تؤثر طفرات ساركوما كيرستن الجرذية (KRAS) تأثيرًا بالغًا على بدء السرطان وتطوره، إذ تعمل كمحركات رئيسية لنمو الخلايا غير المنضبط، وتجنب موت الخلايا المبرمج، وتعزيز بقاء الورم. تُعطّل مسارات الإشارات المتغيرة المرتبطة بطفرات KRAS آليات التنظيم الخلوي، مما يؤدي إلى تطور بعض أنواع السرطان وتطورها. وبصفته محركًا رئيسيًا للسرطان، يلعب KRAS دورًا حاسمًا في تعزيز تكاثر الخلايا السرطانية المتواصل وتثبيط موت الخلايا المبرمج. إن إدراك الدور المحوري لـ KRAS في هذه العمليات يُبرز أهميته كهدف علاجي.
يمكنك الوصول إلى التقرير الكامل على https://www.databridgemarketresearch.com/reports/north-america-kristen-rat-sarcoma-kras-market
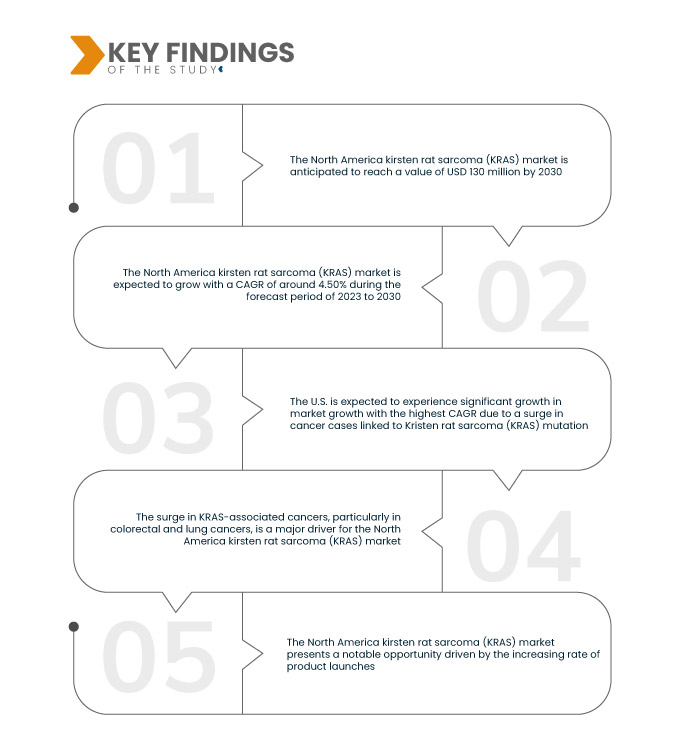
تُحلل شركة داتا بريدج لأبحاث السوق سوق ساركوما فئران كيرستن (KRAS) في أمريكا الشمالية ، والذي بلغ 91.41 مليون دولار أمريكي في عام 2022، ومن المتوقع أن يصل إلى 130 مليون دولار أمريكي بحلول عام 2030، وأن يشهد معدل نمو سنوي مركب قدره 4.50% خلال الفترة المتوقعة من 2023 إلى 2030. ويشهد سوق ساركوما فئران كيرستن (KRAS) في أمريكا الشمالية نموًا بفضل الاستثمارات الضخمة والتقدم المحرز في أنشطة البحث والتطوير، لا سيما في قطاعي الأدوية والتكنولوجيا الحيوية . وتركز هذه المبادرات على كشف التعقيدات الجزيئية لطفرات KRAS، وابتكار تدخلات علاجية دقيقة، مما يعزز الابتكار ويدفع عجلة نمو السوق.
النتائج الرئيسية للدراسة
من المتوقع أن يؤدي تزايد التعاون الاستراتيجي إلى دفع معدل نمو السوق
تُشكل التعاونات الاستراتيجية دافعًا محوريًا في سوق ساركوما كيرستن رات (KRAS) في أمريكا الشمالية، وذلك من خلال تعزيز التآزر بين شركات الأدوية ومؤسسات البحث ومؤسسات الرعاية الصحية. تُمكّن هذه الشراكات من تجميع الموارد والخبرات والبيانات القيّمة بكفاءة، مما يُسرّع من تطوير علاجات فعّالة مُوجّهة لـ KRAS وحلول تشخيصية متقدمة. تُسخّر هذه الجهود التعاونية نقاط القوة الجماعية للتعامل مع تعقيدات أنواع السرطان المرتبطة بـ KRAS، مما يضمن نهجًا أكثر تنسيقًا وتسريعًا للابتكار. تلعب هذه التعاونات دورًا حاسمًا في تشكيل مشهد الأبحاث المُركّزة على KRAS، وتُساهم بشكل كبير في مسار نمو السوق من خلال تعزيز تبادل المعرفة والقدرات المُشتركة.
نطاق التقرير وتقسيم السوق
مقياس التقرير
|
تفاصيل
|
فترة التنبؤ
|
من 2023 إلى 2030
|
سنة الأساس
|
2022
|
السنوات التاريخية
|
2021 (قابلة للتخصيص حتى 2015-2020)
|
الوحدات الكمية
|
الإيرادات بالملايين من الدولارات الأمريكية، والحجم بالوحدات، والتسعير بالدولار الأمريكي
|
القطاعات المغطاة
|
المنتج (الكواشف والمجموعات، أجهزة التحليل، المواد الاستهلاكية)، نوع السرطان (سرطان الرئة، سرطان القولون والمستقيم، سرطان البنكرياس، سرطان الثدي، سرطان البروستاتا، سرطان عنق الرحم، سرطان الجلد، سرطان الكلى، سرطان الدم، سرطان المبيض، وغيرها)، الفئة العمرية (بالغ، أطفال)، التطبيق (تطوير التشخيص الجزيئي، تشخيص الأمراض، اكتشاف الأدوية وتطويرها، الطب الشخصي، التنبؤي، البحث والتطوير، النهج العلاجي/التنبؤات، وغيرها)، المستخدم النهائي (المعاهد الأكاديمية ومعاهد أبحاث السرطان، مختبرات التشخيص، المستشفيات، عيادات الأورام المتخصصة، شركات الأدوية والتكنولوجيا الحيوية، وغيرها)، قناة التوزيع (العطاءات المباشرة، مبيعات التجزئة، موزع تابع لجهة خارجية)
|
الدول المغطاة
|
الولايات المتحدة وكندا والمكسيك في أمريكا الشمالية
|
الجهات الفاعلة في السوق المغطاة
|
BD (الولايات المتحدة)، COSMED srl (إيطاليا)، Abbott (الولايات المتحدة)، F. Hoffmann-La Roche Ltd (سويسرا)، QIAGEN (ألمانيا)، Agilent Technologies Inc. (الولايات المتحدة)، Merck KGaA (ألمانيا)، Carestream Health (الولايات المتحدة)، Illumina, Inc. (الولايات المتحدة)، Thermo Fisher Scientific (الولايات المتحدة)، AbbVie Inc. (الولايات المتحدة)
|
نقاط البيانات التي يغطيها التقرير
|
بالإضافة إلى الرؤى حول سيناريوهات السوق مثل القيمة السوقية ومعدل النمو والتجزئة والتغطية الجغرافية واللاعبين الرئيسيين، فإن تقارير السوق التي تم تنظيمها بواسطة Data Bridge Market Research تتضمن أيضًا تحليلًا متعمقًا من الخبراء وعلم الأوبئة للمرضى وتحليل خطوط الأنابيب وتحليل التسعير والإطار التنظيمي.
|
تحليل القطاعات:
يتم تقسيم سوق ساركوما كيرستن رات (KRAS) في أمريكا الشمالية على أساس المنتج ونوع السرطان والفئة العمرية والتطبيق والمستخدم النهائي وقناة التوزيع.
- على أساس المنتج، يتم تقسيم سوق ساركوما الفئران كيرستن (KRAS) في أمريكا الشمالية إلى الكواشف والمجموعات والمحللات والمواد الاستهلاكية
- على أساس نوع السرطان، يتم تقسيم سوق ساركوما الفئران كيرستن (KRAS) في أمريكا الشمالية إلى سرطان الرئة، وسرطان القولون والمستقيم، وسرطان البنكرياس، وسرطان الثدي، وسرطان البروستاتا، وسرطان عنق الرحم، وسرطان الجلد، وسرطان الكلى، وسرطان الدم، وسرطان المبيض، وغيرها.
- على أساس الفئة العمرية، يتم تقسيم سوق ساركوما كيرستن رات (KRAS) في أمريكا الشمالية إلى البالغين والأطفال
- على أساس التطبيق، يتم تقسيم سوق ساركوما الفئران كيرستن (KRAS) في أمريكا الشمالية إلى تطوير التشخيص الجزيئي، وتشخيص الأمراض، واكتشاف الأدوية وتطويرها، والطب الشخصي، والتنبؤ، والبحث والتطوير، والنهج العلاجي/التنبؤات وغيرها.
- على أساس المستخدم النهائي، يتم تقسيم سوق ساركوما كيرستن رات (KRAS) في أمريكا الشمالية إلى معاهد أكاديمية وأبحاث السرطان، ومختبرات التشخيص، والمستشفيات، وعيادات الأورام المتخصصة، وشركات الأدوية والتكنولوجيا الحيوية وغيرها
- على أساس قناة التوزيع، يتم تقسيم سوق ساركوما كيرستن رات (KRAS) في أمريكا الشمالية إلى عطاءات مباشرة ومبيعات التجزئة وموزعين تابعين لجهات خارجية
اللاعبون الرئيسيون
تعترف شركة Data Bridge Market Research بالشركات التالية باعتبارها اللاعبين الرئيسيين في سوق ساركوما الفئران كيرستن (KRAS) في أمريكا الشمالية: BD (الولايات المتحدة)، COSMED srl (إيطاليا)، Abbott (الولايات المتحدة)، F. Hoffmann-La Roche Ltd (سويسرا)، QIAGEN (ألمانيا)، Agilent Technologies Inc. (الولايات المتحدة)، Merck KGaA (ألمانيا)، Carestream Health (الولايات المتحدة).
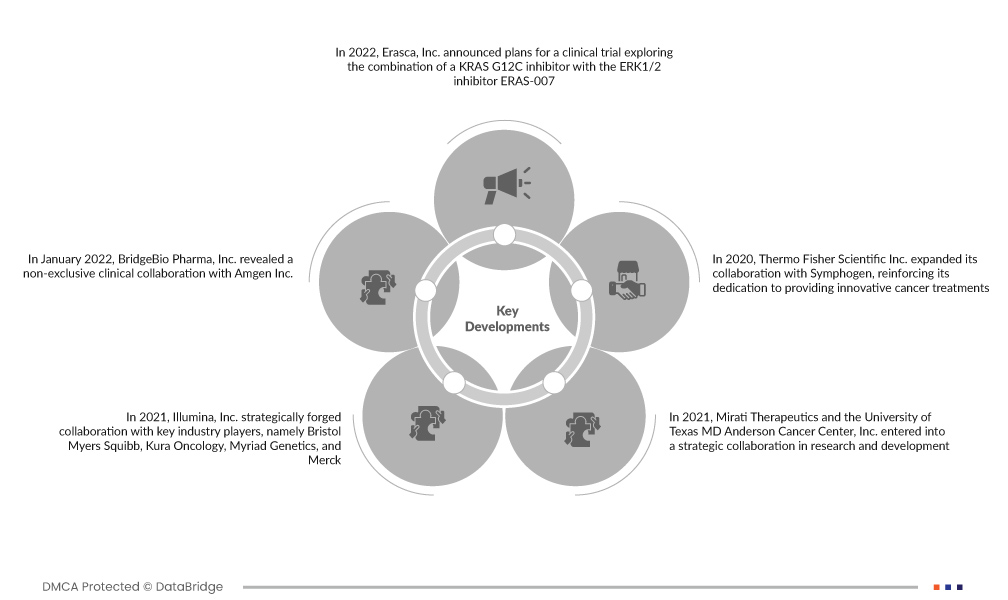
تطورات السوق
- في عام ٢٠٢٢، أعلنت شركة Erasca, Inc. عن خططها لإجراء تجربة سريرية لاستكشاف مزيج مثبط KRAS G12C مع مثبط ERK1/2 ERAS-007. مع التركيز على سرطان الرئة ذو الخلايا غير الصغيرة (NSCLC) وسرطان القولون والمستقيم (CRC) الناتج عن طفرة KRAS G12C، تعكس هذه التجربة نهجًا استراتيجيًا في علم الأورام الدقيق. من خلال استهداف مكونات متعددة من مسار RAS/MAPK، تهدف Erasca إلى تقييم التأثيرات التآزرية المحتملة لهذه المثبطات، مما قد يوسع الخيارات العلاجية لمرضى السرطانات المتحولة إلى KRAS.
- في يناير 2022، كشفت شركة بريدجبيو فارما عن تعاون سريري غير حصري مع شركة أمجين. يهدف هذا التعاون إلى تقييم تركيبة BBP-398، وهو مثبط لـ SHP2، مع LUMAKRAS (سوتوراسيب)، وهو مثبط لـ KRASG12C. تُركز هذه الشراكة على المرضى المصابين بأورام صلبة متقدمة تحمل طفرة KRAS G12C، وتُبرز التزام القطاع باستكشاف العلاجات المركبة. يمكن أن تُمهد هذه التعاونات الطريق لاستراتيجيات علاجية جديدة، وقد تُعزز فعالية مثبطات KRAS الحالية.
- في عام ٢٠٢١، أقامت شركة إيلومينا تعاونًا استراتيجيًا مع جهات فاعلة رئيسية في هذا المجال، وهي بريستول مايرز سكويب، وكورا أونكولوجي، ومايريد جينيتكس، وميرك. مثّل هذا التعاون الاستراتيجي خطوةً محوريةً نحو تطوير أدواتٍ معياريةٍ دقيقةٍ لعلم الأورام. هدفت إيلومينا إلى إحداث ثورةٍ في التنميط الجيني، والمساهمة في علاجاتٍ أكثر فعاليةً ومصممةً خصيصًا للسرطان من خلال تجميع الموارد والخبرات. كان من المتوقع أن يُسهم التآزر الناتج عن هذه الشراكات في رفع دقة التحليل الجيني، وتهيئة إيلومينا لنموٍّ سوقيٍّ كبيرٍ في مجال الطب الدقيق المتطور.
- في عام ٢٠٢١، دخلت شركة ميراتي ثيرابيوتكس ومركز إم دي أندرسون للسرطان بجامعة تكساس في تعاون استراتيجي في مجال البحث والتطوير. يهدف هذا التعاون إلى توسيع نطاق تقييم مثبطات KRAS التجريبية التي تنتجها ميراتي، وهي أداجراسيب (MRTX849) وMRTX1133. يُبرز هذا التقييم، سواءً كعلاج وحيد أو بالاشتراك مع عوامل أخرى، أهمية اتباع مناهج شاملة لعلاج سرطانات KRAS المتحولة. يُركز هذا التعاون على استكشاف طفرات KRAS المتعددة، مما يُظهر التزامًا بمعالجة التباين في سوق KRAS.
- في عام ٢٠٢٠، وسّعت شركة Thermo Fisher Scientific Inc. تعاونها مع شركة Symphogen، معززةً التزامها بتوفير علاجات مبتكرة للسرطان. ركّز هذا التعاون الموسّع على تحسين سير عمل توصيف البروتينات ومراقبة جودة مطياف الكتلة. مكّن هذا التعاون الشركتين من تقديم حلول مطياف كتلة متوافقة مع المعايير لصناعة الأدوية الحيوية، مما لعب دورًا محوريًا في تطوير تقنيات توصيل الأدوية. ومن خلال دمج خبراتهما، سعت Thermo Fisher Scientific وSymphogen إلى المساهمة بشكل كبير في تحسين منهجيات علاج السرطان، وتزويد الصناعة بأدوات متطورة لتوصيل الأدوية بكفاءة.
التحليل الإقليمي
من الناحية الجغرافية، البلدان التي يغطيها تقرير سوق ساركوما كيرستن رات (KRAS) في أمريكا الشمالية هي الولايات المتحدة وكندا والمكسيك في أمريكا الشمالية.
وفقًا لتحليل Data Bridge Market Research:
من المتوقع أن تكون الولايات المتحدة أسرع دولة نموًا في سوق ساركوما كيرستن رات (KRAS) في أمريكا الشمالية خلال الفترة المتوقعة 2023-2030
من المتوقع أن تشهد الولايات المتحدة نموًا كبيرًا في السوق، يتميز بأعلى معدل نمو سنوي مركب (CAGR) خلال الفترة المتوقعة، ويُعزى ذلك في المقام الأول إلى تزايد حالات الإصابة بالسرطان المرتبط بطفرة ساركوما كريستين رات (KRAS). وقد جعل هذا الانتشار من KRAS محورًا رئيسيًا للبحث والتطوير العلاجي. كما تُسهم الاستثمارات الكبيرة التي تُخصصها الدولة للبحث والتطوير في قطاعي الرعاية الصحية والأدوية الحيوية في تعزيز دورها الريادي في تطوير أبحاث السرطان. إضافةً إلى ذلك، يُمثل الانتشار السريع للمنتجات الجديدة في السوق الأمريكية، والتي تُعالج التحديات التي تُمثلها طفرات KRAS، دافعًا رئيسيًا للنمو.
لمزيد من المعلومات التفصيلية حول تقرير سوق ساركوما كيرستن رات (KRAS) في أمريكا الشمالية ، انقر هنا - https://www.databridgemarketresearch.com/reports/global-kirsten-rat-sarcoma-kras-market