Europe Neuropathic Pain Market
Размер рынка в млрд долларов США
CAGR :
% 
 USD
1,123.93 million
USD
2,371.40 million
2022
2030
USD
1,123.93 million
USD
2,371.40 million
2022
2030
| 2023 –2030 | |
| USD 1,123.93 million | |
| USD 2,371.40 million | |
|
|
|
Europe Neuropathic Pain Market, By Type (Spinal Cord Stimulation (SCS) Devices, External Peripheral Nerve Stimulation, Transcutaneous Electrical Nerve Stimulation (Tens) Devices), Product Type (Rechargeable and Non-Rechargeable), Procedure (Invasive and Non-Invasive), Technology (Transcutaneous Electrical Nerve Stimulation, Transcranial Magnetic Stimulation and Respiratory Electrical Stimulation), Modality (Stationary and Portable), Mode of Purchase (Over-the-Counter Devices and Prescription-Based Devices), Pain Type (Peripheral Neuropathy, Entrapment Neuropathy, Phantom Limb Neuropathy, Trigeminal Neuralgia, Post Herpetic Neuralgia (PHN), Post Traumatic Neuropathy, Foot Pain, Coccydynia), Indication (Spinal Stenosis, Chemotherapy-Induced Peripheral Neuropathy, Diabetic Neuropathy, Others), End User (Hospitals, Clinics, Home Healthcare, Ambulatory Surgical Centres and Others), Distribution Channel (Direct Tender, Third Party Distributor and Others) – Industry Trends and Forecast to 2030.
Europe Neuropathic Pain Market Analysis and Size
Increasing R&D expenditure by various market players to create advanced products is expected to drive the growth of the Europe neuropathic pain market. Limitation associated with the medical devices which limits the use of products is expected to act as restraint for the growth of the Europe neuropathic pain market. High risk of neuropathic pain with ageing and rising older population in many countries will create an opportunity for the growth of the Europe neuropathic pain market. Use of traditional therapies in emerging countries due to low health expenditure and disrupted supply chain is expected to act as a challenge for the growth of the Europe neuropathic pain market.
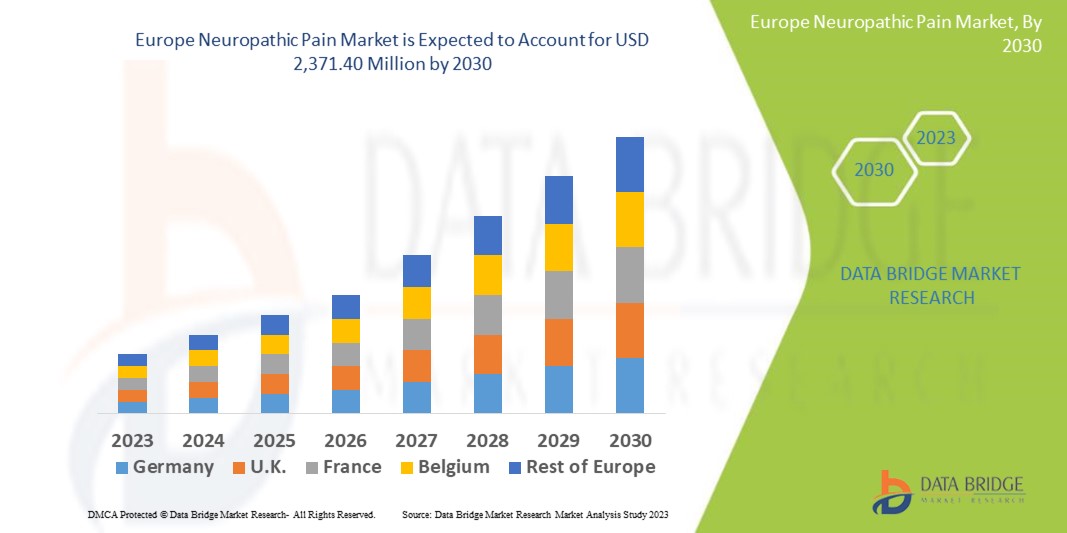
Data Bridge Market Research analyses that the Europe neuropathic pain market, which was USD 1,123.93 million in 2022, is expected to reach USD 2,371.40 million by 2030, and is expected to undergo a CAGR of 9.1% during the forecast period of 2023 to 2030. “Spinal cord stimulation (SCS) devices” segment dominates the type segment of the Europe neuropathic pain market owing to the advancements in technology. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
Europe Neuropathic Pain Market Scope and Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015-2020) |
|
Quantitative Units |
Revenue in USD Million, Volumes in Units, Pricing in USD |
|
Segments Covered |
Type (Spinal Cord Stimulation (SCS) Devices, External Peripheral Nerve Stimulation, Transcutaneous Electrical Nerve Stimulation (Tens) Devices), Product Type (Rechargeable and Non-Rechargeable), Procedure (Invasive and Non-Invasive), Technology (Transcutaneous Electrical Nerve Stimulation, Transcranial Magnetic Stimulation and Respiratory Electrical Stimulation), Modality (Stationary and Portable), Mode of Purchase (Over-the-Counter Devices and Prescription-Based Devices), Pain Type (Peripheral Neuropathy, Entrapment Neuropathy, Phantom Limb Neuropathy, Trigeminal Neuralgia, Post Herpetic Neuralgia (PHN), Post Traumatic Neuropathy, Foot Pain, Coccydynia), Indication (Spinal Stenosis, Chemotherapy-Induced Peripheral Neuropathy, Diabetic Neuropathy, Others), End User (Hospitals, Clinics, Home Healthcare, Ambulatory Surgical Centres and Others), Distribution Channel (Direct Tender, Third Party Distributor and Others) |
|
Countries Covered |
Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe |
|
Market Players Covered |
NEVRO CORP. (U.S.), Boston Scientific Corporation (U.S.), Abbott (U.S.), electroCore, Inc. (U.S.), Polar Medical (England) , B. Braun Melsungen AG (Germany), Omron Healthcare, Inc. (Japan), Cirtec (U.S.), Medtronic (Ireland), Pfizer Inc. (U.S.), Novartis AG (Switzerland), Virpax Pharmaceuticals, Inc. (U.S.), Amgen Inc. (U.S.), Integer Holdings Corporation (U.S.) and AVNS (U.S.) |
|
Market Opportunities |
|
Market Definition
Neuropathic pain is caused by a lesion or disease of the somatosensory system including peripheral fibres (Aβ, Aδ and C fibres) and central neurons and affects 7–10% of the general population. Multiple causes of neuropathic pain have been described and its incidence is likely to increase owing to the aging global population, increased incidence of diabetes mellitus and improved survival from cancer after chemotherapy.
Europe Neuropathic Pain Market Dynamics
Drivers
- Increasing Prevalence of Neuropathic Pain
As the aging population grows and chronic diseases such as diabetes and cancer become more prevalent, the number of people suffering from neuropathic pain is on the rise. This demographic trend drives the demand for neuropathic pain treatments and medications.
- Advances in Medical Research
Ongoing research into the causes and mechanisms of neuropathic pain has led to the development of new treatment options and therapies. As our understanding of neuropathic pain improves, more effective treatments are being developed, creating opportunities for pharmaceutical and biotech companies.
- Novel Drug Development
Pharmaceutical companies are investing in the development of new drugs specifically designed to target neuropathic pain. These drugs aim to provide better pain relief with fewer side effects, improving the quality of life for patients.
Opportunities
- Growing Awareness among Patients
Increased awareness of neuropathic pain and its impact on patients’ lives has led to a greater demand for effective treatments. Patients are becoming more proactive in seeking appropriate care and therapies.
- Rising of Healthcare Expenditure
As healthcare expenditure continues to increase globally, there is more funding available for research, development, and access to neuropathic pain treatments and therapies.
Restraints/Challenges
- Limited Efficacy of Current Treatments
Many existing medications and therapies for neuropathic pain offer only partial relief and can have side effects. This limited efficacy can lead to patient dissatisfaction and the search for alternative treatments.
-
Side Effects and Safety Concerns
Some neuropathic pain medications, including opioids and certain antidepressants, can cause side effects such as drowsiness, dizziness, and addiction. Managing these side effects can be challenging for both patients and healthcare providers.
This Europe neuropathic pain market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the Europe neuropathic pain market Contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
Recent Developments
- In January 2023, Abbott announced that the U.S. Food and Drug Administration (FDA) has approved its Proclaim XR spinal cord stimulation (SCS) system to treat painful diabetic peripheral neuropathy (DPN), a debilitating complication of diabetes
- In July 2022, Vertex Pharmaceuticals Incorporated announced positive Phase 2 results of VX-548 in Acute and Neuropathic Pain by U.S. Food and Drug Administration (FDA)
Europe Neuropathic Pain Market Scope
The Europe neuropathic pain Market is segmented on the basis of type, product type, procedure, technology, modality, mode of purchase, pain type, indication, end user and distribution channel. The growth amongst these segments will help you analyse meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Type
- Spinal Cord Stimulation (SCS) Devices
- External Peripheral Nerve Stimulation
- Transcutaneous Electrical Nerve Stimulation (Tens) Devices
Product Type
- Rechargeable
- Non-Rechargeable
Procedure
- Invasive
- Non-Invasive
Technology
- Transcutaneous Electrical Nerve Stimulation
- Transcranial Magnetic Stimulation
- Respiratory Electrical Stimulation
Modality
- Stationary
- Portable
Mode of Purchase
- Over-the-Counter Devices
- Prescription-Based Devices
Pain Type
- Peripheral Neuropathy
- Entrapment Neuropathy
- Phantom Limb Neuropathy
- Trigeminal Neuralgia
- Post Herpetic Neuralgia (PHN)
- Post Traumatic Neuropathy
- Foot Pain
- Coccydynia
Indication
- Spinal Stenosis
- Chemotherapy-Induced Peripheral Neuropathy
- Diabetic Neuropathy
- Others
End User
- Hospitals
- Clinics
- Home Healthcare
- Ambulatory Surgical Centers
- Others
Distribution Channel
- Direct Tender
- Third Party Distributor
- Others
Europe Neuropathic Pain Market Regional Analysis/Insights
The Europe neuropathic pain market is analysed and market size insights and trends are provided by country, type, product type, procedure, technology, modality, mode of purchase, pain type, indication, end user, and distribution channel as referenced above.
The countries covered in the Europe neuropathic pain market report are Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, and Rest of Europe
Germany dominates the Europe neuropathic pain market because of the strong base of healthcare facilities, strong presence of major players in the market, and rising number of research activities in this region.
Germany is expected to witness significant growth in the Europe neuropathic pain market during the forecast period of 2023 to 2030 due to the increase in government initiatives to promote awareness, rise in medical tourism, growing research activities in the region, availability of massive untapped markets, large population pool, and the growing demand for quality healthcare in the region.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Data points like down-stream and upstream value chain analysis, technical trends and porter's five forces analysis, case studies are some of the pointers used to forecast the market scenario for individual countries. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Healthcare Infrastructure Growth Installed Base and New Technology Penetration
The Europe neuropathic pain market also provides you with detailed market analysis for every country growth in healthcare expenditure for capital equipment, installed base of different kind of products for Europe neuropathic pain market, impact of technology using life line curves and changes in healthcare regulatory scenarios and their impact on the Europe neuropathic pain market. The data is available for historic period 2015-2020.
Competitive Landscape and Europe Neuropathic Pain Market Share Analysis
The Europe neuropathic pain market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to Europe neuropathic pain market.
Some of the major players operating in the Europe neuropathic pain market are:
- NEVRO CORP. (U.S.)
- Boston Scientific Corporation (U.S.)
- Abbott (U.S.)
- electroCore, Inc. (U.S.)
- Polar Medical (England)
- B. Braun Melsungen AG (Germany)
- Omron Healthcare, Inc. (Japan)
- Cirtec (U.S.)
- Medtronic (Ireland)
- Pfizer Inc. (U.S.)
- Novartis AG (Switzerland)
- Virpax Pharmaceuticals, Inc. (U.S.)
- Amgen Inc.(U.S.)
- Integer Holdings Corporation (U.S.)
- AVNS (U.S.)
SKU-
Получите онлайн-доступ к отчету на первой в мире облачной платформе рыночной аналитики
- Интерактивная панель анализа данных
- Панель анализа компании для возможностей с высоким потенциалом роста
- Доступ аналитика-исследователя для настройки и запросов
- Анализ конкурентов с помощью интерактивной панели
- Последние новости, обновления и анализ тенденций
- Используйте возможности сравнительного анализа для комплексного отслеживания конкурентов
Методология исследования
Сбор данных и анализ базового года выполняются с использованием модулей сбора данных с большими размерами выборки. Этап включает получение рыночной информации или связанных данных из различных источников и стратегий. Он включает изучение и планирование всех данных, полученных из прошлого заранее. Он также охватывает изучение несоответствий информации, наблюдаемых в различных источниках информации. Рыночные данные анализируются и оцениваются с использованием статистических и последовательных моделей рынка. Кроме того, анализ доли рынка и анализ ключевых тенденций являются основными факторами успеха в отчете о рынке. Чтобы узнать больше, пожалуйста, запросите звонок аналитика или оставьте свой запрос.
Ключевой методологией исследования, используемой исследовательской группой DBMR, является триангуляция данных, которая включает в себя интеллектуальный анализ данных, анализ влияния переменных данных на рынок и первичную (отраслевую экспертную) проверку. Модели данных включают сетку позиционирования поставщиков, анализ временной линии рынка, обзор рынка и руководство, сетку позиционирования компании, патентный анализ, анализ цен, анализ доли рынка компании, стандарты измерения, глобальный и региональный анализ и анализ доли поставщика. Чтобы узнать больше о методологии исследования, отправьте запрос, чтобы поговорить с нашими отраслевыми экспертами.
Доступна настройка
Data Bridge Market Research является лидером в области передовых формативных исследований. Мы гордимся тем, что предоставляем нашим существующим и новым клиентам данные и анализ, которые соответствуют и подходят их целям. Отчет можно настроить, включив в него анализ ценовых тенденций целевых брендов, понимание рынка для дополнительных стран (запросите список стран), данные о результатах клинических испытаний, обзор литературы, обновленный анализ рынка и продуктовой базы. Анализ рынка целевых конкурентов можно проанализировать от анализа на основе технологий до стратегий портфеля рынка. Мы можем добавить столько конкурентов, о которых вам нужны данные в нужном вам формате и стиле данных. Наша команда аналитиков также может предоставить вам данные в сырых файлах Excel, сводных таблицах (книга фактов) или помочь вам в создании презентаций из наборов данных, доступных в отчете.