Global Induced Pluripotent Stem Cells Market
Размер рынка в млрд долларов США
CAGR :
% 
 USD
2.13 Billion
USD
4.53 Billion
2025
2033
USD
2.13 Billion
USD
4.53 Billion
2025
2033
| 2026 –2033 | |
| USD 2.13 Billion | |
| USD 4.53 Billion | |
|
|
|
|
Global Induced Pluripotent Stem Cells (iPSCs) Market Segmentation, By Cell Source (Skin Cells and Blood Cells), Type (Human IPSCs and Mouse IPSCs), Product (Instruments, Consumables & Kits and Services), Applications (Academic Research, Regenerative Medicine, Cellular Therapy, Toxicology Screening, Drug Discovery and Development, Disease Modelling, Stem Cell Banking, 3D Bio printing and Others), End User (Biotechnology & Pharmaceutical Companies, Research Laboratories, Diagnostic Laboratories and Others), Distribution Channel (Direct Tender and Retail Sales) - Industry Trends and Forecast to 2033
Induced Pluripotent Stem Cells (iPSCs) Market Size
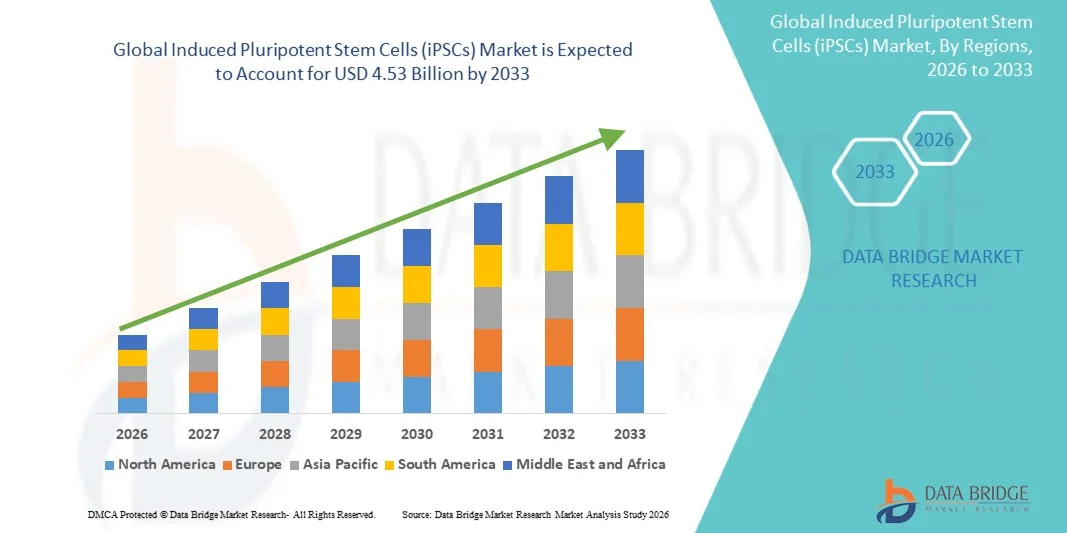
- The global induced pluripotent stem cells (iPSCs) market size was valued at USD 2.13 billion in 2025 and is expected to reach USD 4.53 billion by 2033, at a CAGR of 9.90% during the forecast period
- The market growth is largely fueled by the increasing adoption of advanced cell-based research techniques and rapid technological progress in stem cell biology, leading to expanded applications of induced pluripotent stem cells in disease modeling, drug discovery, and regenerative medicine across research and clinical settings
- Furthermore, the rising demand for personalized medicine, coupled with growing investments in stem cell research and biopharmaceutical development, is establishing iPSCs as a critical tool in modern healthcare innovation. These converging factors are accelerating the uptake of induced pluripotent stem cells (iPSCs) solutions, thereby significantly boosting the market growth
Induced Pluripotent Stem Cells (iPSCs) Market Analysis
- Induced pluripotent stem cells (iPSCs), which are generated by reprogramming adult cells into a pluripotent state, are increasingly vital in biomedical research and regenerative medicine due to their ability to differentiate into multiple cell types and support advanced applications such as disease modeling and drug discovery
- The escalating demand for iPSCs is primarily driven by growing investments in stem cell research, increasing focus on personalized medicine, and expanding applications in toxicity testing, regenerative therapies, and clinical research
- North America dominated the induced pluripotent stem cells (iPSCs) market with a revenue share of approximately 42.6% in 2025, supported by strong R&D funding, advanced healthcare infrastructure, and the presence of leading biotechnology firms. The U.S. continues to lead due to extensive clinical research and innovation in stem cell technologies
- Asia-Pacific is expected to be the fastest-growing region in the induced pluripotent stem cells (iPSCs) market during the forecast period, driven by rising government support, increasing investments in biotechnology, and rapid expansion of research capabilities in countries such as China, Japan, and India
- The Human iPSCs segment held the largest market revenue share of 64.8% in 2025, driven by their extensive application in clinical research, drug discovery, and regenerative medicine
Report Scope and Induced Pluripotent Stem Cells (iPSCs) Market Segmentation
|
Attributes |
Induced Pluripotent Stem Cells (iPSCs) Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
• Thermo Fisher Scientific Inc. (U.S.) |
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Induced Pluripotent Stem Cells (iPSCs) Market Trends
“Growing Adoption in Regenerative Medicine and Disease Modeling”
- A significant and accelerating trend in the global Induced Pluripotent Stem Cells (iPSCs) market is the increasing adoption of iPSCs in regenerative medicine, disease modeling, and drug discovery applications
- iPSCs are being widely utilized to generate patient-specific cell lines, enabling researchers to study disease mechanisms more accurately and develop targeted therapeutic approaches
- For instance, iPSC-derived cells are increasingly used in modeling complex diseases such as neurodegenerative disorders, cardiovascular conditions, and rare genetic diseases
- In addition, advancements in reprogramming technologies and cell culture techniques are improving the efficiency, scalability, and reliability of iPSC production
- The growing use of iPSCs in toxicity testing and personalized medicine is further enhancing their importance in pharmaceutical research and development
- Furthermore, collaborations between research institutions and biotechnology companies are accelerating innovation and commercialization of iPSC-based applications
- This trend is significantly expanding the scope and applications of iPSCs, driving growth in the global market
Induced Pluripotent Stem Cells (iPSCs) Market Dynamics
Driver
“Rising Demand for Personalized Medicine and Drug Discovery”
- The increasing demand for personalized medicine and more effective drug discovery processes is a major driver for the Induced Pluripotent Stem Cells (iPSCs) market
- iPSCs enable the development of patient-specific therapies by allowing researchers to create customized cell models that reflect individual genetic profiles
- For instance, pharmaceutical companies are increasingly using iPSC-based models to screen drug candidates, reduce failure rates in clinical trials, and improve drug safety and efficacy
- In addition, the growing prevalence of chronic and genetic diseases is driving the need for advanced research tools and innovative therapeutic solutions
- Increasing investments in stem cell research, along with supportive government initiatives and funding programs, are further accelerating market growth
- Moreover, the ability of iPSCs to bypass ethical concerns associated with embryonic stem cells is encouraging wider adoption in research and clinical applications
- These factors collectively are significantly contributing to the expansion of the Induced Pluripotent Stem Cells (iPSCs) market
Restraint/Challenge
“High Production Costs and Technical Complexities”
- One of the major challenges in the Induced Pluripotent Stem Cells (iPSCs) market is the high cost associated with cell reprogramming, maintenance, and large-scale production
- The process of generating high-quality iPSCs requires specialized equipment, skilled professionals, and stringent quality control measures, increasing overall operational costs
- For instance, variability in reprogramming efficiency and differentiation potential can affect reproducibility and limit widespread adoption in clinical settings
- In addition, technical challenges related to genomic instability, potential tumorigenicity, and long-term safety concerns remain significant barriers to clinical translation
- Regulatory complexities and the need for extensive validation and clinical trials further delay commercialization of iPSC-based therapies
- Furthermore, limited standardization across protocols and lack of skilled workforce in certain regions can hinder market growth
- Addressing these challenges through technological advancements, cost optimization, and regulatory harmonization will be essential for sustained market expansion
Induced Pluripotent Stem Cells (iPSCs) Market Scope
The market is segmented on the basis of cell source, type, product, application, end user, and distribution channel.
• By Cell Source
On the basis of cell source, the Induced Pluripotent Stem Cells (iPSCs) market is segmented into Skin Cells and Blood Cells. The Skin Cells segment dominated the largest market revenue share of 57.3% in 2025, driven by their ease of accessibility and well-established reprogramming protocols. Skin fibroblasts are widely used due to their high reprogramming efficiency and reliability in generating stable iPSC lines. The segment benefits from extensive use in research applications, particularly in disease modeling and regenerative medicine. In addition, skin cells are relatively easy to obtain through minimally invasive procedures, supporting widespread adoption. The availability of standardized protocols further enhances reproducibility in laboratory settings. Researchers prefer skin-derived iPSCs due to their proven track record in various experimental studies. Increasing investments in stem cell research are also contributing to segment growth. Moreover, the expansion of personalized medicine is boosting demand for patient-specific cell lines. Continuous advancements in reprogramming technologies further strengthen this segment. Overall, accessibility and reliability maintain its dominant position in the market.
The Blood Cells segment is anticipated to witness the fastest growth rate of 22.6% from 2026 to 2033, fueled by the growing preference for less invasive sample collection methods. Blood cells can be obtained through simple procedures, enhancing patient comfort and compliance. The segment is gaining traction due to advancements in reprogramming efficiency and reduced turnaround time. Increasing adoption in clinical research and therapeutic applications is also supporting growth. In addition, blood-derived iPSCs are highly suitable for studying hematological disorders. Technological innovations are improving the quality and scalability of cell production. Rising demand for personalized therapies further accelerates adoption. The segment also benefits from growing research in immunology and regenerative medicine. Increasing collaborations between research institutions and biotech companies support expansion. As minimally invasive techniques gain popularity, this segment is expected to grow rapidly.
• By Type
On the basis of type, the Induced Pluripotent Stem Cells (iPSCs) market is segmented into Human iPSCs and Mouse iPSCs. The Human iPSCs segment held the largest market revenue share of 64.8% in 2025, driven by their extensive application in clinical research, drug discovery, and regenerative medicine. Human iPSCs closely mimic human physiological conditions, making them highly valuable for disease modeling and therapeutic development. The segment benefits from increasing investments in personalized medicine and cell-based therapies. In addition, regulatory support for stem cell research is encouraging adoption. Pharmaceutical companies widely use human iPSCs for drug screening and toxicity testing. The growing focus on precision medicine further supports segment growth. Technological advancements are improving reprogramming efficiency and scalability. Rising prevalence of chronic diseases is increasing demand for innovative treatment approaches. Strong collaboration between academia and industry also drives expansion. Overall, their clinical relevance and broad applicability maintain dominance.
The Mouse iPSCs segment is expected to witness the fastest CAGR of 20.9% from 2026 to 2033, driven by their extensive use in preclinical studies and basic research. Mouse models are essential for understanding disease mechanisms and testing new therapies before human trials. The segment benefits from well-established animal research frameworks and lower costs compared to human studies. Increasing research activities in genetics and developmental biology further support growth. In addition, advancements in gene editing technologies such as CRISPR are enhancing the utility of mouse iPSCs. Academic institutions heavily rely on these models for experimental studies. Growing funding for biomedical research is also contributing to segment expansion. The segment plays a crucial role in early-stage drug development. Continuous innovation in animal models improves research outcomes. As preclinical research continues to expand, this segment is expected to grow steadily.
• By Product
On the basis of product, the Induced Pluripotent Stem Cells (iPSCs) market is segmented into Instruments, Consumables & Kits, and Services. The Consumables & Kits segment dominated the largest market revenue share of 46.5% in 2025, driven by their recurring usage in cell culture, reprogramming, and maintenance processes. These products are essential for routine laboratory operations, ensuring consistent demand. The segment benefits from continuous advancements in reagents and kits that improve efficiency and reliability. Increasing number of research projects involving iPSCs further supports growth. In addition, consumables are widely used across academic and industrial settings. The availability of specialized kits enhances ease of use and standardization. Pharmaceutical companies and research institutions heavily rely on these products. Growing focus on stem cell research is also contributing to higher consumption. The expansion of biotechnology industries further supports demand. Overall, high usage frequency maintains strong market dominance.
The Services segment is anticipated to witness the fastest growth rate of 23.4% from 2026 to 2033, fueled by increasing outsourcing of stem cell research and development activities. Many organizations prefer outsourcing due to cost efficiency and access to specialized expertise. Service providers offer customized solutions such as cell line generation and characterization. The segment benefits from growing collaborations between biotech firms and research institutions. In addition, increasing demand for high-quality and standardized iPSC lines supports growth. Technological advancements are improving service quality and turnaround time. Rising focus on personalized medicine further drives demand for specialized services. The expansion of contract research organizations also contributes to segment growth. Increasing investment in life sciences research supports outsourcing trends. As demand for expertise and scalability rises, this segment is expected to grow rapidly.
• By Application
On the basis of application, the Induced Pluripotent Stem Cells (iPSCs) market is segmented into Academic Research, Regenerative Medicine, Cellular Therapy, Toxicology Screening, Drug Discovery and Development, Disease Modelling, Stem Cell Banking, 3D Bioprinting, and others. The Drug Discovery and Development segment accounted for the largest market revenue share of 34.9% in 2025, driven by the increasing use of iPSCs in screening and validating new drug candidates. iPSCs provide a reliable platform for testing drug efficacy and safety in human-like cellular environments. The segment benefits from rising investments by pharmaceutical companies in innovative drug development. In addition, iPSCs help reduce dependency on animal testing, improving research efficiency. Growing demand for personalized medicine further supports segment growth. Technological advancements are enhancing screening capabilities and accuracy. Increasing prevalence of chronic diseases drives demand for new therapies. Strong collaboration between academia and industry also contributes to expansion. Continuous innovation in cell-based assays further supports growth. Overall, their role in modern drug development ensures dominance.
The Regenerative Medicine segment is expected to witness the fastest CAGR of 24.1% from 2026 to 2033, driven by the growing potential of iPSCs in tissue repair and organ regeneration. Increasing research on stem cell therapies is supporting segment growth. iPSCs offer the ability to generate patient-specific cells, reducing the risk of immune rejection. The segment benefits from rising investments in advanced therapeutic solutions. In addition, advancements in tissue engineering and 3D bioprinting are enhancing application scope. Growing prevalence of chronic and degenerative diseases further drives demand. Regulatory support for regenerative therapies is also encouraging adoption. Pharmaceutical and biotech companies are focusing on developing cell-based treatments. Increasing clinical trials in regenerative medicine contribute to growth. As technology continues to evolve, this segment is expected to expand rapidly.
• By End User
On the basis of end user, the Induced Pluripotent Stem Cells (iPSCs) market is segmented into Biotechnology & Pharmaceutical Companies, Research Laboratories, Diagnostic Laboratories, and others. The Biotechnology & Pharmaceutical Companies segment dominated the largest market revenue share of 51.2% in 2025, driven by extensive use of iPSCs in drug discovery, toxicity testing, and therapeutic development. These companies invest heavily in advanced technologies to accelerate research and innovation. The segment benefits from increasing demand for personalized medicine and targeted therapies. In addition, strong funding and infrastructure support large-scale research activities. Collaborations with academic institutions further enhance innovation. The rising number of clinical trials involving stem cell therapies also supports growth. Companies rely on iPSCs for improving drug development efficiency. Increasing prevalence of chronic diseases further drives demand. Continuous technological advancements strengthen segment capabilities. Overall, strong industrial adoption maintains market dominance.
The Research Laboratories segment is anticipated to witness the fastest growth rate of 22.8% from 2026 to 2033, fueled by increasing academic research activities and government funding. Research institutions play a crucial role in advancing stem cell science and exploring new applications. The segment benefits from growing collaborations with industry players. Increasing number of research projects focused on disease modeling and regenerative medicine supports growth. In addition, availability of grants and funding programs encourages innovation. Technological advancements are improving research capabilities. Rising interest in personalized medicine further drives adoption. Academic institutions are expanding their infrastructure to support stem cell research. The growing focus on translational research also contributes to expansion. As research activities continue to increase, this segment is expected to grow rapidly.
• By Distribution Channel
On the basis of distribution channel, the Induced Pluripotent Stem Cells (iPSCs) market is segmented into Direct Tender and Retail Sales. The Direct Tender segment held the largest market revenue share of 62.4% in 2025, driven by bulk purchasing by large research institutions, biotechnology firms, and pharmaceutical companies. Direct procurement ensures cost efficiency and reliable supply of high-quality products. The segment benefits from long-term contracts and strategic partnerships between suppliers and end users. In addition, direct tendering allows customization of products and services. Increasing large-scale research projects further support demand. Strong supplier relationships enhance procurement efficiency. The segment also benefits from consistent demand from established organizations. Growing investments in life sciences research contribute to expansion. Overall, large-scale purchasing and strong partnerships maintain dominance.
The Retail Sales segment is expected to witness the fastest CAGR of 19.7% from 2026 to 2033, driven by increasing accessibility of iPSC-related products through distributors and online platforms. Smaller research labs and academic institutions prefer retail channels for flexibility and convenience. The segment benefits from the expansion of e-commerce in scientific supplies. Increasing number of startups and small-scale research facilities supports growth. In addition, improved distribution networks enhance product availability. Technological advancements are simplifying procurement processes. Growing awareness about stem cell research further drives demand. Retail channels provide access to a wide range of products. As market accessibility improves, this segment is expected to grow steadily.
Induced Pluripotent Stem Cells (iPSCs) Market Regional Analysis
- North America dominated the induced pluripotent stem cells (iPSCs) market with the largest revenue share of 42.6% in 2025
- Supported by strong R&D funding, advanced healthcare infrastructure, and the presence of leading biotechnology firms. The U.S. continues to lead the region due to extensive clinical research and innovation in stem cell technologies
- The widespread adoption of iPSCs in regenerative medicine, disease modeling, and drug discovery, along with government and private funding initiatives, is further driving market growth in North America.
U.S. Induced Pluripotent Stem Cells (iPSCs) Market Insight
The U.S. induced pluripotent stem cells (iPSCs) market accounted for the largest revenue share in North America in 2025, fueled by extensive clinical research, well-established biotechnology infrastructure, and ongoing innovation in stem cell-based therapies. Increasing applications in personalized medicine, toxicity testing, and disease modeling are accelerating the adoption of iPSCs. Furthermore, collaborations between pharmaceutical companies and research institutions are expanding the development of advanced iPSC-derived therapies, bolstering the U.S. market.
Europe Induced Pluripotent Stem Cells (iPSCs) Market Insight
The Europe induced pluripotent stem cells (iPSCs) market is projected to witness substantial growth throughout the forecast period, driven by rising investments in biotechnology, supportive government initiatives, and increasing use of iPSCs in drug development and disease modeling. Countries such as Germany, France, and the U.K. are witnessing growth in regenerative medicine research, which is supporting market expansion. The adoption of standardized protocols and increased clinical trials further enhance market development.
U.K. Induced Pluripotent Stem Cells (iPSCs) Market Insight
The U.K. induced pluripotent stem cells (iPSCs) market is expected to grow at a noteworthy CAGR during the forecast period, driven by government-backed funding for stem cell research and increasing collaborations between academic institutions and biotechnology companies. The rising focus on personalized medicine and advancements in disease modeling are further contributing to market growth.
Germany Induced Pluripotent Stem Cells (iPSCs) Market Insight
The Germany induced pluripotent stem cells (iPSCs) market is anticipated to expand at a considerable CAGR during the forecast period due to the presence of advanced healthcare infrastructure, strong biotechnology research, and increasing applications of iPSCs in regenerative medicine. Emphasis on innovation and sustainable biotechnology practices is further promoting the adoption of iPSC technologies.
Asia-Pacific Induced Pluripotent Stem Cells (iPSCs) Market Insight
The Asia-Pacific induced pluripotent stem cells (iPSCs) market is poised to grow at the fastest CAGR during the forecast period, driven by rising government support, increasing investments in biotechnology, and the rapid expansion of research capabilities in countries such as China, Japan, and India. In addition, growing awareness of advanced therapeutic options and increasing clinical research activities are accelerating market adoption.
Japan Induced Pluripotent Stem Cells (iPSCs) Market Insight
The Japan induced pluripotent stem cells (iPSCs) market is gaining momentum due to a strong focus on regenerative medicine research, growing biotechnology investments, and increasing government initiatives supporting stem cell therapies. The aging population and demand for innovative treatments are also driving the development and adoption of iPSC technologies.
China Induced Pluripotent Stem Cells (iPSCs) Market Insight
The China induced pluripotent stem cells (iPSCs) market accounted for the largest revenue share in Asia-Pacific in 2025, driven by the expansion of research infrastructure, government-backed biotechnology programs, and increasing prevalence of chronic and genetic diseases. China’s growing investments in regenerative medicine, drug development, and iPSC-based clinical studies are further supporting market growth.
Induced Pluripotent Stem Cells (iPSCs) Market Share
The Induced Pluripotent Stem Cells (iPSCs) industry is primarily led by well-established companies, including:
• Thermo Fisher Scientific Inc. (U.S.)
• Lonza Group AG (Switzerland)
• Merck KGaA (Germany)
• GE Healthcare (U.K.)
• STEMCELL Technologies Inc. (Canada)
• ReproCELL Inc. (Japan)
• Fujifilm Cellular Dynamics, Inc. (U.S.)
• Axol Bioscience Ltd. (U.K.)
• Cellartis AB (Sweden)
• Life Technologies Corporation (U.S.)
• Ncardia BV (Netherlands)
• Astellas Pharma Inc. (Japan)
• Cellular Dynamics International, Inc. (U.S.)
• Cytiva (Sweden)
• Pluristem Therapeutics Inc. (Israel)
Latest Developments in Global Induced Pluripotent Stem Cells (iPSCs) Market
- In September 2021, Fate Therapeutics announced the expansion of its induced pluripotent stem cell (iPSC)-derived natural killer (NK) cell therapy pipeline, advancing multiple clinical programs targeting cancer, reflecting growing commercialization of iPSC-based immunotherapies
- In June 2022, FUJIFILM Cellular Dynamics announced the expansion of its iPSC manufacturing capabilities with new large-scale production facilities, aimed at supporting increasing global demand for clinical-grade iPSC lines used in regenerative medicine and drug discovery
- In October 2022, Evotec SE entered into a strategic collaboration with a major pharmaceutical partner to utilize iPSC-based platforms for drug discovery and disease modeling, strengthening the adoption of iPSC technology in precision medicine research
- In March 2023, Ncardia launched advanced iPSC-derived cardiomyocyte and neuron assay platforms designed to improve drug screening accuracy and reduce late-stage drug failure rates in pharmaceutical development
- In July 2023, European Bank for induced pluripotent Stem Cells expanded its iPSC biobank and distribution services across Europe, enhancing access to high-quality, disease-specific iPSC lines for academic and commercial research applications
- In December 2024, leading biotechnology companies intensified collaborations with pharmaceutical firms to utilize iPSC-derived cells for toxicity testing and drug screening, reflecting a growing shift toward human-relevant models in preclinical research
- In February 2025, U.S. Food and Drug Administration granted Investigational New Drug (IND) clearance for Fertilo, the first iPSC-derived therapy to enter a Phase III clinical trial in the United States, marking a major milestone in clinical translation of iPSC-based therapies
- In January 2025, industry reports highlighted that over 100 clinical trials involving human pluripotent stem cell-based therapies, including iPSCs, were actively ongoing globally, demonstrating rapid progress in clinical adoption and increasing investment in regenerative medicine
SKU-
Получите онлайн-доступ к отчету на первой в мире облачной платформе рыночной аналитики
- Интерактивная панель анализа данных
- Панель анализа компании для возможностей с высоким потенциалом роста
- Доступ аналитика-исследователя для настройки и запросов
- Анализ конкурентов с помощью интерактивной панели
- Последние новости, обновления и анализ тенденций
- Используйте возможности сравнительного анализа для комплексного отслеживания конкурентов
Методология исследования
Сбор данных и анализ базового года выполняются с использованием модулей сбора данных с большими размерами выборки. Этап включает получение рыночной информации или связанных данных из различных источников и стратегий. Он включает изучение и планирование всех данных, полученных из прошлого заранее. Он также охватывает изучение несоответствий информации, наблюдаемых в различных источниках информации. Рыночные данные анализируются и оцениваются с использованием статистических и последовательных моделей рынка. Кроме того, анализ доли рынка и анализ ключевых тенденций являются основными факторами успеха в отчете о рынке. Чтобы узнать больше, пожалуйста, запросите звонок аналитика или оставьте свой запрос.
Ключевой методологией исследования, используемой исследовательской группой DBMR, является триангуляция данных, которая включает в себя интеллектуальный анализ данных, анализ влияния переменных данных на рынок и первичную (отраслевую экспертную) проверку. Модели данных включают сетку позиционирования поставщиков, анализ временной линии рынка, обзор рынка и руководство, сетку позиционирования компании, патентный анализ, анализ цен, анализ доли рынка компании, стандарты измерения, глобальный и региональный анализ и анализ доли поставщика. Чтобы узнать больше о методологии исследования, отправьте запрос, чтобы поговорить с нашими отраслевыми экспертами.
Доступна настройка
Data Bridge Market Research является лидером в области передовых формативных исследований. Мы гордимся тем, что предоставляем нашим существующим и новым клиентам данные и анализ, которые соответствуют и подходят их целям. Отчет можно настроить, включив в него анализ ценовых тенденций целевых брендов, понимание рынка для дополнительных стран (запросите список стран), данные о результатах клинических испытаний, обзор литературы, обновленный анализ рынка и продуктовой базы. Анализ рынка целевых конкурентов можно проанализировать от анализа на основе технологий до стратегий портфеля рынка. Мы можем добавить столько конкурентов, о которых вам нужны данные в нужном вам формате и стиле данных. Наша команда аналитиков также может предоставить вам данные в сырых файлах Excel, сводных таблицах (книга фактов) или помочь вам в создании презентаций из наборов данных, доступных в отчете.