Global Hepatitis E Market
Размер рынка в млрд долларов США
CAGR :
% 
 USD
59.12 Million
USD
86.02 Million
2025
2033
USD
59.12 Million
USD
86.02 Million
2025
2033
| 2026 –2033 | |
| USD 59.12 Million | |
| USD 86.02 Million | |
|
|
|
|
Global Hepatitis E Market Segmentation, By Drug Class (Antiviral Drugs, Interferons, and Others), Route of Administration (Oral, Parenteral, and Others), End-Users (Hospitals, Homecare, Specialty Clinics, and Others), Distribution Channel (Hospital Pharmacy, Online Pharmacy, and Retail Pharmacy)- Industry Trends and Forecast to 2033
Hepatitis E Market Size
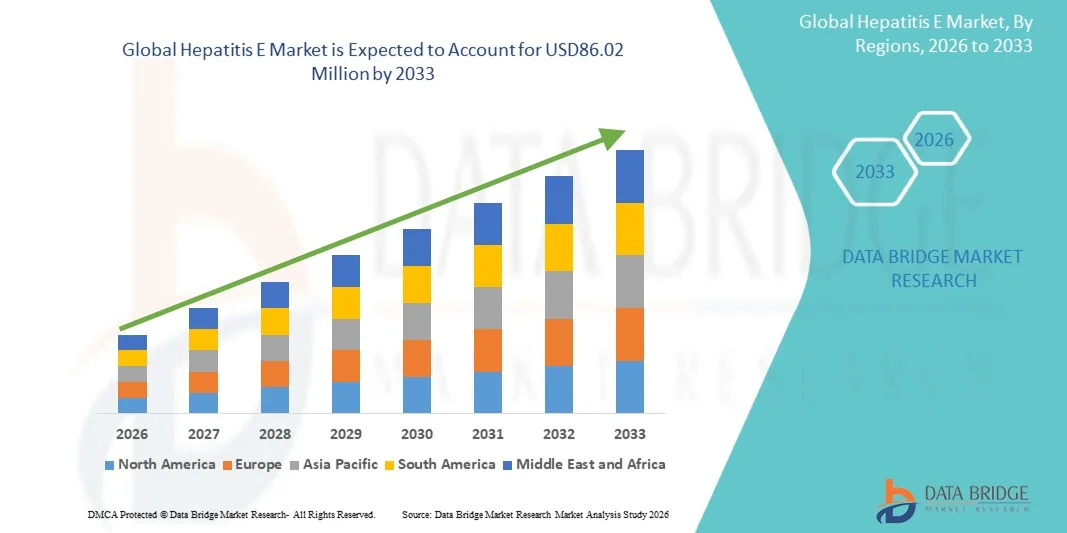
- The global hepatitis E market size was valued at USD 59.12 million in 2025 and is expected to reach USD 86.02 million by 2033, at a CAGR of 4.8% during the forecast period
- The market growth is largely fueled by the increasing prevalence of hepatitis E infections, particularly in developing regions, along with the growing adoption of advanced diagnostic testing technologies and improved disease surveillance systems
- Furthermore, rising awareness regarding waterborne diseases, expanding healthcare infrastructure, and ongoing efforts toward vaccine development and preventive healthcare measures are establishing hepatitis E diagnostics as a critical component of infectious disease management. These converging factors are accelerating the demand for hepatitis E-related solutions, thereby significantly boosting the market’s growth
Hepatitis E Market Analysis
- Hepatitis E, a liver infection caused by the hepatitis E virus (HEV) and primarily transmitted through contaminated water, remains a significant public health concern, particularly in developing regions, driving the need for effective therapeutic options and supportive care treatments
- The escalating demand in the hepatitis E market is primarily fueled by the rising incidence of waterborne infections, increasing awareness about viral hepatitis, and the growing use of antiviral drugs and interferon-based therapies for disease management
- North America dominated the hepatitis E market with the largest revenue share of 38.5% in 2025, characterized by advanced healthcare infrastructure, higher diagnosis and reporting rates, and strong investments in infectious disease research, with the U.S. witnessing increased clinical attention toward hepatitis E, particularly among immunocompromised populations
- Asia-Pacific is expected to be the fastest growing region in the hepatitis E market during the forecast period due to increasing disease prevalence, improving healthcare access, and rising government initiatives to control waterborne infections
- Antiviral drugs segment dominated the hepatitis E market with the largest share of 45.2% in 2025, driven by their effectiveness in managing severe and chronic cases, along with growing clinical adoption
Report Scope and Hepatitis E Market Segmentation
|
Attributes |
Hepatitis E Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
Hepatitis E Market Trends
“Rising Focus on Vaccine Development and Preventive Healthcare”
- A significant and accelerating trend in the global hepatitis E market is the increasing focus on vaccine development and preventive healthcare strategies, particularly in regions with high disease prevalence and limited sanitation infrastructure. This shift is significantly enhancing efforts toward disease control and long-term prevention
- For instance, the HEV 239 vaccine (Hecolin), developed and commercialized in China, has demonstrated strong efficacy in preventing hepatitis E infection and is being evaluated for broader adoption in endemic regions. Similarly, ongoing clinical research is exploring new vaccine candidates to expand global accessibility
- The growing emphasis on preventive healthcare is enabling early intervention strategies such as immunization programs and public health awareness campaigns. For instance, health authorities are increasingly promoting vaccination and hygiene practices to reduce infection rates, particularly in high-risk populations
- The integration of hepatitis E prevention into broader infectious disease control programs is facilitating coordinated healthcare responses. Through centralized public health initiatives, governments can manage hepatitis E alongside other viral infections, improving overall disease monitoring and response efficiency
- This trend toward proactive disease prevention and immunization is fundamentally reshaping public health approaches to hepatitis E. Consequently, companies and research institutions are focusing on expanding vaccine availability and improving access in underserved regions
- The demand for hepatitis E vaccines and preventive solutions is growing steadily across developing and developed regions, as healthcare systems increasingly prioritize disease prevention and population health management
- The rising adoption of molecular diagnostic technologies and point-of-care testing solutions is enhancing early detection capabilities, contributing to improved disease surveillance and timely intervention strategies
Hepatitis E Market Dynamics
Driver
“Growing Need Due to Rising Infection Rates and Public Health Initiatives”
- The increasing prevalence of hepatitis E infections, particularly in regions with poor sanitation, coupled with expanding public health initiatives, is a significant driver for the heightened demand for hepatitis E treatments and preventive solutions
- For instance, in March 2024, the World Health Organization emphasized the need for improved surveillance and control measures for hepatitis E in endemic regions, encouraging governments to strengthen diagnostic and prevention frameworks. Such strategies by global health organizations are expected to drive market growth in the forecast period
- As awareness of hepatitis E transmission and complications increases, healthcare providers are focusing on early diagnosis and effective management, encouraging the use of antiviral therapies and supportive treatments
- Furthermore, the growing investment in healthcare infrastructure and infectious disease control programs is supporting the adoption of advanced treatment and prevention approaches, particularly in emerging economies
- The availability of oral antiviral drugs, improved patient access to healthcare facilities, and government-led awareness campaigns are key factors propelling the adoption of hepatitis E treatment solutions across affected populations. The increasing focus on reducing waterborne diseases and improving sanitation further contributes to market growth
- Rising international funding and support from global health organizations are enabling large-scale hepatitis prevention and control programs, particularly in low- and middle-income countries
- The growing emphasis on maternal and child health is increasing screening and management of hepatitis E infections, especially due to higher risks associated with pregnancy, thereby driving demand for effective treatment options
Restraint/Challenge
“Limited Treatment Options and Regulatory Barriers”
- The lack of widely approved, specific antiviral therapies for hepatitis E, along with regulatory and approval challenges, poses a significant barrier to market expansion. As treatment options remain limited, healthcare providers often rely on supportive care, restricting market growth potential
- For instance, the absence of globally approved vaccines outside select regions has limited widespread immunization efforts, creating disparities in disease prevention and control across different geographies
- Addressing these challenges through increased research and development, regulatory harmonization, and clinical trials is crucial for expanding treatment availability. Companies and research institutions are investing in novel antiviral drugs and vaccine candidates to address unmet needs. In addition, limited awareness in certain regions and underdiagnosis of hepatitis E cases can hinder timely treatment and intervention
- While healthcare infrastructure is improving, gaps in access to diagnostic and treatment services persist in low-income regions, affecting overall disease management outcomes
- Overcoming these challenges through enhanced research investments, improved regulatory pathways, and expanded public health initiatives will be vital for sustained market growth
- Limited commercial incentives for pharmaceutical companies, due to the disease’s higher prevalence in low-income regions, can restrict large-scale investment and slow innovation in this market
- Logistical challenges in vaccine distribution and cold chain management in remote and underserved areas further hinder effective implementation of preventive programs
Hepatitis E Market Scope
The market is segmented on the basis of drug class, route of administration, end-users, and distribution channel.
- By Drug Class
On the basis of drug class, the hepatitis E market is segmented into antiviral drugs, interferons, and others. The antiviral drugs segment dominated the market with the largest market revenue share of 45.2% in 2025, driven by their effectiveness in managing severe and chronic hepatitis E infections, particularly among immunocompromised patients. These drugs are widely preferred due to their targeted action against viral replication and increasing clinical adoption in hospital settings. In addition, ongoing research and off-label use of certain antivirals have further strengthened their position in the market. The availability of oral antiviral formulations and their integration into treatment protocols also contribute to their dominance. Growing physician preference for pharmacological intervention in complicated cases further supports segment growth.
The interferons segment is anticipated to witness the fastest growth rate during the forecast period, fueled by their immunomodulatory effects and increasing exploration in hepatitis E treatment, especially in chronic cases. Interferons enhance the body’s immune response against viral infections, making them a viable option in specific patient populations. Rising clinical trials and research initiatives focused on expanding their therapeutic applications are further boosting demand. Moreover, increasing awareness among healthcare professionals regarding their potential benefits is supporting adoption. The segment is also gaining traction due to advancements in biologics and improved formulation technologies.
- By Route of Administration
On the basis of route of administration, the hepatitis E market is segmented into oral, parenteral, and others. The oral segment dominated the market with the largest market revenue share in 2025, driven by its convenience, ease of administration, and high patient compliance. Oral medications are widely preferred in outpatient settings and for long-term management due to their non-invasive nature. The availability of antiviral drugs in oral form further strengthens this segment’s position. In addition, reduced need for healthcare supervision and lower administration costs contribute to its widespread adoption. Increasing focus on patient-centric treatment approaches also supports the dominance of oral therapies.
The parenteral segment is expected to witness the fastest growth rate during the forecast period, driven by its effectiveness in severe cases requiring immediate and controlled drug delivery. Parenteral administration ensures rapid onset of action and higher bioavailability, making it suitable for hospitalized patients. The growing number of hospital admissions related to acute hepatitis E complications is contributing to segment growth. Furthermore, advancements in injectable biologics and interferon therapies are supporting increased adoption. The segment is also benefiting from improved healthcare infrastructure and access to hospital-based treatments.
- By End-Users
On the basis of end-users, the hepatitis E market is segmented into hospitals, homecare, specialty clinics, and others. The hospitals segment dominated the market with the largest market revenue share in 2025, driven by the high volume of patient admissions for diagnosis and treatment of hepatitis E infections. Hospitals offer advanced diagnostic facilities, skilled healthcare professionals, and access to a wide range of treatment options, making them the primary point of care. The presence of specialized departments for infectious diseases further supports their dominance. In addition, increased hospitalization in severe and high-risk cases contributes significantly to revenue generation. Government investments in hospital infrastructure also play a key role in sustaining segment growth.
The homecare segment is anticipated to witness the fastest growth rate during the forecast period, fueled by the increasing preference for at-home treatment and monitoring, particularly for mild and recovering patients. Homecare solutions offer cost-effectiveness and convenience, reducing the burden on hospital facilities. The availability of oral medications and remote healthcare services is supporting this trend. In addition, the rise of telemedicine and digital health platforms is enabling effective patient management outside traditional healthcare settings. Growing awareness about self-care and disease management is further driving this segment.
- By Distribution Channel
On the basis of distribution channel, the hepatitis E market is segmented into hospital pharmacy, online pharmacy, and retail pharmacy. The hospital pharmacy segment dominated the market with the largest market revenue share in 2025, driven by the direct supply of medications to hospitalized patients and strong integration with clinical care systems. Hospital pharmacies ensure the availability of essential drugs, particularly for acute and severe cases requiring immediate treatment. The reliance on hospital-based treatment pathways further supports this segment’s dominance. In addition, regulatory compliance and controlled drug dispensing contribute to its widespread use. Increasing hospital admissions for infectious diseases also drive demand in this segment.
The online pharmacy segment is expected to witness the fastest growth rate during the forecast period, driven by the rising adoption of e-commerce platforms and increasing demand for convenient medicine purchasing options. Online pharmacies offer easy access to medications, competitive pricing, and home delivery services, enhancing patient convenience. The growing penetration of digital technologies and internet usage is further accelerating this trend. In addition, the shift toward remote healthcare and teleconsultation is supporting online pharmaceutical sales. Expanding regulatory support for e-pharmacies in several regions is also contributing to segment growth.
Hepatitis E Market Regional Analysis
- North America dominated the hepatitis E market with the largest revenue share of 38.5% in 2025, characterized by advanced healthcare infrastructure, higher diagnosis and reporting rates, and strong investments in infectious disease research
- Healthcare providers in the region highly prioritize early detection, effective disease management, and access to advanced treatment options, including antiviral drugs and supportive care for high-risk populations
- This widespread adoption is further supported by strong government initiatives, robust research and development activities, and the presence of well-established healthcare systems, establishing hepatitis E management solutions as a critical component of infectious disease control in both clinical and public health settings
U.S. Hepatitis E Market Insight
The U.S. hepatitis E market captured the largest revenue share within North America in 2025, fueled by advanced diagnostic capabilities and increasing awareness of infectious diseases. Healthcare providers are increasingly prioritizing early detection and management of hepatitis E, particularly among immunocompromised populations such as transplant recipients. The growing focus on public health surveillance, combined with robust demand for antiviral therapies and supportive care, further propels the market. Moreover, the increasing investment in infectious disease research, availability of sophisticated laboratory infrastructure, and improved reporting systems are significantly contributing to the market's expansion. The presence of leading research institutions and strong healthcare spending further strengthens market growth.
Europe Hepatitis E Market Insight
The Europe hepatitis E market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by rising awareness and improved surveillance systems for viral infections. The increase in healthcare spending, coupled with the demand for accurate diagnostic solutions, is fostering the adoption of hepatitis E treatments. European healthcare systems are also focusing on preventive measures, including food safety monitoring and early intervention strategies. The region is experiencing steady growth across hospital and clinical settings, with hepatitis E management becoming an integral part of infectious disease control programs. In addition, government-backed screening initiatives and regulatory support are enhancing market development.
U.K. Hepatitis E Market Insight
The U.K. hepatitis E market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by increasing awareness of viral hepatitis and a strong emphasis on early diagnosis and treatment. In addition, concerns regarding foodborne and waterborne infections are encouraging healthcare providers to enhance screening practices, especially in high-risk groups. The UK’s well-established healthcare infrastructure, alongside its focus on public health initiatives and disease surveillance, is expected to continue to stimulate market growth. Rising adoption of advanced diagnostic technologies and increasing government initiatives further support the expansion of the market.
Germany Hepatitis E Market Insight
The Germany hepatitis E market is expected to expand at a considerable CAGR during the forecast period, fueled by increasing awareness of infectious diseases and the demand for advanced diagnostic and treatment solutions. Germany’s well-developed healthcare infrastructure, combined with its emphasis on research and innovation, promotes the adoption of hepatitis E management strategies. The integration of hepatitis E testing into routine diagnostic frameworks is also becoming increasingly prevalent, particularly in hospital laboratories. In addition, strong regulatory frameworks and increasing healthcare investments are supporting the adoption of effective treatment solutions.
Asia-Pacific Hepatitis E Market Insight
The Asia-Pacific hepatitis E market is poised to grow at the fastest CAGR during the forecast period of 2026 to 2033, driven by high disease prevalence, increasing population, and improving healthcare access in countries such as China, Japan, and India. The region's growing focus on infectious disease control, supported by government initiatives promoting sanitation, clean water access, and vaccination programs, is driving the demand for hepatitis E treatments. Furthermore, as APAC continues to strengthen its healthcare infrastructure, the accessibility of diagnostic and therapeutic solutions is expanding to a wider population base. The rising awareness of waterborne diseases and increasing investments in public health programs are also contributing significantly to market growth.
Japan Hepatitis E Market Insight
The Japan hepatitis E market is gaining momentum due to the country’s advanced healthcare system, aging population, and increasing focus on infectious disease management. The Japanese market places a significant emphasis on early diagnosis and effective treatment, and the adoption of hepatitis E solutions is driven by improved screening practices and technological advancements. The integration of advanced diagnostic technologies, such as molecular testing, is fueling growth. Moreover, Japan's strong research capabilities and government support for healthcare innovation are likely to spur demand for novel treatment and prevention approaches in both clinical and public health sectors.
India Hepatitis E Market Insight
The India hepatitis E market accounted for the largest market revenue share in Asia Pacific in 2025, attributed to the country’s high disease burden, rapid population growth, and increasing awareness of waterborne infections. India represents a significant market for hepatitis E management, with growing demand across hospitals and public health programs. The push towards improved sanitation through government initiatives, expansion of healthcare infrastructure, and rising healthcare expenditure are key factors driving growth. In addition, increasing access to affordable diagnostic and treatment options, along with efforts to enhance disease surveillance, are further propelling the market in India.
Hepatitis E Market Share
The Hepatitis E industry is primarily led by well-established companies, including:
- Xiamen Innovax Biotech Co., Ltd. (China)
- GSK plc (U.K.)
- Merck & Co., Inc. (U.S.)
- Pfizer Inc. (U.S.)
- Sanofi (France)
- Takeda Pharmaceutical Company Limited (Japan)
- Abbott (U.S.)
- F. Hoffmann-La Roche Ltd (Switzerland)
- Siemens Healthineers AG (Germany)
- Bio-Rad Laboratories, Inc. (U.S.)
- DiaSorin S.p.A. (Italy)
- QIAGEN (Netherlands)
- Thermo Fisher Scientific Inc. (U.S.)
- Danaher Corporation (U.S.)
- PerkinElmer, Inc. (U.S.)
- BD (U.S.)
- Grifols, S.A. (Spain)
- Dr. Reddy’s Laboratories Ltd. (India)
- URIHK Pharma Private Limited (India)
What are the Recent Developments in Global Hepatitis E Market?
- In January 2026, Dr. Reddy’s Laboratories announced the launch of its novel recombinant hepatitis E vaccine, Hevaxin, in India, designed for active immunization against hepatitis E virus (HEV) infection in adults aged 18–65 years. The vaccine demonstrated long-lasting immunity and a strong safety profile in clinical evaluations. This launch marks a significant milestone as it addresses a major unmet medical need and supports national hepatitis control programs in a high-burden region
- In August 2025, Urihk Pharmaceuticals introduced a hepatitis E vaccine in India, marking one of the early commercial entries aimed at strengthening preventive healthcare infrastructure. The launch reflects increasing industry focus on vaccine accessibility in emerging economies, where hepatitis E prevalence is high due to sanitation challenges. This development is expected to enhance immunization coverage and reduce disease burden significantly
- In April 2025, the World Health Organization expanded its hepatitis E response strategy by including the hepatitis E vaccine under the International Coordinating Group (ICG) mechanism for outbreak response. This move aims to improve global access to vaccines, especially during emergency situations such as outbreaks in vulnerable regions. It highlights growing institutional support for vaccination as a key preventive measure in hepatitis E management
- In January 2025, researchers reported findings in The Lancet Infectious Diseases indicating that hepatitis E vaccination could remain effective with a reduced two-dose regimen instead of the traditional three-dose schedule. This advancement has the potential to improve patient compliance, lower vaccination costs, and accelerate large-scale immunization programs, particularly in low-resource settings
- In January 2025, a study published in The Lancet Infectious Diseases reported that a two-dose regimen of the Hecolin (HEV 239) vaccine was highly effective in controlling hepatitis E outbreaks, even in high-risk and resource-limited settings. The findings demonstrated strong protection after just two doses, improving feasibility for mass immunization campaigns and emergency outbreak responses
SKU-
Получите онлайн-доступ к отчету на первой в мире облачной платформе рыночной аналитики
- Интерактивная панель анализа данных
- Панель анализа компании для возможностей с высоким потенциалом роста
- Доступ аналитика-исследователя для настройки и запросов
- Анализ конкурентов с помощью интерактивной панели
- Последние новости, обновления и анализ тенденций
- Используйте возможности сравнительного анализа для комплексного отслеживания конкурентов
Методология исследования
Сбор данных и анализ базового года выполняются с использованием модулей сбора данных с большими размерами выборки. Этап включает получение рыночной информации или связанных данных из различных источников и стратегий. Он включает изучение и планирование всех данных, полученных из прошлого заранее. Он также охватывает изучение несоответствий информации, наблюдаемых в различных источниках информации. Рыночные данные анализируются и оцениваются с использованием статистических и последовательных моделей рынка. Кроме того, анализ доли рынка и анализ ключевых тенденций являются основными факторами успеха в отчете о рынке. Чтобы узнать больше, пожалуйста, запросите звонок аналитика или оставьте свой запрос.
Ключевой методологией исследования, используемой исследовательской группой DBMR, является триангуляция данных, которая включает в себя интеллектуальный анализ данных, анализ влияния переменных данных на рынок и первичную (отраслевую экспертную) проверку. Модели данных включают сетку позиционирования поставщиков, анализ временной линии рынка, обзор рынка и руководство, сетку позиционирования компании, патентный анализ, анализ цен, анализ доли рынка компании, стандарты измерения, глобальный и региональный анализ и анализ доли поставщика. Чтобы узнать больше о методологии исследования, отправьте запрос, чтобы поговорить с нашими отраслевыми экспертами.
Доступна настройка
Data Bridge Market Research является лидером в области передовых формативных исследований. Мы гордимся тем, что предоставляем нашим существующим и новым клиентам данные и анализ, которые соответствуют и подходят их целям. Отчет можно настроить, включив в него анализ ценовых тенденций целевых брендов, понимание рынка для дополнительных стран (запросите список стран), данные о результатах клинических испытаний, обзор литературы, обновленный анализ рынка и продуктовой базы. Анализ рынка целевых конкурентов можно проанализировать от анализа на основе технологий до стратегий портфеля рынка. Мы можем добавить столько конкурентов, о которых вам нужны данные в нужном вам формате и стиле данных. Наша команда аналитиков также может предоставить вам данные в сырых файлах Excel, сводных таблицах (книга фактов) или помочь вам в создании презентаций из наборов данных, доступных в отчете.