Global High Drug To Antibody Ratio Adc Therapies Market
시장 규모 (USD 10억)
연평균 성장률 :
% 
 USD
2.12 Billion
USD
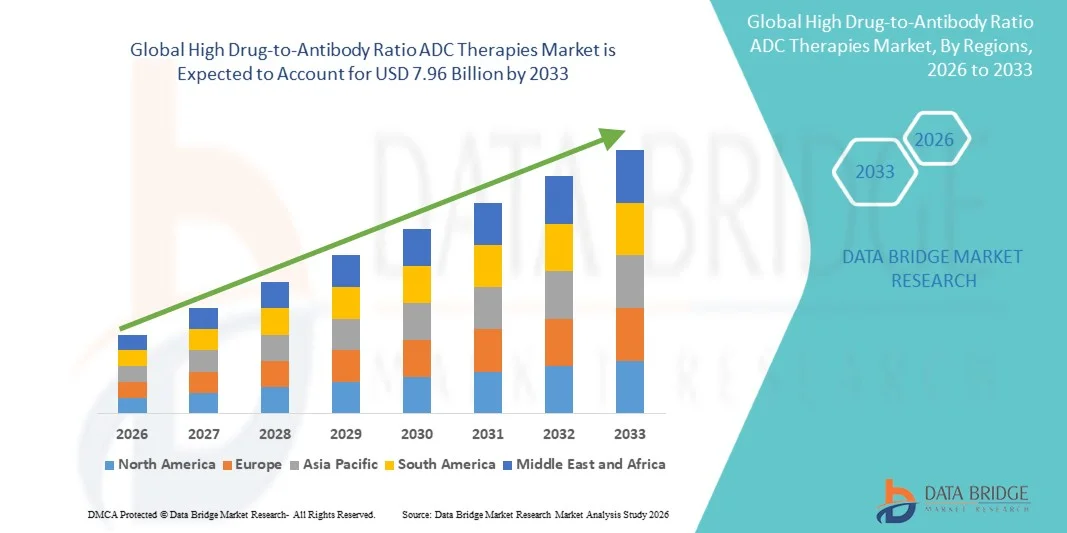
7.96 Billion
2025
2033
USD
2.12 Billion
USD
7.96 Billion
2025
2033
| 2026 –2033 | |
| USD 2.12 Billion | |
| USD 7.96 Billion | |
|
|
|
|
Global High Drug-to-Antibody Ratio ADC Therapies Market Segmentation, Payload Type (Topoisomerase Inhibitors, Microtubule Inhibitors, DNA Damaging Agents 및 기타), 표시 (Breast Cancer, Hematologic Malignancies, Lung Cancer, Colorectal Cancer 및 기타), 개발 단계 : Preclinical, 임상 시험 및 상용 제품) - 산업 동향 및 Fore 2033
높은 약 - 항체 비율 ADC Therapies 시장 크기
- 글로벌 고약-항체 비율 ADC 치료 시장 규모가 가치25억 달러견적 요청50억 달러, 에 의해18.00%의 CAGR예측 기간
- 시장 성장은 고약에 항체 비율 (DAR) 항체-Drug Conjugates (ADCs)의 채택 증가에 의해 크게 연료를 공급하고, 그들의 강화한 치료 효능에 의해 몰아, 종양 세포에 더 높은 세포 독성 하중을 전달하는 능력, 그리고 안정성을 개량하고 떨어져 표적 독성을 감소시키는 연결기 기술에 있는 진보
- 또한, 표적 암 치료에 대한 수요 상승, 혈액 종양 및 고체 종양의 발현 증가, ADC 연구 및 임상 개발의 투자 증가 높은 DAR ADC 치료의 섭취를 가속화하고, 크게 전반적인 시장 성장
높은 약 - 항체 비율 ADC Therapies 시장 분석
- 높은 약 - 항체 비율 (DAR) ADC 치료는 종양 세포에 세포 독성 약의 더 높은 페이로드를 전달하기 위해 설계되었으며, 향상된 효능, 감소 된 체계 독성 및 임상 및 병원 설정에서 채택을 주도하는 능력으로 인해 종양학에서 점점 더 중요합니다.
- 높은 DAR ADC 치료를위한 에스컬레이션 요구 사항은 주로 표적 치료에 중점을 둔 암 전임, ADC 링크를 통해 지속적인 혁신 및 치료 기술을 강화하는 지속적인 혁신에 중점을두고 있으며 치료 결과 및 환자 준수를 향상시킵니다.
- 북미는 2025 년에 약 39.5%의 가장 큰 수익 점유율을 가진 고약에 항체 비율 ADC 치료 시장, 고급 종양 인프라, 높은 연구 및 개발 투자, 강력한 규제 프레임 워크 및 미국의 바이오 기술 스타트업의 존재를 강화했다.
- 아시아 태평양은 예측 기간 동안 가장 빠르게 성장하는 지역이 될 것으로 예상되며, 암 발생률 증가, 의료 인프라 확장, 임상 시험 활동 확대, 중국, 일본 및 인도와 같은 국가의 표적 치료의 채택 증가
- 상업화 된 제품 세그먼트는 FDA 승인 ADC 및 그들의 광범위한 임상 채택의 존재에 의해 구동되는 2025에서 44.3%의 매출 점유율을 가진 시장을 지배했습니다
보고서 범위 및 높은 약물 - 항체 비율 ADC 치료 시장 세그먼트
| 관련 기사 | 높은 약 - 항체 비율 ADC Therapies Key 시장 통찰력 |
| Segments 적용 |
|
| 국가 덮음 | 북아메리카
·
아시아 태평양
중동 및 아프리카
대한민국
|
| 핵심 시장 선수 |
|
| 시장 기회 |
|
| Value 추가 데이터 Infosets | 시장 가치, 성장률, 세그먼트, 지리적 범위 및 주요 플레이어와 같은 시장 시나리오에 대한 통찰력 외에도 데이터 브리지 시장 연구에 의해 큐레이터 시장 보고서는 심층적 인 전문가 분석, 환자 분석, 파이프라인 분석, 가격 분석 및 규제 프레임 워크를 포함합니다. |
높은 약 - 항체 비율 ADC Therapies 시장 동향
"혁신적인 Payload 기술 및 임상 발전을 통한 확장·
- ADC 치료 시장은 높은 약물 - 항체 비율 (DAR) ADC 치료 시장은 안정성과 대상 배달을 유지하면서 더 높은 약물 - 항체 비율을 허용하는 새로운 링크 및 페이로드 기술의 개발입니다. 이 접근법은 치료 효능을 강화하고 치료 가능한 암의 범위를 확대, 특히 hematologic malignancies 및 고체 종양
- 예를 들어, 2024 년 Seagen은 HER2-positive breast 암을 대상으로 한 차세대 높은 DAR ADC 후보를 도입하여 향상된 시토 독성 페이로드 전달을 통해 사전 클리닉 결과를 예측하고 오프 타락 독성을 최소화했습니다. 마찬가지로, Daiichi Sankyo의 trastuzumab deruxtecan의 향상은 항체 특성을 보존하면서 DAR을 증가 시켰습니다. 임상 시험에서 더 높은 효능을 가능하게합니다. 이러한 진보는 임상 결과 및 환자의 관용성을 개선하고, 종양학 센터 전반에 걸쳐 더 넓은 채용을 지원하는 것입니다.
- 또한, 사이트 별 회춘 기술 및 생물 오염 화학에 대한 발전은 항체 안정성, 면역성 위험을 줄이고 약리학 프로파일을 개선하지 않고 DAR를 최적화 할 수 있습니다.
- 더 높은 DAR ADC를 향한 추세는 또한 면역 치료, 체크포인트 억제제 및 표준 의료 화학 물질과 ADC의 성장 중요성을 반영하는 임상 시험의 증가 수에 의해 지원됩니다.
- 자동화된 conjugation 플랫폼과 강화된 정화 방법과 같은 개량한 제조 기술의 통합은 확장 가능한 높 DAR ADCs의 발달을 가속하고, preclinical 테스트에서 임상 신청에 급속한 전환을 가능하게 합니다
- 이 혁신은 공동으로 높은-DAR ADC 개발 투자 및 연구에 대한 기대를 형성하고 있습니다.
높은 약 - 항체 비율 ADC Therapies 시장 역학
관련 기사
"암 치료 및 개선 임상 치료를위한 수요 상승"
- 높은-DAR ADC 치료의 채택을위한 중요한 드라이버 인 정밀 의약품의 성장 수요와 결합 된 암의 글로벌 인산 증가
- 예를 들어, FDA의 Fm-trastuzumab deruxtecan의 가속 승인은 2022 HER2-positive metastatic breast 암에 대한 2022에서 개선 된 진행이없는 생존 및 감소 된 시스템 독성을 포함하여 높은 DAR ADCs의 임상 혜택을 강조했습니다. 마찬가지로 급성 myeloid leukemia의 Gemtuzumab ozogamicin의 지속적인 임상 시험은 이전에 기존 화학 요법에 반응하지 않는 환자의 향상된 효능을 보여줍니다.
- 선택적으로 표적 종양 세포에 있는 ADCs의 이점에 관하여 종양학의 사이에서 인식을 확대하고, 부작용을 최소화하는 것은 더 연료를 공급 시장 성장입니다
- 또한, ADC 연구 및 개발을위한 민간 및 공공 부문 모두에서 자금을 증가, 바이오 제약 회사 및 학술 기관 간의 협력과 함께, 높은 DAR 페이로드 기술에 혁신 가속화
- 어려운 암과 개인화 된 치료 요법을 위한 필요의 상승한 우선권은 ADC 플랫폼에 투자를 계속하고 치료 창과 환자를 최적화하는 초점을 맞추고 있습니다.
스트레인트/Challenge
"높은 개발 비용, 규제 Hurdles 및 안전 Concerns·
- 비교적 높은 비용의 개발 높은-DAR ADC 치료, 복잡한 conjugation 화학, 엄격한 품질 관리 및 비싼 임상 시험, 시장 확장에 상당한 장벽을 포즈
- 예를 들어, 사이트 별 활용 ADC는 전문 시설과 고도로 숙련 된 인력을 필요로하며 작고 중간 크기의 바이오 기술 회사에 대한 고급 투자를 증가시킵니다. 이것은 혁신적인 후보자를 위한 시장 입장을 지연할지도 모릅니다
- FDA 및 EMA와 같은 기관의 엄격한 요구 사항을 포함하여 규제 문제, 안전, 안정성 및 효율성을 위해, 승인 시간을 단축하고 개발 비용을 추가 할 수 있습니다
- 높은-DAR ADC의 잠재적인 off-target 독성 및 면역성에 대한 우려는 장기적인 임상 안전 데이터까지 광범위한 채택을 제한할 수 있습니다.
- 최적화된 제조 공정을 통해 이러한 과제를 해결하고, 강력한 전임 검증 및 규제 전문가들과의 전략적 협력은 높은-DAR ADC 시장의 지속적인 성장을 위해 중요합니다.
높은 약 - 항체 비율 ADC Therapies 시장 범위
시장은 Payload Type, Indication, Development Stage를 기반으로 합니다.
• Payload 유형에 의하여
탑재량 유형의 기초에, 높은 약에 항체 비율 ADC 치료 시장은 Topoisomerase Inhibitors, Microtubule Inhibitors, DNA Damaging Agents 및 다른 사람으로 구분됩니다. Topoisomerase Inhibitors 세그먼트는 표적 암 치료에 입증 된 효능에 의해 구동 2025에서 41.5%의 가장 큰 시장 수익 점유율을 지배하고 떨어져 표적 독성 감소. 이 억제물은 급속하게 종양 세포를 확산하기 위하여 선택적으로 DNA 손상을 유도하기 위하여 그들의 능력 때문에 야기암과 hematologic malignancie에서 넓게 채택됩니다. 제약 회사 Topoisomerase 기반 페이로드를 선호하는 예측 가능한 약리학 및 설치 안전 프로파일. 높은 약에 항체 비율을 가진 이 페이로드의 호환성은 체계적인 부작용을 최소화하면서 강력한 치료 결과를 허용합니다. Topoisomerase ADCs에 대한 임상 성공 및 규제 승인 더 bolster 시장 지배력을 증가. Adoption은 북미 및 유럽에서 특히 높으며, 종양학 파이프라인은 성숙하고 고급 생물 공학 인프라가 존재합니다.
DNA Damaging Agents 세그먼트는 2026년부터 2033년까지 22.1%의 가장 빠른 CAGR를 목격할 것으로 예상되며, 폐와 색소형 모기장과 같은 암을 대상으로 한 지속적인 임상 시험에 의해 연료를 공급합니다. DNA-damaging ADCs는 높은 치료 색인을 제안하고 그들의 임상 매력을 증가시키는 조합 치료를 위해 tailored 할 수 있습니다. 성장은 신중한 링크를 확장하여 가속화되고 항체 기술로 페이로드 배달 효율성을 향상시킵니다. Emerging 시장은 점점 DNA Damaging ADC를 채택하고 정밀 종양학 및 개인화 된 의학 접근법의 잠재력으로 채택하고 있습니다.
• 표시에 의하여
표시의 기초에, 시장은 Breast Cancer, Hematologic Malignancies, Lung Cancer, Colorectal Cancer 및 다른 사람으로 구분됩니다. Breast 암 치료는 2025 년 38.7%의 가장 큰 수익 점유율을 기록했으며 전 세계적으로 높은 인산률과 HER2-positive 및 Triple-negative breast 암 치료의 ADCs의 채택에 참여했습니다. 주요 시장에 있는 호의를 베푸는 reimbursement 정책과 더불어 설치된 임상 의정서, 더 드라이브 dominance. Top-tier 제약 플레이어는 강력한 임상 시험 결과, 환자 인식 확장 및 개인 및 공공 의료 시스템에 채택 때문에 야기 암 ADC에 초점을 맞추고 있습니다.
Hematologic Malignancies 세그먼트는 2026에서 2033까지 21.5%의 가장 빠른 CAGR를 등록 할 것으로 예상되며 백혈병, 림프종 및 여러 myeloma의 증가 우선 순위에 의해 구동됩니다. 암을 타겟팅하는 ADC는 더 높은 응답률과 시스템 독성 감소, 가속된 개발 파이프라인에서 혜택을 제공합니다. 임상 연구는 재발 및 퇴행 환자 인구의 표시를 확장하기 위해 계속. Emerging biologics 및 DNA Damaging Agents 및 Topoisomerase 억제제와 같은 소설 페이로드의 통합은이 부문에서 성장을 가속화합니다.
• 개발 단계로
개발 단계에 따라 시장은 Preclinical, Clinical Trials, Commercialized Products로 구분됩니다. 상업화 된 제품 세그먼트는 FDA 승인 ADC 및 광범위한 임상 채택의 존재에 의해 구동되는 2025에서 44.3%의 매출 점유율을 가진 시장을 지배했습니다. 이 제품은 검증된 안전과 효능 데이터를 제공하여 종양학 및 의료기관의 선호한 선택을 합니다. 상업적 제품도 브랜드 인지도, 보험 적용 및 강력한 유통망 혜택을 제공합니다.
임상 시험 세그먼트는 2026에서 2033에 23.4%의 가장 빠른 CAGR를 목격 할 것으로 예상되며 제약 회사가 공격적으로 ADC 파이프라인을 확장 할 것으로 예상됩니다. LINKER 기술 혁신, 더 높은 약물-to-antibody 비율, 그리고 새로운 탑재 연료 급속한 임상 개발. Biotech 스타트업과 대형 약국의 협업을 통해 획기적인 치료에 대한 규제 인센티브, Biotech 스타트업과 대형 약국 간의 협력을 통해 Emerging 시장은 또한 임상 시험에 참여하고 환자의 접근을 확장하고 차세대 ADC의 글로벌 채택을 가속화합니다.
높은 약 - 항체 비율 ADC Therapies 시장 지역 분석
- 북미는 2025 년에 약 39.5%의 가장 큰 수익 점유율을 가진 고약에 항체 비율 ADC 치료 시장, 고급 종양 인프라, 높은 R & D 투자, 강력한 규제 프레임 워크 및 미국의 바이오 테크 스타트업의 존재를 지배했다.
- 또한 제약 회사 및 학술 암 센터 간의 전략적 파트너십은 새로운 ADC 치료의 가속화 된 개발 및 상용화를 추진하고 있습니다.
- 광범위한 채택은 높은 의료 지출에 의해 더 지원, 전문 종양 센터의 가용성, 그리고 호의를 베푸는 reimbursement 정책, 높은-DAR ADC 치료를위한 최고의 시장으로 북미를 설립
미국 고약 항체 비율 ADC Therapies 시장 통찰력
미국 고약에 항체 비율 ADC 치료 시장은 2025년 북미 내에서 가장 큰 수익 점유율을 차지했으며, 광범위한 임상 시험 활동, 바이오 제약 및 학술 연구 기관 간의 강력한 협력 및 목표 치료의 높은 환자 인식에 의해 연료를 공급했습니다. 예를 들어, HER2-positive breast 암을 위한 fam-trastuzumab deruxtecan의 FDA 승인 및 급성 myeloid 백혈병을 위한 Gemtuzumab ozogamicin의 진행 시험은 혁신적인 ADCs의 지역 급속한 채택을 강조합니다. 강력한 R&D 파이프라인과 Oncology 인프라는 미국 시장을 전파하기 위해 계속되었습니다.
유럽 높은 약 - 항체 비율 ADC Therapies 시장 통찰력
유럽의 높은 약 - 항체 비율 ADC 치료 시장은 예측 기간 동안 실질적인 CAGR에 확장하기 위해 계획되어 암 선명도 상승에 의해 구동되고, 종양 연구에 대한 정부 지원 증가, 및 생체 공학에 대한 엄격한 규제 표준. 예를 들어, 2023 년 Roche와 AstraZeneca는 여러 유럽 종양학 센터를 통해 높은 DAR ADC 임상 프로그램을 시작했으며, 모유 및 폐암의 치료 요구 사항을 해결하는 것을 목표로합니다. 이 지역의 정밀 의학에 중점을두고 표적 치료의 성장 채택은 시장 성장을 더욱 자극 할 것으로 예상됩니다.
U.K. 고약 항체 비율 ADC 치료 시장 통찰력
U.K. 고약 항체 비율 ADC 치료 시장은 예측 기간 동안 주목할만한 CAGR에서 성장할 것으로 예상되며, 암 치료 인프라 확장, 임상 시험 참여 증가 및 혁신적인 생물 공학에 대한 의료 지출이 증가합니다. 예를 들어, U.K. 기반 병원과 글로벌 ADC 개발자 간의 협력은 hematologic malignancies를 대상으로 한 새로운 High-DAR ADC 후보에 대한 평가를 지원하며 접근성과 가속 채택을 강화합니다.
독일 고약 항체 비율 ADC Therapies 시장 통찰력
독일의 높은 약 - 항체 비율 ADC 치료 시장은 예측 기간 동안 상당한 CAGR에서 확장 할 것으로 예상되며, 생명 공학 혁신, 고급 의료 시설 및 임상 연구에 대한 규제 인센티브에 중점을 둡니다. 예를 들어, BioNTech 및 기타 독일 바이오 기술 회사는 ADC 개발 플랫폼에 투자하여 목표의 페이로드 납품을 강화하고 정밀 종양학을 향한 성장 추세를 반영합니다.
아시아 태평양 고약 항체 비율 ADC Therapies 시장 통찰력
아시아 태평양 고약-항체 비율 ADC 치료 시장은 예측 기간 동안 가장 빠른 CAGR에서 성장하기 위해, 암 발산 증가, 의료 인프라 확대, 임상 시험 활동 확대, 중국, 일본 및 인도와 같은 국가에서 대상 치료의 성장 채택. 예를 들어 중국, 여러 ADC 후보의 규제 승인 및 Tier-1 도시의 종양 센터의 확장은 더 넓은 환자 접근을 촉진합니다. 인도에서, 국내 바이오 기술 기업과 글로벌 ADC 개발자 간의 협력은 현지 생산과 임상 연구를 가능하게 합니다. 마찬가지로, 일본은 첨단 기술 종양학 연구에 투자하고 breast, 폐 및 hematologic 암에 대한 혁신적인 치료의 채택은 지역 전역의 시장 성장을 몰고 있습니다.
일본 고약 항체 비율 ADC Therapies 시장 통찰력
일본 고약-항체 비율 ADC 치료 시장은 고급 의료 인프라, 혁신적인 종양학 치료의 조기 채택, 암 인식 증가로 인한 순간을 얻고 있습니다. 예를 들어, 일본의 글로벌 고DAR ADC 임상 시험에 참여하고 정부 지원 연구 보조금의 가용성은 새로운 ADC 치료의 개발 및 섭취를 가속화하고 있습니다.
중국 고약 항체 비율 ADC Therapies 시장 통찰력
중국 높은 약물 - 항체 비율 ADC 치료 시장은 2025 년 아시아 태평양에서 가장 큰 시장 수익 점유율을 차지했으며 암 퇴치, 병원 네트워크를 확장하고 고급 암 치료를 찾는 중급 인구가 성장했습니다. 예를 들어, 상하이 기반 ADC 개발자는 Phase II 및 III 임상 시험에서 여러 높은 DAR ADC 후보를 시작했으며 최첨단 종양 치료의 지역 급속한 채택을 민주화했습니다. Biologics 제조 및 임상 연구를 지원하는 정부 이니셔티브는 더 시장을 추진하고있다.
높은 약 - 항체 비율 ADC Therapies 시장 공유
높은 약 - 항체 비율 ADC 치료 산업은 주로 잘 설립 된 회사에 의해 주도된다 :
- Pfizer Inc. (미국)
- Roche Holding AG (스위스)
- AbbVie Inc. (미국)
- (주)암젠
- Astellas Pharma Inc. (일본)
- Daiichi Sankyo Company(일본)
- Mersana 치료, Inc. (미국)
- Synaffix BV (네덜란드)
- Genmab A/S (덴마크)
- Innovent Biologics, Inc. (중국)
- Wuxi Biologics (중국)
- IGM Biosciences, Inc. (미국)
- RemeGen (주)
Global High Drug-to-Antibody Ratio ADC Therapies Market의 최신 개발
- 2025년 1월, 미국 식품의약품안전청은 Datopotamab Deruxtecan‐dlnk (Datroway), Trop‐2 ‐directed antibody‐drug conjugate에 의해 개발된 Datopotamab Deruxtecan‐dlnk (Datroway), 비정상적인 호르몬 수용체 ‐ positive, HER2‐negative breast 암을 가진 성인을 위한 Daiichi Sankyo와 AstraZeneca에 의해 개발된 ‐directed antibody‐drug conjugate를 승인했습니다.
- 4 월 2025, Datopotamab Deruxtecan (Datroway)는 유럽 연합 (EU)의 마케팅 허가를 받았다. 이 ADC 치료에 대한 추가 확장은 비정상적 또는 메타 정적 인 HR‐positive, HER2‐negative 모유 암 환자에 대한 액세스를 확장
- 6 월 2025에서 미국 FDA는 EGFR의 처리를위한 Datroway를 승인 ‐ small 세포 폐암, 폐암 표시에서이 ADC 치료의 첫 번째 승인을 표시하고 모유 암보다 임상적 영향을 확장
- 6월 2025일, 미국 식품의약품안전청은 다트로웨이(Datroway)를 승인하여, 다른 치료법에 대한 EGFR ‐mutated 종양을 가진 환자를 대상으로 한 옵션을 제공하고 여러 암 유형의 ADCs의 확장 임상 유틸리티를 강조합니다.
- 2025 년 10 월, AstraZeneca와 Daiichi Sankyo는 Datroway가 늦은 단계 임상 시험에서 세 배 중립적인 모유 암을 가진 환자에 있는 전반적인 생존을 크게 개량했다고 보고했습니다, 잠재적 치료 이득과 높은 ‐ 포위 ADC 치료의 임상 진도의 밑에
- 11월 2025일, Day One Biopharmaceuticals는 Mersana Therapeutics를 인수하기로 합의했으며, Mersana의 리드 ADC 자산 Emi‐Le (targeting B7‐H4)를 Day One의 종양학 포트폴리오로 인수하고 고급 ADC 치료의 잠재력에 강한 투자자 신뢰를 신호했습니다.
- 2025년 5월, Radiance Biopharma는 ROR-1-targeted ADC를 위한 독점적인 면허 계약을 입력하고, ADC 공간에 있는 발달 협력을 확장하고 다음 세대 conjugates에 있는 혁신을 지원했습니다
- 6월 2025일, SunRock Biopharma 및 Escugen은 SRB123을 개발하는 파트너십을 발표했습니다. 최초의 ‐ 클래스 CCR9-targeted ADC는 여러 고체 종양을 치료하기 위해 ADC 개발 파이프라인에서 성장하는 협업 및 다양화
SKU-
세계 최초의 시장 정보 클라우드 보고서에 온라인으로 접속하세요
- 대화형 데이터 분석 대시보드
- 높은 성장 잠재력 기회를 위한 회사 분석 대시보드
- 사용자 정의 및 질의를 위한 리서치 분석가 액세스
- 대화형 대시보드를 통한 경쟁자 분석
- 최신 뉴스, 업데이트 및 추세 분석
- 포괄적인 경쟁자 추적을 위한 벤치마크 분석의 힘 활용
연구 방법론
데이터 수집 및 기준 연도 분석은 대규모 샘플 크기의 데이터 수집 모듈을 사용하여 수행됩니다. 이 단계에는 다양한 소스와 전략을 통해 시장 정보 또는 관련 데이터를 얻는 것이 포함됩니다. 여기에는 과거에 수집한 모든 데이터를 미리 검토하고 계획하는 것이 포함됩니다. 또한 다양한 정보 소스에서 발견되는 정보 불일치를 검토하는 것도 포함됩니다. 시장 데이터는 시장 통계 및 일관된 모델을 사용하여 분석하고 추정합니다. 또한 시장 점유율 분석 및 주요 추세 분석은 시장 보고서의 주요 성공 요인입니다. 자세한 내용은 분석가에게 전화를 요청하거나 문의 사항을 드롭하세요.
DBMR 연구팀에서 사용하는 주요 연구 방법론은 데이터 마이닝, 시장에 대한 데이터 변수의 영향 분석 및 주요(산업 전문가) 검증을 포함하는 데이터 삼각 측량입니다. 데이터 모델에는 공급업체 포지셔닝 그리드, 시장 타임라인 분석, 시장 개요 및 가이드, 회사 포지셔닝 그리드, 특허 분석, 가격 분석, 회사 시장 점유율 분석, 측정 기준, 글로벌 대 지역 및 공급업체 점유율 분석이 포함됩니다. 연구 방법론에 대해 자세히 알아보려면 문의를 통해 업계 전문가에게 문의하세요.
사용자 정의 가능
Data Bridge Market Research는 고급 형성 연구 분야의 선두 주자입니다. 저희는 기존 및 신규 고객에게 목표에 맞는 데이터와 분석을 제공하는 데 자부심을 느낍니다. 보고서는 추가 국가에 대한 시장 이해(국가 목록 요청), 임상 시험 결과 데이터, 문헌 검토, 재생 시장 및 제품 기반 분석을 포함하도록 사용자 정의할 수 있습니다. 기술 기반 분석에서 시장 포트폴리오 전략에 이르기까지 타겟 경쟁업체의 시장 분석을 분석할 수 있습니다. 귀하가 원하는 형식과 데이터 스타일로 필요한 만큼 많은 경쟁자를 추가할 수 있습니다. 저희 분석가 팀은 또한 원시 엑셀 파일 피벗 테이블(팩트북)로 데이터를 제공하거나 보고서에서 사용 가능한 데이터 세트에서 프레젠테이션을 만드는 데 도움을 줄 수 있습니다.