXPECTED TO DOMINATE THE EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET AND ASIA-PACIFIC IS EXPECTED TO GROW WITH THE HIGHEST CAGR IN THE FORECAST PERIOD OF 2022 TO 2029
FIGURE 12 INCREASED PREVALENCE OF ORGAN TRANSPLANTATION, RISE IN RECIPIENT PATIENTS, AND PRODUCT APPPROVALS ARE EXPECTED TO DRIVE EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET FROM 2022 TO 2029
FIGURE 13 EPIDEMIOLOGIC EXPOSURE SEGMENT IS EXPECTED TO HAVE THE LARGEST SHARE OF THE EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET FROM 2022 & 2029
FIGURE 14 ASIA-PACIFIC IS THE FASTEST-GROWING MARKET FOR PROPHYLAXIS OF ORGAN REJECTION MANUFACTURERS IN THE FORECAST PERIOD OF 2022 TO 2029
FIGURE 15 EUROPE PROPHYLAXIS OF ORGAN REJECTION: INCIDENCE, 2021
FIGURE 16 EUROPE PROPHYLAXIS OF ORGAN REJECTION: INCIDENCE REGIONAL LEVEL, 2021
FIGURE 17 EUROPE PROPHYLAXIS OF ORGAN REJECTION: PREVALENCE (2018-2021) AT REGIONAL LEVEL SNAPSHOT
FIGURE 18 EUROPE PROPHYLAXIS OF ORGAN REJECTION: SURVIVAL OF PATIENT SNAPSHOT
FIGURE 19 DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES OF EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET
FIGURE 20 INCREASE COUNT OF KIDNEY TRANSPLANTATIONS IN THE U.S., 2020
FIGURE 21 TOTAL NUMBER OF ORGAN TRANSPLANTATION IN THE EUROPE SCENARIO, 2020
FIGURE 22 TOTAL NUMBER OF PATIENTS ON WAITING LIST VS TRANSPLANTS PERFORMED BY ORGAN IN 2020
FIGURE 23 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY CAUSE, 2021
FIGURE 24 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY CAUSE, 2020-2029 (USD MILLION)
FIGURE 25 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY CAUSE, CAGR (2022-2029)
FIGURE 26 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY CAUSE, LIFELINE CURVE
FIGURE 27 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY TREATMENT, 2021
FIGURE 28 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY TREATMENT, 2020-2029 (USD MILLION)
FIGURE 29 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY TREATMENT, CAGR (2022-2029)
FIGURE 30 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY TREATMENT, LIFELINE CURVE
FIGURE 31 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ROUTE OF ADMINISTRATION, 2021
FIGURE 32 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ROUTE OF ADMINISTRATION, 2020-2029 (USD MILLION)
FIGURE 33 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ROUTE OF ADMINISTRATION, CAGR (2022-2029)
FIGURE 34 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ROUTE OF ADMINISTRATION, LIFELINE CURVE
FIGURE 35 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ORGAN, 2021
FIGURE 36 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ORGAN, 2020-2029 (USD MILLION)
FIGURE 37 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ORGAN, CAGR (2022-2029)
FIGURE 38 EUROPE PROPHYLAXIS OF ORGAN REJECTION MARKET: BY ORGAN, LIFELINE CURVE
FIGURE 39 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAR TYPE DE PATIENT, 2021
FIGURE 40 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAR TYPE DE PATIENT, 2020-2029 (EN MILLIONS USD)
FIGURE 41 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAR TYPE DE PATIENT, TCAC (2022-2029)
FIGURE 42 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAR TYPE DE PATIENT, COURBE DE LIGNE DE VIE
FIGURE 43 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAR UTILISATEUR FINAL, 2021
FIGURE 44 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAR UTILISATEUR FINAL, 2020-2029 (EN MILLIONS USD)
FIGURE 45 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAR UTILISATEUR FINAL, TCAC (2022-2029)
FIGURE 46 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAR UTILISATEUR FINAL, COURBE DE LIGNE DE VIE
FIGURE 47 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAR CANAL DE DISTRIBUTION, 2021
FIGURE 48 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAR CANAL DE DISTRIBUTION, 2020-2029 (EN MILLIONS USD)
FIGURE 49 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAR CANAL DE DISTRIBUTION, TCAC (2022-2029)
FIGURE 50 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PAR CANAL DE DISTRIBUTION, COURBE DE LIGNE DE VIE
FIGURE 51 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DES TRANSPLANTATIONS D'ORGANES : INSTANTANÉ (2021)
FIGURE 52 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DES TRANSPLANTATIONS D'ORGANES : PAR PAYS (2021)
FIGURE 53 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DES TRANSPLANTATIONS D'ORGANES : PAR PAYS (2022 ET 2029)
FIGURE 54 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DES TRANSPLANTATIONS D'ORGANES : PAR PAYS (2021 ET 2029)
FIGURE 55 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DES TRANSPLANTATIONS D'ORGANES : PAR CAUSE (2022-2029)
FIGURE 56 MARCHÉ EUROPÉEN DE LA PROPHYLAXIE DU REJET D'ORGANES : PART DE L'ENTREPRISE 2021 (%)
Méthodologie de recherche
La collecte de données et l'analyse de l'année de base sont effectuées à l'aide de modules de collecte de données avec des échantillons de grande taille. L'étape consiste à obtenir des informations sur le marché ou des données connexes via diverses sources et stratégies. Elle comprend l'examen et la planification à l'avance de toutes les données acquises dans le passé. Elle englobe également l'examen des incohérences d'informations observées dans différentes sources d'informations. Les données de marché sont analysées et estimées à l'aide de modèles statistiques et cohérents de marché. De plus, l'analyse des parts de marché et l'analyse des tendances clés sont les principaux facteurs de succès du rapport de marché. Pour en savoir plus, veuillez demander un appel d'analyste ou déposer votre demande.
La méthodologie de recherche clé utilisée par l'équipe de recherche DBMR est la triangulation des données qui implique l'exploration de données, l'analyse de l'impact des variables de données sur le marché et la validation primaire (expert du secteur). Les modèles de données incluent la grille de positionnement des fournisseurs, l'analyse de la chronologie du marché, l'aperçu et le guide du marché, la grille de positionnement des entreprises, l'analyse des brevets, l'analyse des prix, l'analyse des parts de marché des entreprises, les normes de mesure, l'analyse globale par rapport à l'analyse régionale et des parts des fournisseurs. Pour en savoir plus sur la méthodologie de recherche, envoyez une demande pour parler à nos experts du secteur.
Personnalisation disponible
Data Bridge Market Research est un leader de la recherche formative avancée. Nous sommes fiers de fournir à nos clients existants et nouveaux des données et des analyses qui correspondent à leurs objectifs. Le rapport peut être personnalisé pour inclure une analyse des tendances des prix des marques cibles, une compréhension du marché pour d'autres pays (demandez la liste des pays), des données sur les résultats des essais cliniques, une revue de la littérature, une analyse du marché des produits remis à neuf et de la base de produits. L'analyse du marché des concurrents cibles peut être analysée à partir d'une analyse basée sur la technologie jusqu'à des stratégies de portefeuille de marché. Nous pouvons ajouter autant de concurrents que vous le souhaitez, dans le format et le style de données que vous recherchez. Notre équipe d'analystes peut également vous fournir des données sous forme de fichiers Excel bruts, de tableaux croisés dynamiques (Fact book) ou peut vous aider à créer des présentations à partir des ensembles de données disponibles dans le rapport.