Point-of-Care-Tests (POCT) revolutionieren die Gesundheitsversorgung durch schnelle Diagnostik direkt beim Patienten. Die Anwendungsmöglichkeiten erstrecken sich über verschiedene medizinische Bereiche, darunter Kliniken, Krankenhäuser und abgelegene Gebiete. POCT-Geräte bieten benutzerfreundliche Oberflächen und liefern schnelle und präzise Ergebnisse für Erkrankungen wie Diabetes, Herzmarker und Infektionskrankheiten . Diese Funktionen verbessern die Patientenversorgung, indem sie sofortige Behandlungsentscheidungen ermöglichen, Durchlaufzeiten verkürzen und den Zugang zur Gesundheitsversorgung in unterversorgten Regionen erweitern, was letztlich zu optimierten medizinischen Ergebnissen führt.
Vollständigen Bericht abrufen unter https://www.databridgemarketresearch.com/reports/europe-poct-market
Data Bridge Market Research analysiert, dass der europäische Markt für Point-of-Care-Tests (POCT) im Jahr 2021 einen Wert von 6.664,28 Millionen US-Dollar hat und bis 2029 voraussichtlich 13.085,33 Millionen US-Dollar erreichen wird. Dies entspricht einer durchschnittlichen jährlichen Wachstumsrate (CAGR) von 8,8 % im Prognosezeitraum 2022 bis 2029. Point-of-Care-Tests (POCT) liefern schnelle Diagnoseergebnisse direkt beim Patienten und ermöglichen medizinischem Fachpersonal sofortige Behandlungsentscheidungen. Dieser schnelle Zugriff auf wichtige Informationen verbessert die Patientenversorgung, indem Verzögerungen bei Diagnose und Intervention minimiert werden, was zu einer zeitnahen und effektiveren medizinischen Behandlung führt.
Wichtigste Ergebnisse der Studie
Frühdiagnose und Intervention dürften das Marktwachstum ankurbeln
Point-of-Care-Diagnostik (POCT) spielt eine zentrale Rolle bei der Früherkennung von Krankheiten. Sie ermöglicht einen schnellen Behandlungsbeginn und führt letztlich zu besseren Behandlungsergebnissen. Durch die schnelle und sofortige Bereitstellung von Diagnoseinformationen kann medizinisches Fachpersonal umgehend geeignete Therapien einleiten und so Krankheitsverlauf und Komplikationen verhindern. Dieser proaktive Ansatz verbessert die Wirksamkeit medizinischer Interventionen deutlich, verbessert die Patientenprognose und trägt zur Effizienz des gesamten Gesundheitssystems bei.
Berichtsumfang und Marktsegmentierung
Berichtsmetrik
|
Details
|
Prognosezeitraum
|
2022 bis 2029
|
Basisjahr
|
2021
|
Historische Jahre
|
2020 (Anpassbar auf 2014–2019)
|
Quantitative Einheiten
|
Umsatz in Millionen USD, Mengen in Einheiten, Preise in USD
|
Abgedeckte Segmente
|
Produkte (Instrumente, Verbrauchsmaterialien und Reagenzien und Sonstiges), Anwendung (Blutzucker, Herzüberwachung, Gerinnung, Vollblutanalyse, Überwachung der Vitalfunktionen, Tests auf Infektionskrankheiten, Urinanalyse, Cholesterintests und Sonstiges), Endbenutzer (Labore, Krankenhäuser, Kliniken, Ambulanzzentren, häusliche Pflege, Einrichtungen für betreutes Wohnen, Apotheken/Drogerien und Sonstiges), Testmethode (Rezeptbasierte Tests und OTC-Tests), Vertriebskanal (Direktvertrieb, Drittanbieter und Online-Verkauf)
|
Abgedeckte Länder
|
Deutschland, Frankreich, Großbritannien, Niederlande, Schweiz, Belgien, Russland, Italien, Spanien, Türkei, Restliches Europa
|
Abgedeckte Marktteilnehmer
|
Siemens Healthcare GmbH (Deutschland), F. Hoffmann-La Roche Ltd. (Schweiz), Quidel Corporation (USA), Trividia Health, Inc. (USA), Nova Biomedical (USA), PTS Diagnostics (USA), Trinity Biotech (Irland), LumiraDx (Großbritannien), Sekisui Diagnostics (USA), Chembio Diagnostics, Inc. (USA), BD (USA), Thermo Fisher Scientific Inc. (USA), Abbott (USA), Danaher (USA), binx health, inc (USA), QuantuMDx Group Ltd. (Großbritannien), Werfen (Spanien), Abaxis (USA), EKF Diagnostics (Großbritannien), Sysmex Europe SE (Deutschland)
|
Im Bericht behandelte Datenpunkte
|
Neben den Einblicken in Marktszenarien wie Marktwert, Wachstumsrate, Segmentierung, geografische Abdeckung und wichtige Akteure umfassen die von Data Bridge Market Research kuratierten Marktberichte auch eingehende Expertenanalysen, Patientenepidemiologie, Pipeline-Analysen, Preisanalysen und regulatorische Rahmenbedingungen.
|
Segmentanalyse:
Der europäische Markt für Point-of-Care-Tests (POCT) ist nach Produkt, Anwendung, Endbenutzer, Testmethode und Vertriebskanal segmentiert.
- Auf der Grundlage des Produkts ist der Markt in Instrumente, Verbrauchsmaterialien und Reagenzien usw. unterteilt.
- Auf der Grundlage der Anwendung ist der Markt in Blutzucker, Herzüberwachung, Gerinnung, Vollblutanalyse, Überwachung der Vitalfunktionen, Tests auf Infektionskrankheiten, Urinanalyse, Cholesterintests und andere unterteilt.
- Auf der Grundlage des Endverbrauchers ist der Markt in Labore, Krankenhäuser, Kliniken, ambulante Zentren, häusliche Pflege, Einrichtungen für betreutes Wohnen, Apotheken/Drogerien und andere segmentiert.
- Auf der Grundlage der Testmethode wird der Markt in verschreibungspflichtige Tests und OTC-Tests segmentiert.
- Auf der Grundlage des Vertriebskanals ist der Markt in Direktvertrieb, Drittanbietervertrieb und Online-Verkauf segmentiert.
Hauptakteure
Data Bridge Market Research erkennt die folgenden Unternehmen als die wichtigsten europäischen Marktteilnehmer für Point-of-Care-Tests (POCT) an: Siemens Healthcare GmbH (Deutschland), F. Hoffmann-La Roche Ltd. (Schweiz), Quidel Corporation (USA), Trividia Health, Inc. (USA), Nova Biomedical (USA), PTS Diagnostics (USA), Trinity Biotech (Irland), LumiraDx (Großbritannien),
Marktentwicklungen
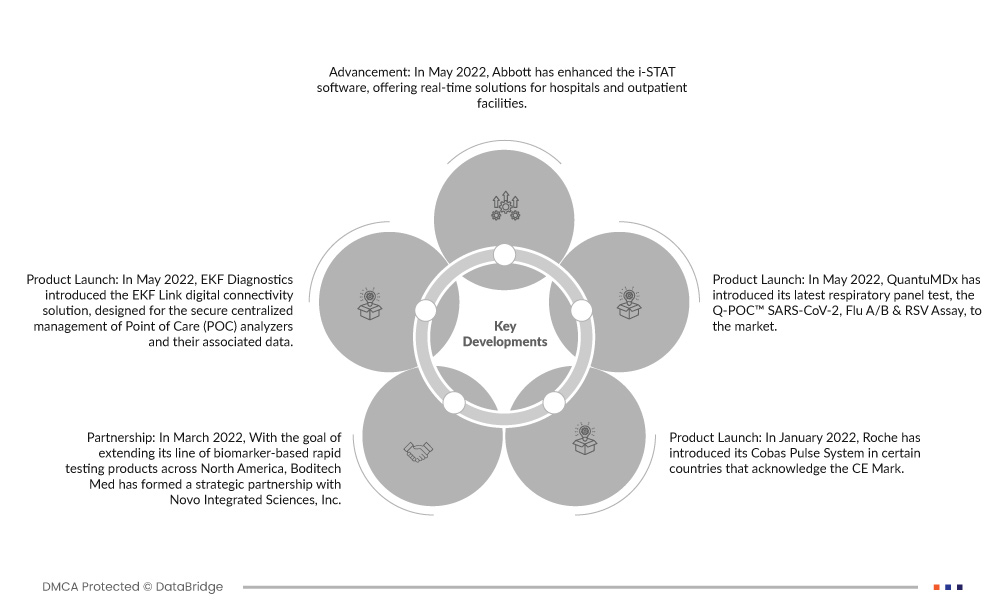
- Im Mai 2022 hat Abbott die i-STAT-Software erweitert und bietet nun Echtzeitlösungen für Krankenhäuser und ambulante Einrichtungen. Die aktualisierte Software zeichnet sich durch hohe Wirksamkeit und Kosteneffizienz aus und unterstützt die Entscheidungsfindung in der Patientenversorgung. So werden effiziente und präzise Gesundheitslösungen gewährleistet.
- Im Mai 2022 stellte EKF Diagnostics die digitale Konnektivitätslösung EKF Link vor, die für die sichere zentrale Verwaltung von Point-of-Care-Analysegeräten (POC) und den zugehörigen Daten entwickelt wurde. Diese Plattform optimiert den Verwaltungsprozess, gewährleistet Datensicherheit und steigert die Effizienz von POC-Testvorgängen.
- Im Mai 2022 hat QuantuMDx seinen neuesten Atemwegstest, den Q-POC™ SARS-CoV-2, Flu A/B & RSV Assay, auf den Markt gebracht.
- Im März 2022 ist Boditech Med mit dem Ziel, seine Produktlinie an Biomarker-basierten Schnelltests in ganz Nordamerika auszuweiten, eine strategische Partnerschaft mit Novo Integrated Sciences, Inc. eingegangen.
- Im Januar 2022 hat Roche sein Cobas Pulse System in einigen Ländern eingeführt, die das CE-Zeichen anerkennen. Dieses fortschrittliche Angebot von Roche Diagnostics stellt die neueste vernetzte Point-of-Care-Lösung für professionelles Blutzuckermanagement dar.
Regionale Analyse
Geografisch betrachtet umfasst der europäische Marktbericht für Point-of-Care-Tests (POCT) folgende Länder: Deutschland, Frankreich, Großbritannien, Niederlande, Schweiz, Belgien, Russland, Italien, Spanien, Türkei und das übrige Europa.
Laut Marktforschungsanalyse von Data Bridge:
Deutschland ist im Prognosezeitraum 2022 – 2029 die dominierende Region im europäischen Point-of-Care-Testing (POCT)-Markt
Im Jahr 2022 dominierte Deutschland den Markt für Point-of-Care-Tests (POCT). Grund dafür ist das wachsende Bewusstsein für die Vorteile von Vor-Ort-Tests. Sie liefern schnelle Ergebnisse, die die Patientenversorgung verbessern, Behandlungsentscheidungen beschleunigen und das Krankheitsmanagement optimieren. Die zunehmende Vertrautheit mit den Möglichkeiten von POCT fördert dessen Akzeptanz als grundlegendes Element moderner Gesundheitspraktiken in ganz Europa.
Für detailliertere Informationen zum Marktbericht für Point-of-Care-Tests (POCT) klicken Sie hier – https://www.databridgemarketresearch.com/reports/europe-poct-market