Global Generic Drug Market
市场规模(十亿美元)
CAGR :
% 
 USD
622.02 Million
USD
1,323.68 Million
2022
2030
USD
622.02 Million
USD
1,323.68 Million
2022
2030
| 2023 –2030 | |
| USD 622.02 Million | |
| USD 1,323.68 Million | |
|
|
|
|
全球仿製藥市場,按類型(簡單仿製藥、超級仿製藥)、品牌(純仿製藥、品牌仿製藥)、適應症(中樞神經系統 (CNS)、心血管、皮膚病學、腫瘤學、呼吸系統等)、給藥途徑(口服、外用、腸外給藥、其他)、最終用戶(醫院、家庭護理、專科診所、其他零售通路(20 年藥店和零售行業。
仿製藥市場分析及規模
世界衛生組織估計,每年有200萬至300萬例非黑色素瘤皮膚癌和13.2萬例黑色素瘤皮膚癌。此外,牛皮癬在全球的盛行率從0.09%到11.43%不等,這是一種嚴重疾病,影響全球至少1億人。事實上,外用藥物是大多數皮膚病的主要治療方法,預計未來幾年先進外用產品市場將會成長。
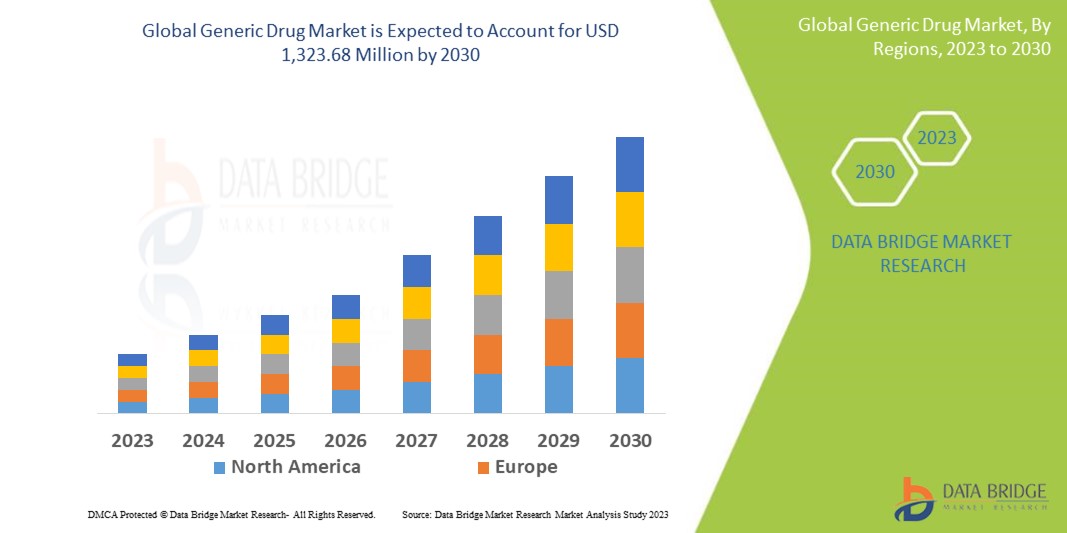
Data Bridge Market Research 分析稱,仿製藥市場規模在 2022 年為 6.2202 億美元,預計到 2030 年將達到 13.2368 億美元,在 2023 年至 2030 年的預測期內複合年增長率為 9.9%。除了對市場價值、成長率、細分、地理覆蓋範圍和主要參與者等市場情景的洞察外,Data Bridge Market Research 策劃的市場報告還包括深度專家分析、患者流行病學、管道分析、定價分析和監管框架。
仿製藥市場範圍和細分
|
報告指標 |
細節 |
|
預測期 |
2023年至2030年 |
|
基準年 |
2022 |
|
歷史歲月 |
2021(可自訂為 2015 - 2020 年) |
|
定量單位 |
收入(百萬美元)、銷售(單位)、定價(美元) |
|
涵蓋的領域 |
類型(簡單仿製藥、超級仿製藥)、品牌(純仿製藥、品牌仿製藥)、適應症(中樞神經系統(CNS)、心血管、皮膚病學、腫瘤學、呼吸系統等)、給藥途徑(口服、外用、腸外給藥、其他)、最終用戶(醫院、家庭護理、專科診所、其他)、經銷商(醫院藥房) |
|
覆蓋國家 |
北美洲的美國、加拿大和墨西哥、德國、法國、英國、荷蘭、瑞士、比利時、俄羅斯、義大利、西班牙、土耳其、歐洲其他地區、中國、日本、印度、韓國、新加坡、馬來西亞、澳洲、泰國、印尼、菲律賓、亞太地區 (APAC) 的其他地區、沙烏地阿拉伯、阿聯酋、南非、埃及、以色列、中東和非洲 (MEA) 的其他地區、其他地區的歐洲地區 |
|
涵蓋的市場參與者 |
Teva Pharmaceuticals Industries Ltd.(以色列)、Mylan NV(美國)、Novartis AG(瑞士)、Pfizer Inc.(美國)、Sun Pharmaceutical Industries Ltd.(印度)、Fresenius SE & Co. KGaA.(德國)、Lupin(印度)、Endo International plc。 PLC.(英國)、STADA Arzneimittel AG(德國)、Eli Lilly and Company(美國)和 Aspen Holdings(南非) |
|
市場機會 |
|
市場定義
仿製藥與品牌藥在多個方面存在差異,例如藥物研發過程中採用的製造流程、輔料和包裝。儘管如此,它們在強度、劑量、品質、安全性、性能和療效方面與品牌藥生物等效。當已上市藥品的專利到期後,仿製藥即可上市。仿製藥不屬於特定製造商,通常受世界各國政府的監管。
仿製藥市場動態
驅動程式
- 人工智慧 (AI) 技術需求的成長將推動行動市場的成長
機器人流程自動化 (RPA)使用人工智慧(AI) 技術來自動化常規的、基於規則的活動。市場的主要營運參與者可以將更多的時間、精力和資源投入到這種自動化的更高價值任務中。仿製藥市場未來幾年將加速發展的關鍵發展之一是使用 RPA 來確保符合法規和標準。製藥公司經常使用 RPA 和其他業務流程自動化工具來執行大量的研發和生產任務。這些都是推動市場成長的特定因素。
- 印度蓬勃發展的製藥業推動印度仿製藥市場
印度是全球最大的醫藥市場之一,擁有多家頂級製藥公司。根據印度政府投資促進與便利化機構Invest India的數據,印度製藥業在產量方面排名全球第三,在價值方面排名全球第十四。印度不僅擁有3,000多家製藥公司和10,500多家生產設施,還擁有美國境外數量最多的符合美國食品藥物管理局(FDA)標準的製藥廠。
機會
- 癌症病例的增加將成為一個機會
根據國際癌症研究機構 (IARC) 2020 年的報告,該報告估計了全球 185 個國家/地區的 36 種癌症的發病率和死亡率,估計 2020 年全球診斷出 1930 萬例新發癌症病例,其中男性超過 1010 萬例,女性超過 930 萬例。此外,全球 RA 網路 2021 年的研究估計,全球有超過 3.5 億人患有關節炎。由於全球老年人口的增長等多種變量,這一數字可能會繼續上升。因此,預計慢性病盛行率的上升將增加對有效治療的需求,從而在整個預測期內推動仿製藥市場的成長。
限制/挑戰
- 嚴格的監管將阻礙成長
FDA 會評估仿製藥的準確性、副作用和其他成分,而嚴格的管控是限制仿製藥擴張的主要因素之一。如果生產商不遵守監管要求,藥品通常會被召回。影響仿製藥品質的關鍵因素包括純度、效力、穩定性和藥物釋放。這些因素應控制在適當的限度、範圍或分佈範圍內,以達到所需的藥品品質。由於政府的嚴格規定,仿製藥需要獲得批准,預計將阻礙市場擴張。
本仿製藥市場報告詳細介紹了近期發展動態、貿易法規、進出口分析、生產分析、價值鏈優化、市場份額、國內和本地市場參與者的影響,並分析了新興收入來源、市場法規變化、戰略市場增長分析、市場規模、品類市場增長、應用領域和市場主導地位、產品審批、產品上市、地域擴張以及市場技術創新等方面的機遇。如需了解更多關於仿製藥市場的信息,請聯繫 Data Bridge 市場研究部門獲取分析師簡報,我們的團隊將協助您做出明智的市場決策,實現市場成長。
COVID-19對仿製藥市場的影響
新冠疫情對仿製藥市場產生了重大影響。新冠疫情爆發後,幾乎所有企業最初都因各種社交隔離規定而遭遇供應鏈中斷。製藥業也未能倖免,這對仿製藥市場造成了不利影響。之後,由於新冠疫情為治療該疾病的藥物研發提供了更多機會,市場對仿製藥的需求增加。美國食品藥物管理局 (FDA) 設立了仿製藥計畫 (Generic Drug Program),透過發送書面訊息、參與開發早期會議以及在整個申請審查過程中明確監管要求等方式,幫助仿製藥生產商開發新產品。
最新動態
- 2020年,專注於開發、生產和銷售高品質品牌藥和仿製藥的美國綜合專科製藥公司ANI Pharmaceuticals, Inc.以5,250萬美元現金收購了Amerigen Pharmaceuticals, Ltd.的商業及在研仿製藥業務。此次交易大大豐富了ANI Pharmaceuticals, Inc.的商業產品組合和後期仿製藥產品線。
全球仿製藥市場範圍
仿製藥市場根據類型、品牌、適應症、給藥途徑、最終用戶和分銷管道進行細分。這些細分市場的成長將幫助您分析行業中成長緩慢的細分市場,並為用戶提供有價值的市場概覽和市場洞察,幫助他們做出策略決策,確定核心市場應用。
類型
- 簡單泛型
- 超級仿製藥
品牌
- 純通用
- 品牌學名藥
適應症
- 中樞神經系統(CNS)
- 心血管
- 皮膚科
- 腫瘤學
- 呼吸系統
- 其他的
給藥途徑
- 口服
- 外用
- 腸外
- 其他的
最終用戶
- 醫院
- 居家護理
- 專科診所
- 其他的
分銷管道
- 醫院藥房
- 網路藥局
- 零售藥局
仿製藥市場區域分析/洞察
對仿製藥市場進行了分析,並根據上述國家、類型、品牌、適應症、給藥途徑、最終用戶和分銷管道提供了市場規模洞察和趨勢。
仿製藥市場報告涵蓋的國家包括北美洲的美國、加拿大和墨西哥、歐洲的德國、法國、英國、荷蘭、瑞士、比利時、俄羅斯、義大利、西班牙、土耳其、歐洲其他地區、中國、日本、印度、韓國、新加坡、馬來西亞、澳洲、泰國、印尼、菲律賓、亞太地區(APAC)的其他地區、沙烏地阿拉伯、阿聯酋、南非、埃及、泰國、中東和其他地區(阿根廷地區的其他地區)。
北美在仿製藥市場佔據主導地位,因為該地區已建立仿製藥審批流程框架,並在研發活動方面處於全球領先地位。
預計亞太地區將在2023年至2030年的預測期內實現最高成長率,這得益於醫療基礎設施的不斷擴張和政府舉措的不斷加強。此外,民眾對醫療狀況的認知度不斷提高,以及該地區人口老化加劇。在亞太地區,中國和印度等國的貢獻超過其他國家。
報告的國家部分還提供了各個市場的影響因素以及國內市場監管變化,這些變化會影響市場的當前和未來趨勢。下游和上游價值鏈分析、技術趨勢、波特五力模型分析以及案例研究等數據點是預測各國市場狀況的一些指標。此外,在對國家/地區數據進行預測分析時,還考慮了全球品牌的存在和可用性,以及它們因本土和國內品牌的激烈競爭或稀缺而面臨的挑戰,國內關稅和貿易路線的影響。
競爭格局和仿製藥市場份額分析
仿製藥市場競爭格局按競爭對手提供詳細資訊。詳細資訊包括公司概況、公司財務狀況、收入、市場潛力、研發投入、新市場計劃、全球佈局、生產基地和設施、生產能力、公司優勢和劣勢、產品發布、產品寬度和廣度以及應用主導地位。以上提供的數據僅與公司在仿製藥市場的重點相關。
仿製藥市場的一些主要參與者包括:
- Teva Pharmaceuticals Industries Ltd.(以色列)
- Mylan NV,(美國)
- 諾華公司(瑞士)
- 輝瑞公司(美國)
- 太陽製藥工業有限公司(印度)
- Fresenius SE & Co. KGaA.,(德國)
- 魯冰花(印度)
- Endo International plc.,(愛爾蘭)
- 奧羅賓多製藥(印度)
- 諾華公司(瑞士)
- Hikma Pharmaceuticals PLC.,(英國)
- STADA Arzneimittel AG(德國)
- 禮來公司(美國)
- 阿斯彭控股(南非)
SKU-
目录
1 引言
1.1 研究目標
1.2 市場定義
1.3 全球仿製藥市場概況
1.4 貨幣和定價
1.5 限制
1.6 覆蓋市場
2 市場區隔
2.1 關鍵要點
2.2 了解全球仿製藥市場規模
2.2.1 供應商定位網格
2.2.2 技術生命線曲線
2.2.3 三腳架資料驗證模型
2.2.4 市場引導
2.2.5 多變量建模
2.2.6 自上而下的分析
2.2.7 挑戰矩陣
2.2.8 應用覆蓋網格
2.2.9 測量標準
2.2.10 供應商份額分析
2.2.11 流行病學
2.2.12 來自關鍵主要訪談的資料點
2.2.13 來自關鍵二級資料庫的資料點
2.3 全球仿製藥市場:研究概況
2.4 假設
3 市場概覽
3.1 驅動程式
3.2 限制
3.3 機遇
3.4 挑戰
4 執行摘要
5 項優質見解
6 流行病學
6.1 各性別發病率
6.2 處理率
6.3 死亡率
6.4 藥物依從性與治療轉換模型
6.5 患者治療成功率
7 產業洞察
7.1 專利分析
7.2 成熟市場的藥物治療率
7.3 人口趨勢:對所有發病率的影響
7.4 患者流程圖
7.5 關鍵定價策略
7.6 關鍵患者入組策略
7.7 與配方化學家的訪談
7.8 其他KOL快照
8 管道分析
8.1 臨床試驗與階段分析
8.2 藥物治療管線
8.3 第三階段候選人
8.4 第二階段候選人
8.5 第一階段候選人
8.6 其他(臨床前和研究)
9 監理框架
10 全球仿製藥市場(按類型)
10.1 概述
10.2 簡單泛型
10.3 超級泛型
11 全球仿製藥市場(按品牌)
11.1 概述
11.2 純仿製藥
11.3 品牌仿製藥
12 全球仿製藥市場(依適應症劃分)
(註:將為所有細分市場和子細分市場提供市場價值、數量和平均售價分析)
12.1 概述
12.2 抗癌藥物
12.2.1 抗腫瘤抗生素
12.2.1.1. 柔紅黴素
12.2.1.1.1. 市場價值(百萬美元)
12.2.1.1.2. 市場交易量(單位)
12.2.1.1.3. 平均售價(ASP)
12.2.1.2. 阿黴素
12.2.1.3. 伊達比星
12.2.1.4. 米托蒽醌
12.2.1.5. 其他
12.2.2 DNA損傷劑
12.2.2.1. 苯丁酸氮芥
12.2.2.2. 環磷醯胺
12.2.2.3. 美法崙
12.2.2.4. 卡鉑
12.2.2.5. 其他
12.2.3 抗代謝藥
12.2.3.1. 甲胺蝶呤
12.2.3.2. 氟達拉濱
12.2.3.3. 阿糖胞苷
12.2.3.4. 其他
12.2.4 DNA修復酶抑制劑
12.2.4.1. 依托泊苷
12.2.4.2. 拓樸替康
12.2.4.3. 其他
12.2.5 抗有絲分裂藥物
12.2.5.1. 長春新鹼
12.2.5.2. 長春花鹼
12.2.5.3. 其他
12.2.6 其他
12.3 中樞神經系統產品
12.3.1 膽鹼酯酶抑制劑
12.3.1.1. 利伐斯的明
12.3.1.2. 多奈哌齊
12.3.1.3. 加蘭他敏
12.3.1.4. 他克林
12.3.1.5. 其他
12.3.2 NMDA 受體拮抗劑
12.3.2.1. 氯胺酮
12.3.2.2. 右美沙芬
12.3.2.3. 美金剛
12.3.2.4. 金剛烷胺
12.3.3 抗癲癇藥
12.3.3.1. 第一代
12.3.3.1.1. 苯巴比妥
12.3.3.1.2. 苯妥英
12.3.3.1.3. 撲米酮
12.3.3.1.4. 乙琥胺
12.3.3.1.5. 卡馬西平
12.3.3.1.6. 氯巴佔
12.3.3.2. 第二代
12.3.3.2.1. 氨己烯酸
12.3.3.2.2. 拉莫三嗪
12.3.3.2.3. 加巴噴丁
12.3.3.2.4. 托吡酯
12.3.3.2.5. 普瑞巴林
12.3.3.2.6. 其他
12.3.3.3. 第三代
12.3.3.3.1. 拉科醯胺
12.3.3.3.2. 盧非醯胺
12.3.3.3.3. 佩拉姆帕內爾
12.3.3.3.4. 其他
12.3.4 抗精神病藥
12.3.4.1. 典型的抗精神病藥
12.3.4.1.1. 氟哌利多
12.3.4.1.2. 洛沙平
12.3.4.1.3. 硫利達嗪
12.3.4.1.4. 莫林酮
12.3.4.1.5. 噻噸
12.3.4.1.6. 氟奮乃靜
12.3.4.1.7. 甲索利達嗪
12.3.4.1.8. 三氟拉嗪
12.3.4.1.9. 奮乃靜
12.3.4.1.10. 氯丙嗪
12.3.4.2. 非典型抗精神病藥
12.3.4.2.1. 阿立哌唑
12.3.4.2.2. 氯氮平
12.3.4.2.3. 齊拉西酮
12.3.4.2.4. 利培酮
12.3.4.2.5. 喹硫平
12.3.4.2.6. 奧氮平
12.3.5 其他
12.4 皮膚科產品
12.4.1 皮質類固醇,依類型
12.4.1.1. 主題
12.4.1.1.1. 莫米松
12.4.1.1.2. 倍他米松
12.4.1.1.3. 氫化可的松
12.4.1.1.4. 丙酸氟替卡松
12.4.1.1.5. 阿氯米松
12.4.1.1.6. 曲安西龍
12.4.1.1.7. 氟輕鬆
12.4.1.1.8. 丙酸氯倍他索
12.4.1.1.9. 其他
12.4.1.2. 口服
12.4.1.2.1. 潑尼松龍
12.4.1.2.2. 潑尼松
12.4.1.2.3. 可的松
12.4.1.2.4. 甲基潑尼松龍
12.4.1.2.5. 其他
12.4.2 類視黃醇
12.4.2.1. 阿維A
12.4.2.2. 阿達帕林
12.4.2.3. 異維他命A酸
12.4.2.4. 其他
12.4.3 抗組織胺藥
12.4.3.1. 賽庚啶
12.4.3.2. 苯海拉明
12.4.3.3. 羥嗪
12.4.3.4. 其他
12.4.4 鈣調磷酸酶抑制劑
12.4.4.1. 他克莫司
12.4.4.2. 吡美莫司
12.4.5 抗感染藥物
12.4.5.1. 抗生素
12.4.5.1.1. 強力黴素
12.4.5.1.2. 雷他帕莫林
12.4.5.1.3. 德拉沙星
12.4.5.1.4. 米諾環素
12.4.5.1.5. 莫匹羅星
12.4.5.1.6. 其他
12.4.5.2. 抗真菌
12.4.5.2.1. 合成
12.4.5.2.1.1 克黴唑
12.4.5.2.1.2 咪康唑
12.4.5.2.1.3 其他
12.4.5.2.2. 咪唑類藥物
12.4.5.2.2.1 酮康唑
12.4.5.2.2.2 氯硝西泮
12.4.5.2.3. 其他
12.4.5.2.4. 其他
12.4.5.3. 抗病毒
12.4.5.3.1. 阿昔洛韋
12.4.5.3.2. 泛昔洛韋
12.4.5.3.3. 伐昔洛韋
12.4.5.3.4. 其他
12.4.5.4. 其他
12.4.6 毛髮生長
12.4.6.1. 米諾地爾
12.4.6.2. 非那雄胺
12.4.6.3. 螺內酯
12.4.6.4. 度他雄胺
12.4.6.5. 其他
12.4.7 其他
12.5 胃腸道產品
12.5.1 瀉藥
12.5.1.1. 滲透性瀉藥
12.5.1.1.1. 戈利特利
12.5.1.1.2. 科萊特
12.5.1.1.3. 聚乙二醇 400
12.5.1.2. 刺激性瀉藥
12.5.1.2.1. 比沙可啶
12.5.1.2.2. 蓖麻油
12.5.1.2.3. 酚酞
12.5.1.2.4. 番瀉葉
12.5.1.3. 容積性瀉藥
12.5.1.3.1. 車前子
12.5.1.3.2. 甲基纖維素
12.5.1.3.3. 聚卡波非
12.5.1.3.4. 其他
12.5.1.4. 潤滑劑和潤膚劑瀉藥
12.5.1.4.1. 礦物油
12.5.1.4.2. 甘油栓
12.5.1.4.3. 其他
12.5.2 抗酸劑
12.5.2.1. 鈉抗酸藥
12.5.2.2. 鈣抗酸藥
12.5.2.3. 鎂抗酸藥
12.5.2.4. 鋁抗酸劑
12.5.2.5. 其他
12.5.3 止瀉藥
12.5.3.1. 地芬諾酯
12.5.3.2. 洛哌丁胺
12.5.3.3. 可待因
12.5.3.4. 其他
12.5.4 H2阻斷劑
12.5.4.1. 法莫替丁
12.5.4.2. 雷尼替丁
12.5.4.3. 其他
12.5.5 質子幫浦抑制劑
12.5.5.1. 奧美拉唑
12.5.5.2. 蘭索拉唑
12.5.5.3. 其他
12.5.6 膽汁酸螯合劑
12.5.6.1. 考來烯胺
12.5.6.2. 考來烯胺
12.5.7 其他
12.6 呼吸產品
12.6.1 支氣管擴張劑
12.6.1.1. 沙丁胺醇
12.6.1.2. 左旋沙丁胺醇
12.6.1.3. 沙美特羅
12.6.1.4. 福莫特羅
12.6.1.5. 其他
12.6.2 皮質類固醇
12.6.2.1. 外消旋腎上腺素
12.6.2.2. 氟替卡松
12.6.2.3. 布地奈德
12.6.2.4. 其他
12.6.3 肥大細胞穩定劑
12.6.3.1. 糠酸莫米松
12.6.3.2. 奈多羅米
12.6.3.3. 其他
12.6.4 白三烯受體拮抗劑
12.6.4.1. 色甘酸鈉
12.6.4.2. 奧馬珠單抗
12.6.4.3. 其他
12.6.5 抗組織胺藥
12.6.5.1. 札魯司特
12.6.5.2. 孟魯司特
12.6.5.3. 齊留通
12.6.5.4. 其他
12.6.6 呼吸興奮劑
12.6.6.1. 氯雷替定
12.6.6.2. 非索非那定
12.6.6.3. 西替利嗪
12.6.6.4. 腎上腺素
12.6.6.5. 其他
12.6.7 肺表面活性劑
12.6.7.1. 多沙普崙
12.6.7.2. 茶鹼
12.6.7.3. 孕酮
12.6.7.4. 咖啡因
12.6.7.5. 其他
12.6.8 氧抗菌劑
12.6.8.1. 棕櫚酸膽磷脂
12.6.8.2. 貝拉克坦
12.6.8.3. 鈣化劑
12.6.8.4. 活性劑阿爾法
12.6.8.5. 其他
12.6.9 其他
12.7 眼科產品
12.7.1 人工淚液
12.7.1.1. 潤膚劑
12.7.1.1.1. 聚乙二醇(PEG)
12.7.1.1.2. 丙二醇
12.7.1.1.3. 甘油
12.7.1.1.4. 聚乙烯醇(PVA)
12.7.1.1.5. 其他
12.7.1.2. 潤膚劑
12.7.1.2.1. 石蠟
12.7.1.2.2. 無水羊毛脂
12.7.1.2.3. 白蠟
12.7.1.2.4. 其他
12.7.1.3. 其他
12.7.2 非類固醇抗發炎藥
12.7.2.1. 溴芬酸
12.7.2.2. 酮咯酸
12.7.2.3. 雙氯芬酸
12.7.2.4. 其他
12.7.3 皮質類固醇
12.7.3.1. 潑尼松龍
12.7.3.2. 氯替潑諾
12.7.3.3. 氟米龍
12.7.3.4. 其他
12.7.4 β受體阻斷劑
12.7.4.1. 左布諾洛爾
12.7.4.2. 噻嗎洛爾
12.7.4.3. 倍他洛爾
12.7.4.4. 其他
12.7.5 前列腺素類似物
12.7.5.1. 比馬前列素
12.7.5.2. 曲伏前列素
12.7.5.3. 拉坦前列素
12.7.5.4. 其他
12.7.6 碳酸酐酶抑制劑
12.7.6.1. 布林佐胺
12.7.6.2. 多佐胺
12.7.7 組合藥物
12.7.7.1. 溴莫尼定/噻嗎洛爾
12.7.7.2. 多佐胺/噻嗎洛爾
12.7.7.3. 苯腎上腺素
12.7.7.4. 丙美卡因
12.7.7.5. 托吡卡胺
12.7.7.6. 其他
12.8 心血管藥物
12.8.1 血管張力素轉換酶抑制劑
12.8.1.1. 貝那普利
12.8.1.2. 卡托普利
12.8.1.3. 馬來酸依那普利
12.8.1.4. 賴諾普利
12.8.1.5. 其他
12.8.2 血管張力素 II 受體拮抗劑(ARBS)
12.8.2.1. 坎地沙坦酯
12.8.2.2. 甲磺酸依普羅沙坦
12.8.2.3. 厄貝沙坦
12.8.2.4. 氯沙坦
12.8.2.5. 其他
12.8.3 抗心律不整藥
12.8.3.1. 胺碘酮
12.8.3.2. 磷酸二異丙吡胺
12.8.3.3. 多非利特
12.8.3.4. 氟卡尼
12.8.3.5. 鹽酸美西律
12.8.3.6. 普魯卡因胺
12.8.3.7. 其他
12.8.4 抗凝血劑
12.8.4.1. 非 VKA 口服抗凝血藥物(NOACS)
12.8.4.1.1. 利伐沙班
12.8.4.1.2. 依度沙班
12.8.4.1.3. 阿哌沙班
12.8.4.1.4. 其他
12.8.4.2. 肝素和低分子肝素
12.8.4.2.1. 達肝素
12.8.4.2.2. 依諾肝素
12.8.4.2.3. 亭扎肝素
12.8.4.2.4. 其他
12.8.4.3. 維生素K拮抗劑
12.8.4.3.1. 華法林
12.8.4.3.2. 苯丙香豆素
12.8.4.3.3. 其他
12.8.4.4. 凝血酶抑制劑
12.8.4.4.1. 比伐盧定
12.8.4.4.2. 阿加曲班
12.8.4.4.3. 達比加群
12.8.4.4.4. 其他
12.8.4.5. 其他
12.8.5 血小板抑制劑
12.8.5.1. 阿斯匹靈
12.8.5.2. 西洛他唑
12.8.5.3. 氯吡格列爾硫酸氫鹽
12.8.5.4. 雙吡達莫
12.8.5.5. 其他
12.8.6 抗高血壓藥
12.8.6.1. 鹽酸可樂定
12.8.6.2. 甲磺酸多沙唑嗪
12.8.6.3. 鹽酸肼屈嗪
12.8.6.4. 甲基多巴
12.8.6.5. 米諾地爾
12.8.6.6. 其他
12.8.7 β受體阻斷劑
12.8.7.1. 鹽酸醋丁洛爾
12.8.7.2. 阿替洛爾
12.8.7.3. 倍他洛爾
12.8.7.4. 比索洛爾
12.8.7.5. 卡維地洛
12.8.7.6. 鹽酸拉貝洛爾
12.8.7.7. 美托洛爾
12.8.7.8. 美托洛爾
12.8.7.9. 納多洛爾
12.8.7.10. 其他
12.8.8 鈣通道阻斷劑
12.8.8.1. 二氫吡啶
12.8.8.1.1. 苯磺酸氨氯地平
12.8.8.1.2. 硝苯地平
12.8.8.1.3. 尼莫地平
12.8.8.1.4. 尼索地平
12.8.8.1.5. 鹽酸尼卡地平
12.8.8.2. 非二氫吡啶
12.8.8.2.1. 鹽酸地爾硫卓
12.8.8.2.2. 鹽酸維拉帕米
12.8.9 利尿劑
12.8.9.1. 噻嗪類利尿劑
12.8.9.1.1. 氯噻酮
12.8.9.1.2. 氫氯噻嗪
12.8.9.1.3. 美托拉宗
12.8.9.1.4. 吲達帕胺
12.8.9.2. 袢利尿劑
12.8.9.2.1. 托拉塞米
12.8.9.2.2. 呋塞米
12.8.9.2.3. 布美他尼
12.8.9.3. 保鉀利尿劑
12.8.9.3.1. 阿米洛利
12.8.9.3.2. 氨苯蝶啶
12.8.9.3.3. 螺內酯
12.8.9.3.4. 依普利酮
12.8.9.4. 其他
12.8.10 脂質藥物
12.8.10.1. 他汀類藥物
12.8.10.1.1. 阿托伐他汀鈣
12.8.10.1.2。氟伐他汀鈉
12.8.10.1.3. 洛伐他汀
12.8.10.1.4. 其他
12.8.10.2. 纖維酸類藥物
12.8.10.2.1. 非諾貝特
12.8.10.2.2. 吉非溴齊
12.8.10.3. 膽汁酸螯合劑
12.8.10.3.1. 鹽酸考來維崙
12.8.10.3.2. 考來烯胺
12.8.10.3.3. 鹽酸考來烯胺
12.8.10.4. 其他脂質藥物
12.8.11 硝酸鹽
12.8.11.1. 口服硝酸甘油
12.8.11.2. 硝酸甘油軟膏
12.8.11.3. 硝酸甘油皮膚貼劑
12.8.11.4. 硝酸甘油舌下片
12.8.11.5. 其他硝酸甘油錠劑、膠囊及噴霧劑
12.8.12 其他
12.9 維生素
12.9.1 維生素B
12.9.2 維生素E
12.9.3 維生素D
12.9.4 維生素C
12.9.5 維生素A
12.9.6 維生素K
12.1 礦物
12.10.1 鈣
12.10.2 鎂
12.10.3 鐵
12.10.4 鉀
12.10.5 鋅
12.10.6 鉻
12.10.7 硒
12.10.8 其他
12.11 其他
13 全球仿製藥市場(依給藥途徑)
13.1 概述
13.2 口頭
13.2.1 固體
13.2.1.1. 固體,依類型
13.2.1.1.1. 片劑
13.2.1.1.2. 膠囊
13.2.1.1.3. 粉末
13.2.1.1.4. 藥丸
13.2.1.1.5. 其他
13.2.1.2. 固體,依劑量
13.2.1.2.1. 少於 100 毫克
13.2.1.2.2.100-500毫克
13.2.1.2.3. 500 毫克 – 1000 毫克
13.2.1.2.4. 超過 1000 毫克
13.2.2 半固態
13.2.2.1. 半固態,依類型
13.2.2.1.1. 凝膠
13.2.2.1.2. 乳液
13.2.2.1.3. 靈丹妙藥
13.2.2.1.4. 其他
13.2.2.2. 半固體,依劑量
13.2.2.2.1. 少於 25 毫升
13.2.2.2.2. 25-50毫升
13.2.2.2.3. 超過 50 毫升
13.2.3 液體
13.2.3.1. 液體,依類型
13.2.3.1.1. 解決方案
13.2.3.1.2. 糖漿
13.2.3.1.3. 其他
13.2.3.2. 液體,依劑量
13.2.3.2.1. 少於 25 毫升
13.2.3.2.2. 25-50毫升
13.2.3.2.3. 超過 50 毫升
13.3 主題
13.3.1 液體
13.3.1.1. 液體,依類型
13.3.1.1.1. 解決方案
13.3.1.1.2. 暫停
13.3.1.2. 液體,依劑量
13.3.1.2.1. 少於 25 毫升
13.3.1.2.2. 25-50毫升
13.3.1.2.3. 超過 50 毫升
13.3.2 半固態
13.3.2.1. 半固態,依類型
13.3.2.1.1. 奶油
13.3.2.1.2. 軟膏
13.3.2.1.3. 凝膠
13.3.2.1.4. 糊劑
13.3.2.1.5. 洗劑
13.3.2.1.6. 其他
13.3.2.2. 半固體,依劑量
13.3.2.2.1. 少於 25 毫克
13.3.2.2.2. 25-50毫克
13.3.2.2.3. 超過 50 毫克
13.3.3 固體
13.3.3.1. 固體,依類型
13.3.3.1.1. 栓劑
13.3.3.1.2. 粉末
13.3.3.2. 固體,依劑量
13.3.3.2.1. 少於1克
13.3.3.2.2. 1GM
13.3.3.2.3. 超過1克
13.3.4 其他
13.4 腸外
13.4.1 腸胃外給藥,依類型
13.4.1.1. 常規藥物傳遞製劑
13.4.1.1.1. 解決方案
13.4.1.1.2. 復溶/凍乾
13.4.1.1.3. 暫停
13.4.1.1.4. 乳劑
13.4.1.1.5. 其他
13.4.1.2. 新型藥物傳遞製劑
13.4.1.2.1. 膠體分散體
13.4.1.2.2. 微粒
13.4.1.2.3. 長效注射劑型
13.4.2 腸胃外給藥,依劑量
13.4.2.1. 1 毫克/毫升
13.4.2.2. 1-5 毫克/毫升
13.4.2.3. 超過5毫克/毫升
13.5 目鏡
13.5.1 眼部,依類型
13.5.1.1. 液體
13.5.1.1.1. 眼藥水
13.5.1.1.2. 噴霧
13.5.1.2. 半固態
13.5.1.2.1. 凝膠
13.5.1.2.2. 軟膏
13.5.2 眼部,依劑量
13.5.2.1. 2.5毫升
13.5.2.2. 5毫升
13.5.2.3. 10 毫升
13.6 鼻內給藥
13.6.1 鼻內給藥,依類型
13.6.1.1. 丟棄
13.6.1.2. 噴霧
13.6.1.3. 粉末
13.6.1.4. 凝膠
13.6.1.5. 其他
13.6.2 鼻內給藥,依劑量
13.6.2.1. 10 毫升
13.6.2.2. 20 毫升
13.6.3 其他
13.6.4
14 全球仿製藥市場(按人口類型劃分)
14.1 概述
14.2 兒科
14.3 成人
14.4 老年病
15 全球仿製藥市場(按購買方式)
15.1 概述
15.2 場外交易
15.3 處方
16 全球仿製藥市場(按最終用戶)
16.1 概述
16.2 醫院
16.2.1 私人
16.2.2 公共
16.3 專科診所
16.4 家庭醫療保健
16.5 門診手術中心
16.6 社區中心
16.7 其他
17 全球仿製藥市場,依分銷管道
17.1 概述
17.2 直接投標
17.3 零售額
17.3.1 醫院藥房
17.3.2 零售藥局
17.3.3 其他
17.4 網路藥局
17.5 其他
18 全球仿製藥市場、公司格局
18.1 公司份額分析:北美
18.2 公司份額分析:歐洲
18.3 公司份額分析:亞太地區
18.4 公司股份分析:南美洲
18.5 合併與收購
18.6 新產品開發與審批
18.7 擴展
18.8 監理變化
18.9 夥伴關係和其他策略發展
19 全球仿製藥市場(按地區)
19.1 全球仿製藥市場,(本章所有細分市場均按國家/地區劃分)
19.2 北美
19.2.1 美國
19.2.2 加拿大
19.2.3 墨西哥
19.3 歐洲
19.3.1 德國
19.3.2 英國
19.3.3 義大利
19.3.4 法國
19.3.5 西班牙
19.3.6 俄羅斯
19.3.7 瑞士
19.3.8 土耳其
19.3.9 比利時
19.3.10 荷蘭
19.3.11 丹麥
19.3.12 瑞典
13.19 波蘭
19.3.14 挪威
2015年3月19日 芬蘭
19.3.16 歐洲其他地區
19.4 亞太地區
19.4.1 日本
19.4.2 中國
19.4.3 韓國
19.4.4 印度
19.4.5 新加坡
19.4.6 泰國
19.4.7 印度尼西亞
19.4.8 馬來西亞
19.4.9 菲律賓
19.4.10 澳大利亞
19.4.11 紐西蘭
19.4.12 越南
19.4.13 台灣
19.4.14 亞太其他地區
19.5 南美洲
19.5.1 巴西
19.5.2 阿根廷
19.5.3 南美洲其他地區
19.6 中東和非洲
19.6.1 南非
19.6.2 埃及
19.6.3 巴林
19.6.4 阿拉伯聯合大公國
19.6.5 科威特
19.6.6 阿曼
19.6.7 卡達
19.6.8 沙烏地阿拉伯
19.6.9 MEA 其他地區
19.7 主要主要見解:按主要國家
20 全球仿製藥市場、公司簡介
20.1 拜耳
20.1.1 公司概況
20.1.2 收入分析
20.1.3 地理分佈
20.1.4 產品組合
20.1.5 近期發展
20.2 賽諾菲
20.2.1 公司概況
20.2.2 收入分析
20.2.3 地理分佈
20.2.4 產品組合
20.2.5 近期發展
20.3 輝瑞公司
20.3.1 公司概況
20.3.2 收入分析
20.3.3 地理存在
20.3.4 產品組合
20.3.5 近期發展
20.4 葛蘭素史克公司
20.4.1 公司概況
20.4.2 收入分析
20.4.3 地理存在
20.4.4 產品組合
20.4.5 最近的發展
20.5 武田製藥株式會社
20.5.1 公司概況
20.5.2 收入分析
20.5.3 地理存在
20.5.4 產品組合
20.5.5 最近的發展
20.6 雅培
20.6.1 公司概況
20.6.2 收入分析
20.6.3 地理存在
20.6.4 產品組合
20.6.5 最近的發展
20.7 諾華公司
20.7.1 公司概況
20.7.2 收入分析
20.7.3 地理存在
20.7.4 產品組合
20.7.5 最近的發展
20.8 太陽製藥工業
20.8.1 公司概況
20.8.2 收入分析
20.8.3 地理存在
20.8.4 產品組合
20.8.5 最近的發展
20.9 梯瓦製藥工業股份有限公司
20.9.1 公司概況
20.9.2 收入分析
20.9.3 地理存在
20.9.4 產品組合
20.9.5 近期發展
20.1 雷迪博士實驗室
20.10.1 公司概況
20.10.2 收入分析
20.10.3 地理存在
20.10.4 產品組合
20.10.5 近期發展
20.11 遠藤製藥公司
20.11.1 公司概況
20.11.2 收入分析
20.11.3 地理存在
20.11.4 產品組合
20.11.5 近期動態
20.12 AMNEAL製藥公司
20.12.1 公司概況
20.12.2 收入分析
20.12.3 地理存在
20.12.4 產品組合
20.12.5 近期動態
20.13 ALKEM 實驗室有限公司
20.13.1 公司概況
20.13.2 收入分析
20.13.3 地理存在
20.13.4 產品組合
20.13.5 近期動態
20.14 第一三共株式會社
20.14.1 公司概況
20.14.2 收入分析
20.14.3 地理存在
20.14.4 產品組合
20.14.5 近期動態
20.15 MYLAN NV(VIATRIS INC.)
20.15.1 公司概況
20.15.2 收入分析
20.15.3 地理存在
20.15.4 產品組合
20.15.5 近期動態
20.16 奧羅賓多製藥
20.16.1 公司概況
20.16.2 收入分析
20.16.3 地理存在
20.16.4 產品組合
20.16.5 近期動態
20.17 ZYDUS 製藥公司
20.17.1 公司概況
20.17.2 收入分析
20.17.3 地理存在
20.17.4 產品組合
20.17.5 近期動態
20.18 魯邦
20.18.1 公司概況
20.18.2 收入分析
20.18.3 地理存在
20.18.4 產品組合
20.18.5 近期動態
20.19 山德士國際有限公司
20.19.1 公司概況
20.19.2 收入分析
20.19.3 地理存在
20.19.4 產品組合
20.19.5 近期動態
20.2 費森尤斯醫藥股份公司
20.20.1 公司概況
20.20.2 收入分析
20.20.3 地理存在
20.20.4 產品組合
20.20.5 近期動態
20.21 賽諾菲
20.21.1 公司概況
20.21.2 收入分析
20.21.3 地理存在
20.21.4 產品組合
20.21.5 近期動態
20.22 CIPLA公司
20.22.1 公司概況
20.22.2 收入分析
20.22.3 地理存在
20.22.4 產品組合
20.22.5 近期動態
20.23 阿斯利康
20.23.1 公司概況
20.23.2 收入分析
20.23.3 地理存在
20.23.4 產品組合
20.23.5 近期動態
20.24 百時美施貴寶
20.24.1 公司概況
20.24.2 收入分析
20.24.3 地理存在
20.24.4 產品組合
20.24.5 近期動態
20.25 PAR製藥
20.25.1 公司概況
20.25.2 收入分析
20.25.3 地理存在
20.25.4 產品組合
20.25.5 最新動態
20.26 HIKMA 製藥公司
20.26.1 公司概況
20.26.2 收入分析
20.26.3 地理存在
20.26.4 產品組合
20.26.5 近期動態
20.27 利潔時
20.27.1 公司概況
20.27.2 收入分析
20.27.3 地理存在
20.27.4 產品組合
20.27.5 近期動態
20.28 百利高
20.28.1 公司概況
20.28.2 收入分析
20.28.3 地理存在
20.28.4 產品組合
20.28.5 近期動態
20.29 大正製藥
20.29.1 公司概況
20.29.2 收入分析
20.29.3 地理存在
20.29.4 產品組合
20.29.5 近期動態
20.3 馬林克羅德
20.30.1 公司概況
20.30.2 收入分析
20.30.3 地理存在
20.30.4 產品組合
20.30.5 最新動態
20.31 安進公司
20.31.1 公司概況
20.31.2 收入分析
20.31.3 地理存在
20.31.4 產品組合
20.31.5 近期動態
20.32 阿瑞娜製藥公司
20.32.1 公司概況
20.32.2 收入分析
20.32.3 地理存在
20.32.4 產品組合
20.32.5 近期發展
20.33 斯塔達藥品股份公司
20.33.1 公司概況
20.33.2 收入分析
20.33.3 地理存在
20.33.4 產品組合
20.33.5 近期發展
20.34 ACCORD HEALTHCARE有限公司
20.34.1 公司概況
20.34.2 收入分析
20.34.3 地理存在
20.34.4 產品組合
20.34.5 近期發展
20.35 ASCENDIS 製藥集團
20.35.1 公司概況
20.35.2 收入分析
20.35.3 地理存在
20.35.4 產品組合
20.35.5 近期發展
20.36 阿爾沃根
20.36.1 公司概況
20.36.2 收入分析
20.36.3 地理存在
20.36.4 產品組合
20.36.5 近期發展
20.37 ANI 製藥公司
20.37.1 公司概況
20.37.2 收入分析
20.37.3 地理存在
20.37.4 產品組合
20.37.5 近期發展
20.38 ACELLA製藥有限責任公司
20.38.1 公司概況
20.38.2 收入分析
20.38.3 地理存在
20.38.4 產品組合
20.38.5 近期發展
20.39 格倫馬克製藥
20.39.1 公司概況
20.39.2 收入分析
20.39.3 地理存在
20.39.4 產品組合
20.39.5 近期發展
20.4 地平線治療公司
20.40.1 公司概況
20.40.2 收入分析
20.40.3 地理存在
20.40.4 產品組合
20.40.5 近期發展
20.41 薩尼斯
20.41.1 公司概況
20.41.2 收入分析
20.41.3 地理存在
20.41.4 產品組合
20.41.5 近期發展
20.42 梅恩製藥
20.42.1 公司概況
20.42.2 收入分析
20.42.3 地理存在
20.42.4 產品組合
20.42.5 近期發展
20.43 大塚製藥
20.43.1 公司概況
20.43.2 收入分析
20.43.3 地理存在
20.43.4 產品組合
20.43.5 近期發展
20.44 瓦克哈特
20.44.1 公司概況
20.44.2 收入分析
20.44.3 地理存在
20.44.4 產品組合
20.44.5 近期發展
20.45 TORQUE PHARMACEUTICALS PVT. 有限公司
20.45.1 公司概況
20.45.2 收入分析
20.45.3 地理存在
20.45.4 產品組合
20.45.5 近期發展
20.46 江蘇恆瑞醫藥股份有限公司
20.46.1 公司概況
20.46.2 收入分析
20.46.3 地理存在
20.46.4 產品組合
20.46.5 近期發展
20.47 克爾卡有限公司
20.47.1 公司概況
20.47.2 收入分析
20.47.3 地理存在
20.47.4 產品組合
20.47.5 近期發展
20.48 HYPERA製藥
20.48.1 公司概況
20.48.2 收入分析
20.48.3 地理存在
20.48.4 產品組合
20.48.5 近期發展
20.49 河北天成藥業有限公司
20.49.1 公司概況
20.49.2 收入分析
20.49.3 地理存在
20.49.4 產品組合
20.49.5 近期發展
20.5 保利製藥公司
20.50.1 公司概況
20.50.2 收入分析
20.50.3 地理存在
20.50.4 產品組合
20.50.5 近期發展
21份相關報告
22 結論
23 問卷
24 關於數據橋市場研究
研究方法
数据收集和基准年分析是使用具有大样本量的数据收集模块完成的。该阶段包括通过各种来源和策略获取市场信息或相关数据。它包括提前检查和规划从过去获得的所有数据。它同样包括检查不同信息源中出现的信息不一致。使用市场统计和连贯模型分析和估计市场数据。此外,市场份额分析和关键趋势分析是市场报告中的主要成功因素。要了解更多信息,请请求分析师致电或下拉您的询问。
DBMR 研究团队使用的关键研究方法是数据三角测量,其中包括数据挖掘、数据变量对市场影响的分析和主要(行业专家)验证。数据模型包括供应商定位网格、市场时间线分析、市场概览和指南、公司定位网格、专利分析、定价分析、公司市场份额分析、测量标准、全球与区域和供应商份额分析。要了解有关研究方法的更多信息,请向我们的行业专家咨询。
可定制
Data Bridge Market Research 是高级形成性研究领域的领导者。我们为向现有和新客户提供符合其目标的数据和分析而感到自豪。报告可定制,包括目标品牌的价格趋势分析、了解其他国家的市场(索取国家列表)、临床试验结果数据、文献综述、翻新市场和产品基础分析。目标竞争对手的市场分析可以从基于技术的分析到市场组合策略进行分析。我们可以按照您所需的格式和数据样式添加您需要的任意数量的竞争对手数据。我们的分析师团队还可以为您提供原始 Excel 文件数据透视表(事实手册)中的数据,或者可以帮助您根据报告中的数据集创建演示文稿。