اضطراب ما بعد الصدمة (PTSD) هو حالة صحية نفسية ناجمة عن تجارب مرعبة، سواءً كانت مباشرة أو كشهود. تشمل أعراضه ذكريات الماضي، والكوابيس، والقلق، والأفكار المُسيطرة حول الحدث. في حين أن معظم الناس يُعانون من صعوبات مؤقتة، فإن الأعراض المُزمنة والشديدة التي تؤثر على الحياة اليومية تُشير إلى الإصابة باضطراب ما بعد الصدمة. يُعد العلاج الفعال أمرًا بالغ الأهمية لتخفيف الأعراض وتحسين الأداء الوظيفي لمن يُعانون من اضطراب ما بعد الصدمة. يُمكن للتدخل المُبكر أن يُحدث فرقًا كبيرًا في عملية التعافي على المدى الطويل.
يمكنك الوصول إلى التقرير الكامل على https://www.databridgemarketresearch.com/reports/germany-post-traumatic-stress-disorder-market
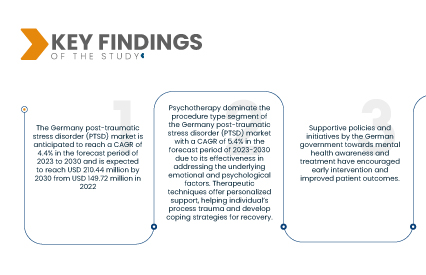
تشير تحليلات شركة داتا بريدج لأبحاث السوق إلى أن سوق اضطراب ما بعد الصدمة (PTSD) في ألمانيا من المتوقع أن ينمو بمعدل نمو سنوي مركب قدره 4.4% خلال الفترة المتوقعة من 2023 إلى 2030، وأن يصل حجمه إلى 210.44 مليون دولار أمريكي بحلول عام 2030، مقارنةً بـ 149.72 مليون دولار أمريكي في عام 2022. وقد أدى تزايد الوعي بالصحة النفسية وأهمية طلب المساعدة لعلاج اضطراب ما بعد الصدمة إلى زيادة وعي الأفراد بأعراضهم والسعي للحصول على التشخيص والعلاج. وقد سهّل هذا الوعي المتزايد التدخلات في الوقت المناسب، مما مكّن من إدارة أفضل ونتائج أفضل للمتضررين من اضطراب ما بعد الصدمة.
النتائج الرئيسية للدراسة
من المتوقع أن يؤدي معالجة الصدمات في المهن عالية الخطورة إلى دفع معدل نمو السوق
أدت الجهود المبذولة لمعالجة ودعم الصدمات النفسية في المهن عالية الخطورة، مثل العاملين في مجال الرعاية الصحية والمستجيبين الأوائل، إلى تحسين إدارة اضطراب ما بعد الصدمة لدى هذه الفئات الضعيفة. غالبًا ما تُعرّض هذه المهن الأفراد لأحداث مؤلمة وصادمة، مما يزيد من خطر إصابتهم باضطراب ما بعد الصدمة. ولمعالجة هذا الوضع، طُبّقت برامج تدريبية متخصصة، وخدمات استشارية، ودعم للصحة النفسية، مما يُساعد المهنيين على التعامل مع العبء العاطفي لعملهم، ويُسهّل التدخل المبكر للوقاية من الآثار النفسية طويلة المدى.
نطاق التقرير وتقسيم السوق
مقياس التقرير
|
تفاصيل
|
فترة التنبؤ
|
من 2023 إلى 2030
|
سنة الأساس
|
2022
|
السنوات التاريخية
|
2021 (قابلة للتخصيص حتى 2015-2020)
|
الوحدات الكمية
|
الإيرادات بالملايين من الدولارات الأمريكية، والحجم بالوحدات، والتسعير بالدولار الأمريكي
|
القطاعات المغطاة
|
النوع (اضطراب ما بعد الصدمة التفارقي، اضطراب ما بعد الصدمة غير المعقد، اضطراب ما بعد الصدمة المصاحب، اضطرابات أخرى)، نوع العلاج (الأدوية، العلاج النفسي، أجهزة البرمجيات)، طريقة الإعطاء (عن طريق الفم، عن طريق الحقن، اضطرابات أخرى)، الفئة العمرية (الأطفال، البالغون، كبار السن)، المستخدم النهائي (المستشفيات، العيادات الخارجية، مراكز الصحة النفسية، الرعاية الصحية المنزلية ، اضطرابات أخرى)، قناة التوزيع (صيدلية المستشفى، صيدلية التجزئة، صيدلية الإنترنت وغيرها)
|
الجهات الفاعلة في السوق المغطاة
|
شركة AbbVie Inc. (الولايات المتحدة)، شركة Viatris Inc. (الولايات المتحدة)، شركة Pfizer Inc. (الولايات المتحدة)، شركة Eli Lilly and Company (الولايات المتحدة)، شركة Takeda Pharmaceutical Company Limited (اليابان)، شركة Aurobindo Pharma (الهند)، شركة AstraZeneca (المملكة المتحدة)، شركة Cipla Inc. (الهند)، شركة Lupin (الهند)، شركة Accord Healthcare GmbH (ألمانيا)، شركة Dr. Reddy's Laboratories Ltd. (الهند)، شركة Zydus Pharmaceuticals, Inc. (الولايات المتحدة)، مجموعة شركات GlaxoSmithKline (المملكة المتحدة)، شركة Teva Pharmaceutical Industries Ltd. (إسرائيل)، شركة Alkermes (الولايات المتحدة)، شركة Novartis AG (سويسرا) وغيرها.
|
نقاط البيانات التي يغطيها التقرير
|
بالإضافة إلى الرؤى حول سيناريوهات السوق مثل القيمة السوقية ومعدل النمو والتجزئة والتغطية الجغرافية واللاعبين الرئيسيين، فإن تقارير السوق التي تم تنظيمها بواسطة Data Bridge Market Research تتضمن أيضًا تحليلًا متعمقًا من الخبراء وعلم الأوبئة للمرضى وتحليل خطوط الأنابيب وتحليل التسعير والإطار التنظيمي.
|
تحليل القطاعات:
يتم تقسيم سوق اضطراب ما بعد الصدمة (PTSD) في ألمانيا على أساس النوع ونوع العلاج وطريقة الإدارة والفئة العمرية والمستخدم النهائي وقناة التوزيع.
- يُقسّم سوق اضطراب ما بعد الصدمة (PTSD) في ألمانيا، حسب نوعه، إلى اضطراب ما بعد الصدمة الانفصالي، واضطراب ما بعد الصدمة غير المعقد، واضطراب ما بعد الصدمة المصاحب، وغيرها. يهيمن اضطراب ما بعد الصدمة غير المعقد على فئة أنواع هذا السوق بمعدل نمو سنوي مركب قدره 5.5% خلال الفترة المتوقعة من 2023 إلى 2030، نظرًا لانتشاره بين الأفراد الذين تعرضوا لصدمة واحدة. ويتيح عرض أعراضه البسيط نسبيًا إمكانية تحديده في الوقت المناسب واتخاذ تدخلات علاجية محددة، مما يُسهم في تحسين نتائج العلاج.
في عام 2023، تهيمن اضطرابات ما بعد الصدمة غير المعقدة على شريحة النوع في سوق اضطرابات ما بعد الصدمة في ألمانيا
في عام ٢٠٢٣، يهيمن اضطراب ما بعد الصدمة غير المعقد على سوق أنواع اضطراب ما بعد الصدمة في ألمانيا، نظرًا لشيوعه بين الأفراد الذين تعرضوا لصدمة واحدة. ويتيح عرض أعراضه بوضوح الكشف المبكر والتدخلات الهادفة، مما يؤدي إلى تحسين نتائج العلاج بمعدل نمو سنوي مركب قدره ٥.٥٪ خلال الفترة المتوقعة من ٢٠٢٣ إلى ٢٠٣٠.
- بناءً على نوع الإجراء، يُقسّم سوق اضطراب ما بعد الصدمة (PTSD) في ألمانيا إلى أدوية، وعلاج نفسي، وأجهزة برمجية. يهيمن العلاج النفسي على شريحة الإجراءات في سوق اضطراب ما بعد الصدمة (PTSD) في ألمانيا، بمعدل نمو سنوي مركب قدره 5.4% خلال الفترة المتوقعة 2023-2030، نظرًا لفعاليته في معالجة العوامل العاطفية والنفسية الكامنة. تقدم التقنيات العلاجية دعمًا شخصيًا، مما يساعد الأفراد على تجاوز الصدمة وتطوير استراتيجيات للتعافي.
- بناءً على طريقة الإعطاء، يُقسّم سوق علاج اضطراب ما بعد الصدمة (PTSD) في ألمانيا إلى علاجات فموية، وحقنية، وغيرها. يهيمن العلاج الفموي على شريحة طرق الإعطاء الإجرائية في سوق علاج اضطراب ما بعد الصدمة (PTSD) في ألمانيا، بمعدل نمو سنوي مركب قدره 4.7% خلال الفترة المتوقعة 2023-2030، نظرًا لسهولة استخدامه وألفته لدى المرضى. وتُفضّل سهولة بلع الدواء، مما يجعله طريقة مقبولة على نطاق واسع ومتاحة للإعطاء لمختلف العلاجات والأدوية.
في عام 2023، ستسيطر شركة Oral على قطاع طرق إدارة نوع الإجراء في سوق اضطراب ما بعد الصدمة (PTSD) في ألمانيا
في عام ٢٠٢٣، سيطر العلاج الفموي على سوق علاج اضطراب ما بعد الصدمة (PTSD) في ألمانيا بفضل سهولة استخدامه وألفة المرضى به. يُعدّ تناول الدواء عن طريق البلع مريحًا ومقبولًا على نطاق واسع وسهل المنال، مما يجعله الخيار الأمثل لمختلف العلاجات والأدوية بمعدل نمو سنوي مركب قدره ٤.٧٪ خلال الفترة المتوقعة ٢٠٢٣-٢٠٣٠.
- بناءً على الفئة العمرية، يُقسّم سوق اضطراب ما بعد الصدمة (PTSD) في ألمانيا إلى فئات عمرية: الأطفال، والبالغون، وكبار السن. يهيمن البالغون على شريحة الفئات العمرية في سوق اضطراب ما بعد الصدمة (PTSD) في ألمانيا، بمعدل نمو سنوي مركب قدره 4.9% خلال الفترة المتوقعة 2023-2030، نظرًا لتعرضهم المتزايد للأحداث الصادمة المحتملة بمرور الوقت. ومع التقدم في السن، يزداد احتمال التعرض لمواقف مُحفّزة، مما يؤدي إلى ارتفاع معدل انتشار اضطراب ما بعد الصدمة بين هذه الفئة العمرية.
- بناءً على المستخدم النهائي، يُقسّم سوق اضطراب ما بعد الصدمة (PTSD) في ألمانيا إلى مستشفيات، وعيادات خارجية، ومراكز صحة نفسية، وخدمات رعاية منزلية، وغيرها. تهيمن مراكز الصحة النفسية على شريحة المستخدمين النهائيين في سوق اضطراب ما بعد الصدمة في ألمانيا، بمعدل نمو سنوي مركب قدره 5.5% خلال الفترة المتوقعة 2023-2030، وذلك بفضل ارتباطها المباشر بمرافق الرعاية الصحية. وبصفتها المصدر الرئيسي لأدوية علاج اضطراب ما بعد الصدمة، تضمن هذه المراكز سرعة الوصول إليها وتوفير إرشادات متخصصة، مما يؤدي إلى توزيع فعال ومناسب للأدوية على المرضى.
- بناءً على قنوات التوزيع، يُقسّم سوق اضطراب ما بعد الصدمة (PTSD) في ألمانيا إلى صيدليات المستشفيات، وصيدليات التجزئة، والصيدليات الإلكترونية. تهيمن صيدليات المستشفيات على قطاع قنوات التوزيع في سوق اضطراب ما بعد الصدمة (PTSD) في ألمانيا بمعدل نمو سنوي مركب قدره 4.8% خلال الفترة المتوقعة 2023-2030، وذلك بفضل الرعاية الشاملة التي تقدمها للأفراد المصابين بهذا الاضطراب. وتُمكّن خبرتها ومواردها وتركيزها على الصحة النفسية من وضع خطط علاج مُصممة خصيصًا ودعم مُستهدف، مما يضمن إدارة فعّالة وتعافيًا فعالًا للمرضى.
اللاعبون الرئيسيون
تعترف شركة Data Bridge Market Research بالشركات التالية باعتبارها اللاعبين الرئيسيين في سوق اضطراب ما بعد الصدمة (PTSD) في ألمانيا: AbbVie Inc. (الولايات المتحدة)، Viatris Inc. (الولايات المتحدة)، Pfizer Inc. (الولايات المتحدة)، Eli Lilly and Company (الولايات المتحدة)، Takeda Pharmaceutical Company Limited (اليابان).
تطورات السوق
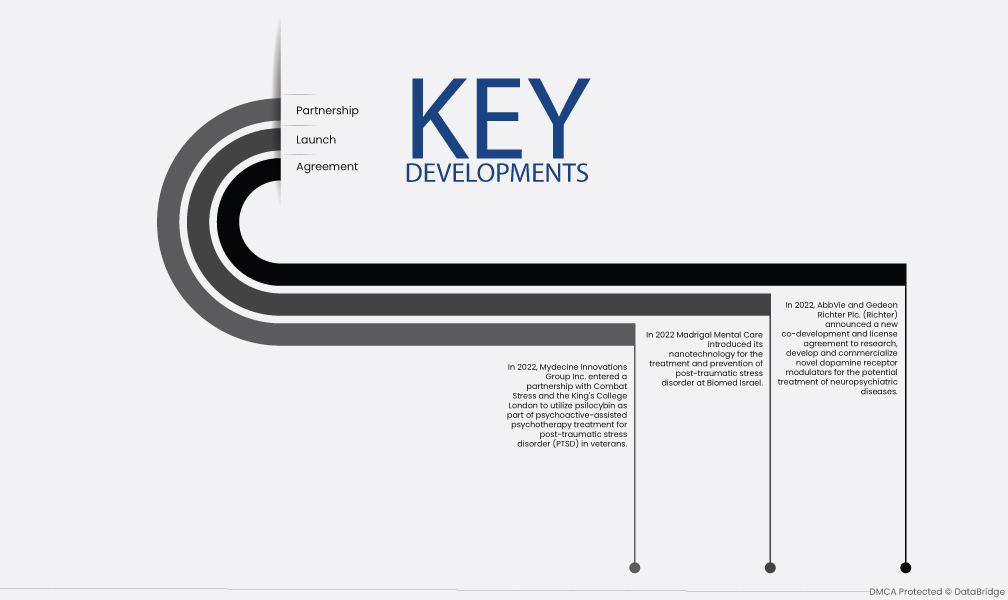
- في عام 2022، أعلنت شركة AbbVie وشركة Gedeon Richter Plc. (Richter) عن اتفاقية جديدة للتطوير المشترك والترخيص للبحث والتطوير وتسويق منظمات مستقبلات الدوبامين الجديدة لعلاج الأمراض العصبية والنفسية المحتملة.
- في عام 2022، قدمت Madrigal Mental Care تقنيتها النانوية لعلاج والوقاية من اضطراب ما بعد الصدمة في Biomed Israel.
- في عام 2022، دخلت شركة Mydecine Innovations Group Inc. في شراكة مع Combat Stress وKing's College London لاستخدام السيلوسيبين كجزء من العلاج النفسي بمساعدة العلاج النفسي لاضطراب ما بعد الصدمة (PTSD) لدى المحاربين القدامى.
لمزيد من المعلومات التفصيلية حول تقرير سوق اضطراب ما بعد الصدمة في ألمانيا (PTSD) ، انقر هنا - https://www.databridgemarketresearch.com/reports/germany-post-traumatic-stress-disorder-market