北美 CRISPR 基因检测和诊断市场,按类别(第 1 类 - 多种效应蛋白和第 2 类 - 单个 CrRNA 结合蛋白)、产品和服务(产品和服务)、应用(生物医学诊断、基因组工程、药物发现、农业应用和其他)、工作流程(样品制备、预扩增、CrRNA、Cas 酶和传感)、最终用户(医院、诊断中心、生物技术公司、学术和研究机构等)、分销渠道(直接招标、零售销售)划分的行业趋势和预测到 2029 年
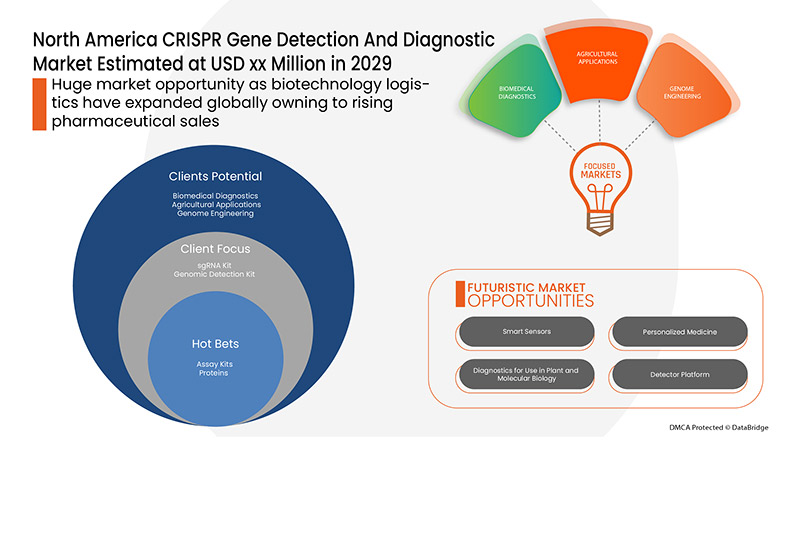
市场定义和见解
CRISPR 是一种基因组编辑工具,它允许研究人员轻松改变 DNA 序列和修改基因功能。它有许多潜在的应用,包括纠正遗传缺陷以及治疗和预防疾病传播。基于 CRISPR 的诊断方法已用于许多生物医学应用,例如检测传染性和非传染性疾病的核酸生物标志物以及检测遗传疾病。CRISPR 中的检测试剂盒由两部分组成:一种称为 Cas9 的蛋白质和一种向导 RNA,即一串具有特定遗传密码的核酸分子。

该 CRISPR-Cas9 系统已针对哺乳动物细胞进行了修改。我们可以通过非同源末端连接 (NHEJ) 引入移码突变,从而引入特定于我们感兴趣的基因的引导序列 (sgRNA) 来敲除特定基因,或者产生敲入突变。
CRISPR-Cas 9 系统扩大了基因和细胞治疗的诊断和服务范围。制药公司在研发方面投入巨资以开发新产品,大量基因和细胞治疗药物进入早期开发阶段。市场参与者的投资将为急需治疗的患者提供安全有效的治疗方案。
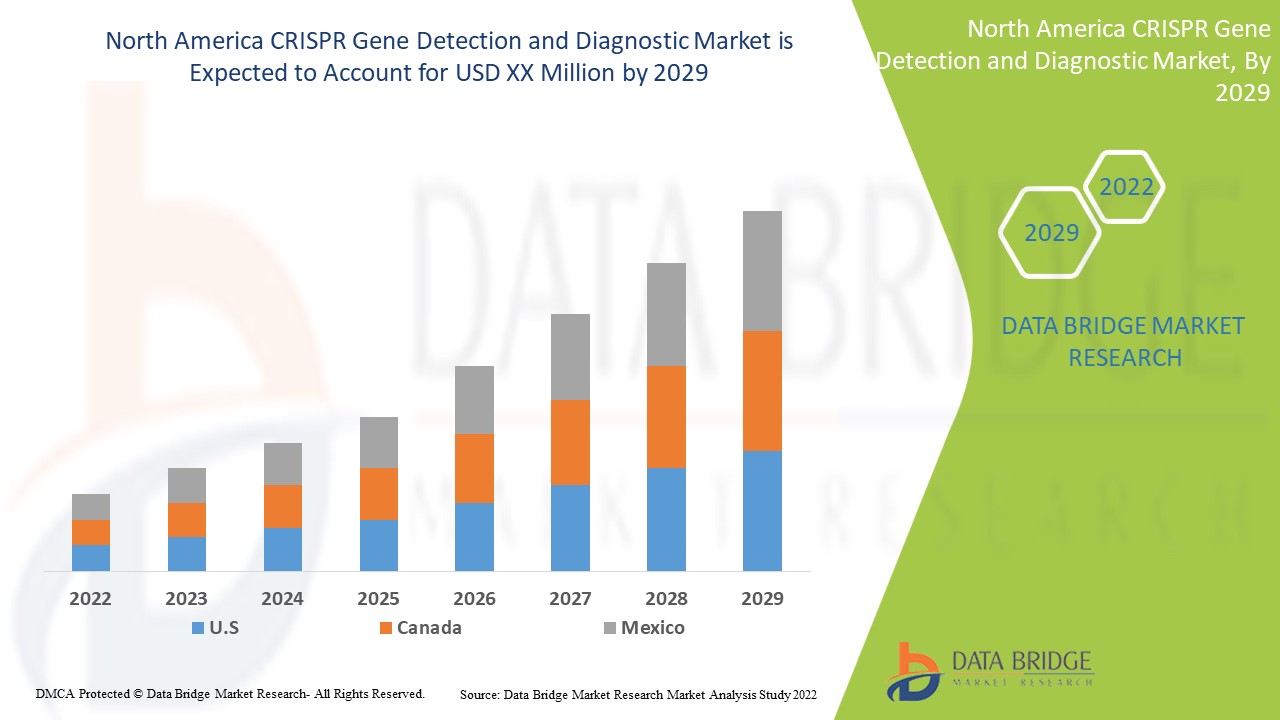
北美 CRISPR 基因检测和诊断具有支持作用,旨在减轻症状的严重程度。Data Bridge Market Research 分析,在 2022 年至 2029 年的预测期内,CRISPR 基因检测和诊断市场将以 19.6% 的复合年增长率增长。
|
报告指标 |
细节 |
|
预测期 |
2022 至 2029 年 |
|
基准年 |
2021 |
|
历史岁月 |
2020(可定制至 2019 - 2014) |
|
定量单位 |
收入(百万美元),定价(美元) |
|
涵盖的领域 |
按类别(第 1 类 - 多种效应蛋白和第 2 类 - 单个 CrRNA 结合蛋白)、产品和服务(产品和服务)、应用(生物医学诊断、基因组工程、药物发现、农业应用等)、工作流程(样品制备、预扩增、CrRNA、Cas 酶和传感)、最终用户(医院、诊断中心、生物技术公司、学术和研究机构等)分销渠道(直接招标、零售) |
|
覆盖国家 |
美国、加拿大和墨西哥 |
|
涵盖的市场参与者 |
GenScript、OriGene Technologies, Inc.、Applied StemCell、GeneCopoeia, Inc.、Agilent Technologies, Inc.、Synthego、BioVision Inc.、Hera Biolabs、Cellecta, Inc.、New England Biolabs、10x Genomics、addgene CasTag Biosciences、Merck KGaA、Integrated DNA Technologies, Inc.(Danaher 的子公司)、Thermo Fisher Scientific Inc. 等 |
北美 CRISPR 基因检测和诊断市场动态
驱动程序
- 慢性病患病率和发病率上升
慢性病是常见的疾病,每3个成年人中就有1人患有慢性病,慢性病影响着广大人民群众的健康和生活质量。
CRISPR 是成簇的规律间隔的短回文重复序列的缩写。近年来,CRISPR 已成为基因编辑的主导工具,用于改变细胞中 DNA 的特定序列。CRISPR 在亨廷顿氏病、肌肉营养不良症、癌症和高胆固醇的研究和治疗中具有重要用途。
例如,
- 2021 年,NORD(美国罕见疾病组织)的数据显示,杜氏肌营养不良症 (DMD) 的诊断发病率很高。杜氏肌营养不良症 (DMD) 是一种常见的遗传病,全球每 3,500 名男性新生儿中就有 1 名患有此病
- 研发投资增加
基因编辑技术,例如 CRISPR-Cas 9 系统,已经扩大了基因和细胞治疗的诊断和服务范围。制药公司在研发方面投入大量资金来开发新产品,大量基因和细胞治疗药物进入早期开发阶段。市场参与者的投资将实现为急需的患者提供安全有效的治疗的目标。
例如,
- 2022 年 2 月,Synthego 筹集了 2 亿美元用于研发,以推动基于 CRISPR 的药物从早期研究到临床的开发。Synthego 将利用 E 轮融资的投资金额来加速创建 CRISPR 诊断和服务
CRISPR 基因诊断的资金可用性
CRISPR 基因诊断和研究由美国国立卫生研究院 (NIH) 预算资助。私营部门也资助 CRISPR 基因检测和研究,但这种投资通常发生在测试和开发阶段,然后是初始基础研究。由于基因组编辑是一个如此新的领域,必须有一个公正的政府机构对其进行监督;FDA 谨慎而彻底,但他们在无休止地争取资金,进行长期投资,使付款与潜在的未来受益者保持一致,这将进一步促进 CRISPR 基因检测和诊断市场的增长。
此外,CRISPR 基因诊断技术的进步、公共和私人组织不断增加的宣传活动以及政府资金的增加都是扩大 CRISPR 基因检测市场的因素。其他因素,如对有效疗法的需求增加以及对及时诊断的认识不断提高,将对 CRISPR 基因检测和诊断市场的增长率产生积极影响。此外,高可支配收入、慢性病数量增加、生活方式的改变将导致 CRISPR 基因检测和诊断市场的扩大。
机会
- 医疗支出的增加
此外,研发活动的增加以及政府和私人组织投资的增加将为市场增长率带来新的机遇。
- 市场参与者的战略主动性
由于对慢性病的及时治疗,美国对 CRISPR 基因检测和诊断的需求有所增加。这些有利因素增加了对药物的需求,为了满足市场需求,大大小小的市场参与者都在采用各种策略。
主要参与者还试图制定具体的策略,例如产品发布、收购、批准、扩张和合作,以确保业务顺利进行,避免风险,并增加市场销售的长期增长。
例如,
- 2021 年 5 月,Horizon Discovery Ltd. 在沃尔瑟姆扩展了基因调节产品组合,推出了首个合成单向导 RNA 和正在申请专利的用于 CRISPR 干扰的 dcas9 抑制剂。产品组合的扩展增加了合成向导 RNA 产品组合在美国和英国地区的销售和收入,并增加了与市场参与者的合作
此外,有效疗法的推出和持续的临床试验将为 2022-2029 年预测期内的 CRISPR 基因检测和诊断市场提供有利机会。此外,当前未满足的大量需求和医疗技术的发展将在未来提高 CRISPR 基因检测和诊断市场的增长率。
限制/挑战
然而,CRISPR 诊断的高成本和使用 CRISPR 诊断时面临的风险将阻碍 CRISPR 基因检测和诊断市场的增长率。此外,使用 MRI 设备时产生的风险也将阻碍 CRISPR 基因检测和诊断市场的增长。缺乏熟练的专业知识和法规将在上述预测期内进一步挑战市场。
- 基于 CRISPR 的诊断成本上升
基于 CRISPR 的疗法具有巨大的潜力,但成本较高。大多数基因组编辑疗法需要更长的开发和生产时间,因此成本上升。此外,与 CRISPR 基因检测和诊断相关的检测试剂盒和药物适用于大部分人群。这些成本被转嫁给患者。因此,目前的高成本预计未来将呈下降趋势。
例如,
- 2021 年 7 月,据 Integrated DNA Technologies, Inc. 称,首个基于 CRISPR 的 SARS-CoV-2 商业化诊断检测试剂盒(包括逆转录 LAMP (RT-LAMP) 作为预扩增)目前每反应价格为 30.15 美元
CRISPR 基因检测和诊断市场报告提供了最新发展、贸易法规、进出口分析、生产分析、价值链优化、市场份额、国内和本地市场参与者的影响的详细信息,分析了新兴收入领域的机会、市场法规的变化、战略市场增长分析、市场规模、类别市场增长、应用领域和主导地位、产品批准、产品发布、地域扩展、市场技术创新。如需了解有关 CRISPR 基因检测和诊断市场的更多信息,请联系 Data Bridge Market Research 获取分析师简报,我们的团队将帮助您做出明智的市场决策,实现市场增长。
患者流行病学分析
根据Globocan的研究,2020年乳腺癌的发病率较高,约为11.7%,其次是肺癌(11.40%)、结直肠癌(10.00%),宫颈癌和食道癌的发病率较低。
CRISPR 基因检测和诊断市场还为您提供有关患者分析、预后和治疗的详细市场分析。患病率、发病率、死亡率、依从率是报告中提供的一些数据变量。分析流行病学对市场增长的直接或间接影响,以创建更稳健、更队列的多元统计模型,用于预测增长期的市场。
COVID-19 对CRISPR 基因检测和诊断市场的影响
COVID-19 给市场带来了负面影响。疫情期间的封锁和隔离使诊断管理和治疗变得复杂。无法获得常规和药物管理的医疗设施将进一步影响市场。社会隔离会增加压力、绝望和社会支持,所有这些都可能导致疫情期间抗惊厥药物依从性的降低。
近期发展
- 2020 年 8 月,SHERLOCK BIOSCIENCES 宣布与 Dartmouth-Hitchcock Health 合作,开展 SHERLOCK 新冠病毒诊断试剂盒临床试验。该试剂盒已获得美国食品药品监督管理局 (FDA) 的紧急使用授权 (EUA) 紧急批准。
北美 CRISPR 基因检测和诊断市场范围
CRISPR 基因检测和诊断市场分为六个部分:类别、产品和服务、应用、工作流程、最终用户和分销渠道。这些部分之间的增长将帮助您分析行业中微弱的增长部分,并为用户提供有价值的市场概览和市场洞察,帮助他们做出战略决策,确定核心市场应用。
班级
- 第 1 类 - 多重效应蛋白
- 第 2 类 - 单个 CrRNA 结合蛋白
根据类别,CRISPR 基因检测和诊断市场分为第 1 类 - 多种效应蛋白和第 2 类 - 单个 CrRNA 结合蛋白。
产品与服务
- 产品
- 服务
根据产品和服务,CRISPR 基因检测和诊断市场细分为产品和服务。
应用
- 生物医学诊断
- 基因组工程
- 药物研发
- 农业应用
- 其他的
根据应用,CRISPR 基因检测和诊断市场细分为生物医学诊断、基因组工程、药物发现、农业应用和其他。
工作流
- 样品制备
- 前置放大
- 肌动蛋白RNA
- Cas酶
- 传感
根据工作流程,CRISPR 基因检测和诊断市场细分为样品制备、预扩增、CrRNA、Cas 酶和传感。
最终用户
- 医院
- 诊断中心
- 生物科技公司
- 学术及研究机构
- 其他的
根据最终用户,CRISPR 基因检测和诊断市场分为医院、诊断中心、生物技术公司、学术和研究机构等。
分销渠道
- 直接招标
- 零售销售
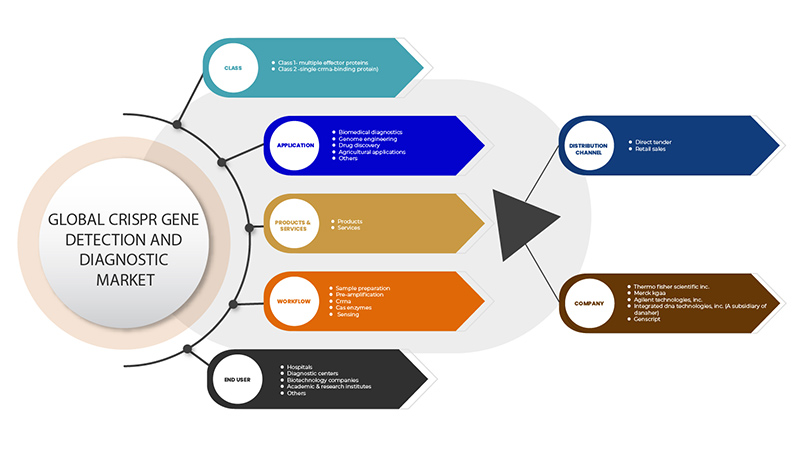
根据分销渠道,CRISPR 基因检测和诊断市场分为直接招标和零售。
CRISPR 基因检测和诊断市场区域分析/见解
对北美 CRISPR 基因检测和诊断市场进行了分析,并按上述地区、类别、产品和服务、应用、工作流程、最终用户和分销渠道提供了市场规模洞察和趋势。
CRISPR 基因检测和诊断市场报告涵盖的国家包括美国、加拿大和墨西哥。
由于医疗支出的增加,美国在 CRISPR 基因检测和诊断市场占据主导地位。
报告的国家部分还提供了影响单个市场因素和国内市场法规变化,这些变化影响了市场的当前和未来趋势。新销售、替代销售、国家人口统计、疾病流行病学和进出口关税等数据点是用于预测单个国家市场情景的一些主要指标。此外,在提供国家数据的预测分析时,还考虑了北美品牌的存在和可用性以及由于来自本地和国内品牌的激烈或稀少的竞争而面临的挑战、销售渠道的影响。
竞争格局和CRISPR 基因检测与诊断市场份额分析
北美 CRISPR 基因检测和诊断市场竞争格局按竞争对手提供详细信息。详细信息包括公司概况、公司财务状况、产生的收入、市场潜力、研发投资、新市场计划、北美业务、生产基地和设施、生产能力、公司优势和劣势、产品发布、产品宽度和广度、应用主导地位。以上提供的数据点仅与公司对 CRISPR 基因检测和诊断市场的关注有关。
CRISPR 基因检测和诊断市场的一些主要参与者包括 GenScript、OriGene Technologies, Inc.、Applied StemCell、GeneCopoeia, Inc.、Agilent Technologies, Inc.、Synthego、BioVision Inc.、Hera Biolabs、Cellecta, Inc.、New England Biolabs、10x Genomics、addgene CasTag Biosciences、Merck KGaA、Integrated DNA Technologies, Inc.(丹纳赫的子公司)、Thermo Fisher Scientific Inc. 等。
SKU-
目录
1 引言
1.1 研究目標
1.2 市場定義
1.3 北美CRISPR基因檢測與診斷市場概況
1.4 貨幣和定價
1.5 限制
1.6 覆蓋市場
2 北美 CRISPR 基因檢測與診斷市場:細分
2.1 覆蓋市場
2.2 地理範圍
研究需時 2.3 年
2.4 DBMR三腳架資料驗證模型
2.5 與關鍵意見領袖的初步訪談
2.6 多變量建模
2.7 艙段生命線曲線
2.8 DBMR 市場地位網格
2.9 供應商份額分析
2.1 市場最終用戶覆蓋網格
2.11 第二手資料
3 執行摘要
4 項優質見解
4.1 佩爾斯特
4.2 波特五力模型
5 智慧財產權格局(專利格局)
6 流行病學
7 北美 CRISPR 基因檢測與診斷市場:監管情景
8 北美 CRISPR 基因檢測和診斷市場管道分析(CRISPR DIAGNOSTICS)
9 市場概覽
9.1 驅動程式
9.1.1 慢性病盛行率和發生率上升
9.1.2 研發投入增加
9.1.3 CRISPR基因診斷的資金可用性
9.1.4 CRISPR基因診斷GMP認證核准數量增加
9.1.5 基於 CRISPR 的診斷臨床試驗的興起
9.2 限制
9.2.1 基於 CRISPR 的診斷成本上升
9.2.2 使用 CRISPR 診斷面臨的風險
9.2.3 與 CRISPR 基因檢測和診斷研究相關的倫理問題
9.2.4 替代方案的可用性
9.3 機遇
9.3.1 市場參與者的策略主動性
9.3.2 醫療支出成長
9.3.3 基於 CRISPR 的診斷技術進步的出現
9.4 挑戰
9.4.1 缺乏 CRISPR 診斷所需的熟練專業人員
9.4.2 嚴格的監管
10 北美 CRISPR 基因檢測和診斷市場(按類別)
10.1 概述
10.2 2類單一crRNA結合蛋白
10.2.1 生物醫學診斷
10.2.2 農業應用
10.2.3 基因組工程
10.2.4 藥物研發
10.2.5 其他
10.3 1類多效應蛋白
11 北美 CRISPR 基因檢測和診斷市場(按產品和服務)
11.1 概述
11.2 產品
11.2.1 檢測試劑盒
11.2.1.1 SGRNA 試劑盒
11.2.1.2 基因組檢測試劑盒
11.2.1.3 其他
11.2.2 蛋白質
11.2.2.1 CAS9
11.2.2.2 CPF1
11.2.2.3 其他
11.2.3 質粒和載體
11.2.4 庫
11.2.5 控制套件
11.2.6 配送系統產品
11.2.7 設計工具
11.2.8 基因組 RNA
11.2.9 HDR 阻斷器
11.2.9.1 疊氮胸苷
11.2.9.2 三氟胸苷
11.2.9.3 其他
11.2.9.4 其他
11.3 服務
11.3.1 G-RNA設計
11.3.2 細胞系工程
11.3.3 微生物基因編輯
11.3.4 DNA合成
11.3.5 其他
12 北美 CRISPR 基因檢測與診斷市場(按應用)
12.1 概述
12.2 生物醫學診斷
12.2.1 癌症
12.2.2 血液疾病
12.2.3 遺傳性疾病
12.2.4 肌肉萎縮症
12.2.5 愛滋病
12.2.6 神經退化性疾病
12.2.7 其他
12.3 農業應用
12.4 基因組工程
12.4.1 細胞系工程
12.4.2 人類幹細胞
12.5 藥物研發
12.6 其他
13 北美 CRISPR 基因檢測和診斷市場(按工作流程)
13.1 概述
13.2 CRRNA
13.3 CAS酶
13.4 預放大
13.4.1 聚合酶連鎖反應
13.4.2 燈
13.4.3 機器人流程自動化
13.5 樣品製備
13.6 感測
13.6.1 螢光探針
13.6.2 比色法
14 北美 CRISPR 基因檢測和診斷市場(按最終用戶)
14.1 概述
14.2 生技公司
14.3 學術與研究機構
14.4 診斷中心
14.5 醫院
14.6 其他
15 北美 CRISPR 基因檢測和診斷市場(按分銷管道)
15.1 概述
15.2 直接投標
15.3 零售額
16 北美 CRISPR 基因檢測與診斷市場(按地區)
16.1 北美
16.1.1 美國
16.1.2 加拿大
16.1.3 墨西哥
17 北美 CRISPR 基因檢測與診斷市場:公司格局
17.1 公司份額分析:北美
18 SWOT分析
19 公司簡介
19.1 賽默飛世爾科技公司
19.1.1 公司概況
19.1.2 收入分析
19.1.3 公司股份分析
19.1.4 產品組合
19.1.5 近期發展
19.2 默克集團
19.2.1 公司概況
19.2.2 收入分析
19.2.3 公司股份分析
19.2.4 產品組合
19.2.5 近期發展
19.3 安捷倫科技公司
19.3.1 公司概況
19.3.2 收入分析
19.3.3 公司股份分析
19.3.4 產品組合
19.3.5 最近的發展
19.4 綜合 DNA 技術公司(丹納赫的子公司)
19.4.1 公司概況
19.4.2 收入分析
19.4.3 公司股份分析
19.4.4 產品組合
19.4.5 近期發展
19.5 金斯瑞
19.5.1 公司概況
19.5.2 收入分析
19.5.3 公司股份分析
19.5.4 產品組合
19.5.5 最近的發展
19.6 10X基因組學
19.6.1 公司概況
19.6.2 收入分析
19.6.3 產品組合
19.6.4 近期發展
19.7 應用幹細胞
19.7.1 公司概況
19.7.2 產品組合
19.7.3 最近的發展
19.8 添加基因
19.8.1 公司概況
19.8.2 產品組合
19.8.3 最近的發展
19.9 生物視覺公司
19.9.1 公司概況
19.9.2 產品組合
19.9.3 最近的發展
19.1 Cellecta公司
19.10.1 公司概況
19.10.2 產品組合
19.10.3 近期發展
19.11 CAS 標籤生物科學
19.11.1 公司概況
19.11.2 產品組合
19.11.3 最近的發展
19.12 基因工程有限公司
19.12.1 公司概況
19.12.2 產品組合
19.12.3 最近的發展
19.13 地平線探索有限公司
19.13.1 公司概況
19.13.2 產品組合
19.13.3 近期發展
19.14 HERA 生物實驗室
19.14.1 公司概況
19.14.2 產品組合
19.14.3 最近的發展
19.15 新英格蘭生物實驗室
19.15.1 公司概況
19.15.2 產品組合
19.15.3 近期發展
19.16 ORIGENE 技術公司
19.16.1 公司概況
19.16.2 產品組合
19.16.3 最近的發展
19.17 合成器
19.17.1 公司概況
19.17.2 產品組合
19.17.3 近期發展
19.18 寶生物株式會社
19.18.1 公司概況
19.18.2 收入分析
19.18.3 產品組合
19.18.4 最近的發展
19.19 TOOLGEN公司
19.19.1 公司概況
19.19.2 產品組合
19.19.3 最近的發展
20份問卷
21份相關報告
表格列表
TABLE 1 PIPELINE ANALYSIS FOR NORTH AMERICA CRISPR GENE THERAPEUTICS
TABLE 2 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY CLASS, 2020-2029 (USD MILLION)
TABLE 3 NORTH AMERICA CLASS-2 SINGLE CRRNA-BINDING PROTEIN IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 4 NORTH AMERICA CLASS-2 SINGLE CRRNA-BINDING PROTEIN IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 5 NORTH AMERICA CLASS-1 MULTIPLE EFFECTOR PROTEINS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 6 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCTS AND SERVICES, 2020-2029 (USD MILLION)
TABLE 7 NORTH AMERICA PRODUCTS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 8 NORTH AMERICA PRODUCTS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 9 NORTH AMERICA ASSAY KITS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 10 NORTH AMERICA PROTEINS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 11 NORTH AMERICA HDR BLOCKERS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 12 NORTH AMERICA SERVICES IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 13 NORTH AMERICA SERVICES IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 14 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 15 NORTH AMERICA BIOMEDICAL DIAGNOSTICS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 16 NORTH AMERICA BIOMEDICAL DIAGNOSTICS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 17 NORTH AMERICA AGRICULTURAL APPLICATIONS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 18 NORTH AMERICA GENOME ENGINEERING IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 19 NORTH AMERICA GENOME ENGINEERING IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 20 NORTH AMERICA DRUG DISCOVERYIN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 21 NORTH AMERICA OTHERS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 22 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 23 NORTH AMERICA CRRNA IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 24 NORTH AMERICA CAS ENZYME IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 25 NORTH AMERICA PRE-AMPLIFICATION IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 26 NORTH AMERICA PRE-AMPLIFICATION IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 27 NORTH AMERICA SAMPLE PREPARATION IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 28 NORTH AMERICA SENSING IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 29 NORTH AMERICA SENSING IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 30 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 31 NORTH AMERICA BIOTECHNOLOGY COMPANIES IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 32 NORTH AMERICA ACADEMIC AND RESEARCH INSTITUTES IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 33 NORTH AMERICA DIAGNOSTIC CENTERS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 34 NORTH AMERICA HOSPITALS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 35 NORTH AMERICA OTHERS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 36 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 37 NORTH AMERICA DIRECT TENDER IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 38 NORTH AMERICA RETAIL SALES IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY REGION, 2020-2029 (USD MILLION)
TABLE 39 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY COUNTRY, 2020-2029 (USD MILLION)
TABLE 40 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY CLASS, 2020-2029 (USD MILLION)
TABLE 41 NORTH AMERICA CLASS-2 SINGLE CRRNA-BINDING PROTEIN IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 42 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCTS AND SERVICES, 2020-2029 (USD MILLION)
TABLE 43 NORTH AMERICA PRODUCTS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 44 NORTH AMERICA ASSAY KITS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 45 NORTH AMERICA HDR BLOCKERS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 46 NORTH AMERICA PROTEINS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 47 NORTH AMERICA SERVICES IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 48 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 49 NORTH AMERICA GENOME ENGINEERING IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 50 NORTH AMERICA BIOMEDICAL DIAGNOSTICS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 51 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 52 NORTH AMERICA PRE-AMPLIFICATION IN GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 53 NORTH AMERICA SENSING IN GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 54 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 55 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 56 U.S. CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY CLASS, 2020-2029 (USD MILLION)
TABLE 57 U.S. CLASS-2 SINGLE CRRNA-BINDING PROTEIN IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 58 U.S. CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCTS AND SERVICES, 2020-2029 (USD MILLION)
TABLE 59 U.S. PRODUCTS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 60 U.S. ASSAY KITS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 61 U.S. HDR BLOCKERS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 62 U.S. PROTEINS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 63 U.S. SERVICES IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 64 U.S. CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 65 U.S. GENOME ENGINEERING IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 66 U.S. BIOMEDICAL DIAGNOSTICS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 67 U.S. CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 68 U.S. PRE-AMPLIFICATION IN GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 69 U.S. SENSING IN GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 70 U.S. CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 71 U.S. CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 72 CANADA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY CLASS, 2020-2029 (USD MILLION)
TABLE 73 CANADA CLASS-2 SINGLE CRRNA-BINDING PROTEIN IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 74 CANADA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCTS AND SERVICES, 2020-2029 (USD MILLION)
TABLE 75 CANADA PRODUCTS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 76 CANADA ASSAY KITS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 77 CANADA HDR BLOCKERS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 78 CANADA PROTEINS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 79 CANADA SERVICES IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 80 CANADA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 81 CANADA GENOME ENGINEERING IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 82 CANADA BIOMEDICAL DIAGNOSTICS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 83 CANADA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 84 CANADA PRE-AMPLIFICATION IN GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 85 CANADA SENSING IN GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 86 CANADA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 87 CANADA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
TABLE 88 MEXICO CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY CLASS, 2020-2029 (USD MILLION)
TABLE 89 MEXICO CLASS-2 SINGLE CRRNA-BINDING PROTEIN IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 90 MEXICO CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCTS AND SERVICES, 2020-2029 (USD MILLION)
TABLE 91 MEXICO PRODUCTS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 92 MEXICO ASSAY KITS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 93 MEXICO HDR BLOCKERS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 94 MEXICO PROTEINS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY PRODUCT TYPE, 2020-2029 (USD MILLION)
TABLE 95 MEXICO SERVICES IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY TYPE, 2020-2029 (USD MILLION)
TABLE 96 MEXICO CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 97 MEXICO GENOME ENGINEERING IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 98 MEXICO BIOMEDICAL DIAGNOSTICS IN CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY APPLICATION, 2020-2029 (USD MILLION)
TABLE 99 MEXICO CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 100 MEXICO PRE-AMPLIFICATION IN GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 101 MEXICO SENSING IN GENE DETECTION AND DIAGNOSTIC MARKET, BY WORKFLOW, 2020-2029 (USD MILLION)
TABLE 102 MEXICO CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY END USER, 2020-2029 (USD MILLION)
TABLE 103 MEXICO CRISPR GENE DETECTION AND DIAGNOSTIC MARKET, BY DISTRIBUTION CHANNEL, 2020-2029 (USD MILLION)
图片列表
FIGURE 1 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: SEGMENTATION
FIGURE 2 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: DATA TRIANGULATION
FIGURE 3 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: DROC ANALYSIS
FIGURE 4 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: NORTH AMERICA VS REGIONAL MARKET ANALYSIS
FIGURE 5 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: DBMR POSITION GRID
FIGURE 8 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: VENDOR SHARE ANALYSIS
FIGURE 9 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: END USER COVERAGE GRID
FIGURE 10 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: SEGMENTATION
FIGURE 11 NORTH AMERICA IS ANTICIPATED TO DOMINATE THE NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET AND ASIA-PACIFIC IS ESTIMATED TO BE GROWING WITH THE HIGHEST CAGR IN THE FORECAST PERIOD OF 2022 TO 2029
FIGURE 12 INCREASED INCIDENCE OF CHRONIC DISEASES, RISE IN TECHNOLOGICAL ADVANCEMENTS IN CRISPR DIAGNOSTICS, AND GOVERNMENT FUNDING FOR THE DEVELOPMENT OF CRISPR DETECTION KITS ARE EXPECTED TO DRIVE THE NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET FROM 2022 TO 2029
FIGURE 13 CLASS SEGMENT IS EXPECTED TO HAVE THE LARGEST SHARE OF THE NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET FROM 2022 & 2029
FIGURE 14 NORTH AMERICA CRISPR GENE PATENT SCENARIO, BY APPLICATION
FIGURE 15 CRISPR PATENT LANDSCAPE AND NUMBER OF APPLICATIONS OF NEW PATENT FAMILIES FILED WORLDWIDE, 2001 TO 2019
FIGURE 16 DRIVERS, RESTRAINTS, OPPORTUNITIES, AND CHALLENGES OF THE NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET
FIGURE 17 INCIDENCE OF VARIOUS TYPES OF CANCER IN 2020
FIGURE 18 PREVALENCE OF HUNTINGTON’S DISEASE IN 2019
FIGURE 19 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: BY CLASS, 2021
FIGURE 20 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: BY CLASS, 2022-2029 (USD MILLION)
FIGURE 21 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: BY CLASS, CAGR (2022-2029)
FIGURE 22 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: BY CLASS, LIFELINE CURVE
FIGURE 23 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: BY PRODUCTS AND SERVICES, 2021
FIGURE 24 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: BY PRODUCTS AND SERVICES, 2022-2029 (USD MILLION)
FIGURE 25 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: BY PRODUCTS AND SERVICES, CAGR (2022-2029)
FIGURE 26 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: BY PRODUCTS AND SERVICES, LIFELINE CURVE
FIGURE 27 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: BY APPLICATION, 2021
FIGURE 28 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: BY APPLICATION, 2022-2029 (USD MILLION)
FIGURE 29 NORTH AMERICA CRISPR GENE DETECTION AND DIAGNOSTIC MARKET: BY APPLICATION, CAGR (2022-2029)
圖30 北美CRISPR基因檢測與診斷市場:依應用、生命線曲線
圖 31 北美 CRISPR 基因檢測與診斷市場:按工作流程,2021 年
圖 32 北美 CRISPR 基因檢測與診斷市場:按工作流程,2022-2029 年(百萬美元)
圖 33 北美 CRISPR 基因檢測與診斷市場:依工作流程、複合年增長率(2022-2029)
圖34 北美CRISPR基因檢測與診斷市場:依工作流程、生命線曲線
圖 35 北美 CRISPR 基因檢測和診斷市場:按最終用戶劃分,2021 年
圖 36 北美 CRISPR 基因檢測與診斷市場:按最終用戶劃分,2022-2029 年(百萬美元)
圖 37 北美 CRISPR 基因檢測和診斷市場:按最終用戶劃分,複合年增長率(2022-2029)
圖38 北美CRISPR基因檢測與診斷市場:依最終用戶、生命線曲線
圖 39 北美 CRISPR 基因檢測與診斷市場:按分銷管道,2021 年
圖40 北美CRISPR基因檢測與診斷市場:按分銷管道,2022-2029年(百萬美元)
圖 41 北美 CRISPR 基因檢測與診斷市場:按分銷管道、複合年增長率(2022-2029)
圖42 北美CRISPR基因檢測與診斷市場:依通路、生命線曲線
圖43 北美CRISPR基因檢測與診斷市場:快照(2021年)
圖44 北美CRISPR基因檢測與診斷市場:依國家(2021年)
圖 45 北美 CRISPR 基因檢測和診斷市場:按國家/地區(2022 年和 2029 年)
圖 46 北美 CRISPR 基因檢測與診斷市場:依國家(2021 年和 2029 年)
圖 47 北美 CRISPR 基因檢測與診斷市場:按類別(2022-2029)
圖48 北美CRISPR基因檢測與診斷市場:2021年公司佔有率(%)
研究方法
数据收集和基准年分析是使用具有大样本量的数据收集模块完成的。该阶段包括通过各种来源和策略获取市场信息或相关数据。它包括提前检查和规划从过去获得的所有数据。它同样包括检查不同信息源中出现的信息不一致。使用市场统计和连贯模型分析和估计市场数据。此外,市场份额分析和关键趋势分析是市场报告中的主要成功因素。要了解更多信息,请请求分析师致电或下拉您的询问。
DBMR 研究团队使用的关键研究方法是数据三角测量,其中包括数据挖掘、数据变量对市场影响的分析和主要(行业专家)验证。数据模型包括供应商定位网格、市场时间线分析、市场概览和指南、公司定位网格、专利分析、定价分析、公司市场份额分析、测量标准、全球与区域和供应商份额分析。要了解有关研究方法的更多信息,请向我们的行业专家咨询。
可定制
Data Bridge Market Research 是高级形成性研究领域的领导者。我们为向现有和新客户提供符合其目标的数据和分析而感到自豪。报告可定制,包括目标品牌的价格趋势分析、了解其他国家的市场(索取国家列表)、临床试验结果数据、文献综述、翻新市场和产品基础分析。目标竞争对手的市场分析可以从基于技术的分析到市场组合策略进行分析。我们可以按照您所需的格式和数据样式添加您需要的任意数量的竞争对手数据。我们的分析师团队还可以为您提供原始 Excel 文件数据透视表(事实手册)中的数据,或者可以帮助您根据报告中的数据集创建演示文稿。