Global Lateral Flow Assay Market
Taille du marché en milliards USD
TCAC :
% 
 USD
9.27 Billion
USD
17.53 Billion
2025
2033
USD
9.27 Billion
USD
17.53 Billion
2025
2033
| 2026 –2033 | |
| USD 9.27 Billion | |
| USD 17.53 Billion | |
|
|
|
|
Global Lateral Flow Assay Market Segmentation, By Product Type (Laparoscopes, Energy Devices, Insufflators, Suction/Irrigation Systems, Closure Devices, Hand Instruments, Access Devices, Laparoscopic Scissors, Laparoscopic Hooks and Accessories), Application (General Surgery, Bariatric Surgery, Gynaecological Surgery, Urological Surgery, Colorectal Surgery, Paediatric Surgery and Others), End User (Hospitals, Ambulatory Surgical Centres and Specialty Clinics)- Industry Trends and Forecast to 2033
Lateral Flow Assay Market Size
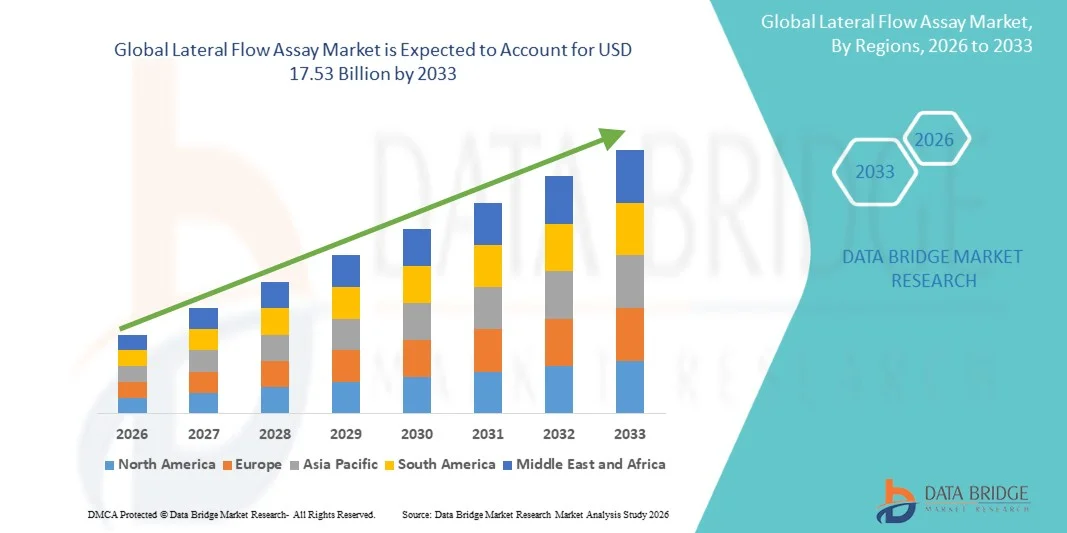
- The global lateral flow assay market size was valued at USD 9.27 billion in 2025 and is expected to reach USD 17.53 billion by 2033, at a CAGR of 8.29% during the forecast period
- The market growth is largely fueled by the increasing demand for rapid point-of-care diagnostics, rising prevalence of infectious diseases, and expanding applications in clinical testing, food safety, and environmental monitoring, leading to broader adoption across healthcare and non-healthcare sectors
- Furthermore, continuous technological advancements in assay sensitivity, multiplex detection capabilities, and user-friendly formats, along with growing emphasis on early disease detection and decentralized testing solutions, are establishing lateral flow assays as a preferred rapid diagnostic platform. These converging factors are accelerating the uptake of lateral flow assay products, thereby significantly boosting the industry's growth
Lateral Flow Assay Market Analysis
- Lateral flow assays, offering rapid, paper-based diagnostic testing for the qualitative or semi-quantitative detection of analytes in biological samples, are increasingly vital tools in modern healthcare, veterinary diagnostics, food safety, and environmental testing due to their speed, cost-effectiveness, portability, and ease of use in decentralized settings
- The escalating demand for lateral flow assays is primarily fueled by the rising prevalence of infectious and chronic diseases, increasing need for point-of-care testing, growing demand for rapid diagnostics in remote and resource-limited areas, and heightened awareness of early disease detection and screening programs
- North America dominated the lateral flow assay market with the largest revenue share of 38.64% in 2025, characterized by advanced healthcare infrastructure, strong presence of leading diagnostic manufacturers, and high adoption of rapid testing solutions, with the U.S. witnessing substantial demand across clinical diagnostics, home testing kits, and infectious disease screening initiatives supported by regulatory approvals and continuous product innovation
- Asia-Pacific is expected to be the fastest growing region in the lateral flow assay market during the forecast period due to expanding healthcare access, rising government initiatives for disease control, increasing investments in diagnostic infrastructure, and growing awareness regarding early and rapid testing solutions
- Energy Devices segment dominated the lateral flow assay market with a market share of 41.8% in 2025, driven by the growing preference for minimally invasive surgical procedures, technological advancements in electrosurgical and ultrasonic systems, and increasing demand for efficient tissue sealing and dissection solutions across various surgical applications
Report Scope and Lateral Flow Assay Market Segmentation
|
Attributes |
Lateral Flow Assay Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
Lateral Flow Assay Market Trends
“Rising Adoption of Multiplex and Digital-Integrated Rapid Testing Solutions”
- A significant and accelerating trend in the global lateral flow assay market is the integration of multiplex detection capabilities and digital connectivity features, enabling simultaneous identification of multiple biomarkers with improved result interpretation and data management across decentralized healthcare settings
- For instance, several diagnostic manufacturers have introduced multiplex rapid test kits capable of detecting multiple respiratory pathogens within a single device, while smartphone-enabled readers allow users to capture, store, and transmit results for remote consultation and epidemiological tracking
- Advanced assay design and digital reader integration enable improved sensitivity, quantitative analysis, automated result interpretation, and reduced human error, while connected platforms support real-time data sharing with healthcare providers and public health systems for faster response and monitoring
- The seamless incorporation of lateral flow assays with digital health platforms and telemedicine services facilitates centralized disease surveillance and patient management, allowing healthcare professionals to monitor testing outcomes alongside other clinical data through unified digital ecosystems
- This trend toward more accurate, connected, and user-friendly rapid diagnostics is reshaping expectations for point-of-care testing solutions. Consequently, companies are developing next-generation lateral flow platforms with enhanced analytical performance, AI-assisted interpretation, and compatibility with mobile health applications
- The demand for digitally integrated and multiplex lateral flow assays is growing rapidly across hospitals, clinics, and home-care environments, as healthcare systems increasingly prioritize rapid decision-making, decentralized diagnostics, and improved patient engagement
- Growing investment in research and development activities aimed at improving assay stability, shelf life, and cost efficiency is strengthening product accessibility in emerging and resource-limited markets
Lateral Flow Assay Market Dynamics
Driver
“Increasing Demand for Rapid Point-of-Care and Decentralized Diagnostics”
- The rising global burden of infectious and chronic diseases, coupled with the growing emphasis on early diagnosis and decentralized healthcare delivery, is a significant driver for the heightened demand for lateral flow assays
- For instance, multiple healthcare systems have expanded rapid testing programs in community clinics and home-care settings to improve early detection and reduce hospital burden, accelerating procurement of point-of-care lateral flow diagnostic kits
- As healthcare providers seek faster clinical decision-making tools, lateral flow assays offer advantages such as minimal sample preparation, short turnaround times, portability, and ease of use without the need for complex laboratory infrastructure
- Furthermore, increasing government initiatives supporting screening programs, outbreak preparedness, and rural healthcare access are encouraging the integration of rapid diagnostic kits into primary care and mobile health units
- The convenience of on-site testing, reduced dependency on centralized laboratories, and the ability to deliver immediate results for timely treatment decisions are key factors propelling adoption across hospitals, diagnostic centers, and home settings. The growing trend toward self-testing and consumer-driven healthcare further contributes to market expansion
- Expanding reimbursement coverage for point-of-care diagnostics in several developed markets is further supporting adoption among healthcare providers and patients
- Rising awareness among consumers regarding preventive healthcare and routine screening is increasing the utilization of rapid testing kits for early disease detection
Restraint/Challenge
“Limited Sensitivity in Certain Applications and Regulatory Stringency”
- Concerns regarding comparatively lower sensitivity and specificity in some lateral flow assays, particularly when compared with laboratory-based molecular diagnostics, pose a significant challenge to broader clinical acceptance in critical care scenarios
- For instance, in cases requiring ultra-high analytical accuracy, confirmatory testing through polymerase chain reaction (PCR) or other advanced laboratory techniques is often recommended, limiting standalone reliance on certain rapid test formats
- Addressing performance variability through improved antibody design, enhanced signal amplification technologies, and rigorous quality control measures is crucial for strengthening clinician confidence and expanding application scope
- In addition, stringent regulatory approval processes and evolving compliance requirements across different regions can delay product launches and increase development costs for manufacturers
- While technological advancements are steadily improving assay performance and reliability, concerns over false positives or false negatives in specific use cases may restrain adoption in highly regulated healthcare environments
- Overcoming these challenges through continuous innovation, robust clinical validation, harmonized regulatory pathways, and user education on appropriate test utilization will be vital for sustained market growth
- Intense market competition and price pressure, particularly from low-cost manufacturers, can impact profit margins and limit investment capacity for smaller players
- Supply chain disruptions affecting raw materials such as antibodies and membranes may create production delays and affect product availability in certain regions
Lateral Flow Assay Market Scope
The market is segmented on the basis of product type, application, and end user.
- By Product Type
On the basis of product type, the market is segmented into laparoscopes, energy devices, insufflators, suction/irrigation systems, closure devices, hand instruments, access devices, laparoscopic scissors, laparoscopic hooks and accessories. The energy devices segment dominated the market with the largest market revenue share of 41.8% in 2025, driven by their critical role in tissue cutting, coagulation, and sealing during minimally invasive surgical procedures. These devices enhance surgical precision, reduce blood loss, and shorten operative time, making them indispensable across multiple laparoscopic interventions. The growing preference for advanced bipolar and ultrasonic energy systems further strengthens their adoption. Hospitals increasingly invest in technologically advanced energy platforms to improve surgical outcomes and patient safety. In addition, rising volumes of minimally invasive procedures worldwide continue to reinforce the dominance of this segment.
The laparoscopes segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by continuous technological advancements in high-definition imaging, 3D visualization, and improved optical systems. Modern laparoscopes provide enhanced clarity and depth perception, enabling surgeons to perform complex procedures with greater accuracy. Increasing demand for minimally invasive diagnostic and therapeutic procedures is accelerating procurement of advanced laparoscopic systems. The expansion of ambulatory surgical centres and specialty clinics is also contributing to higher adoption rates. Furthermore, ongoing innovations focused on compact, lightweight, and ergonomically designed laparoscopes are expected to drive strong future growth in this segment.
- By application
On the basis of application, the market is segmented into general surgery, bariatric surgery, gynaecological surgery, urological surgery, colorectal surgery, paediatric surgery and others. The general surgery segment dominated the market with the largest revenue share in 2025, primarily due to the high volume of laparoscopic procedures performed for conditions such as appendectomies, cholecystectomies, and hernia repairs. General surgery benefits significantly from minimally invasive approaches that reduce hospital stays and postoperative complications. The widespread availability of trained surgeons and standardized laparoscopic protocols further supports segment dominance. Healthcare institutions continue to prioritize laparoscopic general surgeries to enhance patient recovery outcomes. The consistent rise in lifestyle-related disorders requiring surgical intervention also contributes to sustained demand within this segment.
The bariatric surgery segment is expected to witness the fastest growth during the forecast period, driven by the increasing global prevalence of obesity and related metabolic disorders. Minimally invasive laparoscopic techniques are widely preferred in bariatric procedures due to reduced complication rates and faster recovery times. Growing awareness regarding weight management and supportive reimbursement frameworks in developed regions are encouraging more patients to opt for surgical solutions. Advancements in laparoscopic instruments tailored specifically for bariatric interventions further strengthen segment growth. In addition, rising investments in specialized obesity treatment centres are anticipated to accelerate adoption across emerging markets.
- By End User
On the basis of end user, the market is segmented into hospitals, ambulatory surgical centres and specialty clinics. The hospitals segment dominated the market with the largest revenue share in 2025, attributed to the availability of advanced surgical infrastructure and skilled healthcare professionals. Hospitals handle a higher volume of complex and emergency laparoscopic procedures, necessitating comprehensive instrument inventories. Strong capital investment capacity enables hospitals to procure technologically advanced laparoscopic equipment and energy systems. Furthermore, integrated perioperative care and postoperative monitoring facilities make hospitals the preferred choice for major minimally invasive surgeries. The presence of multidisciplinary surgical teams also supports sustained dominance of this segment.
The ambulatory surgical centres segment is projected to register the fastest growth rate from 2026 to 2033, supported by the increasing shift toward outpatient and same-day surgical procedures. These centres offer cost-effective treatment options with reduced hospital stay durations, attracting both patients and payers. Technological advancements enabling safe minimally invasive surgeries in outpatient settings are further driving adoption. The growing emphasis on healthcare cost optimization and operational efficiency contributes significantly to segment expansion. Moreover, rising patient preference for convenient and faster recovery environments is expected to propel demand within ambulatory surgical centres over the forecast period.
Lateral Flow Assay Market Regional Analysis
- North America dominated the lateral flow assay market with the largest revenue share of 38.64% in 2025, characterized by advanced healthcare infrastructure, strong presence of leading diagnostic manufacturers, and high adoption of rapid testing solutions
- Healthcare providers in the region highly value the speed, ease of use, and reliability offered by lateral flow assays, particularly for infectious disease testing, chronic condition monitoring, and home-based diagnostic applications
- This widespread adoption is further supported by favorable reimbursement policies, significant investments in research and development, a strong presence of leading diagnostic manufacturers, and the growing preference for decentralized testing solutions, establishing lateral flow assays as a preferred rapid diagnostic platform across clinical and non-clinical settings
U.S. Lateral Flow Assay Market Insight
The U.S. lateral flow assay market captured the largest revenue share of 82% in 2025 within North America, fueled by strong demand for rapid point-of-care diagnostics and widespread adoption of home-based testing solutions. Healthcare providers and consumers are increasingly prioritizing early disease detection through convenient, quick-result diagnostic kits. The growing preference for decentralized healthcare delivery, combined with robust reimbursement frameworks and government-backed screening programs, further propels the lateral flow assay industry. Moreover, the increasing integration of digital readers and mobile health applications for result tracking and telehealth consultations is significantly contributing to the market's expansion.
Europe Lateral Flow Assay Market Insight
The Europe lateral flow assay market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by stringent regulatory standards for diagnostic testing and the escalating need for rapid infectious disease screening across healthcare settings. The increase in aging population, coupled with expanding preventive healthcare initiatives, is fostering the adoption of rapid diagnostic kits. European healthcare systems are also drawn to the cost-effectiveness and operational efficiency these assays offer. The region is experiencing significant growth across hospitals, diagnostic laboratories, and home-care environments, with lateral flow assays being incorporated into both routine screening programs and emergency preparedness frameworks.
U.K. Lateral Flow Assay Market Insight
The U.K. lateral flow assay market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by the strong emphasis on public health surveillance and rapid community-based testing initiatives. In addition, concerns regarding infectious disease outbreaks and the need for timely diagnostics are encouraging healthcare providers and individuals to adopt rapid testing solutions. The UK’s established healthcare infrastructure, alongside its robust research ecosystem, is expected to continue to stimulate market growth. Furthermore, the increasing utilization of self-testing kits and pharmacy-based rapid diagnostics is reinforcing adoption across the country.
Germany Lateral Flow Assay Market Insight
The Germany lateral flow assay market is expected to expand at a considerable CAGR during the forecast period, fueled by increasing awareness of preventive healthcare and the demand for reliable, high-quality diagnostic technologies. Germany’s advanced healthcare infrastructure, combined with its strong focus on medical innovation and quality standards, promotes the adoption of rapid diagnostic assays in both clinical and non-clinical settings. The integration of lateral flow assays with digital health platforms is also becoming increasingly prevalent, with a strong preference for accurate, privacy-compliant solutions aligning with local regulatory expectations.
Asia-Pacific Lateral Flow Assay Market Insight
The Asia-Pacific lateral flow assay market is poised to grow at the fastest CAGR of 23% during the forecast period of 2026 to 2033, driven by increasing healthcare expenditure, rising prevalence of infectious diseases, and expanding access to diagnostic services in countries such as China, Japan, and India. The region's growing focus on strengthening primary healthcare infrastructure, supported by government-led disease control initiatives, is driving the adoption of rapid diagnostic solutions. Furthermore, as APAC emerges as a major manufacturing hub for diagnostic kits and components, the affordability and accessibility of lateral flow assays are expanding to a broader population base.
Japan Lateral Flow Assay Market Insight
The Japan lateral flow assay market is gaining momentum due to the country’s advanced healthcare system, aging population, and strong emphasis on early disease detection. The Japanese market places significant importance on rapid and accurate diagnostics, and the adoption of lateral flow assays is driven by the increasing need for efficient screening in hospitals and community clinics. The integration of rapid testing kits with digital monitoring systems is fueling growth. Moreover, Japan's demographic trends are likely to spur demand for convenient, easy-to-use diagnostic solutions in both home-care and institutional settings.
India Lateral Flow Assay Market Insight
The India lateral flow assay market accounted for the largest market revenue share in Asia Pacific in 2025, attributed to the country's expanding healthcare infrastructure, rising public health awareness, and growing demand for affordable diagnostic solutions. India stands as one of the fastest-growing markets for rapid testing kits, and lateral flow assays are becoming increasingly popular in hospitals, diagnostic centers, and rural health programs. The push towards strengthening primary healthcare services and large-scale screening initiatives, alongside strong domestic manufacturing capabilities, are key factors propelling the market in India.
Lateral Flow Assay Market Share
The Lateral Flow Assay industry is primarily led by well-established companies, including:
- Abbott (U.S.)
- Thermo Fisher Scientific Inc. (U.S.)
- F. Hoffmann-La Roche Ltd (Switzerland)
- Danaher (U.S.)
- BIOMÉRIEUX (France)
- Siemens Healthineers AG (Germany)
- BD (U.S.)
- Hologic Inc. (U.S.)
- PerkinElmer, Inc. (U.S.)
- QIAGEN (Netherlands)
- Bio-Rad Laboratories, Inc. (U.S.)
- QuidelOrtho Corporation (U.S.)
- Abingdon Health plc (U.K.)
- DCN Dx (U.S.)
- Bioassay Works LLC (U.S.)
- Lumos Diagnostics Holdings Limited (U.S.)
- NG BIOTECH (France)
- Akonni Biosystems, Inc. (U.S.)
- Chembio Diagnostics, Inc. (U.S.)
- Trinity Biotech plc (Ireland)
What are the Recent Developments in Global Lateral Flow Assay Market?
- In November 2025, Abingdon Health PLC launched seaweed-based lateral flow housings to reduce plastic waste from diagnostic test cassettes, providing a sustainable alternative to conventional plastic for lateral flow assays and helping customers lower the environmental impact of mass rapid test production
- In October 2025, Abingdon Health USA Inc reported progress on five new contract development projects underway at its Madison facilities, including development of duplex lateral flow tests and veterinary lateral flow products, highlighting expanded R&D and manufacturing capabilities in the U.S. lateral flow assay sector
- In March 2024, Gold Standard Diagnostics launched the SENSIStrip Gluten PowerLine Lateral Flow Device, a highly sensitive allergen detection test that minimizes false negatives due to a hook line design, enabling rapid gluten residue detection in food matrices and rinse samples within minutes
- In November 2023, Abbott Laboratories launched a new line of rapid lateral flow assays designed for quick and accurate infectious disease diagnosis, expanding the company’s diagnostic portfolio and strengthening point-of-care testing capabilities globally
- In May 2023, Gold Standard Diagnostics added 23 new allergen lateral flow devices to its product portfolio, expanding the range of rapid food allergen tests for beta-lactoglobulin, fish, lupin, ovalbumin, Brazil nut, and more
SKU-
Accédez en ligne au rapport sur le premier cloud mondial de veille économique
- Tableau de bord d'analyse de données interactif
- Tableau de bord d'analyse d'entreprise pour les opportunités à fort potentiel de croissance
- Accès d'analyste de recherche pour la personnalisation et les requêtes
- Analyse de la concurrence avec tableau de bord interactif
- Dernières actualités, mises à jour et analyse des tendances
- Exploitez la puissance de l'analyse comparative pour un suivi complet de la concurrence
Méthodologie de recherche
La collecte de données et l'analyse de l'année de base sont effectuées à l'aide de modules de collecte de données avec des échantillons de grande taille. L'étape consiste à obtenir des informations sur le marché ou des données connexes via diverses sources et stratégies. Elle comprend l'examen et la planification à l'avance de toutes les données acquises dans le passé. Elle englobe également l'examen des incohérences d'informations observées dans différentes sources d'informations. Les données de marché sont analysées et estimées à l'aide de modèles statistiques et cohérents de marché. De plus, l'analyse des parts de marché et l'analyse des tendances clés sont les principaux facteurs de succès du rapport de marché. Pour en savoir plus, veuillez demander un appel d'analyste ou déposer votre demande.
La méthodologie de recherche clé utilisée par l'équipe de recherche DBMR est la triangulation des données qui implique l'exploration de données, l'analyse de l'impact des variables de données sur le marché et la validation primaire (expert du secteur). Les modèles de données incluent la grille de positionnement des fournisseurs, l'analyse de la chronologie du marché, l'aperçu et le guide du marché, la grille de positionnement des entreprises, l'analyse des brevets, l'analyse des prix, l'analyse des parts de marché des entreprises, les normes de mesure, l'analyse globale par rapport à l'analyse régionale et des parts des fournisseurs. Pour en savoir plus sur la méthodologie de recherche, envoyez une demande pour parler à nos experts du secteur.
Personnalisation disponible
Data Bridge Market Research est un leader de la recherche formative avancée. Nous sommes fiers de fournir à nos clients existants et nouveaux des données et des analyses qui correspondent à leurs objectifs. Le rapport peut être personnalisé pour inclure une analyse des tendances des prix des marques cibles, une compréhension du marché pour d'autres pays (demandez la liste des pays), des données sur les résultats des essais cliniques, une revue de la littérature, une analyse du marché des produits remis à neuf et de la base de produits. L'analyse du marché des concurrents cibles peut être analysée à partir d'une analyse basée sur la technologie jusqu'à des stratégies de portefeuille de marché. Nous pouvons ajouter autant de concurrents que vous le souhaitez, dans le format et le style de données que vous recherchez. Notre équipe d'analystes peut également vous fournir des données sous forme de fichiers Excel bruts, de tableaux croisés dynamiques (Fact book) ou peut vous aider à créer des présentations à partir des ensembles de données disponibles dans le rapport.