Europe Endoscopic Hemostasis Market
Размер рынка в млрд долларов США
CAGR :
% 
 USD
675.69 Million
USD
1,183.22 Million
2025
2033
USD
675.69 Million
USD
1,183.22 Million
2025
2033
| 2026 –2033 | |
| USD 675.69 Million | |
| USD 1,183.22 Million | |
|
|
|
|
Европейский рынок эндоскопического гемостаза по типу продукта (механические устройства гемостаза, термические устройства, актуальные агенты и инъекционные средства и другие), процедура (верхняя желудочно-кишечная эндоскопия, нижняя желудочно-кишечная эндоскопия, бронхоскопический гемостаз и другие), применение (желудочно-кишечное кровотечение, не желудочно-кишечное кровотечение, лечение травм и другие), конечный пользователь (больницы, центры амбулаторной хирургии, специализированные клиники и другие), канал распределения (прямые продажи и косвенные продажи) - отраслевые тенденции и прогноз до 2033 года
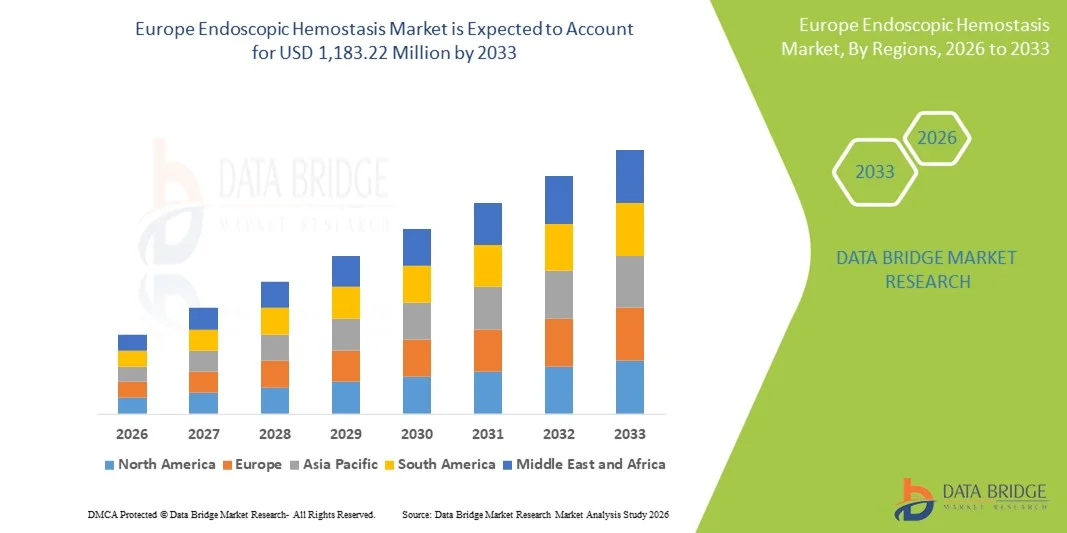
Размер рынка эндоскопического гемостаза в Европе
- Ожидается, что европейский рынок эндоскопических гемостазов достигнет$1 183,22 млн.К 2033 году из675,69 млн. долларов СШАв 2025 году, с существенным ростомCAGR 7,7%в прогнозном периоде 2026-2033 гг.
- Европейский рынок эндоскопического гемостаза демонстрирует устойчивый и устойчивый рост, обусловленный растущей распространенностью желудочно-кишечных расстройств, увеличением заболеваемости колоректальным раком, пептическими язвами и желудочно-кишечными кровотечениями, а также растущим внедрением минимально инвазивных эндоскопических процедур по всему региону.
- Рост рынка также поддерживается строгими клиническими рекомендациями, сильным акцентом на безопасность пациентов и процедурную эффективность, а также растущим спросом на передовые гемостатические технологии, которые улучшают процедурные результаты и снижают уровень осложнений. Непрерывные достижения в разработке эндоскопических устройств, включая улучшенные клипсы, системы коагуляции, спреи и комбинированную терапию, повышают точность, простоту использования и клиническую эффективность.
Европейский эндоскопический анализ рынка гемостаза
- Эндоскопические продукты гемостаза становятся все более важными в экосистеме здравоохранения Европы, позволяя клиницистам эффективно управлять желудочно-кишечными кровотечениями во время диагностических и терапевтических эндоскопических процедур. Эти устройства играют жизненно важную роль в улучшении процедурной безопасности, клинических результатов и выздоровления пациентов, обеспечивая быстрый и надежный контроль кровотечения. Эндоскопические решения гемостаза широко используются в ключевых клинических условиях, включая больницы, специализированные клиники и амбулаторные хирургические центры, для таких применений, как кровотечение из язвенной болезни, колоректальные процедуры, кровотечение после полипэктомии и кровоизлияние, связанное с опухолью.
- Расширение инфраструктуры здравоохранения, увеличение объемов эндоскопических процедур и внедрение минимально инвазивных подходов к лечению подпитывают высокий спрос на устройства для эндоскопического гемостаза в Европе. Поставщики медицинских услуг все чаще используют передовые гемостатические технологии для повышения эффективности процедур, снижения частоты осложнений, сокращения пребывания в больнице и улучшения общих результатов лечения пациентов, тем самым поддерживая устойчивый рост рынка.
- Кроме того, строгие клинические рекомендации, повышение внимания к безопасности пациентов и растущий контроль со стороны регулирующих органов ускоряют принятие передовых и клинически доказанных решений для эндоскопического гемостаза. Производители медицинских устройств вкладывают значительные средства в исследования и разработки для разработки инновационных продуктов, которые предлагают улучшенную точность, простоту использования и совместимость с современными эндоскопическими платформами, в то же время удовлетворяя меняющимся нормативным стандартам и требованиям к клинической производительности.
- Германия, по прогнозам, возглавит европейский рынок эндоскопического гемостаза в 2026 году, составляя 15,89% доли регионального рынка. Это лидерство поддерживается хорошо налаженной инфраструктурой здравоохранения страны, большим объемом эндоскопических процедур, сильным внедрением передовых медицинских технологий и присутствием ведущих производителей медицинских устройств. Благоприятные рамки возмещения расходов, квалифицированная клиническая рабочая сила и постоянные инвестиции в инновации в области здравоохранения еще больше укрепляют доминирующее положение Германии на европейском рынке.
- Дания является самой быстрорастущей страной на рынке эндоскопического гемостаза в Европе, чему способствует раннее внедрение цифровых медицинских и передовых эндоскопических платформ, сильная интеграция диагностики с помощью искусственного интеллекта и эндоскопических процедур с визуальным управлением, а также высокий уровень участия клинических исследований в желудочно-кишечной и интервенционной эндоскопии. Хорошо структурированные реферальные пути страны, акцент на дневном уходе и амбулаторном эндоскопическом лечении и быстрое коммерческое использование клиницистами гемостатических устройств следующего поколения еще больше ускоряют процедурные объемы. Кроме того, сотрудничество между больницами, академическими учреждениями и производителями медицинских устройств, наряду с упорядоченными сроками одобрения регулирующих органов для инновационных технологий, продолжают позиционировать Данию как ведущий рынок ранних адоптеров, стимулируя более быстрый рост по сравнению с другими европейскими странами.
- Сегмент устройств механического гемостаза доминирует на европейском рынке эндоскопического гемостаза, на который приходится 43,86% рынка в 2025 году. Это доминирование обусловлено широким клиническим внедрением эндоскопических клипсов и устройств лигирования полос из-за их доказанной эффективности, простоты развертывания, экономической эффективности и пригодности для широкого спектра показаний желудочно-кишечного кровотечения. Их сильный профиль безопасности и совместимость со стандартными эндоскопическими процедурами продолжают поддерживать устойчивый спрос в медицинских учреждениях.
Сфера охвата и сегментация рынка эндоскопического гемостаза Европы
| Атрибуты | Эндоскопический рынок гемостаза Европы |
| Сегменты покрыты |
|
| Страны, охваченные | Европа
|
| Ключевые игроки рынка |
|
| Рыночные возможности |
|
| Информационные наборы данных с добавленной стоимостью | В дополнение к информации о рыночных сценариях, таких как рыночная стоимость, темпы роста, сегментация, географическое покрытие и основные игроки, рыночные отчеты, курируемые Data Bridge Market Research, также включают критерии отбора поставщиков, технологическое продвижение, патентный анализ, нормативную базу, отслеживание инноваций и стратегический анализ тарифов и воздействия на рынок. |
Европейские тенденции рынка эндоскопического гемостаза
Расширение применения передовых минимально инвазивных технологий эндоскопического гемостаза
- Устойчивое расширение эндоскопических и минимально инвазивных процедур по всей Европе является основным фактором, способствующим более широкому внедрению эндоскопических устройств гемостаза. Поскольку частота желудочно-кишечных кровотечений, колоректальных расстройств и связанных с ними состояний продолжает расти, поставщики медицинских услуг все чаще полагаются на передовые решения гемостаза для обеспечения эффективного контроля кровотечения, процедурной безопасности и улучшения результатов лечения пациентов в больших объемах клинических условий.
- Устройства эндоскопического гемостаза играют решающую роль в обеспечении клинической эффективности, безопасности пациента и процедурной надежности во время диагностической и терапевтической эндоскопии. Обеспечивая быстрый контроль кровотечения, снижая риски осложнений и поддерживая минимально инвазивные подходы к лечению, эти устройства помогают оптимизировать процедурные рабочие процессы, одновременно улучшая время восстановления и общее качество медицинской помощи в медицинских учреждениях.
- Растущее внедрение передовых эндоскопических технологий, включая визуализацию высокой четкости, терапевтическую эндоскопию и комбинированные методы гемостаза, увеличило спрос на высокоэффективные гемостатические решения. Инновации в механических зажимах, системах тепловой коагуляции, инъекционной терапии и актуальных гемостатических средствах повышают процессуальную точность, показатели успеха лечения и уверенность клинициста, поддерживая развивающуюся клиническую практику.
- Поставщики медицинских услуг все чаще используют эндоскопические устройства гемостаза для удовлетворения растущих нормативных требований, стандартов безопасности и клинических показателей. Строгие клинические рекомендации, растущий акцент на безопасность пациентов и уход, основанный на результатах, и растущий спрос на минимально инвазивные вмешательства побуждают больницы и клиники интегрировать передовые технологии гемостаза, которые обеспечивают согласованность, соответствие и высококачественную доставку медицинской помощи.
- В целом, расширяющийся масштаб эндоскопических процедур, клинических инноваций и развития инфраструктуры здравоохранения позиционирует устройства эндоскопического гемостаза как важный компонент современной желудочно-кишечной помощи. Эти решения поддерживают процедурную эффективность, безопасность пациентов, соблюдение нормативных требований и устойчивый рост в развивающемся эндоскопическом ландшафте Европы, что позволяет легко повторно использовать это понимание в разных регионах путем адаптации динамики системы здравоохранения и моделей принятия.
Динамика рынка эндоскопического гемостаза в Европе
водитель
Рост случаев желудочно-кишечного кровотечения
- Растущая частота желудочно-кишечных кровотечений была установлена как основополагающая сила, стимулирующая рост на мировом рынке эндоскопического гемостаза. По мере того, как распространенность верхних и нижних желудочно-кишечных кровотечений увеличивается во всем мире, спрос на минимально инвазивные терапевтические вмешательства на основе эндоскопии был усилен. Эндоскопический гемостаз предлагает критические клинические преимущества, включая быстрый контроль кровотечения, снижение потребности в хирургическом вмешательстве, более низкие требования к переливанию и более короткое пребывание в больнице, тем самым позиционируя его как метод лечения первой линии при остром и хроническом лечении желудочно-кишечных кровотечений. Следовательно, эскалация случаев желудочно-кишечных кровотечений, вызванная старением населения, более высокой распространенностью заболеваний печени, использованием антикоагулянтов и задержкой доступа к медицинской помощи во время системных сбоев в здравоохранении, привела к увеличению объемов процедур и более широкому внедрению передовых эндоскопических устройств гемостаза в больницах и эндоскопических центрах по всему миру.
- Например,
- В сентябре 2021 года Medscape сообщила, что верхнее желудочно-кишечное кровотечение происходило с частотой примерно 100 случаев на 100 000 населения ежегодно и оставалось одной из наиболее распространенных причин экстренного госпитализации, подчеркивая постоянно высокое бремя заболевания, требующее эндоскопического вмешательства.
- В июне 2023 года журнал Gastroenterology Research сообщил, что смертность, связанная с верхним желудочно-кишечным кровотечением в США, увеличилась в период с 2012 по 2021 год, причем более резкое увеличение наблюдалось в последующие годы, что указывает на ухудшение клинических результатов и усиление требований к лечению.
- В июне 2023 года издание StatPearls Publishing заявило, что желудочно-кишечное кровотечение продолжает представлять собой частую глобальную медицинскую чрезвычайную ситуацию, при этом срочная эндоскопическая диагностика и гемостатическое лечение остаются критически важными для снижения смертности и заболеваемости.
- В январе 2025 года журнал клинической медицины сообщил, что у пациентов с прогрессирующим заболеванием печени наблюдалась значительно более высокая частота массивных эпизодов желудочно-кишечных кровотечений, усиливая связь между распространенностью хронических заболеваний и повышенной потребностью в эндоскопическом гемостазе.
- В сентябре 2025 года PubMed Central подчеркнул, что верхнее желудочно-кишечное кровотечение остается опасным для жизни осложнением среди населения с хроническими заболеваниями печени, поддерживая повышенный спрос на эндоскопические терапевтические процедуры.
- Глобальная эскалация заболеваемости желудочно-кишечными кровотечениями прочно утвердилась в качестве постоянного двигателя структурного роста рынка эндоскопического гемостаза. Непрерывный рост острых кровотечений в сочетании с расширением популяций, затронутых хроническими заболеваниями печени, использованием антитромботических препаратов и возрастной желудочно-кишечной патологией, создает устойчивое и нециклическое требование для эндоскопического контроля кровотечения. Поскольку клинические рекомендации все чаще отдают приоритет эндоскопической терапии как лечению первой линии, структурно усиливается зависимость от технологий гемостаза для экстренного вмешательства, профилактики рецидивов и управления осложнениями. Кроме того, улучшение показателей выживаемости расширяет мониторинг пациентов и повторяет циклы вмешательства, тем самым умножая пожизненный процедурный спрос. Эта динамика закрепляет принятие эндоскопического гемостаза близко к глобальным эпидемиологическим тенденциям, позиционируя этот драйвер как долгосрочную основу для расширения рынка в развитых и новых системах здравоохранения.
Сдержанность/вызов
Высокая стоимость и техническая сложность эндоскопических гемостазов
- Несмотря на растущее клиническое внедрение, европейский рынок эндоскопического гемостаза продолжает сталкиваться со структурными ограничениями из-за высокой стоимости и технической сложности передовых эндоскопических устройств гемостаза. Эти технологии часто требуют значительных первоначальных капиталовложений для закупки оборудования, постоянных расходов на расходные материалы и специализированного обслуживания. Кроме того, эффективное использование гемостатических эндоскопических устройств требует передовой подготовки врачей, квалифицированного вспомогательного персонала и сложной инфраструктуры больницы, что ограничивает внедрение в чувствительные к затратам системы здравоохранения. Государственные больницы в странах с низким и средним уровнем дохода и даже объекты с ограниченным бюджетом в развитых регионах часто сталкиваются с барьерами, связанными с доступностью, разрывами в возмещении и готовностью рабочей силы. В результате сохраняется неравномерный доступ и более медленное проникновение передовых технологий эндоскопического гемостаза, что ограничивает расширение рынка.
- Для силы,
- В ноябре 2022 года, согласно высокой стоимости желудочно-кишечной эндоскопии, процедуры и оборудование являются основным сдерживающим фактором. Например, TNE стоит 125,90 евро за процедуру, в то время как эндоскопия полости рта стоит 184,10 евро, а MACE стоит 407,10 евро. Кроме того, обслуживание и переработка оборудования увеличивают стоимость, а гибкие эндоскопы стоят около 79 330 евро, что делает процедуры дорогими в целом.
- В июне 2024 года Science direct подчеркнула, что высокая стоимость желудочно-кишечной эндоскопии усугубляется в странах с низким и средним уровнем дохода (СНД и LMIC) из-за отсутствия местных средств обслуживания и ремонта. Области, требующие ремонта, должны быть отправлены за границу, что требует значительных затрат и задержек. Кроме того, дешевые подержанные и китайские эндоскопы часто не имеют надлежащего обслуживания и технической поддержки.
- В октябре 2025 года BMJ Open Gastroenterology опубликовала исследование по микрозатратам, показывающее, что в больнице Национальной службы здравоохранения Великобритании общая стоимость повторного использования желудочно-кишечных эндоскопов составила 107,34 фунта стерлингов, при этом капитальные затраты и расходы на техническое обслуживание являются основными факторами затрат, подчеркивая экономические барьеры для широкого внедрения эндоскопического оборудования в учреждениях общественного здравоохранения.
- В августе 2024 года научный обзор ScienceDirect сообщил, что закупка, техническое обслуживание и связанные с этим логистические расходы на оборудование для эндоскопии остаются основным препятствием для развития и поддержания услуг эндоскопии в странах с низким и средним уровнем дохода из-за высокой стоимости устройства и отсутствия инфраструктуры.
- В феврале 2025 года, по данным науки, высокая стоимость желудочно-кишечной эндоскопии очевидна в различных исследованиях, особенно для скрининга и наблюдения. Например, в то время как общий скрининг населения может быть экономически неэффективным в западных регионах, целевое наблюдение для групп высокого риска, таких как группы с метаплазией желудочного кишечника, все еще может быть экономически эффективным, с ICER в диапазоне от 20 739,1 до 98 402,2 долларов США за QALY.
- Собранные данные ясно указывают на то, что высокая стоимость и техническая сложность, связанные с желудочно-кишечной эндоскопией и эндоскопическими устройствами гемостаза, представляют собой постоянное ограничение роста рынка. Значительные капиталовложения в закупку оборудования, повышенные затраты на процедуру и текущие расходы, связанные с обслуживанием, переработкой и ремонтом, значительно увеличивают общую нагрузку на системы здравоохранения. Эти проблемы еще более обостряются в регионах с низким и средним уровнем дохода, где ограниченная техническая инфраструктура и отсутствие местных возможностей обслуживания задерживают принятие и ограничивают процедурный потенциал. Даже в развитых системах здравоохранения соображения экономической эффективности влияют на стратегии скрининга и ограничивают широкое внедрение. В совокупности эти экономические и операционные ограничения замедляют проникновение передовых технологий эндоскопического гемостаза, усиливая доступность и техническую сложность как структурные барьеры для расширения рынка.
Европейский рынок эндоскопических гемостазов
Европейский рынок эндоскопического гемостаза подразделяется на пять ключевых сегментов: тип продукта, процедура, приложение, конечный пользователь и канал распределения.
- Тип продукта
На основе типа продукта европейский рынок эндоскопического гемостаза сегментирован на механические устройства гемостаза, тепловые устройства, актуальные агенты и инъекционные средства и другие. Согласно прогнозам, в 2026 году сегмент устройств для механического гемостаза будет доминировать на европейском рынке эндоскопического гемостаза с наибольшей долей рынка 43,89% из-за широкого клинического предпочтения в достижении немедленного, контролируемого и длительного прекращения кровотечения во время эндоскопических вмешательств. Механические решения, такие как зажимы и устройства бандажирования, обычно предпочитают за их способность обеспечивать точное закрытие сосуда, не вызывая повреждения термальной ткани, тем самым снижая скорость кровоизлияния и осложнения после процедуры. Их применимость в широком спектре сценариев кровотечения, включая пептическую язву, варикозное кровоизлияние и кровотечение после полипэктомии, привела к постоянно высокому использованию как в чрезвычайных, так и в выборных эндоскопических условиях. Сильная зависимость от механического гемостаза как терапевтического подхода первой линии подчеркивает его существенный вклад в общие доходы рынка и укрепляет его доминирующее положение в ландшафте типа продукта в течение прогнозируемого периода.
Сегмент актуальных агентов и инъекций является самым быстрорастущим сегментом на европейском рынке эндоскопического гемостаза, регистрируя CAGR 8,1%. Этот рост в первую очередь обусловлен ростом заболеваемости желудочно-кишечными (ЖК) нарушениями кровотечений, увеличением принятия минимально инвазивных эндоскопических процедур и клиническим предпочтением быстродействующих, простых в применении гемостатических растворов во время чрезвычайных и выборных вмешательств.
- По процедуре
На основе процедуры рынок сегментирован на верхнюю желудочно-кишечную эндоскопию, нижнюю желудочно-кишечную эндоскопию, бронхоскопический гемостаз и другие. По прогнозам, в 2026 году верхний сегмент эндоскопии желудочно-кишечного тракта будет доминировать на европейском рынке эндоскопического гемостаза с долей рынка 43,62% из-за его широкого клинического принятия в качестве основного процедурного подхода для лечения острых и рецидивирующих желудочно-кишечных кровотечений. Верхняя эндоскопия ЖКТ широко используется для диагностики и немедленного терапевтического контроля кровоточащих язв, варикозного кровоизлияния и поражений Dieulafoy, где быстрое гемостатическое вмешательство клинически важно. Ожидается, что высокая частота процедур в отделениях неотложной помощи и больницах высшего звена в сочетании с сильной руководящей поддержкой раннего эндоскопического вмешательства сохранит лидирующие позиции на рынке. Его продолжающееся доминирование отражается в его значительной доле на рынке и устойчивой траектории роста до 2033 года, что указывает на постоянный спрос как в развитых, так и в развивающихся системах здравоохранения.
Нижний сегмент эндоскопии желудочно-кишечного тракта является самым быстрорастущим сегментом на европейском рынке эндоскопического гемостаза, регистрируя CAGR 8,1%. Этот рост обусловлен растущей распространенностью более низких желудочно-кишечных кровотечений, включая колоректальный рак, воспалительные заболевания кишечника, дивертикулярные заболевания и геморроидальные кровотечения, а также растущим внедрением процедур скрининга и диагностической колоноскопии по всей Европе.
- Подача заявления
На основе применения рынок сегментирован на желудочно-кишечные кровотечения, не желудочно-кишечные кровотечения, управление травмами и другие. В 2026 году сегмент желудочно-кишечных кровотечений, по прогнозам, будет доминировать на европейском рынке эндоскопического гемостаза с долей рынка 71,57% из-за высокой глобальной распространенности язвенной болезни, варикоз пищевода и колоректальных злокачественных новообразований, требующих эндоскопического контроля кровотечения. Желудочно-кишечное кровотечение остается наиболее распространенным показанием для эндоскопических процедур гемостаза, обеспечивая последовательное использование механических, термических и актуальных гемостатических растворов в больницах и амбулаторных учреждениях. Ожидается, что критическая потребность в быстром контроле кровотечения для снижения заболеваемости, продолжительности пребывания в больнице и смертности укрепит устойчивый спрос в этом сегменте применения. Его большая доля в общей рыночной стоимости подчеркивает центральную роль желудочно-кишечных показаний в формировании общей динамики рынка в течение прогнозируемого периода.
Сегмент управления травмами является самым быстрорастущим сегментом на европейском рынке эндоскопического гемостаза, регистрируя CAGR в 8,3%. Этот рост обусловлен увеличением частоты травматических травм, включая травмы живота, случайные травмы и экстренные случаи желудочно-кишечного кровотечения, а также растущим предпочтением минимально инвазивных эндоскопических вмешательств для достижения быстрого контроля кровотечения.
- конечным пользователем
На базе конечного потребителя рынок сегментирован на больницы, центры амбулаторной хирургии, специализированные клиники и другие. По прогнозам, в 2026 году сегмент больниц будет доминировать на европейском рынке эндоскопического гемостаза с самой большой долей рынка 52,69% из-за концентрации передовой инфраструктуры эндоскопии, квалифицированных гастроэнтерологов и возможностей неотложной помощи в больницах. Сложные случаи кровотечения, в том числе тяжелое верхнее и нижнее желудочно-кишечное кровоизлияние, в основном управляются в государственных и частных больницах, где имеются всесторонние диагностические и интервенционные ресурсы. Более высокий приток пациентов, большие объемы процедур и установленные рамки закупок еще больше усиливают спрос на больничные устройства и расходные материалы для эндоскопического гемостаза. Ожидается, что эта структурная зависимость от стационарного ухода сохранит лидирующие позиции сегмента до 2033 года, несмотря на постепенный рост в амбулаторных условиях.
Сегмент центров амбулаторной хирургии является самым быстрорастущим сегментом на европейском рынке эндоскопического гемостаза, регистрируя CAGR 8,1%. Этот рост обусловлен переходом эндоскопических процедур из больниц в амбулаторные учреждения, растущим спросом на экономически эффективные и минимально инвазивные вмешательства и растущим внедрением однодневной диагностической и терапевтической эндоскопии по всей Европе.
- канал распределения
На основе канала распределения рынок сегментируется на прямые продажи и косвенные продажи, а косвенные продажи далее сегментируются на онлайн- и офлайн-каналы. По прогнозам, в 2026 году сегмент косвенных продаж будет доминировать на европейском рынке эндоскопических гемостазов с наибольшей долей рынка 58,48%, поскольку закупки в основном осуществляются через дистрибьюторов, групповые закупочные организации и региональные сети медицинских поставок. Косвенные каналы широко предпочтительны из-за их способности предлагать комплексные продукты, поддержку управления запасами и более широкий географический охват, особенно на развивающихся рынках и децентрализованных системах здравоохранения. Больницы и амбулаторные центры часто полагаются на дистрибьюторские источники, чтобы обеспечить постоянную доступность устройств критического гемостаза при оптимизации затрат на закупку. Ожидается, что эта структура распределения продолжит стимулировать более широкое внедрение косвенных каналов продаж в течение прогнозируемого периода.
Сегмент косвенных продаж является самым быстрорастущим каналом дистрибуции на европейском рынке эндоскопического гемостаза, регистрируя CAGR 7,9%. Этот рост обусловлен растущей ролью сторонних дистрибьюторов, групповых закупочных организаций (GPO) и региональных поставщиков медицинских устройств в улучшении доступа на рынок в различных европейских системах здравоохранения.
Европейский эндоскопический гемостаз Региональный анализ рынка
- Германия представляет собой один из наиболее важных рынков для эндоскопических гемостазов, поддерживаемый передовой инфраструктурой здравоохранения, сильной производственной базой медицинских устройств и высоким уровнем внедрения минимально инвазивных эндоскопических процедур. Постоянные клинические инновации в гастроэнтерологии, большой объем диагностических и терапевтических эндоскопий, а также строгие клинические и нормативные стандарты способствуют постоянному внедрению передовых механических, термических и актуальных решений гемостаза в больницах и специализированных центрах ухода.
- В Великобритании растет спрос на технологии эндоскопического гемостаза, поскольку растущая распространенность желудочно-кишечных расстройств, старение населения и растущий акцент на минимально инвазивных подходах к лечению меняют клиническую практику. Медицинские работники все больше внимания уделяют повышению безопасности пациентов, снижению осложнений, связанных с процедурами, и повышению клинической эффективности, что ускоряет внедрение надежных и экономически эффективных устройств гемостаза в государственных и частных медицинских учреждениях.
- Франция продолжает выступать в качестве ключевого центра роста рынка эндоскопического гемостаза, что обусловлено сильным клиническим опытом в гастроэнтерологии, расширением доступа к передовым эндоскопическим услугам и увеличением внимания к ориентированному на пациента и ориентированному на результат уходу. Акцент страны на клиническом качестве, безопасности и технологических инновациях побуждает учреждения здравоохранения принимать передовые решения гемостаза, которые улучшают процедурные результаты, поддерживают минимально инвазивные вмешательства и соответствуют меняющимся нормативным и медицинским стандартам.
Немецкий рынок эндоскопического гемостаза
Немецкий рынок эндоскопического гемостаза набирает обороты благодаря большому объему желудочно-кишечных эндоскопических процедур и раннему внедрению передовых методов терапевтической эндоскопии. Немецкие больницы и специализированные клиники уделяют большое внимание клинической точности, процедурной надежности и выбору устройств на основе фактических данных, что обеспечивает постоянный спрос на высокопроизводительные механические и энергетические гемостазы. Кроме того, наличие ведущих производителей медицинских изделий, хорошо зарекомендовавших себя программ клинической подготовки, а также строгих нормативных требований и стандартов качества способствует быстрому внедрению технологически совершенных устройств гемостаза. Ориентация Германии на процедурную стандартизацию, безопасность пациентов и оптимизацию результатов укрепляет ее позиции на европейском рынке, ориентированном на технологии и инновации..
Эндоскопический гемостаз в Великобритании Market Insight
Рынок эндоскопического гемостаза в Великобритании продолжает расширяться, поскольку поставщики медицинских услуг отдают приоритет минимально инвазивным путям лечения, эффективности в эндоскопических блоках и уменьшению осложнений, связанных с процедурой. Растущая частота желудочно-кишечных кровотечений в сочетании с растущим спросом на услуги эндоскопии NHS ускоряет внедрение экономически эффективных, простых в использовании устройств гемостаза, которые поддерживают высокую процедурную пропускную способность. Сильный акцент на клинических рекомендациях, ценностном уходе и стандартизированных протоколах лечения формирует решения о покупке, в то время как растущее использование амбулаторных и эндоскопических настроек дневного ухода еще больше поддерживает спрос. Эти факторы коллективно позиционируют рынок Великобритании как один, обусловленный доступом, эффективностью и масштабируемым клиническим внедрением, а не концентрацией производства устройств..
Доля рынка эндоскопического гемостаза в Европе
Отрасль специализированных пищевых ингредиентов в основном возглавляется хорошо зарекомендовавшими себя компаниями, в том числе:
- Микротехнологическая эндоскопия (Китай)
- Taewoong Medical Co., Ltd. (Южная Корея)
- Ovesco Endoscopy AG (Германия)
- Apollo Endosurgery, Inc. (США)
- Argon Medical Devices, Inc. (США)
- Olympus Corporation (Япония)
- Бостонская научная корпорация (США)
- CONMED Corporation (США)
- Medtronic (Ирландия)
- Кук (США)
- ERBE Elektromedizin GmbH (Германия)
- Karl Storz SE & Co. KG (Германия)
- Pentax Medical (Япония)
- Endoskopie Technik Gerhard (Германия)
- Merit Medical Systems, Inc. (США)
- Diversatek, Inc. (США)
- STERIS plc (Великобритания)
- B. Braun SE (Германия)
- Duomed Group (Бельгия)
Последние разработки на европейском рынке эндоскопических гемостазов
- В декабре 2025 года Olympus утроила свои обязательства по корпоративному венчурному фонду, запустив Olympus Innovation Ventures Fund II с дополнительными 150 миллионами долларов для инвестиций в стартапы MedTech, ориентированные на эндоскопию, диагностику, цифровое здравоохранение и связанные с ними инновационные области для укрепления долгосрочного роста и технологического лидерства.
- В октябре 2025 года Boston Scientific объявила о соглашении о приобретении частной компании Nalu Medical, Inc., специализирующейся на имплантируемых технологиях нейростимуляции при хронической боли. Приобретение было направлено на укрепление портфеля нейромодуляции Boston Scientific, ускорение инноваций в управлении болью и расширение возможностей лечения пациентов с хроническими болями.
- В октябре 2025 года корпорация CONMED объявила о стратегическом выходе из своего гастроэнтерологического бизнеса, отказавшись от своих продуктовых линеек GI и связанных с ними активов в рамках реорганизации портфеля, чтобы сосредоточиться на основных хирургических и ортопедических решениях. Этот шаг был позиционирован для оптимизации фокуса продуктов компании, активизации инвестиций в быстрорастущие процедурные области и улучшения долгосрочных профилей доходов и маржи путем перераспределения ресурсов в сторону флагманских платформ устройств CONMED и новых технологий.
- В августе 2025 года Управление по санитарному надзору за качеством пищевых продуктов и медикаментов США (FDA) одобрило первое в своем роде клиническое исследование ADVANCE EVERO 18 с эверолимусным покрытием PTA баллонного катетера, что позволило оценить его безопасность и эффективность при лечении симптоматического заболевания периферических артерий. Это решение стало важной вехой для технологии воздушных шаров с лекарственным покрытием, поддерживая продвижение Куком эндоваскулярной терапии следующего поколения, направленной на снижение рестеноза и улучшение долгосрочной проходимости сосудов у пациентов с артериальной болезнью нижних конечностей.
- В июле руководящие принципы ESGE по актуальным гемостатическим агентам сообщили об остановке крови Анкаферда с 73%-100% показателями гемостаза для источников кровотечения ЖКТ, таких как язвы и злокачественные опухоли при эндоскопическом использовании.
SKU-
Получите онлайн-доступ к отчету на первой в мире облачной платформе рыночной аналитики
- Интерактивная панель анализа данных
- Панель анализа компании для возможностей с высоким потенциалом роста
- Доступ аналитика-исследователя для настройки и запросов
- Анализ конкурентов с помощью интерактивной панели
- Последние новости, обновления и анализ тенденций
- Используйте возможности сравнительного анализа для комплексного отслеживания конкурентов
Содержание
1 Введение
1.1 Цели исследования
1.2 Маркетологическое определение
1.3 ОБЩЕСТВО ЕВРОПЕЙСКОЙ ЭНДОСКОПИЧЕСКОЙ ГЕМОСТАССКОЙ МАРКЕТ
1.4 Ограничения
1.5 МАРКЕТЫ
2 МАРКЕТНАЯ СЕГМЕНТАЦИЯ
2.1 Приняты меры
2.2 ГЕОГРАФИЧЕСКАЯ СКОПА
2,3 года, присланные на обучение
2.4 КУРРЕНСИЯ И ПРИЧИНА
2.5 DBMR TRIPOD DATA VALIDATION
2.6 Первичное Интервью с ключевыми лидерами
2.7 МУЛЬТИВАРИАТНОЕ МОДЕЛЛирование
2.8 DBMR MARKET POSITION GRID
2.9 ВЕНДОР ДЕЛАЕТ АНАЛИЗИС
2.1 МАРКЕТНЫЙ КОВЕРАЖНЫЙ ГРИД ПОЛЬЗОВАТЕЛЯ
2.11 Вторичные источники
2.12 Предложения
3 ИСПОЛНИТЕЛЬНАЯ РЕЗЮМЕ
4 Премиум Впечатления
4.1 Портеры пять сил Аналитика
4.2 Брендовый обзор
4.2.1 КОМПАРАТИВНЫЙ АНАЛИЗ КЛЮЧЕВЫХ ИГРАТЕЛЕЙ ЕВРОПЫ
4.2.1.1 КОМПАРАТИВНЫЙ НАРАТИВ БРЭНДА (АНАЛИТИЧЕСКИЙ ОБЗОР)
4.2.2 ПРОДУКТ ВО ВНИМАНИЕ БРАНДА - ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИС
4.2.2.1 ПРОДУКТНЫЙ ОБЗОР — ЭНДОСКОПические решения по гемостазу
4.2.2.2 КЛИНИЧЕСКОЕ ВЗГЛЯДЕНИЕ - ВЛИЯНИЕ НА КЛИНИЧЕСКОЕ ВОПРОСЫ ЕВРОПЫ
4.3 Покупка потребителями
4.3.1 Клиническая эффективность и результаты лечения
4.3.2 СТОИМОСТЬ И СООТВЕТСТВЕННОСТЬ
4.3.3 Сотрудничество и интеграция
4.3.4 Требования к обучению и простота использования
4.3.5 Соблюдение политики в области здравоохранения
4.3.6 ПРОЦУРЕНТНЫЕ ПРОЦЕССЫ И ЗАПАДНЫЕ СОГЛАШЕНИЯ
4.3.7 ВЛИЯНИЕ КЛИНИЧЕСКИХ СТАКЕХОЛДЕРОВ
4.3.8 ПРАВИЛА ПОКУПАТЕЛЯ
4.3.9 Включение
4.4 Патентный анализ
4.4.1 КАЧЕСТВО И СИЛЬНОСТЬ ПАТЕНТА - ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИС
4.4.2 СТРАНА ПАТЕНТНАЯ ЗЕМЛЯ
4.4.3 ИС-стратегия и управление
4.4.4 Лицензирование и сотрудничество
4.5 RAW MATERIAL COVERAGE
4.5.1 МЕТАЛЛЬНЫЕ КОМПОНЕНТЫ И АЛЛОЙСКИЕ СИСТЕМЫ
4.5.2 Полимерные и синтетические материалы
4.5.3 Естественные и биосопутствующие материалы
4.5.4 ЭЛЕКТРИЧЕСКИЕ И ТЕРМИЧЕСКИЕ СИСТЕМЫ
4.5.5 ПРЕДСТАВЛЯЕМЫЕ АКЦЕССОРЫ И МАТЕРИАЛЫ ДЛЯ ЛЮДЕЙ
4.6 ТЕХНОЛОГИЧЕСКИЕ ПРИНЯТИЯ В ЕВРОПЕ ЭНДОСКОПИЧЕСКИЙ МАРКЕТ ГЕМОСТАЗА
4.6.1 Интенсивные инструменты механического гемостазиса
4.6.2 УПРАВЛЕНИЕ ТЕРМИЧЕСКИХ СОГЛАШЕНИЙ И ЭНЕРГИЧЕСКИХ СИСТЕМ
4.6.3 ТЕПИЧЕСКИЕ ГЕМОСТАТИЧЕСКИЕ АГЕНТЫ
4.6.4 ИНТЕГРАЦИЯ С АДЖУНКТИВНЫМИ ДИАГНОСТИЧЕСКИМИ ТЕХНОЛОГИЯМИ
4.6.5 Разработка многофункциональных и производственных решений
4.6.6 Усовершенствованные системы ЭРГОНОМИКИ ПОЛЬЗОВАТЕЛЯ И ДЕПЛОЙМЕНТОВ РЕШЕНИЯ
4.6.7 Включение
4.7 ЦЕННЫЙ АНАЛИЗ
4.7.1 Проверка
4.7.2 Сырые материалы и компоненты
4.7.3 DEVICE DESIGN, R&D & MANUFACTURING
4.7.4 Процессинг, завершение и стерилизация
4.7.5 Упаковка и размещение
4.7.6 ЛОГИСТИКА, ДИСТРИБУЦИЯ И КОММЕРЦИАЛИЗАЦИЯ
4.7.7 Включение
4.8 Критерии отбора вендоров
4.8.1 КЛИНИЧЕСКАЯ ПЕРФОРМА
4.8.2 СОСТОЯНИЕ ПРАВИТЕЛЬСТВА И ПРИНЯТИЙ
4.8.3 ПРОГРАММНЫЕ СТОИМОСТИ И ОБЩИЕ ПРАВИЛА
4.8.4 Вспомогательный ростовщичество и логистика
4.8.5 Поддержка, обучение и клиническое обучение
4.8.6 СИСТЕМЫ КАЧЕСТВА И ПРЕДОСТАВЛЕНИЯ ПРАВИЛА
4.8.7 РЕПУТАЦИЯ, СЛУЖБА И ПОСТ-МАРКЕТНАЯ ПЕРФОРМАЦИЯ
4.8.8 Инновации и будущее
4.9 Оценка КОМПАНИИ
4.1 ПРОФИТ МАРЖИН СКЕНАРИО
4.10.1 Водители, осуществляющие маргинальное движение
4.10.2 ЦЕННОСТЬ
4.10.3 Региональные различия
4.10.4 Будущие СЦЕНАРИИ
4.11 Изменение климата
4.11.1 ОБЩИЕ КОНЦЕРНЫ
4.11.2 Ответы промышленных предприятий
4.11.3 Роль правительства
4.11.4 Аналитические рекомендации
4.12 ИННОВАЦИОННЫЙ ТРАКСЕР И СТРАТЕГИЧЕСКИЙ АНАЛИЗ
4.12.1 ВАЖНЫЕ ДЕЛА И СТРАТЕГИЧЕСКИЕ АНАЛИЗЫ
4.12.1.1 Совместные мероприятия
4.12.1.2 МЕРГЕРЫ И КАКУИСИЦИИ
4.12.1.3 Лицензирование и партнерство
4.12.1.4 Сотрудничество в области технологии
4.12.2 СТРАТЕГИЧЕСКИЕ ДАЙВЕНЦИИ
4.12.3 Стадия развития
4.12.4 ТИМЕЛИНЫ И МИЛЕСТОНЫ
4.12.5 Информационные стратегии и методологии
4.12.6 ОБЯЗАТЕЛЬСТВО РИСКА И ИМЕНИЕ
4.12.7 Будущее
4.13 ГРАНИЧЕСКИЙ АНАЛИЗ
4.13.1 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ И СИЛЬНОСТИ
4.13.2 Обязательные и коагуляционные решения
4.13.3 Практика регулирования и возмещения
4.14 Вспомогательный анализ цепи
4.14.1 Проверка
4.14.2 ЛОГИСТИЧЕСКИЙ КОСТ СКЕНАРИО
4.14.3 ВАЖНОСТЬ ЛОГИСТИЧЕСКИХ УСЛУГ-ПРОВИДЕНТОВ
4.14.4 Вывод
5 ТАРИФОВ И ИМПАКТ НА МАРКЕТ
5.1 Проверка
5.2 Коэффициент ТАРИФФ (S) В ТОП-5 РЫНКОВ СТРАНЫ
5.3 ВНИМАНИЕ: Локальное производство V/S ВАЖНОЕ ОТНОШЕНИЕ
5.4 Критерии выбора вендоров
5.5 ПРИМЕЧАНИЕ В СУПЛЕННОЙ ЦЕПЕ
5.5.1 Сырьевое материальное обеспечение
5.5.2 Производство и производство
5.5.3 ЛОГИСТИКА И ДИСТРИБУЦИЯ
5.5.4 Ценообразование и позиция рынка
5.6 Индустриальные участники: ПРОАКТИВНЫЕ ДВИЖЕНИЯ
5.6.1 Оптимизация цепочки поставок
5.6.2 Совместные мероприятия
5.7 Последствия для цен
5.8 ИНКЛИНАЦИЯ ПРАВИЛА
5.8.1 Геополитическое положение
5.8.2 Торговые Партнёрские Партнёрские Компании
5.8.2.1 Соглашения о свободной торговле
5.8.2.2 УСТАНОВЛЕНИЯ АЛЛИЯНСОВ
5.8.3 СТАТУСНАЯ АККРЕДИТАЦИЯ (ВКЛЮЧАЯ MFTN)
5.8.4 Гостевой путь исправления
5.8.4.1 ВАЖНЫЕ СХЕМЫ ДЛЯ УСТАНОВЛЕНИЯ ПРОИЗВОДСТВА
5.8.4.2 УСТАНОВЛЕНИЕ ОЭЗ/МЕЖДУНАРОДНЫХ ПАРКОВ
6 КОНВЕРАЦИЯ ПРАВИЛА
6.1 Продукты
6.2 Удостоверенные стандарты
6.3 Стандарты безопасности
6.3.1 МАТЕРИАЛЬНАЯ РУКОВОДКА И СТОРОГА
6.3.2 Транспорт и меры предосторожности
6.3.3 Идентификационные данные ГАЗАРД
7 МАРКЕТНЫЙ ОБЗОР
7.1 Водители
7.1.1 Разрыв в каскадах для искусственного кровотока
7.1.2 Растущее население, ведущее в нетрезвом виде требование в отношении ОПАСНОСТИ
7.1.3 Принятие минимально инвазивных эндоскопических процедур
7.1.4 ПРОГРАММЫ РАЗВИВАНИЯ И ПРОГРАММЫ ПРОГРАММЫ СКРЫТИЯ ДЛЯ ГАСТРОИНТЕСТИНАЛЬНОЙ ЭНДОСКОПИИ
7.2 УВЕДОМЛЕНИЯ
7.2.1 Высокая стоимость и техническое соответствие ЭНДОСКОПИЧЕСКИХ ГЕМОСТАЗИСОВ
7.2.2 Ограниченная осведомленность в странах с низким уровнем дохода
7.3 Положения
7.3.1 ТЕХНОЛОГИЧЕСКИЕ ПРИНЯТИЯ В ГЕМОСТАССКИХ РЕШЕНИЯХ
7.3.2 ОБЕСПЕЧЕНИЕ В РАЗВИВАЮЩИХ РЫНКАХ (Азия-Пацифик, Латинская Америка)
7.3.3 Растущее принятие решений по эндоскопии домашних хозяйств и капсул
7.4 Вызовы
7.4.1 Вопросы возмещения расходов на эндоскопические процедуры
7.4.2 УСЛУГИ В ЭНДОСКОПНОЙ РЕПРЕССИИ И СТЕРИЛИЗАЦИОННЫХ ПРОЦЕССАХ
8 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ
8.1 Проверка
8.2 Механические гемостазы
8.2.1 МЕХАНИЧЕСКИЕ РЕШЕНИЯ ГЕМОСТАЗА В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ТИПУ
8.2.1.1 Гемостатические отклонения
8.2.1.1.1 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАЗИСНЫХ РЕШЕНИЯХ, ПО ТИПУ
8.2.1.1.1.1 КЛИПЫ СПОСОБНОСТИ
8.2.1.1.1.2 КЛИПЫ СЛЕДУЮЩИХ СКОРОН (ОТС)
8.2.1.1.2 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАЗИСНЫХ РЕШЕНИЯХ МАТЕРИАЛЬНЫЙ
8.2.1.1.2.1 БЕЗОПАСНЫЙ СТЕЛЬ
8.2.1.1.2.2 Титаниум
8.2.1.2 Обязательные устройства
8.2.1.2.1 ОБЯЗАТЕЛЬНЫЕ УСЛОВИЯ В МЕХАНИЧЕСКИХ ГЕМОСТАЗИСНЫХ УСЛОВИЯХ МАРКЕТ
8.2.1.2.1.1 Вариативные стычки
8.2.1.2.1.2 Устройства для Эзофагической Обвязки
8.2.1.3 Гемостатические силы
8.2.1.4 Другие
8.2.2 МЕХАНИЧЕСКИЕ РЕШЕНИЯ ГЕМОСТАЗА В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ
8.2.2.1 Северная Америка
8.2.2.2 Европа
8.2.2.3 ASIA-PACIFIC
8.2.2.4 Южная Америка
8.2.2.5 Средний Восток и Африка
8.3 Термические решения
8.3.1 ТЕМРАЛЬНЫЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ТИПУ
8.3.1.1 Коагуляторы ПЛАСМА АРГОН
8.3.1.2 Проблемы с подогревом
8.3.1.3 Биполярные решения по регулированию
8.3.1.3.1 Биполярная коагуляция на рынке цифровых устройств, по типу энергии
8.3.1.3.1.1 Монополярный
8.3.1.3.1.2 Биполярный
8.3.1.3.2 Биполярная коагуляция на рынке цифровых устройств
8.3.1.3.2.1 СВЕЩАНИЕ ЖИЗНИ
8.3.1.3.2.2 Меньшее кровотечение
8.3.1.4 Другие
8.3.2 ТЕМРАЛЬНЫЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО РЕГИОНУ
8.3.2.1 Северная Америка
8.3.2.2 Европа
8.3.2.3 ASIA-PACIFIC
8.3.2.4 Южная Америка
8.3.2.5 Средний Восток и Африка
8.4 Топические агенты и препятствия
8.4.1 ТЕПИЧЕСКИЕ АГЕНТЫ И ИНЖЕКТИВЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ
8.4.1.1 Введение Эпинефрина
8.4.1.2 ГЕМОСТАТИЧЕСКАЯ СВЯЗЬ (ВОЛОС)
8.4.1.3 Гемостатические поля
8.4.1.4 ФИБРИНСКИЕ СИЛАНСЫ
8.4.1.5 СКЛЕРОСИВНЫЕ АГЕНТЫ
8.4.2 ТЕПИЧЕСКИЕ АГЕНТЫ И ОТВЕТСТВЕННОСТИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ
8.4.2.1 Северная Америка
8.4.2.2 Европа
8.4.2.3 ASIA-PACIFIC
8.4.2.4 Южная Америка
8.4.2.5 Средний Восток и Африка
8.5 Другие
8.5.1 ДРУГИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ
8.5.1.1 Северная Америка
8.5.1.2 Европа
8.5.1.3 ASIA-PACIFIC
8.5.1.4 Южная Америка
8.5.1.5 Средний Восток и Африка
9 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРЕДСТАВЛЕННОМУ типу
9.1 Проверка
9.2 ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ
9.2.1 ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ТИПУ
9.2.1.1 УПРАВЛЕНИЕ КЛЮЧЕВЫМ КЛЮЧЕМ
9.2.1.2 Вариабельное кровотечение
9.2.1.3 ДЬЕУЛАФОЙ ЛИЗИОН
9.2.2 ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ
9.2.2.1 Северная Америка
9.2.2.2 Европа
9.2.2.3 ASIA-PACIFIC
9.2.2.4 Южная Америка
9.2.2.5 Средний Восток и Африка
9.3 ГЛАВНАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ
9.3.1 ГЛАВНАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ТИПУ
9.3.1.1 Колоническое кровотечение
9.3.1.2 Диверсионное кровотечение
9.3.2 ГЛАВНАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО РЕГИОНУ
9.3.2.1 Северная Америка
9.3.2.2 Европа
9.3.2.3 ASIA-PACIFIC
9.3.2.4 Южная Америка
9.3.2.5 Средний Восток и Африка
9.4 Бронхоскопический гемостаз
9.4.1 БРОНКОСКОПНЫЙ ГЕМОСТАЗ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТ
9.4.1.1 Северная Америка
9.4.1.2 Европа
9.4.1.3 ASIA-PACIFIC
9.4.1.4 Южная Америка
9.4.1.5 Средний Восток и Африка
9.5 Другие
9.5.1 ДРУГИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ
9.5.1.1 Северная Америка
9.5.1.2 Европа
9.5.1.3 ASIA-PACIFIC
9.5.1.4 Южная Америка
9.5.1.5 Средний Восток и Африка
10 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ
10.1 Проверка
10.2 Гастроинтестинальное кровотечение
10.2.1 Гастроинтестинальное кровотечение в эндоскопическом гемостазисе, по типу
10.2.1.1 ПЕПТИЧЕСКОЕ КЛЮЧЕНИЕ
10.2.1.2 Эзофагические различия
10.2.1.3 Контрагенты
10.2.2 ГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРОДУКТНОМУ ТИПУ
10.2.2.1 Решения по механическому гемостазу
10.2.2.2 Технические решения
10.2.2.3 ТЕПИЧЕСКИЕ АГЕНТЫ И НЕПРЯМОСТИ
10.2.2.4 Другие
10.2.3 ГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО РЕГИОНУ
10.2.3.1 Северная Америка
10.2.3.2 Европа
10.2.3.3 ASIA-PACIFIC
10.2.3.4 Южная Америка
10.2.3.5 Средний Восток и Африка
10.3 Негастроинтестинальное кровотечение
10.3.1 Негастроинтестинальное кровотечение в эндоскопическом гемостазисе, по типу
10.3.1.1 NASAL BLEEDING (EPISTAXIS)
10.3.1.2 Постхирургическое кровотечение
10.3.2 Негастроинтестинальное кровотечение в эндоскопическом гемостазисе, по типу продукта
10.3.2.1 Решения по механическому гемостазу
10.3.2.2 Термические решения
10.3.2.3 ТЕПИЧЕСКИЕ АГЕНТЫ И НЕОБХОДИМОСТИ
10.3.2.4 Другие
10.3.3 Негастроинтестинальное кровотечение в эндоскопическом гемостазном рынке, Регион, 2018-2033 годы (USD THOUSAND)
10.3.3.1 Северная Америка
10.3.3.2 Европа
10.3.3.3 ASIA-PACIFIC
10.3.3.4 Южная Америка
10.3.3.5 Средний Восток и Африка
10.4 УПРАВЛЕНИЕ ТРАУМой
10.4.1 УПРАВЛЕНИЕ ТРАУМОЙ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТОВ
10.4.1.1 Технические решения по гемостазу
10.4.1.2 ТЕРМИЧЕСКИЕ УСЛОВИЯ
10.4.1.3 ТЕПИЧЕСКИЕ АГЕНТЫ И НЕОБХОДИМОСТИ
10.4.1.4 Другие
10.4.2 УПРАВЛЕНИЕ ТРАУМОЙ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО РЕГИОНУ
10.4.2.1 Северная Америка
10.4.2.2 Европа
10.4.2.3 ASIA-PACIFIC
10.4.2.4 Южная Америка
10.4.2.5 Средний Восток и Африка
10.5 Другие
10.5.1 ДРУГИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ
10.5.1.1 Северная Америка
10.5.1.2 Европа
10.5.1.3 ASIA-PACIFIC
10.5.1.4 Южная Америка
10.5.1.5 Средний Восток и Африка
11 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПОЛЬЗОВАТЕЛЬ
11.1 Проверка
11.2 Основы
11.2.1 ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, ПО ТИПУ
11.2.1.1 ПУБЛИКА
11.2.1.2 Приватный
11.2.2 ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО РЕГИОНУ
11.2.2.1 Северная Америка
11.2.2.2 Европа
11.2.2.3 ASIA-PACIFIC
11.2.2.4 Южная Америка
11.2.2.5 Средний Восток и Африка
11.3 АМБУЛАТОРНЫЕ ЦЕНТРЫ ИСКУССТВ
11.3.1 АМБУЛАТОРНЫЕ ЦЕНТРЫ ИССЛЕДОВАТЕЛЕЙ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО РЕГИОНУ
11.3.1.1 Северная Америка
11.3.1.2 Европа
11.3.1.3 ASIA-PACIFIC
11.3.1.4 Южная Америка
11.3.1.5 Средний Восток и Африка
11.4 СПЕЦИАЛЬНАЯ КЛИНИКА
11.4.1 СПЕЦИАЛЬНАЯ КЛИНИКА В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО РЕГИОНУ
11.4.1.1 Северная Америка
11.4.1.2 Европа
11.4.1.3 ASIA-PACIFIC
11.4.1.4 Южная Америка
11.4.1.5 Средний Восток и Африка
11.5 Другие
11.5.1 ДРУГИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ
11.5.1.1 Северная Америка
11.5.1.2 Европа
11.5.1.3 ASIA-PACIFIC
11.5.1.4 Южная Америка
11.5.1.5 Средний Восток и Африка
12 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, КАНАЛ ДИСТРИБУЦИИ
12.1 Проверка
12.2 Непрямые продажи
12.2.1 Непрямые продажи в эндоскопическом гемостазе, по типу
12.2.1.1 ФЛИН
12.2.1.2 Онлайн
12.2.2 Непрямые продажи на рынке эндоскопических гемостазов
12.2.2.1 Северная Америка
12.2.2.2 Европа
12.2.2.3 ASIA-PACIFIC
12.2.2.4 Южная Америка
12.2.2.5 Средний Восток и Африка
12.3 Прямые продажи
12.3.1 ПРАВИЛЬНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО РЕГИОНУ
12.3.1.1 Северная Америка
12.3.1.2 Европа
12.3.1.3 ASIA-PACIFIC
12.3.1.4 Южная Америка
12.3.1.5 Средний Восток и Африка
13 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ
13.1 Европа
13.1.1 Германия
13.1.2 Великобритания.
13.1.3 Франция
13.1.4 Италия
13.1.5 Испания
13.1.6 Планы
13.1.7 БЕЛГИУМ
13.1.8 Швейцария
13.1.9 СВЕДЕН
13.1.10 Денмарк
13.1.11 Норвежский
13.1.12 Финляндия
13.1.13 Россия
13.1.14 Тюркей
13.1.15 ПЕСТ ЕВРОПЫ
14 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ: КОМПАНИЯ ЛЭНДСКАП
14.1 КОМПАНИЯ ДЛЯ АНАЛИЗА: ГЛОБАЛ
15 СВОТ АНАЛИЗ
16 КОМПАНИЯ ПОЛУЧИТЕЛЬСТВА
16.1 OLYMPUS CORPORATION
16.1.1 КОМПАНИЯ СНАПШОТ
16.1.2 РЕВЕНУАЛЬНЫЙ АНАЛИЗ
16.1.3 КОМПАНИЯ ДЛЯ АНАЛИЗА
16.1.4 ПРОДУКТ ПОРТФОЛИО
16.1.5 ПРОЕКТ РАЗВИТИЯ
16.2 Бостонская научная корпорация
16.2.1 КОМПАНИЯ СНАПШОТ
16.2.2 РЕВЕННЫЙ АНАЛИЗ
16.2.3 КОМПАНИЯ ДЛЯ АНАЛИЗА
16.2.4 ПРОДУКТ ПОРТФОЛИО
16.2.5 ПРОЕКТ РАЗВИТИЯ
16.3 СРЕДНАЯ КОРПОРАЦИЯ
16.3.1 КОМПАНИЯ СНАПШОТ
16.3.2 РЕВЕННЫЙ АНАЛИЗ
16.3.3 КОМПАНИЯ ДЛЯ АНАЛИЗА
16.3.4 ПРОДУКТ ПОРТФОЛИО
16.3.5 РАЗВИТИЕ ПРИЛОЖЕНИЯ
16.4 Медронический
16.4.1 КОМПАНИЯ СНАПШОТ
16.4.2 РЕВЕННЫЙ АНАЛИЗ
16.4.3 КОМПАНИЯ ДЛЯ АНАЛИЗА
16.4.4 ПРОДУКТ ПОРТФОЛИО
16.4.5 РАЗВИТИЕ ПРИЛОЖЕНИЯ
16.5 Кука
16.5.1 КОМПАНИЯ СНАПШОТ
16.5.2 КОМПАНИЯ ДЛЯ АНАЛИЗА
16.5.3 ПРОДУКТ ПОРТФОЛИО
16.5.4 ПРОЕКТ РАЗВИТИЯ
16.6 Анкаферд
16.6.1 КОМПАНИЯ СНАПШОТ
16.6.2 ПРОДУКТ ПОРТФОЛИО
16.6.3 РЕКОМЕНДАЦИЯ
16.7 B. BRAUN SE
16.7.1 КОМПАНИЯ СНАПШОТ
16.7.2 ПРОДУКТ ПОРТФОЛИО
16.7.3 РЕКОМЕНДАЦИЯ
16.8 CREO MEDICAL GMBH
16.8.1 КОМПАНИЯ СНАПШОТ
16.8.2 ПРОДУКТ ПОРТФОЛИО
16.8.3 ПРОЕКТ РАЗВИТИЯ
16.9 Диверсатек, ИНК.
16.9.1 КОМПАНИЯ СНАПШОТ
16.9.2 ПРОДУКТ ПОРТФОЛИО
16.9.3 РЕКОМЕНДАЦИЯ
16.1 Дуомедная группа
16.10.1 КОМПАНИЯ СНАПШОТ
16.10.2 ПРОДУКТ ПОРТФОЛИО
16.10.3 РЕКОМЕНДАЦИЯ
16.11 ENDOCLOT PLUS, INC.
16.11.1 КОМПАНИЯ СНАПШОТ
16.11.2 ПРОДУКТ ПОРТФОЛИО
16.11.3 РЕКОМЕНДАЦИЯ
16.12 ЭРБЕ ЭЛЕКТРОМЕДИЗИН ГМБХ
16.12.1 КОМПАНИЯ СНАПШОТ
16.12.2 ПРОДУКТ ПОРТФОЛИО
16.12.3 ПРОЕКТ РАЗВИТИЯ
16.13 ФУЖИФИЛЬМА ХОЛДИНГСКАЯ КОРПОРАЦИЯ
16.13.1 КОМПАНИЯ СНАПШОТ
16.13.2 Ревеню АНАЛИЗ
16.13.3 ПРОДУКТ ПОРТФОЛИО
16.13.4 ПРОЕКТ РАЗВИТИЯ
16.14 Джонсон и Джонсон (Этикон)
16.14.1 КОМПАНИЯ СНАПШОТ
16.14.2 Ревеню АНАЛИЗ
16.14.3 ПРОДУКТ ПОРТФОЛИО
16.14.4 РЕКОМЕНДАЦИЯ
16.15 KARL STORZ SE & CO. KG, TUTTLINGEN
16.15.1 КОМПАНИЯ СНАПШОТ
16.15.2 ПРОДУКТ ПОРТФОЛИО
16.15.3 РЕКОМЕНДАЦИЯ
16.16 Медитация С.Р.Л.
16.16.1 КОМПАНИЯ СНАПШОТ
16.16.2 ПРОДУКТ ПОРТФОЛИО
16.16.3 ПРОЕКТ РАЗВИТИЯ
16.17 Микротехническая эндоскопия
16.17.1 КОМПАНИЯ СНАПШОТ
16.17.2 ПРОДУКТ ПОРТФОЛИО
16.17.3 ПРОЕКТ РАЗВИТИЯ
16.18 МТВ Эндоскопи Мануфакту
16.18.1 КОМПАНИЯ СНАПШОТ
16.18.2 ПРОДУКТ ПОРТФОЛИО
16.18.3 РЕКОМЕНДАЦИЯ
16.19 OVESCO ENDOSCOPY AG
16.19.1 КОМПАНИЯ СНАПШОТ
16.19.2 ПРОДУКТ ПОРТФОЛИО
16.19.3 РЕКОМЕНДАЦИЯ
16.2 PENTAX MEDICAL
16.20.1 КОМПАНИЯ СНАПШОТ
16.20.2 Решение спора
16.20.3 ПРОЕКТ РАЗВИТИЯ
16.21 СТЕРИС
16.21.1 КОМПАНИЯ СНАПШОТ
16.21.2 РЕВЕННЫЙ АНАЛИЗ
16.21.3 ПРОДУКТ ПОРТФОЛИО
16.21.4 ПРОЕКТ РАЗВИТИЯ
17 КОМПАНИЯ ПОЛУЧАЕТ ДИСТРИБУТОР
17.1 Решения БОСТОН IVY HEALTHCARE ограничены
17.1.1 КОМПАНИЯ СНАПШОТ
17.1.2 ПРОДУКТ ПОРТФОЛИО
17.1.3 ПРОЕКТ РАЗВИТИЯ
17.2 Кардинальное здоровье
17.2.1 КОМПАНИЯ СНАПШОТ
17.2.2 РЕВЕННЫЙ АНАЛИЗ
17.2.3 ПРОДУКТ ПОРТФОЛИО
17.2.4 РЕКОМЕНДАЦИЯ
17.3 Генри Шейн, ИНК.
17.3.1 КОМПАНИЯ СНАПШОТ
17.3.2 РЕВЕННЫЙ АНАЛИЗ
17.3.3 ПРОДУКТ ПОРТФОЛИО
17.3.4 РЕКОМЕНДАЦИЯ
17.4 МЦКЕССОН МЕДИЦИНСКО-СУРГИЧЕСКИЙ ИНК.
17.4.1 КОМПАНИЯ СНАПШОТ
17.4.2 ПРОДУКТ ПОРТФОЛИО
17.4.3 РЕКОМЕНДАЦИЯ
17,5 МФО Медикал
17.5.1 КОМПАНИЯ СНАПШОТ
17.5.2 ПРОДУКТ ПОРТФОЛИО
17.5.3 НОВЫЙ ДЕВЕПМЕНТ
18 вопросов
19 Связанные поправки
Список таблиц
СТАТЬЯ 1 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 2 ЕВРОПЕЙСКИЕ МЕХАНИЧЕСКИЕ ГЕМОСТАССКИЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 3 ЕВРОПЕЙСКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 4 ЕВРОПЕЙСКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ, МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 5 ЕВРОПЕЙСКИЕ ОБЯЗАТЕЛЬНЫЕ УСЛОВИЯ В МЕХАНИЧЕСКОМ ГЕМОСТАССКОМ УСЛУГЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 6 ЕВРОПЕЙСКИЕ МЕХАНИЧЕСКИЕ ГЕМОСТАЗИСНЫЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПРИ РЕГИОНЕ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 7 ЕВРОПЕЙСКИЕ ТЕРМИЧЕСКИЕ УБЕЖДЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 8 ЕВРОПЕЙСКАЯ БИПОЛАРНАЯ КОЛУЛЯЦИЯ В РЫНОКЕ ТЕРМИЧЕСКИХ РЕШЕНИЙ, ПО ЭНЕРГЕТИЧЕСКОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 9 ЕВРОПЕЙСКАЯ БИПОЛАРНАЯ КОНФЕРЕНЦИЯ В МАРКЕ ТРАНСПОРТНЫХ СРЕДСТВ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 10 ЕВРОПЕЙСКИЕ ТЕРМИЧЕСКИЕ УБЕЖДЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО РЕГИОНУ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 11 ЕВРОПЕЙСКИЕ ТЕМНЫЕ АГЕНТЫ И НЕПРЯМОСТИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 12 ЕВРОПЕЙСКИХ ТЕМНЫХ АГЕНТОВ И ИНЖЕКТАБЛЕЙ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПРИ РЕГИОНЕ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 13 ЕВРОПЫ ДРУГИХ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, ПРИ РЕГИОНЕ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 14 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 15 ЕВРОПЕЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 16 ЕВРОПЕЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПРИ РЕГИОНЕ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 17 ЕВРОПЕЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 18 ЕВРОПЕЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПРИ РЕГИОНЕ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 19 ЕВРОПЕЙСКИЙ БРОНКОСКОПИЧЕСКИЙ ГЕМОСТАЗ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПРИ РЕГИОНЕ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 20 ЕВРОПЕЙ ДРУГИХ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПРИ РЕГИОНЕ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 21 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 22 EUROPE GASTROINTESTINAL BLEEDING IN ENDOSCOPIC HEMOSTASIS MARKET, BYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 23 EUROPE GASTROINTESTINAL BLEEDING IN ENDOSCOPIC HEMOSTASIS MARKET, BY PRODUCT TYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 24 EUROPE GASTROINTESTINAL BLEEDING IN ENDOSCOPIC HEMOSTASIS MARKET, BY REGION, 2018-2033, (USD THOUSAND)
СТАТЬЯ 25 ЕВРОПЕЙСКОЕ НЕГАСТРОИНТЕСТИНОЕ КРОВЛЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 26 ЕВРОПЕЙСКОЕ НЕГАСТРОИНТЕСТИЧЕСКОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 27 EUROPE NON-GASTROINTESTINAL BLEEDING IN ENDOSCOPIC HEMOSTASIS MARKET, BY REGION, 2018-2033, (USD THOUSAND)
СТАТЬЯ 28 ЕВРОПЕЙСКОЕ ТРАУМНОЕ УПРАВЛЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 29 ЕВРОПЕЙСКОЕ ТРАУМНОЕ УПРАВЛЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПРИ РЕГИОНЕ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 30 ЕВРОПЫ ДРУГИХ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, ПРИ РЕГИОНЕ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 31 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 32 ЕВРОПЕЙСКИЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 33 ЕВРОПЕЙСКИЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПРИ РЕГИОНЕ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 34 ЕВРОПЕЙСКИЕ АМБУЛАТОРНЫЕ ЦЕНТРЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО РЕГИОНУ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 35 ЕВРОПЕЙСКАЯ СПЕЦИАЛИТИЧЕСКАЯ КЛИНИКА В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО РЕГИОНУ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 36 ЕВРОПЫ ДРУГИХ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, ПО РЕГИОНУ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 37 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО РАЗРАБОТКИ ЧАННЕЛЯ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 38 ЕВРОПЕЙСКИЕ ИНДИРЕКТИВНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 39 ЕВРОПЕЙСКИЕ ИНДИРЕКТНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПРИ РЕГИОНЕ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 40 ЕВРОПЕЙСКИЕ ПРАВИЛЬНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПРИ РЕГИОНЕ, 2018-2033, (USD THOUSAND)
СТАТЬЯ 41 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ, СТРАНА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 42 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 43 ЕВРОПЕЙСКИЕ МЕХАНИЧЕСКИЕ ГЕМОСТАССКИЕ ДЕВИЦИИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 44 ЕВРОПЕЙСКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 45 ЕВРОПЕЙСКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ, МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 46 ЕВРОПЕЙСКИЕ ОБЯЗАТЕЛЬНЫЕ УСЛОВИЯ В МЕХАНИЧЕСКОМ ГЕМОСТАССКОМ УСЛУГЕ, СТАТЬЯ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 47 ЕВРОПЕЙСКИЕ ТЕРМИЧЕСКИЕ РЕШЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 48 ЕВРОПЕЙСКАЯ БИПОЛАРНАЯ СОГЛАШЕНИЕ В РЫНОКЕ ТРАНСПОРТНЫХ РЕШЕНИЙ, ПО ЭНЕРГИЧЕСКОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 49 ЕВРОПЕЙСКАЯ БИПОЛАРНАЯ СОГЛАШЕНИЕ В МАРКЕ ТРАНСПОРТНЫХ СРЕДСТВ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 50 ЕВРОПЕЙСКИХ ТЕМНЫХ АГЕНТОВ И ИНЖЕКТИВОВ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 51 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 52 ЕВРОПЕЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 53 ЕВРОПЕЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 54 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 55 ЕВРОПЕЙСКИЙ ГАСТРОИНТЕСТИНАЛЬНЫЙ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТ, ВЫБОР, 2018-2033 (USD THOUSAND)
СТАТЬЯ 56 EUROPE GASTROINTESTINAL BLEEDING IN ENDOSCOPIC HEMOSTASIS MARKET, BY PRODUCT TYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 57 ЕВРОПЕЙСКОЕ НЕГАСТРОИНТЕСТИЧЕСКОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 58 EUROPE NON-GASTROINTESTINAL BLEEDING IN ENDOSCOPIC HEMOSTASIS MARKET, BY PRODUCT TYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 59 ЕВРОПЕЙСКОЕ ТРАУМНОЕ УПРАВЛЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 60 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 61 ЕВРОПЕЙСКИЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 62 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, КАНАЛ ДИСТРИБУЦИИ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 63 ЕВРОПЕЙСКИЕ ИНДИРЕКТИВНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 64 ГЕРМАНИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 65 ГЕРМАНИЙСКИЕ МЕХАНИЧЕСКИЕ ГЕМОСТАССКИЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 66 ГЕРМАНИЙСКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 67 ГЕРМАНИЙСКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 68 ОБЯЗАТЕЛЬНЫХ УБЕЖДЕНИЙ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ УБЕЖДЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 69 ГЕРМАНИЙСКИЕ ТЕРМИЧЕСКИЕ УБЕЖДЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 70 ГЕРМАНИЙСКАЯ БИПОЛАРНАЯ КОЛУЛЯЦИЯ В МАРКЕ ТЕРМИЧЕСКИХ РЕШЕНИЙ, ПО ЭНЕРГИЧЕСКОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 71 ГЕРМАНИЙСКАЯ БИПОЛАРНАЯ КОЛУЛЯЦИЯ В МАРКЕ ТРАНСПОРТНЫХ РЕШЕНИЙ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 72 ГЕРМАНИЙСКИЕ ТОПНЫЕ АГЕНТЫ И ИНЖЕКТИВЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 73 ГЕРМАНИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 74 ГЕРМАНИЯ ПОСЛЕ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 75 ГЕРМАНИЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 76 ГЕРМАНИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 77 ГЕРМАНИЯ ГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ВЫБОР, 2018-2033 (USD THOUSAND)
СТАТЬЯ 78 ГЕРМАНИЯ ГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 79 ГЕРМАНИЯ НЕГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ВЫБОР, 2018-2033 (USD THOUSAND)
СТАТЬЯ 80 ГЕРМАНИЯ НЕГАСТРОИНТЕСТИНАЛЬНОЕ СКЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 81 УПРАВЛЕНИЕ ГЕРМАНИИ ТРАУМЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 82 ГЕРМАНИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 83 ГЕРМАНИЙСКИЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 84 ГЕРМАНИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО РАЗРАБОТКИ ЧАННЕЛЯ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 85 ГЕРМАНИИ НЕДИРЕКТИВНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 86 ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАСИСКИЙ МАРКЕТ, В ПРИМЕЧАНИИ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 87 МЕХАНИЧЕСКИЕ ГЕМОСТАССКИЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 88 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 89 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ, МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 90 ОБЯЗАТЕЛЬНЫХ УБЕЖДЕНИЙ В МЕХАНИЧЕСКОМ ГЕМОСТАЗСКОМ УБЕЖДЕНИИ, КАЖДЫЙ ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 91 ТЕРМИЧЕСКИЕ УБЕЖДЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 92 БИПОЛАРНАЯ КОАГУЛЯЦИЯ В ТЕМНЫХ РЕШЕНИЯХ МАРКЕТ, ПО ЭНЕРГИЧЕСКОМУ ТИПУ, 2018-2033 (USD THOUSAND)
ПРИМЕЧАНИЕ 93 БИПОЛАРНАЯ СОГЛАШЕНИЕ В МАРКЕ ТЕМНЫХ УБЕЖДЕНИЙ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 94 ТОРГОВЫЕ АГЕНТЫ И ИНЖЕКТИВЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 95 ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
96 U.K. UPPER GASTROINTESTINAL ENDOSCOPY IN ENDOSCOPIC HEMOSTASIS MARKET, BYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 97 ГОСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 98 БЕЗОПАСНЫЙ ГЕМОСТАССКИЙ МАРКЕТ, ПОСТАНОВЛЕНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 99 ГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 100 ГАСТРОИНТЕСТИНАЛЬНОЕ СВЕТСТВО В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
Таблица 101 Великобритания Негастроинтестинальное кровотечение в эндоскопическом гемостазисе Маркет, Тип, 2018-2033 (USD THOUSAND)
СТАТЬЯ 102 Великобритания Негастроинтестинальное кровотечение в эндоскопическом гемостазисе, по типу продукта, 2018-2033 годы (USD THOUSAND)
СТАТЬЯ 103 УПРАВЛЕНИЕ ТРАУМЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 104 Британский эндоскопический гемостаз Маркет, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 105 ХОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 106 БЕЗОПАСНЫЙ ГЕМОСТАСИСКИЙ МАРКЕТ, КАНАЛ ДИСТРИБУЦИИ, 2018-2033 (USD THOUSAND)
Таблица 107 Непрямые продажи в эндоскопическом гемостазе, Тип, 2018-2033 (USD THOUSAND)
СТАТЬЯ 108 FRANCE ENDOSCOPIC HEMOSTASIS MARKET, BY PRODUCT TYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 109 ФРАНСОВЫЕ МЕХАНИЧЕСКИЕ ГЕМОСТАССКИЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 110 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ ФРАНЦИИ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 111 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ ФРАНЦИИ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ, МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 112 ФРАНСОВЫЕ ОБЯЗАТЕЛЬНЫЕ УСЛОВИЯ В МЕХАНИЧЕСКОМ ГЕМОСТАССКОМ УСЛУГЕ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 113 ФРАНЦЕВСКИЕ ТЕРМИЧЕСКИЕ РЕШЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 114 БИПОЛАРНАЯ КОНФЕРЕНЦИЯ ФРАНЦИИ В МАРКЕ ТРАНСПОРТНЫХ РЕШЕНИЙ, ПО ЭНЕРГИЧЕСКОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 115 БИПОЛАРНАЯ КОЛУЛЯЦИЯ ФРАНЦИИ В МАРКЕ ТРАНСПОРТНЫХ РЕШЕНИЙ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 116 ФРАНЦИИ ТОПИЧЕСКИЕ АГЕНТЫ И ИНЖЕКТИВЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 117 FRANCE ENDOSCOPIC HEMOSTASIS MARKET, BY PROCEDURE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 118 FRANCE UPPER GASTROINTESTINAL ENDOSCOPY IN ENDOSCOPIC HEMOSTASIS MARKET, BYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 119 ГОСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 120 ФРАНЦИЯ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 121 ФРАНЦИЯ ГАСТРОИНТЕСТИНАЛЬНОГО КЛЕЧЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 122 ФРАНЦИЯ ГАСТРОИНТЕСТИНАЛЬНОГО КЛЕЧЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 123 ФРАНЦИЯ НЕГАСТРОИНТЕСТИНАЛЬНОГО КЛЕЧЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 124 ФРАНЦИЯ НЕГАСТРОИНТЕСТИНАЛЬНОГО КЛЕЧЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 125 FRANCE TRAUMA MANAGEMENT IN ENDOSCOPIC HEMOSTASIS MARKET, BY PRODUCT TYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 126 ФРАНЦИЯ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 127 ФРАНЦЕВСКИЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 128 ФРАНЦИЯ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПО РАЗРАБОТКИ ЧАННЕЛЯ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 129 ИНДИРЕКТИВНЫЕ ПРОДАЖИ ФРАНЦИИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 130 ИТАЛИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 131 ИТАЛИЙСКИЕ МЕХАНИЧЕСКИЕ ГЕМОСТАССКИЕ ДЕВИЦИИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 132 ИТАЛИЙСКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 133 ИТАЛИЙСКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ, МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 134 ИТАЛЬНЫЕ ОБЯЗАТЕЛЬСТВА В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 135 ИТАЛИЙСКИЕ ТЕРМИЧЕСКИЕ УБЕЖДЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 136 ИТАЛИЙСКАЯ БИПОЛАРНАЯ КОЛУЛЯЦИЯ В МАРКЕ ТЕРМИЧЕСКИХ РЕШЕНИЙ, В ЭНЕРГИЧЕСКОМ ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 137 ИТАЛИЙСКАЯ БИПОЛАРНАЯ КОЛУЛЯЦИЯ В МАРКЕ ТРАНСПОРТНЫХ СРЕДСТВ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 138 ИТАЛИЙСКИЕ ТОПНЫЕ АГЕНТЫ И ИНЖЕКТИВЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 139 ИТАЛИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 140 ИТАЛИЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 141 ИТАЛИЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 142 ИТАЛИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 143 ИТАЛИЙСКИЙ ГАСТРОИНТЕСТИНАЛЬНЫЙ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 144 ИТАЛИЙСКИЙ ГАСТРОИНТЕСТИНАЛЬНЫЙ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 145 ИТАЛИЯ НЕГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ВЫБОР, 2018-2033 (USD THOUSAND)
СТАТЬЯ 146 ИТАЛИЯ НЕГАСТРОИНТЕСТИНАЛЬНОЕ СКЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 147 ИТАЛИЙСКОЕ ТРАУМНОЕ УПРАВЛЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 148 ИТАЛИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 149 ИТАЛИЙСКИЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, ВЫБОР, 2018-2033 (USD THOUSAND)
СТАТЬЯ 150 ИТАЛИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, КАНАЛ ДИСТРИБУЦИИ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 151 ИТАЛИЙСКИЕ ИНДИРЕКТИВНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 152 Испания ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАСС МАРКЕТ, В ТЕМЕ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 153 Испания МЕХАНИЧЕСКИЕ ГЕМОСТАЗИСНЫЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
Таблица 154 Испания ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 155 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ, МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 156 ИСПОЛНИТЕЛЬНЫЕ ОБЯЗАТЕЛЬНЫЕ УСЛОВИЯ В МЕХАНИЧЕСКОМ ГЕМОСТАССКОМ УСЛУГЕ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 157 ИСПОЛНИТЕЛЬНЫЕ ТЕРМИЧЕСКИЕ УБЕЖДЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 158 ИСПАНИЯ БИПОЛАРНАЯ КОЛУЛЯЦИЯ В МАРКЕ ТРАНСПОРТНЫХ УБЕЖДЕНИЙ, В ЭНЕРГИЧЕСКОМ ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 159 ИСПАНИЯ БИПОЛАРНАЯ КОНФЕРЕНЦИЯ В МАРКЕ ТРАНСПОРТНЫХ СРЕДСТВ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
Таблица 160 Испания Топические АГЕНТЫ И ИНЖЕКТИБЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 161 Испания ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАСС МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 162 Испания ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 163 ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 164 Испания ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАСС МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 165 Испания ГАСТРОИНТЕСТИНАЛЬНОЕ СКЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 166 Испания ГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
Таблица 167 Испания Негастроинтестинальное кровотечение в эндоскопическом гемостазисе Маркет, Тип, 2018-2033 (USD THOUSAND)
СТАТЬЯ 168 Испания НЕГАСТРОИНТЕСТИНАЛЬНОЕ СКЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 169 УПРАВЛЕНИЕ ИСПАНИИ ТРАУМЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
Таблица 170 Испания ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАСС МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 171 Испания ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 172 Испания ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАСС МАРКЕТ, КАНАЛ ДИСТРИБУЦИИ, 2018-2033 (USD THOUSAND)
Таблица 173 Испания Индиректные продажи в эндоскопическом гемостазе, Тип, 2018-2033 (USD THOUSAND)
СТАТЬЯ 174 ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 175 НЕТЕРЛАНДОВ МЕХАНИЧЕСКИХ ГЕМОСТАЗОВ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 176 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 177 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ, МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 178 ОБЯЗАТЕЛЬНЫХ УБЕЖДЕНИЙ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ УБЕЖДЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 179 ТЕРМИЧЕСКИЕ РЕШЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, ВЫБОР, 2018-2033 (USD THOUSAND)
СТАТЬЯ 180 НЕТЕРЛАНДОВ БИПОЛАРНАЯ КОНФЕРЕНЦИЯ В МАРКЕ ТЕРМИЧЕСКИХ РЕШЕНИЙ, ПО ТЕМЕ ЭНЕРГИИ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 181 НИДЕРЛАНДСКАЯ БИПОЛАРНАЯ КОЛУЛЯЦИЯ В РЫНОКЕ ТЕРМИЧЕСКИХ РЕШЕНИЙ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 182 ТОПИЧЕСКИЕ АГЕНТЫ И ИНЖЕКТИВЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 183 ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 184 НЕТЕРЛАНДЫ ПОСЛЕ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 185 НИДЕРЛАНДОВ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 186 ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 187 ГЕЛЛАНДЫ ГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 188 ГАСТРОИНТЕСТИНАЛЬНОЕ СКЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 189 НЕГЕРЛАНДЫ НЕГАСТРОИНТЕСТИНАЛЬНОЕ СКЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 190 НЕГЕРЛАНДЫ НЕГАСТРОИНТЕСТИНАЛЬНОЕ СКЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 191 NETHERLANDS TRAUMA MANAGEMENT IN ENDOSCOPIC HEMOSTASIS MARKET, BY PRODUCT TYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 192 ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 193 ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 194 ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, КАНАЛ ДИСТРИБУЦИИ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 195 НИДЕРЛАНДЫ ИНДИРЕКТИВНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 196 БЕЛЬГИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 197 БЕЛЬГИЙСКИЕ МЕХАНИЧЕСКИЕ ГЕМОСТАССКИЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 198 БЕЛГИЙСКИХ ГЕМОСТАТИЧЕСКИХ КЛИПОВ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 199 БЕЛГИЙСКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 200 БЕЛГИУМНЫХ ОБЯЗАТЕЛЬСТВ В МЕХАНИЧЕСКОМ ГЕМОСТАЗСКОМ РЕШЕНИИ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 201 БЕЛГИУМНЫЕ ТЕРМИЧЕСКИЕ РЕШЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
TABLE 202 BELGIUM BIPOLAR COAGULATION IN THERMAL DEVICES MARKET, BY ENERGY TYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 203 БЕЛЬГИЙСКАЯ БИПОЛАРНАЯ СОГЛАШЕНИЕ В МАРКЕ ТРАНСПОРТНЫХ СРЕДСТВ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
TABLE 204 BELGIUM TOPICAL AGENTS & INJECTIBLES IN ENDOSCOPIC HEMOSTASIS MARKET, BYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 205 БЕЛГИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
TABLE 206 BELGIUM UPPER GASTROINTESTINAL ENDOSCOPY IN ENDOSCOPIC HEMOSTASIS MARKET, BYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 207 БЕЛЬГИЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 208 БЕЛГИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
TABLE 209 BELGIUM GASTROINTESTINAL BLEEDING IN ENDOSCOPIC HEMOSTASIS MARKET, BYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 210 БЕЛГИЙСКИЙ ГАСТРОИНТЕСТИНАЛЬНЫЙ БЛЕД В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 211 БЕЛГИУМ НЕГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 212 БЕЛГИЯ НЕГАСТРОИНТЕСТИНАЛЬНОЕ СКЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 213 УПРАВЛЕНИЕ БЕЛЬГИЕМ ТРАУМА В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 214 БЕЛЬГИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 215 БЕЛГИЙСКИХ ГОСПИТАЛОВ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, СТАТЬЯ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 216 БЕЛГИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, КАННЕЛЬ ДИСТРИБУЦИИ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 217 БЕЛГИЙСКИЕ ИНДИРЕКТИВНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 218 СВИТЦЕРЛЯНДСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 219 СВИТЦЕРЛЯНДСКИЕ МЕХАНИЧЕСКИЕ ГЕМОСТАЗИСНЫЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 220 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 221 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАТЕРИАЛ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 222 СВАЙЦЕРЛЯНДСКИЕ ОБЯЗАТЕЛЬСТВА В МЕХАНИЧЕСКОМ ГЕМОСТАЗСКОМ РЕШЕНИИ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 223 СВИТЦЕРЛЯНДСКИЕ ТЕРМИЧЕСКИЕ УБЕЖДЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 224 СВИТЦЕРЛЯНДСКАЯ БИПОЛАРНАЯ КОНФЕРЕНЦИЯ В РЫНОКЕ ТЕРМИЧЕСКИХ РЕШЕНИЙ, ПО ЭНЕРГИЧЕСКОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 225 СВИТЦЕРЛЯНДСКАЯ БИПОЛАРНАЯ КОНФЕРЕНЦИЯ В МАРКЕ ТРАНСПОРТНЫХ РЕШЕНИЙ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 226 СВИТЦЕРЛЯНДСКИЕ ТОПИЧЕСКИЕ АГЕНТЫ И ИНЖЕКТИВЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 227 СВИТЦЕРЛЯНДСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 228 СВИТЦЕРЛЯНДСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 229 СВИТЦЕРЛЯНДСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
Таблица 230 СВИТЦЕРЛЯНДСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
Таблица 231 СВИТЦЕРЛЯНДСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ КЛЕЧАЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 232 СВИТЦЕРЛЯНДСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ КЛИВКА В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 233 СВИТЦЕРЛАНДСКАЯ НЕГАСТРОИНТЕСТИНАЯ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 234 СВИТЦЕРЛАНДСКАЯ НЕГАСТРОИНТЕСТИНАЯ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 235 УПРАВЛЕНИЕ СВИТЦЕРЛЯНДСКОЙ ТРАУМЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 236 СВИТЦЕРЛЯНДСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
Таблица 237 СВИТЦЕРЛЯНДСКИЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 238 СВИТЦЕРЛЯНДСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, КАНАЛ ДИСТРИБУЦИИ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 239 СВИТЦЕРЛЯНДСКИЕ ИНДИРЕКТИВНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
Таблица 240 СВЕДЕННЫЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 241 СВЕДЕННЫЕ МЕХАНИЧЕСКИЕ ГЕМОСТАССКИЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
Таблица 242 СВЕДЕННЫЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
Таблица 243 СВЕДЕННЫЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
Таблица 244 СВЕДЕННЫЕ ОБЯЗАТЕЛЬСТВА В МЕХАНИЧЕСКОМ ГЕМОСТАЗСКОМ РЕШЕНИИ МАРКЕТ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 245 СВЕДЕННЫЕ ТЕРМИЧЕСКИЕ РЕШЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 246 СВЕДЕННАЯ БИПОЛАРНАЯ КОЛУЛЯЦИЯ В МАРКЕ ТРАНСПОРТНЫХ РЕШЕНИЙ, ПО ЭНЕРГИЧЕСКОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 247 СВЕДЕННАЯ БИПОЛАРНАЯ КОНФЕРЕНЦИЯ В МАРКЕ ТРАНСПОРТНЫХ СРЕДСТВ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 248 СВЕДЕННЫЕ ТОПНЫЕ АГЕНТЫ И ИНЖЕКТИБЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
Таблица 249 СВЕДЕНСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
Таблица 250 СВЕДЕННАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
Таблица 251 СВЕДЕННАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТ, КИПЕ, 2018-2033 (USD THOUSAND)
Таблица 252 СВЕДЕННЫЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
Таблица 253 СВЕДЕНСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ КЛЕЧАЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ТИП, 2018-2033 (USD THOUSAND)
Таблица 254 СВЕДЕНСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ БЛЕДОВАНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
Таблица 255 СВЕДЕННАЯ НЕГАСТРОИНТЕСТИНАЛЬНАЯ КРОВЬ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ТИП, 2018-2033 (USD THOUSAND)
Таблица 256 СВЕДЕННАЯ НЕГАСТРОИНТЕСТИНАЛЬНАЯ КЛЕЧАЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 257 СВЕДЕННОЕ ТРАУМНОЕ УПРАВЛЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
Таблица 258 СВЕДЕННЫЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
Таблица 259 СВЕДЕННЫЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 260 СВЕДЕННЫЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО РАЗРАБОТКИ ЧАННЕЛЯ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 261 СВЕДЕННАЯ ИНДИРЕКТНАЯ ПРОДАЖА В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 262 ДАНМАРК ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 263 ДЕНМАРКОВЫЕ МЕХАНИЧЕСКИЕ ГЕМОСТАЗИСНЫЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 264 ДЕНМАРК ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 265 ДЕНМАРКОВЫЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 266 ДЕНМАРКОВЫЕ ОБЯЗАТЕЛЬСТВА В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ ОБЕСПЕЧЕНИЯХ, СТАТЬЯ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 267 ТЕМРАЛЬНЫЕ ДЕВИЦИИ ДЕНМАРКА В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
268 DENMARK BIPOLAR COAGULATION IN THERMAL DEVICES MARKET, BY ENERGY TYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 269 ДЕНМАРКОВСКАЯ БИПОЛАРНАЯ КОЛУЛЯЦИЯ В РЫНОКЕ ТРАНСПОРТНЫХ СРЕДСТВ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 270 DENMARK TOPICAL AGENTS & INJECTIBLES IN ENDOSCOPIC HEMOSTASIS MARKET, BYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 271 ДАНМАРК ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАСС МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 272 DENMARK UPPER GASTROINTESTINAL ENDOSCOPY IN ENDOSCOPIC HEMOSTASIS MARKET, BYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 273 ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 274 ДЕНМАРК ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 275 ДЕНМАРК ГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 276 ДЕНМАРК ГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 277 ДЕНМАРК НЕГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 278 DENMARK NON-GASTROINTESTINAL BLEEDING IN ENDOSCOPIC HEMOSTASIS MARKET, BY PRODUCT TYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 279 УПРАВЛЕНИЕ ДЕНМАРКА ТРАУМА В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 280 ДАНМАРК ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 281 ДЕНМАРКОВЫЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 282 DENMARK ENDOSCOPIC HEMOSTASIS MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD THOUSAND)
СТАТЬЯ 283 ИНДИРЕКТИВНЫЕ ПРОДАЖИ ДЕНМАРКА В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 284 СЕВЕРНО-ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 285 Норвежские МЕХАНИЧЕСКИЕ РЕШЕНИЯ ГЕМОСТАЗА В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
Таблица 286 Норвежские гемостатические клипы в МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, КИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 287 ЕВРОПЕЙСКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
Таблица 288 Норвежские ОБЯЗАТЕЛЬНЫЕ УСЛУГИ В МЕХАНИЧЕСКОМ ГЕМОСТАЗСКОМ УСЛУГЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 289 НОРВИЧЕСКИЕ ТЕРМИЧЕСКИЕ РЕШЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 290 Норвежская биполярная коагуляция в АРКЕТНЫХ РЕШЕНИЯХ, ИНЕРГИЧЕСКИЙ ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 291 Норвежская биполярная коагуляция на рынке традиционных устройств, 2018-2033 (USD THOUSAND)
TABLE 292 NORWAY TOPICAL AGENTS & INJECTIBLES IN ENDOSCOPIC HEMOSTASIS MARKET, BYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 293 СЕВЕРНО-ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 294 Норвежская ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 295 Норвежская ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 296 Норвежский эндоскопический гемостазовый рынок, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 297 Норвежское ГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 298 Норвежское ГАСТРОИНТЕСТИНАЛЬНОЕ СКЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 299 Норвежское негастроинтестинальное кровотечение в эндоскопическом гемостазисе, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 300 НЕГАСТРОИНТЕСТИНАЛЬНОЕ СКЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПРИ ПРОДУКТНОМ ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 301 Норвежское ТРАУМНОЕ УПРАВЛЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
Таблица 302 Норвежский эндоскопический гемостаз Маркет, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 303 НОРВЫЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 304 Норвежский эндоскопический гемостазовый рынок, КАНАЛ ДИСТРИБУЦИИ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 305 Норвежские непрямые продажи в эндоскопическом гемостазе, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 306 ФИНЛЯНДСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 307 Финляндские МЕХАНИЧЕСКИЕ ГЕМОСТАССКИЕ УСЛОВИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 308 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 309 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ, МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 310 ФИНЛАНДСКИЕ ОБЯЗАТЕЛЬСТВА В МЕХАНИЧЕСКОМ ГЕМОСТАССКОМ РЕШЕНИИ МАТЕРИАЛЬНЫХ РЕШЕНИЙ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 311 ФИНЛАНДСКИЕ ТЕРМИЧЕСКИЕ РЕШЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 312 ФИНЛАНДСКАЯ БИПОЛАРНАЯ КОЛУЛЯЦИЯ В МАРКЕ ТЕРМИЧЕСКИХ РЕШЕНИЙ, ПО ТЕМЕ ЭНЕРГИИ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 313 ФИНЛАНДСКАЯ БИПОЛАРНАЯ КОЛУЛЯЦИЯ В МАРКЕ ТРАНСПОРТНЫХ СРЕДСТВ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 314 ФИНЛАНДСКИЕ ТОПНЫЕ АГЕНТЫ И ИНЖЕКТИВЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 315 ФИНЛАНДСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 316 ФИНЛАНДСКАЯ ГОСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 317 ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 318 Финляндский ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 319 ФИНЛЯНДСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ КЛЕЧАЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 320 ФИНЛЯНДСКИЙ ГАСТРОИНТЕСТИНАЛЬНЫЙ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 321 ФИНЛАНДСКАЯ НЕГАСТРОИНТЕСТИНАЛЬНАЯ КЛЕЧАЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 322 ФИНЛАНДСКАЯ НЕГАСТРОИНТЕСТИНАЛЬНАЯ БЛЕДОВАНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 323 ФИНЛАНДСКОЕ ТРАУМНОЕ УПРАВЛЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 324 Финляндский эндоскопический гемостазовый рынок, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 325 ФИНЛАНДСКИЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 326 ФИНЛАНДСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ, КАНАЛ ДИСТРИБУЦИИ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 327 ИНДИРЕКТНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, ВЫБОР, 2018-2033 (USD THOUSAND)
СТАТЬЯ 328 РОССИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 329 РОССИЙСКИЕ МЕХАНИЧЕСКИЕ ГЕМОСТАССКИЕ РЕШЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 330 РОССИЙСКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 331 ГЕМОСТАТИЧЕСКИЕ КЛИПЫ РОССИИ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ, МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 332 РОССИЙСКИЕ ОБЯЗАТЕЛЬНЫЕ УСЛОВИЯ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ УСЛОВИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 333 РОССИЙСКИЕ ТЕРМИЧЕСКИЕ РЕШЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, ВЫБОР, 2018-2033 (USD THOUSAND)
СТАТЬЯ 334 БИПОЛАРНОЕ РОССИЙСКОЕ СОГЛАШЕНИЕ В МАРКЕ ТРАНСПОРТНЫХ РЕШЕНИЙ, ПО ЭНЕРГЕТИЧЕСКОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 335 РОССИЙСКАЯ БИПОЛАРНАЯ КОНФЕРЕНЦИЯ В ТЕМНЫХ РАЗВИТИЯХ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 336 РОССИЙСКИЕ ТОПНЫЕ АГЕНТЫ И ИНЖЕКТИВЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 337 РОССИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 338 РУССКАЯ ГОСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 339 РОССИЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, КИТАЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 340 РОССИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 341 РОССИЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ КЛЕЧАЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ВЫБОР, 2018-2033 (USD THOUSAND)
СТАТЬЯ 342 РОССИЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ КЛЕЧАЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 343 РОССИЙСКОЕ НЕГАСТРОИНТЕСТИНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, КИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 344 РОССИЯ НЕГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 345 УПРАВЛЕНИЕ РОССИЙСКОЙ ТРАУМЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 346 РОССИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 347 РУССКИЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 348 РОССИЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО РАЗРАБОТКИ ЧАННЕЛЬ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 349 РОССИЙСКИЕ ИНДИРЕКТИВНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, ВЫБОР, 2018-2033 (USD THOUSAND)
СТАТЬЯ 350 ТУРКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 351 МЕХАНИЧЕСКИЕ РЕШЕНИЯ ГЕМОСТАЗА В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 352 ТУРКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 353 ТУРКИЕ ГЕМОСТАТИЧЕСКИЕ КЛИПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 354 ТУРКИВЫЕ ОБЯЗАТЕЛЬНЫЕ УБЕЖДЕНИЯ В МЕХАНИЧЕСКИХ ГЕМОСТАЗИСНЫХ УБЕЖДЕНИЯХ МАРЕТ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 355 ТЕРМИЧЕСКИЕ РЕШЕНИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 356 ТУРЦИЯ БИПОЛАРНОЙ КОАГУЛЯЦИИ В РЫНОКЕ ТРАНСПОРТНЫХ РЕШЕНИЙ, ПО ЭНЕРГИЧЕСКОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 357 ТУРЦИЯ БИПОЛАРНОЙ КОАГУЛЯЦИИ В МАРКЕ ТРАНСПОРТНЫХ СРЕДСТВ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 358 ТОРГОВЫЕ АГЕНТЫ ТУРЦИИ И ИНЖЕКТИВЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАССКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 359 ТУРКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
TABLE 360 TURKEY UPPER GASTROINTESTINAL ENDOSCOPY IN ENDOSCOPIC HEMOSTASIS MARKET, BYPE, 2018-2033 (USD THOUSAND)
СТАТЬЯ 361 ТЮРКЕЙСКАЯ ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 362 ТУРЦИЯ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 363 ТЮРКИЙ ГАСТРОИНТЕСТИНАЛЬНЫЙ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТ, ВЫБОР, 2018-2033 (USD THOUSAND)
СТАТЬЯ 364 ТУРКИЙ ГАСТРОИНТЕСТИНАЛЬНЫЙ БЛЕД В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 365 ТЮРКЕЙ НЕГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 366 ТЮРКЕЙ НЕГАСТРОИНТЕСТИНАЛЬНОЕ КЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 367 УПРАВЛЕНИЕ ТРАУМА ТУРЦИИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 368 МАРКЕТ ЭНДОСКОПИЧЕСКОГО ГЕМОСТАЗА, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 369 ТЮРКЕЙСКИЕ ГОСПИТАЛЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 370 ТЮРКЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИСКИЙ МАРКЕТ, ПО РАЗРАБОТКЕ ЧАННЕЛЯ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 371 ИНДИРЕКТИВНЫЕ ПРОДАЖИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 372 РЕСТ ЕВРОПЫ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 373 РЕШЕНИЯ ЕВРОПЕЙСКИХ МЕХАНИЧЕСКИХ ГЕМОСТАЗОВ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 374 ПЕРЕХОД ГЕМОСТАТИЧЕСКИХ КЛИПОВ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ МАРКЕТ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 375 РЕСТ ГЕМОСТАТИЧЕСКИХ КЛИПОВ ЕВРОПЫ В МЕХАНИЧЕСКИХ ГЕМОСТАССКИХ РЕШЕНИЯХ, МАТЕРИАЛЬНЫЙ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 376 РЕШЕНИЕ ЕВРОПЕЙСКИХ ОБЯЗАТЕЛЬСТВ В МЕХАНИЧЕСКОМ ГЕМОСТАЗСКОМ РЕШЕНИИ, ТАЙП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 377 РЕШЕНИЕ ЕВРОПЕЙСКИХ ТЕРМИЧЕСКИХ УБЕЖДЕНИЙ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 378 ПРЕДОСТАВЛЕНИЕ ЕВРОПЕЙСКОЙ БИПОЛАРНОЙ СОГЛАШЕННОСТИ В РЫНОКЕ ТРАНСПОРТНЫХ РЕШЕНИЙ, ПО ЭНЕРГИЧЕСКОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 379 ПРЕДОСТАВЛЕНИЕ ЕВРОПЕЙСКОЙ БИПОЛАРНОЙ СОГЛАШЕНИЯ В МАРКЕ ТРАНСПОРТНЫХ СРЕДСТВ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 380 ПЕРЕДЫШЛЕНИЯ ТОРГОВЫХ АГЕНТОВ ЕВРОПЫ И ИНЖЕКТИВОВ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 381 РЕСТ ЕВРОПЫ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОЦЕДУРЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 382 ПРОСТОКА ЕВРОПЕЙСКОЙ ГАСТРОИНТЕСТИНАЛЬНОЙ ЭНДОСКОПИИ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕТЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 383 ПЕРЕХОД ГАСТРОИНТЕСТИНАЛЬНАЯ ЭНДОСКОПИЯ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 384 РЕСТ ЕВРОПЫ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПРИМЕЧАНИЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 385 РЕСТ ГАСТРОИНТЕСТИНАЛЬНОГО СВЕТА ЕВРОПЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 386 ПРЕДОСТАВЛЕНИЕ ГАСТРОИНТЕСТИНАЛЬНОГО КЛЕЧЕНИЯ ЕВРОПЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 387 РЕСТ ЕВРОПЫ НЕГАСТРОИНТЕСТИНАЛЬНОЕ СКЛЕЧЕНИЕ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ТИП, 2018-2033 (USD THOUSAND)
СТАТЬЯ 388 ПРЕДОСТАВЛЕНИЕ ЕВРОПЫ НЕГАСТРОИНТЕСТИНАЛЬНЫМ КЛЕЧЕНИЕМ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРИМЕЧАНИЮ ПРОДУКТА, 2018-2033 (USD THOUSAND)
СТАТЬЯ 389 РЕШЕНИЕ ПРОДУКТОВ ТРАУМЫ ЕВРОПЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, ПО ПРОДУКТНОМУ ТИПУ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 390 РЕСТ ЕВРОПЫ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, К концу использования, 2018-2033 (USD THOUSAND)
СТАТЬЯ 391 ПЕРЕДАЧА ЕВРОПЕЙСКИХ ГОСПИТАЛОВ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗСКОМ МАРКЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 392 РЕСТ ЕВРОПЫ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО РАЗРАБОТКИ ЧАННЕЛЯ, 2018-2033 (USD THOUSAND)
СТАТЬЯ 393 ОБЕСПЕЧЕНИЯ ИНДИРЕКТОВ ЕВРОПЫ В ЭНДОСКОПИЧЕСКОМ ГЕМОСТАЗЕ, В ТИПЕ, 2018-2033 (USD THOUSAND)
Список рисунков
ФИГРАФИЯ 1 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: СЕГМЕНТАЦИЯ
ФИГРАФИЯ 2 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: ТРИАНГУляция данных
ФИГРА 3 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ: ДРОК АНАЛИЗИС
ФИГРА 4 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: СТРАНАЯ МАРКЕТНАЯ АНАЛИЗА
ФИГРАФИЯ 5 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: КОМПАНИЯ ИССЛЕДОВАНИЕ АНАЛИЗА
ФИГРАФИЯ 6 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: ИНТЕРВЬЮ-ДЕМОГРАФИКА
ФИГРА 7 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: МУЛЬТИВАРИАТНАЯ МОДЕЛЛИНГ
8 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: ДБМР МАРКЕТ ПОСИЦИОННЫЙ ГРИД
ФИГРАФИЯ 9 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: ВЕНДОР ДЕЛАТЬ АНАЛИЗ
Рисунок 10 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: МАРКЕТ ЭНД-ЮЗЕР КОВЕРАГ ГРИД
ФИГРАФИЯ 11 ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ: СЕГМЕНТАЦИЯ
ФИГРАФИЯ 12 ДВА СЕГМЕНТА КОМПРИЗИРОВАНИЯ РЕСПУБЛИЧЕСКОГО ЭНДОСКОПИЧЕСКОГО ГЕМОСТАССКОГО МАРКЕТА ПО ПРОДУКТНОМУ ТИПУ
ФИГРА 13 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: ИСПОЛНИТЕЛЬНАЯ РЕЗЮМЕ
ФИГРА 14 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: СТРАТЕГИЧЕСКИЕ РЕШЕНИЯ
ФИГРА 15 ВОССТАНОВЛЕНИЕ ЭНДОСКОПИЧЕСКИХ ГЕМОСТАЗОВ, ОПРЕДЕЛЯЕМЫХ ДВИГАТЬ РОСТ ЭНДОСКОПИЧЕСКИХ ГЕМОСТАЗОВ ЕВРОПЫ С 2026 по 2033 год
16 Тип 2 ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАЗИЗМ ОПЫТАЕТСЯ СОГЛАСИТЬСЯ ДЛЯ ЛАРГЕСТИЧЕСКОГО РАЗВИТИЯ ЭНДОСКОПИЧЕСКИХ ГЕМОСТАЗОВ ЕВРОПЫ В 2026 и 2033 годах
ФИГРА 17 КОМПАНИЯ ОЦЕНКА КВАДРАНТА
ФИГРА 18 ДРОК АНАЛИЗ
ФИГРАФИЯ 19 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРОДУКТНОМУ ТИПУ, 2025
Рисунок 20 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ: ПО ПРОДУКТНОМУ ТИПУ, КАГР (2025-2032)
ФИГРА 21 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: ПО ПРОДУКТНОМУ ТИПУ, КАГР (2025-2032)
ФИГРАФИЯ 22 ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ: ПО ПРОДУКТНОМУ ТИПУ, ЛИФЕЛИНА КУРВЕ (2025-2032)
ФИГРАЛ 23 ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ, ПО ПРЕДСТАВЛЕННОМУ типу, 2025
ФИГРА 24 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: ПО ПРОЦЕДУРЕ, 2026-2033 (USD THOUSAND)
25 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: ПО ПРОЦЕДУРЕ, 2026-2033 (USD THOUSAND)
ФИГРАФИЯ 26 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ, ПО ПРИМЕЧАНИЮ, 2025
ФИГРА 27 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ: ПРИМЕЧАНИЕ, 2026-2033 (USD THOUSAND)
ФИГРАНИЯ 28 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЯ ГЕМОСТАССКАЯ МАРКЕТ: ПРИМЕЧАНИЕ, 2026-2033 (USD THOUSAND)
ФИГРА 29 ЕВРОПЕЙСКИЙ ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ: ПРИМЕЧАНИЕ, 2026-2033 (USD THOUSAND)
ФИГРА 30 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ, К концу использования, 2025
ФИГРА 31 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: К концу использования, 2026-2033 (USD THOUSAND)
ФИГРА 32 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: К концу использования, 2026-2033 (USD THOUSAND)
ФИГРА 33 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: К концу использования, 2026-2033 (USD THOUSAND)
FIGURE 34 EUROPE ENDOSCOPIC HEMOSTASIS MARKET, BY DISTRIBUTION CHANNEL, 2025
ФИГРА 35 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: КАНАЛ ДИСТРИБУЦИИ, 2026-2033 (USD THOUSAND)
ФИГРА 36 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: ПО РАЗРАБОТКЕ CHANNEL, 2026-2033 (USD THOUSAND)
ФИГРА 37 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: С помощью ДИСТРИБУЦИОННОГО КАНАЛА, 2026-2033 (USD THOUSAND)
ФИГРА 38 ЭНДОСКОПИЧЕСКИЙ ГЕМОСТАССКИЙ МАРКЕТ: СНАПШОТ (2025)
ФИГРА 39 ЕВРОПЕЙСКАЯ ЭНДОСКОПИЧЕСКАЯ ГЕМОСТАССКАЯ МАРКЕТ: КОМПАНИЯ ДЛЯ 2025 (%)
Методология исследования
Сбор данных и анализ базового года выполняются с использованием модулей сбора данных с большими размерами выборки. Этап включает получение рыночной информации или связанных данных из различных источников и стратегий. Он включает изучение и планирование всех данных, полученных из прошлого заранее. Он также охватывает изучение несоответствий информации, наблюдаемых в различных источниках информации. Рыночные данные анализируются и оцениваются с использованием статистических и последовательных моделей рынка. Кроме того, анализ доли рынка и анализ ключевых тенденций являются основными факторами успеха в отчете о рынке. Чтобы узнать больше, пожалуйста, запросите звонок аналитика или оставьте свой запрос.
Ключевой методологией исследования, используемой исследовательской группой DBMR, является триангуляция данных, которая включает в себя интеллектуальный анализ данных, анализ влияния переменных данных на рынок и первичную (отраслевую экспертную) проверку. Модели данных включают сетку позиционирования поставщиков, анализ временной линии рынка, обзор рынка и руководство, сетку позиционирования компании, патентный анализ, анализ цен, анализ доли рынка компании, стандарты измерения, глобальный и региональный анализ и анализ доли поставщика. Чтобы узнать больше о методологии исследования, отправьте запрос, чтобы поговорить с нашими отраслевыми экспертами.
Доступна настройка
Data Bridge Market Research является лидером в области передовых формативных исследований. Мы гордимся тем, что предоставляем нашим существующим и новым клиентам данные и анализ, которые соответствуют и подходят их целям. Отчет можно настроить, включив в него анализ ценовых тенденций целевых брендов, понимание рынка для дополнительных стран (запросите список стран), данные о результатах клинических испытаний, обзор литературы, обновленный анализ рынка и продуктовой базы. Анализ рынка целевых конкурентов можно проанализировать от анализа на основе технологий до стратегий портфеля рынка. Мы можем добавить столько конкурентов, о которых вам нужны данные в нужном вам формате и стиле данных. Наша команда аналитиков также может предоставить вам данные в сырых файлах Excel, сводных таблицах (книга фактов) или помочь вам в создании презентаций из наборов данных, доступных в отчете.