O mercado global de comprimidos inteligentes é um setor em rápido crescimento no setor da saúde. As pílulas inteligentes, equipadas com sensores e tecnologia sem fios, oferecem monitorização em tempo real, diagnóstico e administração direcionada de medicamentos. Melhoram a adesão do paciente, permitem uma medicina personalizada e facilitam a monitorização remota do paciente. Com os avanços tecnológicos e a crescente prevalência de doenças crónicas, o mercado é impulsionado por fatores como melhores resultados em saúde, relação custo-benefício e procura por soluções de saúde não invasivas.
Obtenha um relatório de amostra gratuito: https://databridgemarketresearch.com/reports/global-smart-pills-market
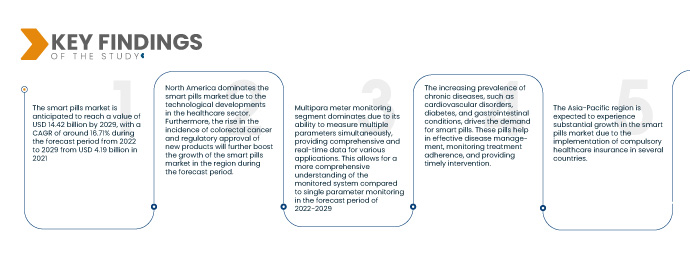
A Data Bridge Market Research analisa que o mercado das pílulas inteligentes deverá crescer a um valor de 14,42 mil milhões de dólares até 2029, com um CAGR de cerca de 16,71% durante o período previsto de 2022 a 2029, de 4,19 mil milhões de dólares em 2021. As pílulas inteligentes permitem a monitorização em tempo real do trato gastrointestinal e fornecem dados valiosos aos profissionais de saúde. Isto permite o atendimento remoto ao paciente, uma vez que os profissionais de saúde podem rever as imagens e os dados captados para tomar diagnósticos e decisões de tratamento precisas. A crescente procura por serviços de monitorização remota e telemedicina está a impulsionar a adoção de comprimidos inteligentes.
Principais conclusões do estudo
O foco na medicina personalizada deverá impulsionar a taxa de crescimento do mercado
As pílulas inteligentes desempenham um papel crucial na viabilização da medicina personalizada, fornecendo dados em tempo real sobre a resposta do doente à medicação. Estes dados ajudam os profissionais de saúde a adaptar os planos de tratamento e a otimizar a terapêutica com base nas necessidades individuais dos doentes. Monitorizando fatores como a absorção do medicamento, a adesão e a resposta fisiológica, as pílulas inteligentes permitem ajustes nas dosagens e nos regimes de tratamento. Esta abordagem personalizada melhora a eficácia do tratamento, minimiza os efeitos secundários e melhora os resultados para os doentes, levando a intervenções de saúde mais precisas e direcionadas.
Âmbito do Relatório e Segmentação de Mercado
Métrica de Reporte
|
Detalhes
|
Período de previsão
|
2022 a 2029
|
Ano base
|
2021
|
Anos Históricos
|
2020 (personalizável para 2014-2019)
|
Unidades quantitativas
|
Receita em milhões de dólares americanos, volumes em unidades, preços em dólares americanos
|
Segmentos abrangidos
|
Tipo (monitorização multiparâmetro, monitorização de parâmetro único), indicação de doença (hemorragia gastrointestinal oculta, doença de Crohn, tumores do intestino delgado, doença celíaca, síndromes de polipose hereditária, perturbações mentais), aplicação (endoscopia por cápsula, administração de medicamentos, monitorização de doentes oncológicos), utilizador final (hospital, centros de diagnóstico, outros)
|
Países abrangidos
|
EUA, Canadá e México na América do Norte, Alemanha, França, Reino Unido, Holanda, Suíça, Bélgica, Rússia, Itália, Espanha, Turquia, Resto da Europa na Europa, China, Japão, Índia, Coreia do Sul, Singapura, Malásia, Austrália, Tailândia, Indonésia, Filipinas, Resto da Ásia-Pacífico (APAC) na Ásia-Pacífico (APAC), Arábia Saudita, Emirados Árabes Unidos, África do Sul, Egito, Israel, Resto do Médio Oriente e África (MEA) como parte do Médio Oriente e África (MEA), Brasil, Argentina e Resto da América do Sul como parte da América do Sul.
|
Participantes do mercado abrangidos
|
ACAMP (Canadá), BDD Ltd (Coreia do Sul), Boston Scientific Corporation (EUA), Cerner Corporation (EUA), FUJIFILM Holdings Corporation (Japão), GENERAL ELECTRIC COMPANY (EUA), MEDTRONIC (Irlanda), Olympus Corporation (Japão), PENTAX Medical (Japão), Proteus Digital Health (EUA), entre outras.
|
Pontos de dados abordados no relatório
|
Para além dos insights sobre os cenários de mercado, tais como o valor de mercado, a taxa de crescimento, a segmentação, a cobertura geográfica e os principais participantes, os relatórios de mercado selecionados pela Data Bridge Market Research incluem também análises aprofundadas de especialistas, epidemiologia dos doentes, análise de pipeline, análise de preços e estrutura regulamentar.
|
Análise de Segmentos:
O mercado das pílulas inteligentes está segmentado com base no tipo, indicação da doença, aplicação e utilizador final
- Com base no tipo, o mercado de comprimidos inteligentes está segmentado em monitorização multiparâmetro e monitorização de parâmetro único. O segmento de monitorização de contadores multiparâmetros domina devido à sua capacidade de medir vários parâmetros em simultâneo, fornecendo dados abrangentes e em tempo real para várias aplicações. Isto permite uma compreensão mais abrangente do sistema monitorizado em comparação com a monitorização de parâmetro único no período previsto de 2022-2029.
- Com base na indicação da doença, o mercado das pílulas inteligentes está segmentado em hemorragia gastrointestinal oculta, doença de Crohn, tumores do intestino delgado, doença celíaca, síndromes de polipose hereditária e perturbações mentais. O sangramento gastrointestinal oculto predomina na indicação da doença devido à sua importância como fonte oculta de hemorragia gastrointestinal que requer um diagnóstico e monitorização precisos. A endoscopia por cápsula é comummente utilizada para detetar e avaliar hemorragias gastrointestinais ocultas, contribuindo para o seu domínio no período previsto de 2022-2029.
- Com base na aplicação, o mercado das pílulas inteligentes está segmentado em endoscopia por cápsula , administração de medicamentos e monitorização de doentes oncológicos. O segmento da endoscopia por cápsula domina o segmento de aplicação do mercado devido à sua natureza não invasiva e capacidade de fornecer uma visualização abrangente do trato gastrointestinal, auxiliando no diagnóstico e monitorização de vários distúrbios e doenças digestivas no período previsto de 2022-2029.
Em 2022, o segmento da endoscopia por cápsula domina o segmento de aplicação do mercado
Em 2022, o segmento de endoscopia por cápsula domina o segmento de aplicação do mercado devido à sua natureza não invasiva, capacidade de captar imagens detalhadas do trato gastrointestinal e à sua ampla utilização para diagnosticar distúrbios gastrointestinais no período previsto de 2022-2029.
- Com base no utilizador final, o mercado das pílulas inteligentes está segmentado em hospitais, centros de diagnóstico e outros. O segmento hospitalar domina o segmento de utilizadores finais do mercado devido ao seu papel principal na prestação de cuidados de saúde e à sua ampla utilização de ferramentas de diagnóstico de ponta para o atendimento e monitorização de doentes no período previsto de 2022 a 2029.
Em 2022, o segmento hospitalar domina o segmento dos utilizadores finais do mercado
Em 2022, o segmento hospitalar dominará o segmento de utilizadores finais do mercado devido ao seu papel como prestadores de cuidados de saúde primários e à sua ampla utilização de tecnologias avançadas de diagnóstico para fins de monitorização e cuidados ao doente no período previsto de 2022 a 2029.
Principais jogadores
A Data Bridge Market Research reconhece as seguintes empresas como os principais participantes do mercado de comprimidos inteligentes: Cerner Corporation (EUA), FUJIFILM Holdings Corporation (Japão), GENERAL ELECTRIC COMPANY (EUA), MEDTRONIC (Irlanda), Olympus Corporation (Japão), PENTAX Medical (Japão)
Desenvolvimento de Mercado
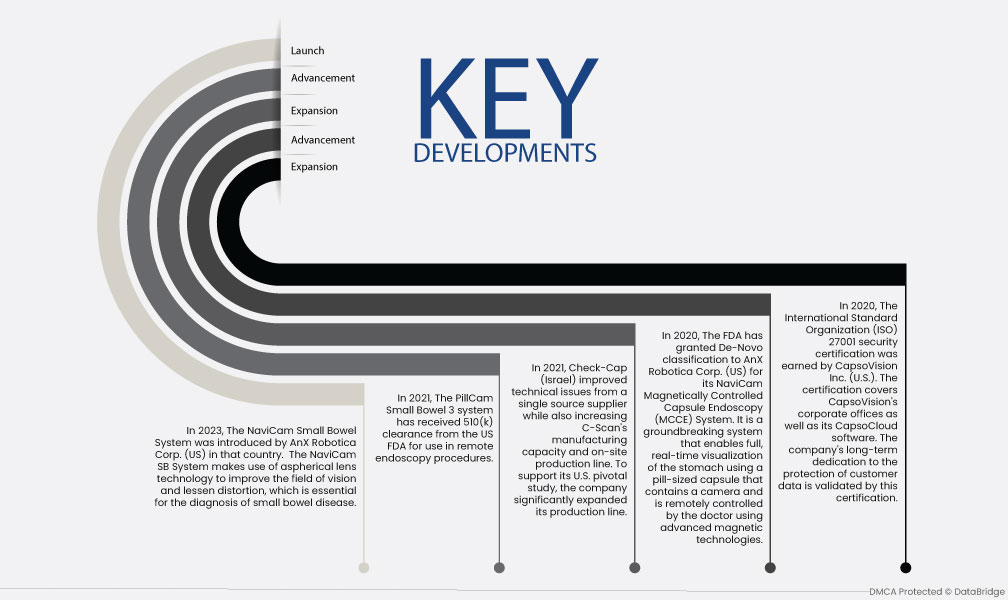
- Em 2023, o sistema NaviCam para o intestino delgado foi introduzido pela AnX Robotica Corp. (EUA) naquele país. O sistema NaviCam SB utiliza a tecnologia de lentes asféricas para melhorar o campo de visão e diminuir a distorção, o que é essencial para o diagnóstico de doenças do intestino delgado.
- Em 2021, o sistema PillCam Small Bowel 3 recebeu a autorização 510(k) da FDA dos EUA para utilização em procedimentos de endoscopia remota.
- Em 2021, a Check-Cap (Israel) melhorou os problemas técnicos de um único fornecedor e, ao mesmo tempo, aumentou a capacidade de fabrico e a linha de produção no local da C-Scan. Para apoiar o seu estudo fundamental nos EUA, a empresa expandiu significativamente a sua linha de produção.
- Em 2020, a FDA concedeu a classificação De-Novo à AnX Robotica Corp. (EUA) para o seu sistema de endoscopia por cápsula controlada magneticamente (MCCE) NaviCam. É um sistema inovador que permite a visualização completa e em tempo real do estômago utilizando uma cápsula do tamanho de um comprimido que contém uma câmara e é controlada remotamente pelo médico através de tecnologias magnéticas avançadas.
- Em 2020, a certificação de segurança ISO 27001 da International Standards Organization foi obtida pela CapsoVision Inc. (EUA). A certificação abrange os escritórios corporativos da CapsoVision, bem como o seu software CapsoCloud. A dedicação a longo prazo da empresa à proteção dos dados dos clientes é validada por esta certificação.
Análise Regional
Geograficamente, os países abrangidos pelo relatório de mercado de comprimidos inteligentes são os EUA, Canadá e México na América do Norte, Alemanha, França, Reino Unido, Holanda, Suíça, Bélgica, Rússia, Itália, Espanha, Turquia, Resto da Europa na Europa, China, Japão, Índia, Coreia do Sul, Singapura, Malásia, Austrália, Tailândia, Indonésia, Filipinas, Resto da Ásia-Pacífico (APAC) na Ásia-Pacífico (APAC), Arábia Saudita, Emirados Árabes Unidos, África do Sul, Egito, Israel, Resto do Médio Oriente e África (MEA) como parte do Médio Oriente e África (MEA), Brasil, Argentina e Resto da América do Sul como parte da América do Sul.
De acordo com a análise de pesquisa de mercado da Data Bridge:
A América do Norte é a região dominante no mercado de comprimidos inteligentes durante o período previsto de 2022 a 2029
O domínio da América do Norte no mercado das pílulas inteligentes pode ser atribuído aos seus desenvolvimentos tecnológicos avançados no setor da saúde. A região testemunhou um progresso significativo no desenvolvimento e adoção de tecnologias de pílulas inteligentes. Além disso, a crescente incidência de cancro colorretal e a aprovação regulamentar de novos produtos de comprimidos inteligentes contribuem para o crescimento do mercado. Estes fatores criam um ambiente favorável para a expansão do mercado de comprimidos inteligentes na América do Norte durante o período previsto.
Estima-se que a Ásia-Pacífico seja a região com o crescimento mais rápido no mercado de comprimidos inteligentes no período previsto de 2022 a 2029.
Espera-se que a região Ásia-Pacífico experimente um crescimento substancial no mercado das pílulas inteligentes devido à implementação de um seguro de saúde obrigatório em vários países. Com a expansão da cobertura de saúde, mais indivíduos têm acesso a serviços e tratamentos médicos, incluindo comprimidos inteligentes. Este maior acesso a recursos de saúde e cobertura de seguro impulsiona a procura de tecnologias de saúde inovadoras, como as pílulas inteligentes, levando a um crescimento significativo do mercado na região da Ásia-Pacífico.
Para obter informações mais detalhadas sobre o relatório de mercado de comprimidos inteligentes , clique aqui – https://www.databridgemarketresearch.com/reports/global-smart-pills-market