Global Cytokine Storm Targeted Immunotherapy Market
Tamanho do mercado em biliões de dólares
CAGR :
% 
 USD
1.12 Billion
USD
4.62 Billion
2025
2033
USD
1.12 Billion
USD
4.62 Billion
2025
2033
| 2026 –2033 | |
| USD 1.12 Billion | |
| USD 4.62 Billion | |
|
|
|
|
Segmentação do Mercado de Imunoterapia Meta da Citocina Global por Segmentação do Mercado da Tempestade Global de Citocina (anticorpos monoclonais, corticosteróides, inibidores da JAK, inibidores da Interleucina, inibidores do TNF, agentes imunossupressores, medicamentos anti- inflamatórios de pequenas moléculas, terapêuticas de purificação sanguínea e terapêuticas à base de células), Indicação de Doença (infecções virais graves, sepse e choque séptico, complicações relacionadas com a COVID-19, doenças auto- imunes e inflamatórias, síndrome de libertação de citoquinas induzida pela terapêutica CAR-T, síndrome de dificuldade respiratória aguda, rejeição de transplante de órgãos e perturbações hematológicas), Utilizador Final (Hospitales, clínicas especializadas, centros de tratamento do cancro, institutos de investigação e ambulatórios)- Tendências e Previsão da Indústria para 2033
Imunoterapia com destino à tempestade de citocinaTamanho do Mercado
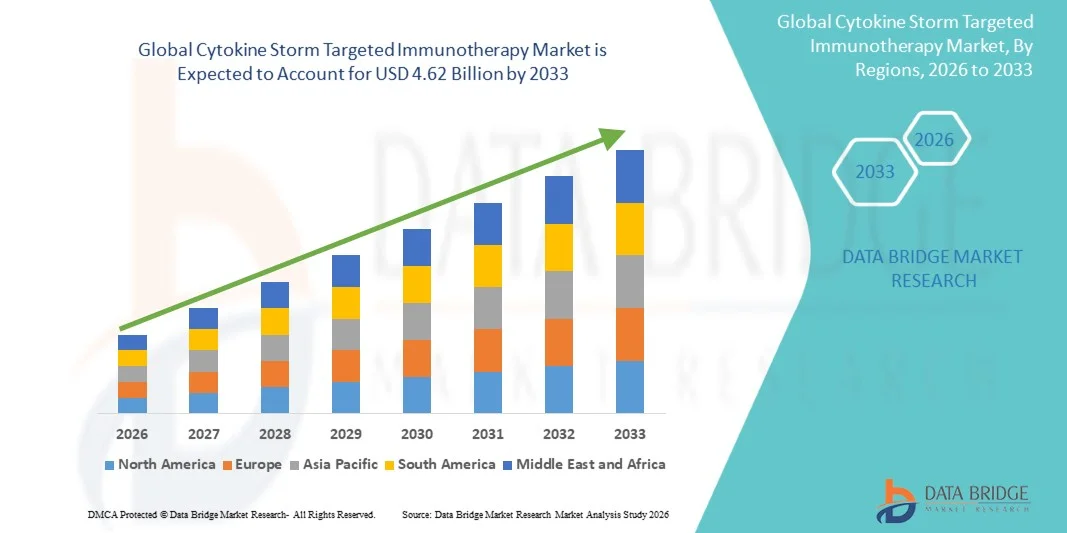
- O mercado global de imunoterapia alvo da tempestade de citocinas foi avaliado emUSD 1,12 mil milhões em 2025e espera-se alcançar4.62 mil milhões de USD até 2033, emCAGR de 19,40%durante o período de previsão
- O crescimento do mercado é amplamente alimentado pela crescente incidência de respostas imunológicas inflamatórias graves, como sepse, complicações do COVID-19 e síndrome de liberação de citocinas induzidas pela terapia com CAR-T, levando ao aumento da demanda por tratamentos imunomodulatórios direcionados em ambientes de cuidados críticos
- Além disso, avanços crescentes em biologics, inibidores da interleucina, e terapias alvo da via JAK-STAT, juntamente com pesquisas clínicas em desregulação imunológica e medicina de precisão, estão estabelecendo o manejo da tempestade de citocinas como uma área de foco chave na imunoterapia
Imunoterapia com destino à tempestade de citocinaAnálise de mercado
- As imunoterapias alvo da tempestade de citocina, projetadas para modular as respostas imunes hiperativas através da inibição de citocinas, regulação da via JAK-STAT e outros mecanismos imunomodulatórios, são cada vez mais críticas no manejo de condições inflamatórias potencialmente fatais, comosépsis,infecções virais graves, síndrome de libertação de citocinas induzida pela terapêutica com CAR-T, esíndrome do desconforto respiratório agudoem ambientes hospitalares e de cuidados intensivos
- A crescente demanda por imunoterapia alvo de tempestades de citocinas é alimentada principalmente pelo aumento da carga global de doenças infecciosas, adoção crescente de métodos biológicos avançados e abordagens de imunoterapia de precisão, e aumento da necessidade clínica de regulação imunológica rápida em pacientes de cuidados críticos com inflamação sistêmica grave.
- A América do Norte dominou o mercado de imunoterapia alvo da tempestade de citocinas com a maior parcela de receita de 42,5% em 2025, impulsionada por forte infraestrutura de saúde, adoção precoce de biológicos avançados, altas taxas de internação em UTI e presença de empresas líderes em biotecnologia e farmacêuticas em desenvolvimento ativoinibidores da interleucinae inibidores de JAK para doenças inflamatórias e imunomediadas nos EUA.
- Asia-Pacific é esperado para ser a região de crescimento mais rápido no mercado de imunoterapia alvo tempestade de citocinas durante o período de previsão devido ao aumento dos gastos com saúde, aumentando a incidência dedoenças infecciosas, ampliação da infraestrutura de cuidados críticos e adoção crescente de terapias biológicas avançadas em economias emergentes
- O segmento de inibidores da interleucina dominou o mercado de imunoterapia alvo de tempestades de citocinas com uma parcela significativa de mercado de 38,6% em 2025, devido à sua forte eficácia clínica no bloqueio de vias inflamatórias chave, como IL-6 e IL-1, que desempenham um papel central na progressão da tempestade de citocinas e danos aos órgãos associados.
Âmbito do relatório eSegmentação do Mercado de Imunoterapia com Tempestade de Citocina
|
Atributos |
Chave de imunoterapia orientada para tempestade de citocinaPerspectivas de mercado |
|
Segmentos Cobertos |
|
|
Países abrangidos |
América do Norte · U.S. · Canadá · México Europa · Alemanha · França · U.K. · Países Baixos · Suíça · Bélgica · Rússia · Itália · Espanha · Turquia · Resto da Europa Ásia- Pacífico · China · Japão · Índia · Coreia do Sul · Singapura · Malásia · Austrália · Tailândia · Indonésia · Filipinas · Resto da Ásia-Pacífico Médio Oriente e África · Arábia Saudita · U.A.E. · África do Sul · Egito · Israel · Resto do Oriente Médio e África América do Sul · Brasil · Argentina · Resto da América do Sul |
|
Jogadores do mercado chave |
|
|
Oportunidades de Mercado |
· Expansão da imunoterapia de precisão e tratamento orientado por biomarcadores · Aumento do uso de protocolos de manejo de tempestades de citocinas em CAR-T e outras imunoterapias de câncer |
|
Informações sobre o Valor Adicionado |
Além dos insights sobre cenários de mercado, como valor de mercado, taxa de crescimento, segmentação, cobertura geográfica e principais atores, os relatórios de mercado curados pela Data Bridge Market Research também incluem análise de especialistas em profundidade, epidemiologia do paciente, análise de pipelines, análise de preços e marco regulatório |
Tendências do mercado de imunoterapia direcionadas para a tempestade da citocina
“Expansão de Biologias de Precisão e Terapias de Bloqueio de Citocinas”
- Uma tendência significativa e acelerada no mercado global de imunoterapia alvo da tempestade de citocinas é a adoção crescente de biológicos de precisão, como inibidores da interleucina e moduladores da via JAK-STAT para o controle rápido das respostas imunes hiperinflamatórias em ambientes críticos de cuidados e oncologia.
- Por exemplo, inibidores da IL-6, como o tocilizumab, são amplamente utilizados em condições inflamatórias graves, incluindo tempestades de citocinas relacionadas com COVID-19 e síndrome de liberação de citocinas induzida pela terapia com CAR-T, demonstrando forte relevância clínica na imunomodulação de emergência
- Avanços na imunologia estão possibilitando o desenvolvimento de terapias alvo de próxima geração que suprimem seletivamente a sinalização excessiva de citocinas, preservando a função imune essencial, reduzindo assim o risco de mortalidade em pacientes com sepse e SDRA.
- A integração dos diagnósticos baseados em biomarcadores com a imunoterapia está facilitando a detecção precoce de tempestades de citocinas e possibilitando estratégias personalizadas de tratamento baseadas em perfis inflamatórios de citocinas e gravidade da doença.
- Essa tendência de modulação imunológica altamente direcionada e baseada em evidências está reformulando protocolos de tratamento em unidades de terapia intensiva, levando a decisões de intervenção mais rápidas e melhora dos resultados de sobrevida do paciente em condições hiperinflamatórias.
- O aumento dos ensaios clínicos e a expansão de dutos para novos agentes alvo de citocinas estão acelerando a inovação e fortalecendo o futuro cenário terapêutico para o manejo de tempestades de citocinas
- A crescente colaboração entre empresas de biotecnologia e instituições de saúde está a aumentar as capacidades de investigação e a acelerar o desenvolvimento de soluções avançadas de imunoterapia
Dinâmica do Mercado de Imunoterapia com Alvo de Tempestade de Citocina
Controlador
“Cidência crescente de graves doenças inflamatórias e infecciosas que conduzem a demanda de tratamento”
- A prevalência crescente de quadros inflamatórios graves, como sepse, síndrome do desconforto respiratório agudo e síndrome de liberação de citocinas relacionada à terapia CAR-T, é um grande fator de aceleração da demanda por imunoterapias alvo de citocinas globalmente
- Por exemplo, em abril de 2025, vários hospitais expandiram o uso de IL-6 e inibidores de JAK em protocolos de terapia intensiva para o manejo de complicações hiperinflamatórias potencialmente fatais em pacientes com terapia viral e de câncer
- O aumento da carga de doenças infecciosas emergentes e surtos virais recorrentes está aumentando as taxas de hospitalização e criando forte necessidade clínica de terapias de supressão imunológica rápida em ambientes de cuidados críticos
- Além disso, a adoção crescente de imunoterapias avançadas de câncer, como a terapia com CAR-T, está contribuindo para maior incidência de síndrome de liberação de citocinas, aumentando ainda mais a demanda por drogas imunomoduladoras direcionadas
- O aumento do investimento em cuidados de saúde na infraestrutura de cuidados intensivos e a expansão de protocolos de tratamento baseados em biologics estão fortalecendo a integração de terapias tempestade de citocinas em práticas de cuidados hospitalares padrão
- Aumentar a conscientização dos profissionais de saúde sobre a intervenção precoce e o manejo de tempestades de citocinas está melhorando os resultados do tratamento e as taxas de adoção da terapia de condução
- Ampliar o apoio regulatório e as autorizações de uso de emergência para imunoterapias críticas durante crises de saúde estão acelerando o acesso ao mercado e a captação
Restrição/Desafio
“Alto custo de tratamento e complexidade de segurança das imunoterapias avançadas”
- Preocupações relacionadas aos altos custos de tratamento e à pouca acessibilidade de terapias biológicas avançadas e de anticorpos monoclonais representam um desafio significativo para a adoção mais ampla de imunoterapias direcionadas para tempestades de citocinas, especialmente no desenvolvimento de sistemas de saúde
- Por exemplo, imunoterapias premium, como inibidores de IL-6 e inibidores de JAK, muitas vezes requerem administração e monitoramento hospitalares especializados, tornando-os menos acessíveis para ambientes de saúde de baixo recurso
- Desafios de segurança incluindo risco de imunossupressão, infecções secundárias e reações imunológicas adversas criam hesitação entre os clínicos ao selecionar terapias agressivas de bloqueio de citocinas para pacientes críticos
- Além disso, vias de aprovação regulatória complexas e rigorosos requisitos de validação clínica para novos medicamentos imunomoduladores atrasam a entrada no mercado e limitam a comercialização rápida de terapias emergentes.
- Consciência limitada e capacidade diagnóstica inconsistente para detecção precoce de tempestades de citocinas em algumas regiões restringem ainda mais o início oportuno do tratamento e reduzem a eficácia global da terapia
- Superar esses desafios por meio de otimização de custos, melhor perfil de segurança e diretrizes clínicas ampliadas será essencial para a penetração sustentada do mercado e adoção terapêutica mais ampla.
- Reembolso limitado para biologics de alto custo em vários sistemas de saúde restringe ainda mais o acesso do paciente e retarda as taxas de adoção
- Restrições da cadeia de suprimentos e complexidades de fabricação associadas à produção biológica podem impactar a disponibilidade consistente de terapias avançadas direcionadas a citocinas
Alcance do Mercado de Imunoterapia com Alvo na Tempestade de Citocina
O mercado é segmentado com base no tipo de terapia, indicação de doença e usuário final.
- Por tipo de terapia
Com base no tipo de terapia, o mercado de imunoterapia alvo da tempestade de citocinas é segmentado em anticorpos monoclonais, corticosteroides, inibidores de JAK, inibidores de interleucina, inibidores de TNF, imunossupressores, fármacos anti-inflamatórios de pequena molécula, terapias de purificação sanguínea e terapias de base celular. O segmento de inibidores da interleucina dominou o mercado com a maior parcela de receita de mercado de 38,6% em 2025, impulsionada pela sua forte eficácia clínica no direcionamento das principais vias inflamatórias, como IL-6 e IL-1, que são centrais na progressão da tempestade de citocinas. Essas terapias são amplamente adotadas em ambientes hospitalares para o manejo de respostas inflamatórias graves em condições como a síndrome de liberação de citocinas induzida pela terapia COVID-19 e pela terapia com CAR-T. Seu rápido início de ação e capacidade de reduzir significativamente o risco de mortalidade em pacientes críticos reforçam ainda mais sua dominância. Além disso, o aumento da aprovação regulatória e a ampla familiaridade do médico com os inibidores da interleucina estão contribuindo para sua demanda sustentada. A disponibilidade de biológicos bem estabelecidos e fortes evidências clínicas que apoiam seu uso reforça ainda mais sua posição de liderança no mercado.
Prevê-se que o segmento de inibidores JAK testemunhe a taxa de crescimento mais rápida durante o período de previsão, alimentado pela sua capacidade de modular múltiplas vias de sinalização de citocinas simultaneamente através do mecanismo JAK-STAT. Essas terapias estão ganhando força devido à sua opção de administração oral e efeitos imunomodulatórios mais amplos em comparação com os biológicos alvo único. O aumento dos ensaios clínicos e a expansão das indicações de inibidores de JAK em condições inflamatórias e autoimunes estão acelerando ainda mais sua adoção. Seu uso crescente no manejo de infecções virais graves e complicações de tempestade de citocinas está impulsionando a demanda em mercados desenvolvidos e emergentes. Além disso, avanços em terapias com pequenas moléculas direcionadas e melhores perfis de segurança estão aumentando a confiança do médico em seu uso. A flexibilidade nos protocolos de tratamento e o potencial para terapias combinadas também contribuem para sua rápida trajetória de crescimento.
- Por indicação de doença
Com base na indicação da doença, o mercado é segmentado em infecções virais graves, sepse e choque séptico, complicações relacionadas ao COVID-19, doenças autoimunes e inflamatórias, síndrome de liberação de citocinas induzida pela terapia CAR-T, síndrome do desconforto respiratório agudo, rejeição de transplante de órgãos e distúrbios hematológicos. O segmento de infecções virais graves dominou o mercado com maior participação de receita em 2025, impulsionado pela alta carga global de doenças infecciosas e pela incidência significativa de tempestades de citocinas em surtos virais. Condições como COVID-19 e influenza têm destacado a necessidade crítica de intervenções imunomodulatórias rápidas, levando ao aumento da adoção de terapias direcionadas. Hospitais e unidades de terapia intensiva dependem fortemente de inibidores de citocinas para gerenciar respostas inflamatórias graves em pacientes infectados. A disponibilidade de protocolos de tratamento estabelecidos e as autorizações de uso de emergência durante crises de saúde corroboraram ainda mais a dominância desse segmento. Além disso, o monitoramento contínuo dos riscos de mutação viral continua a sustentar a demanda por terapias efetivas de tempestade de citocinas.
Espera-se que o segmento da síndrome de liberação de citocinas induzida pela terapia CAR-T testemunhe a taxa de crescimento mais rápida durante o período de previsão, impulsionada pela rápida expansão das imunoterapias avançadas de câncer globalmente. À medida que as terapias de CAR-T se tornam mais utilizadas para neoplasias hematológicas, a incidência da síndrome de liberação de citocinas está aumentando, necessitando de soluções eficazes de manejo. Imunoterapias direcionadas, como inibidores da IL-6, estão se tornando componentes essenciais dos protocolos de tratamento do CAR-T. Os investimentos crescentes na investigação em oncologia e o aumento das aprovações de terapias de base celular estão a alimentar ainda mais o crescimento deste segmento. Além disso, aumentar a conscientização dos oncologistas sobre estratégias de intervenção precoce é melhorar os resultados dos pacientes e impulsionar a adoção da terapia. A integração do manejo da tempestade de citocinas nas vias de tratamento do câncer também está apoiando o crescimento a longo prazo.
- Por Usuário Final
Com base no usuário final, o mercado é segmentado em hospitais, clínicas especializadas, centros de tratamento do câncer, institutos de pesquisa e ambientes de atendimento ambulatorial. O segmento hospitalar dominou o mercado com a maior participação de receita em 2025, impulsionado pelo alto volume de pacientes críticos que necessitavam de terapia intensiva e intervenção imunoterapia imediata. Os hospitais estão equipados com infraestrutura avançada, incluindo UTIs e sistemas especializados de monitoramento, possibilitando a administração efetiva de terapias de tempestades de citocinas. A disponibilidade de profissionais de saúde capacitados e o acesso a uma ampla gama de biológicos contribuem ainda mais para sua dominância. Além disso, os hospitais servem como centros primários para o manejo de infecções graves, sepse e complicações relacionadas ao câncer, garantindo uma demanda consistente de imunoterapias direcionadas. A integração de protocolos de tratamento avançados e capacidades de resposta a emergências reforça a sua posição de liderança no mercado.
O segmento de centros de tratamento do câncer é esperado para testemunhar a taxa de crescimento mais rápida durante o período de previsão, alimentado pela adoção crescente de terapias com células CAR-T e outros tratamentos oncológicos avançados. Esses centros estão cada vez mais incorporando o manejo da tempestade de citocinas como um componente crítico do cuidado ao câncer, particularmente para pacientes submetidos à imunoterapia. O aumento dos investimentos em infra-estruturas de oncologia e em instalações de tratamento especializadas está a apoiar o crescimento deste segmento. Além disso, o crescente foco no tratamento personalizado do câncer e melhor monitoramento do paciente está aumentando a demanda por imunoterapias direcionadas. A expansão de redes dedicadas de atenção ao câncer e o aumento da colaboração com empresas de biotecnologia aceleram ainda mais o crescimento do segmento. À medida que a imunoterapia do câncer continua evoluindo, espera-se que o papel dos centros de tratamento especializados no manejo de tempestades de citocinas se expanda significativamente.
Análise Regional do Mercado de Imunoterapia com Alvo na Tempestade de Citocina
- A América do Norte dominou o mercado de imunoterapia alvo da tempestade de citocinas com a maior parcela de receita de 42,5% em 2025, impulsionada por forte infraestrutura de saúde, adoção precoce de biológicos avançados, altas taxas de internação em UTI e presença de empresas líderes em farmacêuticas e biotecnologias desenvolvendo ativamente inibidores da interleucina e inibidores da JAK para condições inflamatórias e imunomediadas nos EUA.
- Os profissionais de saúde da região priorizam altamente tratamentos de resposta rápida, eficácia clínica e soluções imunomodulatórias avançadas para o manejo de condições como sepse, síndrome de liberação de citocinas induzida pela terapia com CAR-T e infecções virais graves.
- Esta adoção generalizada é ainda apoiada por gastos substanciais em saúde, uma forte presença de empresas farmacêuticas e de biotecnologia líderes, e crescente investimento em pesquisas de cuidados críticos e imunoterapia, estabelecendo terapias de citocinas direcionadas como componentes essenciais dos modernos protocolos de tratamento hospitalar
U.S. Cytokine Tempestade Meta Imunoterapia Mercado Insight
O mercado de imunoterapia alvo da tempestade de citocinas dos EUA capturou a maior parcela de receita de cerca de 79% em 2025 na América do Norte, alimentada pela forte presença de infraestrutura de saúde avançada e pela rápida adoção de imunoterapias biológicas e de precisão. Os profissionais de saúde estão cada vez mais priorizando o manejo efetivo de quadros inflamatórios graves, como sepse, síndrome de liberação de citocinas induzida pela terapia com CAR-T e síndrome do desconforto respiratório agudo. A crescente preferência por inibidores alvo de citocinas, aliada à demanda robusta por tratamentos de resposta rápida e cuidados baseados em UTI, impulsiona ainda mais o mercado. Além disso, o aumento da pesquisa clínica, as aprovações do FDA e a presença de empresas farmacêuticas líderes estão contribuindo significativamente para a expansão do mercado.
Europe Cytokine Storm Targeted Immunotherapy Market Insight
Projeta-se que o mercado europeu de imunoterapia alvo da tempestade de citocinas se expanda em um CAGR substancial ao longo do período de previsão, impulsionado principalmente por fortes quadros regulatórios e pela crescente necessidade de manejo efetivo de condições inflamatórias críticas. O aumento dos gastos em saúde, aliado à demanda por biológicos avançados, está promovendo a adoção de terapias direcionadas a citocinas. Os sistemas de saúde europeus estão também centrados na melhoria dos resultados dos doentes através de abordagens precoces de intervenção e de medicina de precisão. A região vem experimentando crescimento significativo em ambientes hospitalares e de atenção especializada, sendo incorporadas imunoterapias tanto nos protocolos de tratamento padrão quanto nas estratégias de cuidados críticos avançados.
U.K. Cytokine tempestade alvo Immunoterapia Mercado Insight
Prevê-se que o mercado de imunoterapia alvo da tempestade de citocinas do Reino Unido cresça em um notável CAGR durante o período de previsão, impulsionado pelo aumento da carga de doenças infecciosas e inflamatórias e pela demanda por soluções de tratamento avançadas. Além disso, as preocupações com as reações imunológicas graves e os resultados dos pacientes estão incentivando os profissionais de saúde a adotar imunoterapias direcionadas. Prevê-se que a forte infra-estrutura de cuidados de saúde do Reino Unido, juntamente com o seu foco na investigação clínica e na inovação, continue a estimular o crescimento do mercado.
Alemanha Cytokine Storm Targeted Immunotherapy Market Insight
Espera-se que o mercado de imunoterapia alvo da tempestade de citocinas da Alemanha se expanda em um considerável CAGR durante o período de previsão, alimentado pelo aumento da conscientização de tratamentos avançados de imunoterapia e da demanda por soluções de saúde de alta qualidade. A infra-estrutura médica bem desenvolvida da Alemanha, combinada com a sua ênfase na inovação e na investigação, promove a adopção de terapias orientadas para as citocinas, especialmente em ambientes hospitalares. A integração dos biológicos avançados em protocolos de tratamento também vem se tornando cada vez mais prevalente, com forte preferência por terapias efetivas e baseadas em evidências, alinhadas com os padrões locais de saúde.
Visão de Mercado da Imunoterapia com Alvo na Tempestade Citocina Ásia-Pacífico
O mercado de imunoterapia alvo da tempestade de citocinas Ásia-Pacífico está pronto para crescer no CAGR mais rápido de 23% durante o período de previsão de 2026 a 2033, impulsionado pelo aumento dos gastos com saúde, aumento da incidência de doenças infecciosas e expansão da infraestrutura de cuidados críticos em países como China, Japão e Índia. O crescente foco da região na melhoria do acesso à saúde, apoiado por iniciativas governamentais, está impulsionando a adoção de imunoterapias avançadas. Além disso, à medida que a Ásia-Pacífico surge como uma região-chave para a pesquisa clínica e a produção biológica, a acessibilidade e a acessibilidade de terapias específicas estão se expandindo para uma população mais ampla de pacientes.
Japão Cytokine Tempestade Meta Immunoterapia Mercado Insight
O mercado de imunoterapia alvo da tempestade de citocinas do Japão está ganhando impulso devido ao sistema avançado de saúde do país, rápida adoção tecnológica e crescente foco na medicina de precisão. O mercado japonês coloca ênfase significativa no manejo efetivo de condições inflamatórias graves, e a adoção de terapias alvo de citocinas é impulsionada pelo crescente uso de biológicos avançados. A integração das imunoterapias com os protocolos de tratamento hospitalares é combustível para o crescimento. Além disso, é provável que o envelhecimento da população japonesa estimule a demanda por soluções de tratamento eficientes e confiáveis em ambientes de cuidados agudos e crônicos.
Índia Cytokine tempestade alvo imunoterapia mercado Insight
O mercado de imunoterapia alvo da tempestade de citocinas da Índia representou a maior participação de receita de mercado na Ásia Pacífico em 2025, atribuída à expansão da infraestrutura de saúde do país, aumento da carga de doenças infecciosas e adoção crescente de soluções de tratamento avançadas. A Índia é um mercado em rápido crescimento de biologias e imunoterapias, com crescente utilização em hospitais e centros de cuidados críticos. O impulso para melhorar o acesso aos cuidados de saúde e a disponibilidade de opções de tratamento eficazes em termos de custos, juntamente com o aumento das capacidades farmacêuticas nacionais, são factores fundamentais que impulsionam o mercado na Índia.
Quota de Mercado de Imunoterapia Metada por Tempestade de Citocina
A indústria de imunoterapia alvo da tempestade Cytokine é liderada principalmente por empresas bem estabelecidas, incluindo:
- F. Hoffmann-La Roche Ltd (Suíça)
- Genentech, Inc. (EUA)
- Chugai Pharmaceutical Co., Ltd. (Japão)
- Sanofi (França)
- Regeneron Pharmaceuticals, Inc. (EUA)
- Novartis AG (Suíça)
- Pfizer Inc. (EUA)
- Eli Lilly and Company (EUA)
- Bristol Myers Squibb Company (EUA)
- AbbVie Inc. (EUA)
- AstraZeneca PLC (U.K.)
- GSK plc (UK)
- Amgen Inc. (EUA)
- Takeda Pharmaceutical Company Limited (Japão)
- Astellas Pharma Inc. (Japão)
- Merck KGaA (Alemanha)
- Celltrion Inc. (Coreia do Sul)
- Fresenius Kabi AG (Alemanha)
- Biogen Inc. (EUA)
Quais são os recentes desenvolvimentos no mercado global de imunoterapia com destino à tempestade de citocina
- Em agosto de 2025, a Food and Drug Administration (FDA) dos EUA expandiu a aprovação do tocilizumab-anoh (Avtozma), um biossimiliar ao Actemra, para incluir o tratamento da síndrome de liberação de citocinas (SCR) em pacientes adultos e pediátricos submetidos à terapia CAR-T. Este marco regulatório alinha o biossimiliar com o biológico de referência e amplia significativamente o acesso às opções de tratamento alvo de tempestades de citocinas, apoiando a adoção clínica mais ampla em ambientes de imunoterapia
- Em julho de 2025, novas pesquisas clínicas destacaram avanços nas estratégias de manejo multidisciplinar para a síndrome de liberação de citocinas associada à terapia com células CAR-T, incluindo melhor estratificação de risco e protocolos de tratamento profilático. Esses desenvolvimentos estão aumentando os perfis de segurança e otimizando os resultados terapêuticos, reforçando a importância da imunoterapia direcionada no manejo de respostas imunes graves.
- Em janeiro de 2025, Celltrion recebeu aprovação da FDA dos EUA para Avtozma (tocilizumab-anoh), um inibidor biossimiliar de IL-6 usado no tratamento de doenças de tempestade de citocinas, incluindo CRS induzida pela terapia CAR-T e complicações COVID-19. Esta aprovação marcou um passo fundamental para aumentar a concorrência no segmento biológico e melhorar a acessibilidade e a acessibilidade das imunoterapias de citocinas em nível global.
- Em junho de 2024, a Agência Europeia de Medicamentos (EMA) concedeu a aprovação de biossimilares tocilizumab, tais como Tofidence para múltiplas indicações, incluindo condições relacionadas com a tempestade de citocinas, reforçando a disponibilidade de inibidores da interleucina em toda a Europa. Essa expansão suporta uma integração mais ampla das terapias alvo de citocinas em protocolos de tratamento padrão para doenças inflamatórias e imunomediadas.
- Em março de 2022, o FDA dos EUA aprovou Carvykti (ciltacabtagene autoleucel), uma terapia de células CAR-T dirigida pela BCMA para mieloma múltiplo, que está intimamente associada com os requisitos de controle da síndrome de liberação de citocinas. Essa aprovação avançou significativamente a paisagem da imunoterapia, aumentando a necessidade de estratégias efetivas de mitigação de tempestades de citocinas e aumentando a demanda por tratamentos imunomodulatórios direcionados
SKU-
Obtenha acesso online ao relatório sobre a primeira nuvem de inteligência de mercado do mundo
- Painel interativo de análise de dados
- Painel de análise da empresa para oportunidades de elevado potencial de crescimento
- Acesso de analista de pesquisa para personalização e customização. consultas
- Análise da concorrência com painel interativo
- Últimas notícias, atualizações e atualizações Análise de tendências
- Aproveite o poder da análise de benchmark para um rastreio abrangente da concorrência
Metodologia de Investigação
A recolha de dados e a análise do ano base são feitas através de módulos de recolha de dados com amostras grandes. A etapa inclui a obtenção de informações de mercado ou dados relacionados através de diversas fontes e estratégias. Inclui examinar e planear antecipadamente todos os dados adquiridos no passado. Da mesma forma, envolve o exame de inconsistências de informação observadas em diferentes fontes de informação. Os dados de mercado são analisados e estimados utilizando modelos estatísticos e coerentes de mercado. Além disso, a análise da quota de mercado e a análise das principais tendências são os principais fatores de sucesso no relatório de mercado. Para saber mais, solicite uma chamada de analista ou abra a sua consulta.
A principal metodologia de investigação utilizada pela equipa de investigação do DBMR é a triangulação de dados que envolve a mineração de dados, a análise do impacto das variáveis de dados no mercado e a validação primária (especialista do setor). Os modelos de dados incluem grelha de posicionamento de fornecedores, análise da linha de tempo do mercado, visão geral e guia de mercado, grelha de posicionamento da empresa, análise de patentes, análise de preços, análise da quota de mercado da empresa, normas de medição, análise global versus regional e de participação dos fornecedores. Para saber mais sobre a metodologia de investigação, faça uma consulta para falar com os nossos especialistas do setor.
Personalização disponível
A Data Bridge Market Research é líder em investigação formativa avançada. Orgulhamo-nos de servir os nossos clientes novos e existentes com dados e análises que correspondem e atendem aos seus objetivos. O relatório pode ser personalizado para incluir análise de tendências de preços de marcas-alvo, compreensão do mercado para países adicionais (solicite a lista de países), dados de resultados de ensaios clínicos, revisão de literatura, mercado remodelado e análise de base de produtos . A análise de mercado dos concorrentes-alvo pode ser analisada desde análises baseadas em tecnologia até estratégias de carteira de mercado. Podemos adicionar quantos concorrentes necessitar de dados no formato e estilo de dados que procura. A nossa equipa de analistas também pode fornecer dados em tabelas dinâmicas de ficheiros Excel em bruto (livro de factos) ou pode ajudá-lo a criar apresentações a partir dos conjuntos de dados disponíveis no relatório.