Global Lysosomal Storage Disorder Drugs Market
Размер рынка в млрд долларов США
CAGR :
% 
 USD
10.76 Billion
USD
21.93 Billion
2024
2032
USD
10.76 Billion
USD
21.93 Billion
2024
2032
| 2025 –2032 | |
| USD 10.76 Billion | |
| USD 21.93 Billion | |
|
|
|
Global Lysosomal Storage Disorder Drugs Market Segmentation, By Type of Disorder Gaucher DiseaseFabry DiseasePompe Disease, Mucopolysaccharidosis (MPS), Niemann-Pick DiseaseKrabbe Disease, and Others), Type (Enzyme Replacement Therapy (ERT), Substrate Reduction Therapy (SRT), Chaperone Therapy, and Others), Drugs (Imiglucerase, Agalsidase Beta, Idursulfase, Alglucosidase Alpha, Velaglucerase, Taliglucerase Alfa, Laronidase, Agalsidase Alpha, Galsulfase, Avalglucosidase Alfa, and Others), Route of Administration (Intravenous (IV), Subcutaneous (SC), Oral, and Others), Age Group (Pediatric, Adults, and Geriatric), Gender (Male and Female), Distribution Channel (Hospital Pharmacies, Drugs Stores and Retail Pharmacies, and Online Pharmacies) - Industry Trends and Forecast to 2032
Lysosomal Storage Disorder Drugs Market Analysis
The global lysosomal storage disorder drugs market encompasses the commercial sector for pharmaceuticals developed to diagnose, treat, and manage various lysosomal storage disorders, which are rare genetic conditions typically characterized by the accumulation of undigested substances within lysosomes due to enzyme deficiencies. This market includes a range of therapeutic products, such as enzyme replacement therapies, substrate reduction therapies, and gene therapies, aimed at addressing the diverse clinical manifestations of LSDs, such as Gaucher disease, Fabry disease, and Pompe disease. With increasing awareness, advancements in research and technology, and a growing number of pipeline therapies, this market is poised for significant expansion. In addition, rising healthcare expenditure and initiatives to improve access to rare disease treatments further drive market growth, presenting both opportunities and challenges for pharmaceutical companies and healthcare providers.
Lysosomal Storage Disorder Drugs Market Size
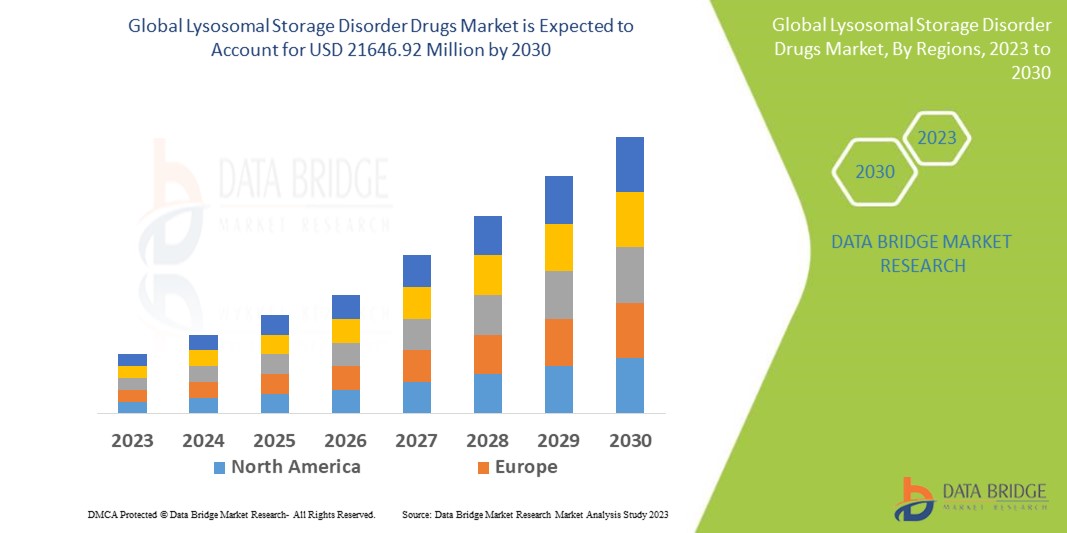
Global lysosomal storage disorder drugs market size was valued at USD 10.76 billion in 2024 and is projected to reach USD 21.93 billion by 2032, growing with a CAGR of 9.4% during the forecast period of 2025 to 2032. In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include import export analysis, production capacity overview, production consumption analysis, price trend analysis, climate change scenario, supply chain analysis, value chain analysis, raw material/consumables overview, vendor selection criteria, PESTLE Analysis, Porter Analysis, and regulatory framework.
Lysosomal Storage Disorder Drugs Market Trends
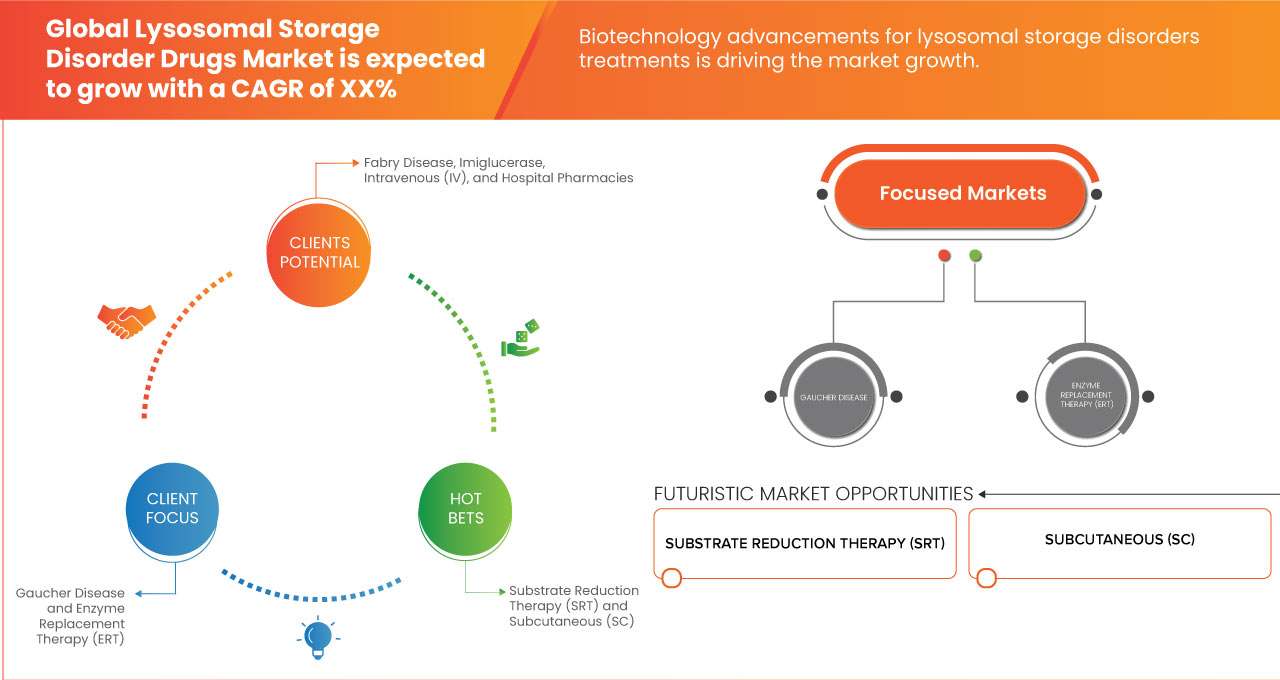
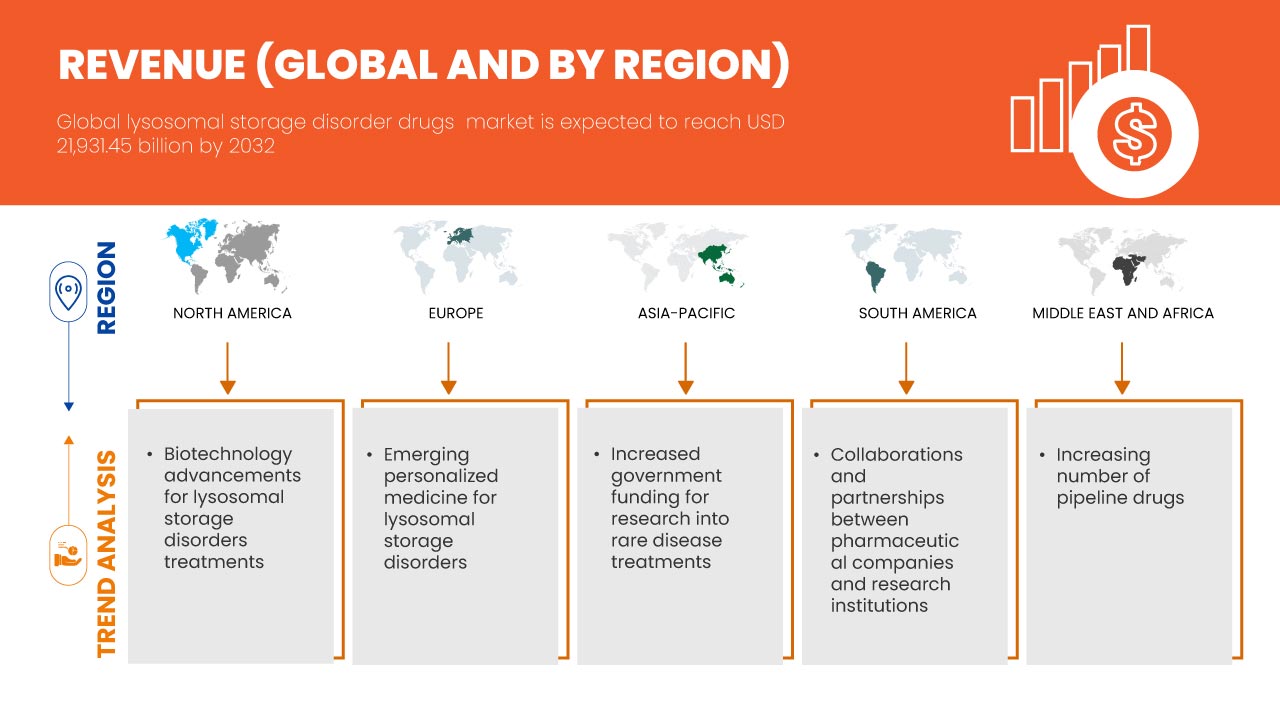
“Biotechnology Advancements for Lysosomal Storage Disorders Treatments”
Advancements in biotechnology have played a pivotal role in transforming the treatment landscape for Lysosomal Storage Disorders (LSDs), providing patients with more effective and tailored treatment options. Enzyme Replacement Therapy (ERT) has revolutionized the treatment of Lysosomal Storage Disorders (LSDs) by directly addressing the enzyme deficiencies that cause these conditions. In LSD patients, the lack of specific enzymes leads to the accumulation of toxic substances in cells, damaging organs and tissues. ERT works by administering synthetic enzymes to compensate for the missing ones, improving metabolic functions and alleviating symptoms. Over the years, ERT has advanced significantly, with new, more effective formulations and delivery methods that improve enzyme absorption and reduce side effects. These innovations have resulted in better clinical outcomes, this trend slowed disease progression, reduced organ damage, and enhanced patient quality of life.
Report Scope and Lysosomal Storage Disorder Drugs Market Segmentation
|
Attributes |
Lysosomal Storage Disorder Drugs Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
U.S., Canada, Mexico, Germany, U.K., Italy, France, Spain, Russia, Switzerland, Turkey, Belgium, Netherlands, Denmark, Sweden, Poland, Norway, Finland, rest of Europe, Japan, China, South Korea, India, Singapore, Thailand, Indonesia, Malaysia, Philippines, Australia, New Zealand, Vietnam, Taiwan, rest of Asia-Pacific, Brazil, Argentina, rest of South America, South Africa, Egypt, Bahrain, United Arab Emirates, Kuwait, Oman, Qatar, Saudi Arabia, and rest of Middle East and Africa |
|
Key Market Players |
Sanofi (France), BioMarin (U.S.), Pfizer Inc. (U.S.), Amicus Therapeutics, Inc. (U.S.), Takeda Pharmaceutical Company Limited (Japan), Ultragenyx Pharmaceutical Inc. (U.S.), Orchard Therapeutics plc (U.K.), Spur Therapeutics (U.K.), Sangamo Therapeutics (U.S.), Protalix BioTherapeutics Inc. (Israel), CHIESI Farmaceutici S.p.A. (Italy), Forge Biologics (U.S.), Denali Therapeutics (U.S.), REGENXBIO INC. (U.S.), and JCR Pharmaceuticals Co., Ltd. (Japan) among others |
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include import export analysis, production capacity overview, production consumption analysis, price trend analysis, climate change scenario, supply chain analysis, value chain analysis, raw material/consumables overview, vendor selection criteria, PESTLE Analysis, Porter Analysis, and regulatory framework. |
Lysosomal Storage Disorder Drugs Market Definition
Lysosomal storage disorder drugs refer to a category of pharmaceuticals specifically developed to treat Lysosomal Storage Disorders (LSDs), which are a group of rare genetic conditions caused by deficiencies in specific enzymes responsible for breaking down complex molecules within lysosomes. These drugs aim to address the underlying metabolic abnormalities associated with LSDs by either replacing the missing enzymes (enzyme replacement therapy), by enhancing the body's ability to produce the enzymes, or by modifying the substrates that accumulate due to enzyme deficiency (substrate reduction therapy). By improving enzymatic function or reducing the toxic build-up of substrates, these treatments can alleviate symptoms, slow disease progression, and ultimately improve the quality of life for individuals affected by these challenging conditions.
Lysosomal Storage Disorder Drugs Market Dynamics
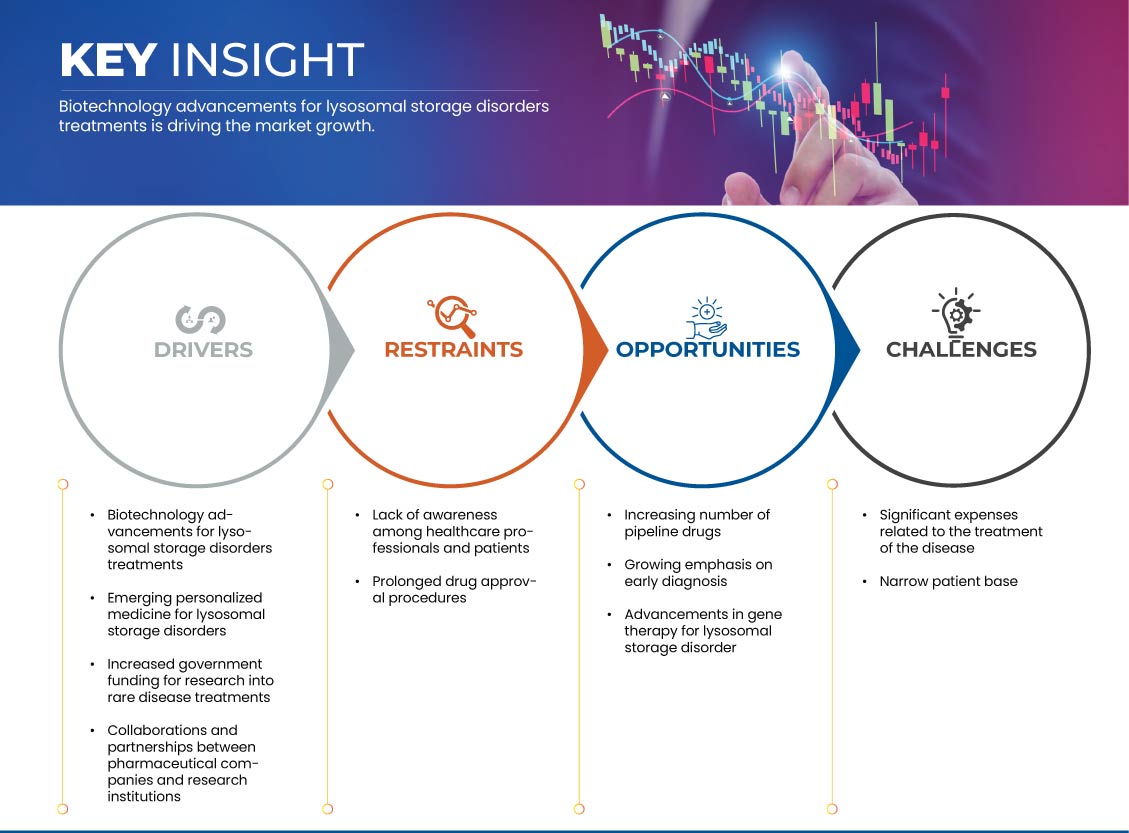
Drivers
- Emerging Personalized Medicine for Lysosomal Storage Disorders
Emerging personalized medicine is transforming the landscape of treatment for Lysosomal Storage Disorders (LSDs) by offering therapies specifically tailored to the individual genetic profiles of patients. LSDs are caused by genetic mutations that affect enzyme function, and these mutations can vary widely between patients. Personalized treatments involve analyzing a patient’s unique genetic makeup to develop a more targeted approach, optimizing the efficacy of therapies such as enzyme replacement or gene therapy. This precision allows for better matching of treatments with the patient's specific needs, minimizing side effects and enhancing therapeutic outcomes. As advancements in genetic testing and technology progress, healthcare providers are able to identify the most suitable interventions, potentially improving response rates and overall treatment success. The shift toward personalized medicine is accelerating the development of customized LSD treatments, which promise to revolutionize disease management. With these innovations, the global lysosomal storage disorders drug market is witnessing increased demand for more effective, individualized treatment options. Personalized medicine is thus a key driver of market growth, as it boosts treatment effectiveness and enhances patient outcomes, fostering greater confidence in therapeutic options.
For instance,
In February 2023, according to the article published by NCBI, Emerging personalized medicine uses an individual’s genetic profile to guide decisions related to the prevention, diagnosis, and treatment of diseases. This approach enables more precise and effective therapies for Lysosomal Storage Disorders (LSDs), tailoring treatments to specific genetic mutations. As personalized medicine becomes more prevalent, it drives the demand for targeted LSD therapies, fueling growth in the global lysosomal storage disorders drug market.
- Increased Government Funding for Research into Rare Disease Treatments
Government funding and grants for research into rare disease treatments are crucial for advancing the development of therapies for conditions such as Lysosomal Storage Disorders (LSDs). Rare diseases, often lacking commercial viability due to small patient populations, pose significant challenges in drug development. To address this, governments worldwide are increasingly allocating funds to incentivize research in these underserved areas. Financial support in the form of grants, subsidies, and tax credits helps pharmaceutical companies and research institutions overcome the high costs and risks associated with developing treatments for rare diseases. Furthermore, governments often fast-track regulatory processes for these treatments, recognizing the urgent need for solutions. By reducing the financial burden on researchers and companies, these funding mechanisms enable the exploration of innovative therapies, such as enzyme replacement and gene therapies that would otherwise face significant barriers to development.
For instance,
In August 2023, according to the article published by National Organization for Rare Diseases, The National Organization for Rare Disorders announced over USD 100,000 in grant funding for rare disease research, highlighting increased government support for advancing treatments. These funds enable researchers to explore innovative therapies for rare diseases like Lysosomal Storage Disorders (LSDs). Such financial backing accelerates drug development, driving growth in the global LSD drug market by fostering new treatment options.
Opportunities
- Increasing Number of Pipeline Drugs
The increasing number of drugs in the pipeline for Lysosomal Storage Disorders (LSDs) represents a significant opportunity for the global lysosomal storage disorders drugs market, potentially leading to improved treatment options and enhanced patient outcomes. As researchers and pharmaceutical companies enhance their understanding of these disorders, they are developing a diverse array of novel therapies, including gene therapies, substrate reduction therapies, and small molecule drugs. This expanded pipeline reflects a growing recognition of the unmet medical needs within the LSD community and promises to offer patients a wider variety of treatment modalities tailored to their specific conditions. The introduction of innovative therapies can enhance patient adherence to treatment, reduce disease burden, and ultimately improve the quality of life for individuals living with these disorders.
For instance,
In June 2024, Ultragenyx announced the Planning to file for accelerated approval of UX111 for the treatment of Sanfilippo Syndrome Type A (MPS IIIA). UX111 is a novel in vivo gene therapy in Phase 1/2/3 development for Sanfilippo syndrome type A (MPS IIIA), a rare fatal lysosomal storage disease with no approved treatment that primarily affects the central nervous system.
- Growing Emphasis on Early Diagnosis
The growing emphasis on early diagnosis of Lysosomal Storage Disorders (LSDs) presents a significant opportunity for the global lysosomal storage disorders drugs market by facilitating timely interventions that can greatly enhance patient outcomes. As healthcare systems place greater emphasis on early detection through improved screening programs, genetic testing, and advancements in diagnostic technologies, more patients are likely to be diagnosed at an earlier stage of their diseases. This early intervention allows for better management of symptoms but increases the potential efficacy of existing and emerging therapies. The result of this shift is a broader patient base that requires and can benefit from treatment options, ultimately driving demand within the lysosomal storage disorders drug market.
For instance,
In February 2023, according to an article, ‘Lysosomal storage disorders: from biology to the clinic with reference to India’, early diagnosis is the most critical part of the management of LSDs as it provides the opportunity for therapeutic intervention, precise genetic counselling, prenatal diagnosis and a better outcome for the patient.
Restraints/Challenges
- Lack of Awareness Among Healthcare Professionals and Patients
The lack of awareness among healthcare professionals and patients about Lysosomal Storage Disorders (LSDs) presents a significant barrier to early diagnosis, treatment, and effective management. Many healthcare providers, especially in regions with limited exposure to rare diseases, often fail to recognize the symptoms of LSDs, which are diverse and overlap with other more common conditions. This lack of knowledge leads to delayed diagnoses, misdiagnosis, and ineffective treatment, exacerbating the severity of the disease. For patients, especially those in resource-poor or underserved areas, the lack of awareness prevents early intervention, leaving them unaware of potential therapies. The complexity and rarity of LSDs further complicate the situation, as patients and healthcare professionals may not fully understand the importance of early treatment or the availability of specialized care options. Fewer patients are diagnosed and treated in time, leading to suboptimal clinical outcomes. This lack of awareness limits the demand for LSD-specific treatments and hinders the overall growth of the global lysosomal storage disorders drugs market.
For instance,
In January 2024, according to the article published by Wiley, there’s a significant diagnostic delay of around 15 years from symptom onset to diagnosis is common in adult LSD cases due to overlapping clinical phenotypes and varying severity. This delay highlights the lack of awareness among healthcare professionals, particularly those treating adult patients. Such restraints hinder early detection and timely treatment, restricting the growth of the global lysosomal storage disorders drugs market.
- Prolonged Drug Approval Procedures
Lengthy drug approval processes present a significant restraint for the global lysosomal storage disorder drugs market, slowing the introduction of much-needed therapies. The process for obtaining regulatory approval for new drugs, particularly for rare diseases like LSDs, involves numerous clinical trials, extensive documentation, and rigorous assessments by regulatory bodies such as the FDA and EMA. These approval timelines are often extended due to the need for a thorough evaluation of safety, efficacy, and potential long-term effects. In the case of LSDs, the rarity and complexity of these diseases add another layer of challenge, as there is a limited patient population for clinical trials, making it difficult to generate robust data. The need for specialized treatments and the lack of established standards for these rare diseases complicates the approval process. Consequently, drug developers face prolonged waiting periods, delays in bringing new treatments to market, and additional costs, which can be discouraging for pharmaceutical companies. As a result, the lengthy approval processes slow the availability of innovative therapies for patients, reducing market growth and hindering the accessibility of effective treatments for LSDs. This regulatory delay ultimately acts as a major restraint for the global lysosomal storage disorders drugs market.
For instance,
In August 2024, according to the article published by Drugs.com, The research, development, and approval process for drugs typically takes 12 to 15 years. This extended timeline delays the introduction of new therapies and increases costs for pharmaceutical companies. As a result, the lengthy drug approval process restrains the growth of the global LSD drug market by limiting the speed of treatment availability.
This market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
Lysosomal Storage Disorder Drugs Market Scope
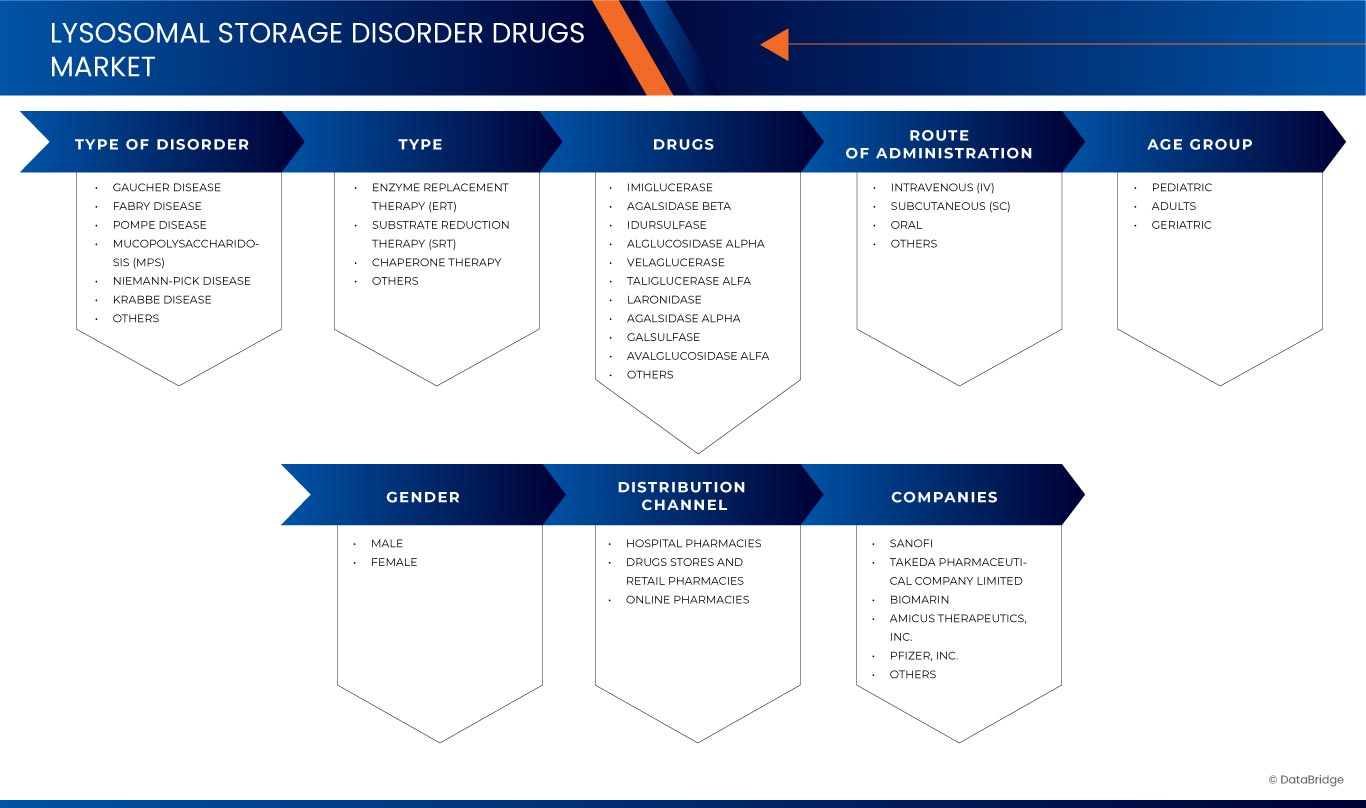
The market is segmented on the basis of by type of disorder, type, drugs, route of administration, age group, gender, and distribution channel. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Type of Disorder
- Gaucher Disease
- Type 1
- Type 3
- Type 2
- Fabry Disease
- Pompe Disease
- Infantile-Onset Pompe
- Late-Onset Pompe
- Mucopolysaccharidosis (MPS)
- MPS I
- MPS II
- MPS IV
- MPS VI
- MPS III
- Niemann-Pick Disease
- Type C
- Type B
- Type A
- Krabbe Disease
- Others
Type
- Enzyme Replacement Therapy (ERT)
- Substrate Reduction Therapy (SRT)
- Chaperone Therapy
- Others
Drugs
- Imiglucerase
- Agalsidase Beta
- Idursulfase
- Alglucosidase Alpha
- Velaglucerase
- Taliglucerase Alfa
- Laronidase
- Agalsidase Alpha
- Galsulfase
- Avalglucosidase Alfa
- Others
Route of Administration
- Intravenous (IV)
- Subcutaneous (SC)
- Oral
- Others
Age Group
- Pediatric
- Adults
- Geriatric
Gender
- Male
- Female
Distribution Channel
- Hospital Pharmacies
- Drugs Stores and Retail Pharmacies
- Online Pharmacies
Lysosomal Storage Disorder Drugs Market Regional Analysis
The market is analyzed and market size insights and trends are provided by type of disorder, type, drugs, route of administration, age group, gender, and distribution channel as referenced above.
The countries covered in the market are U.S., Canada, Mexico, Germany, U.K., Italy, France, Spain, Russia, Switzerland, Turkey, Belgium, Netherlands, Denmark, Sweden, Poland, Norway, Finland, rest of Europe, Japan, China, South Korea, India, Singapore, Thailand, Indonesia, Malaysia, Philippines, Australia, New Zealand, Vietnam, Taiwan, rest of Asia-Pacific, Brazil, Argentina, rest of South America, South Africa, Egypt, Bahrain, United Arab Emirates, Kuwait, Oman, Qatar, Saudi Arabia, and rest of Middle East and Africa.
North America is expected to dominate the global lysosomal storage disorder drugs market due to its advanced healthcare infrastructure, high adoption of drugs, significant research and development investments, and a large patient population.
Asia-Pacific is expected to be the fastest growing due to increasing healthcare infrastructure, rising prevalence of lysosomal storage diseases, and growing awareness about advanced therapies.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Data points like down-stream and upstream value chain analysis, technical trends and porter's five forces analysis, case studies are some of the pointers used to forecast the market scenario for individual countries. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Lysosomal Storage Disorder Drugs Market Share
The market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to market.
Lysosomal Storage Disorder Drugs Market Leaders Operating in the Market Are:
- Sanofi (France)
- BioMarin (U.S.)
- Pfizer Inc. (U.S.)
- Amicus Therapeutics, Inc. (U.S.)
- Takeda Pharmaceutical Company Limited (Japan)
- Ultragenyx Pharmaceutical Inc. (U.S.)
- Orchard Therapeutics plc (U.K.)
- Spur Therapeutics (U.K.)
- Sangamo Therapeutics (U.S.)
- Protalix BioTherapeutics Inc. (Israel)
- Chiesi Farmaceutici S.p.A. (Italy)
- Forge Biologics (U.S.)
- Denali Therapeutics (U.S.)
- REGENXBIO INC. (U.S.)
- JCR Pharmaceuticals Co., Ltd. (Japan)
Latest Developments in Lysosomal Storage Disorder Drugs Market
- In October 2024, Amicus Therapeutics announced a settlement regarding its drug Galafold (migalastat), which treats Fabry disease. The settlement resolves ongoing patent disputes and confirms the continuation of the drug’s marketing without litigation interference. This agreement with multiple parties aims to provide stability for Galafold’s availability and development while protecting intellectual property rights
- In June 2024, Amicus Therapeutics was honored with the Prix Galien UK Award for its innovative treatment, Pombiliti (miglustat), for the management of Fabry disease. The award recognizes excellence in pharmaceutical innovation and emphasizes the impact of Pombiliti in improving the lives of patients with rare genetic conditions. This accolade highlights Amicus’ leadership in rare disease therapies
- In April 2024, Forge Biologics announced it would present nine times at the ASGCT 27th Annual Meeting in May 2024, including a late-breaking oral presentation and three technical sessions. Presentations will cover process development, molecular advancements, and clinical updates, including a significant clinical result for FBX-101 in Krabbe disease
- In November 2023, Chiesi Group has been reaccredited as a Great Place to Work-Certified organization across multiple regions, including Italy, Australia, the US, and others. With an 85% response rate from employees, Chiesi achieved an 83% overall satisfaction rate, reflecting its commitment to fostering a positive, inclusive, and collaborative work environment focused on employee well-being and growth
- In January 2024, Denali Therapeutics Inc., a biopharmaceutical company developing therapies to cross the blood-brain barrier for treating neurodegenerative and lysosomal storage diseases, announced progress and milestones for 2024. CEO Ryan Watts, Ph.D., highlighted these developments during a corporate presentation at the 42nd Annual J.P. Morgan Healthcare Conference on January 9th
SKU-
Получите онлайн-доступ к отчету на первой в мире облачной платформе рыночной аналитики
- Интерактивная панель анализа данных
- Панель анализа компании для возможностей с высоким потенциалом роста
- Доступ аналитика-исследователя для настройки и запросов
- Анализ конкурентов с помощью интерактивной панели
- Последние новости, обновления и анализ тенденций
- Используйте возможности сравнительного анализа для комплексного отслеживания конкурентов
Методология исследования
Сбор данных и анализ базового года выполняются с использованием модулей сбора данных с большими размерами выборки. Этап включает получение рыночной информации или связанных данных из различных источников и стратегий. Он включает изучение и планирование всех данных, полученных из прошлого заранее. Он также охватывает изучение несоответствий информации, наблюдаемых в различных источниках информации. Рыночные данные анализируются и оцениваются с использованием статистических и последовательных моделей рынка. Кроме того, анализ доли рынка и анализ ключевых тенденций являются основными факторами успеха в отчете о рынке. Чтобы узнать больше, пожалуйста, запросите звонок аналитика или оставьте свой запрос.
Ключевой методологией исследования, используемой исследовательской группой DBMR, является триангуляция данных, которая включает в себя интеллектуальный анализ данных, анализ влияния переменных данных на рынок и первичную (отраслевую экспертную) проверку. Модели данных включают сетку позиционирования поставщиков, анализ временной линии рынка, обзор рынка и руководство, сетку позиционирования компании, патентный анализ, анализ цен, анализ доли рынка компании, стандарты измерения, глобальный и региональный анализ и анализ доли поставщика. Чтобы узнать больше о методологии исследования, отправьте запрос, чтобы поговорить с нашими отраслевыми экспертами.
Доступна настройка
Data Bridge Market Research является лидером в области передовых формативных исследований. Мы гордимся тем, что предоставляем нашим существующим и новым клиентам данные и анализ, которые соответствуют и подходят их целям. Отчет можно настроить, включив в него анализ ценовых тенденций целевых брендов, понимание рынка для дополнительных стран (запросите список стран), данные о результатах клинических испытаний, обзор литературы, обновленный анализ рынка и продуктовой базы. Анализ рынка целевых конкурентов можно проанализировать от анализа на основе технологий до стратегий портфеля рынка. Мы можем добавить столько конкурентов, о которых вам нужны данные в нужном вам формате и стиле данных. Наша команда аналитиков также может предоставить вам данные в сырых файлах Excel, сводных таблицах (книга фактов) или помочь вам в создании презентаций из наборов данных, доступных в отчете.