The psychedelic drugs market is experiencing a resurgence in interest and applications, primarily in the field of mental health and therapy. Psychedelics, such as psilocybin and MDMA, are being explored for their potential to treat depression, anxiety, PTSD, and addiction. The dominating segment within this market is psychedelic-assisted therapy, where these substances are used in conjunction with therapeutic practices to address various mental health conditions. This holistic approach shows promise in revolutionizing mental healthcare and promoting overall well-being.
Access Full Report at https://www.databridgemarketresearch.com/reports/us-psychedelic-drugs-market
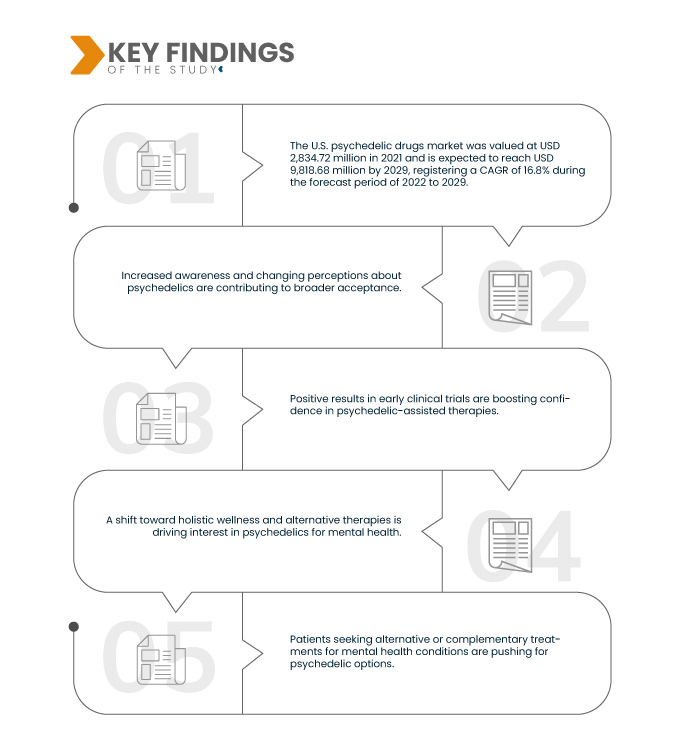
Data Bridge Market Research analyses that the U.S. Psychedelic Drugs Market was valued at USD 2,834.72 million in 2021 and is expected to reach USD 9,818.68 million by 2029, registering a CAGR of 16.8% during the forecast period of 2022 to 2029. The rising incidence of mental health conditions such as depression and PTSD has intensified the demand for innovative treatment approaches. Conventional treatments often have limitations, prompting the search for novel, more effective options, such as psychedelic-assisted therapies, to address the growing mental health crisis.
Key Findings of the Study
Regulatory changes are expected to drive the market's growth rate
Evolving regulations signify a notable shift in how psychedelic substances are perceived and studied. These changes enable researchers and scientists to conduct more extensive and rigorous clinical trials involving psychedelics. The relaxation of restrictions has provided an opportunity to explore the therapeutic potential of these substances in treating mental health disorders. This regulatory shift acknowledges the need for innovative solutions and encourages scientific exploration to unlock the promising benefits of psychedelics within a controlled and safe framework.
Report Scope and Market Segmentation
Report Metric
|
Details
|
Forecast Period
|
2022 to 2029
|
Base Year
|
2021
|
Historic Years
|
2020 (Customizable to 2014-2019)
|
Quantitative Units
|
Revenue in USD Million, Volumes in Units, Pricing in USD
|
Segments Covered
|
Source (Synthetic, Natural), Type (Empathogens, Dissociatives, Others), Drugs (Gamma-Hydroxybutyric Acid, Ketamine, Psilocybin, Others), Application (Narcolepsy, Treatment Resistant Depression, Major Depressive Disorder, Opiate Addiction, Post-Traumatic Stress Disorder, Others), Route of Administration (Oral, Inhalation, Injectable), End User (Hospitals, Specialty Clinics, Homecare, Others), Distribution Channel (Hospitals Pharmacy, Retail Pharmacy, Online Pharmacy)
|
Countries Covered
|
U.S., Canada and Mexico in North America.
|
Market Players Covered
|
Avadel (Ireland), Celon Pharma S.A. (Poland), Johnson & Johnson Private Limited (U.S.), Hikma Pharmaceuticals PLC (U.K.), Amneal Pharmaceuticals LLC. (U.S.), NeuroRx, Inc. (U.S.), Jazz Pharmaceuticals, Inc. (Ireland), COMPASS (U.K.), Develco Pharma Schweiz AG (Switzerland), Douglas Pharmaceuticals Limited (New Zealand)
|
Data Points Covered in the Report
|
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
|
Segment Analysis:
The U.S. psychedelic drugs market is segmented on the basis of source, type, drugs, application, route of administration, end user, and distribution channel.
- On the basis of source, the U.S. psychedelic drugs market is segmented into synthetic, and natural.
- On the basis of type, the U.S. psychedelic drugs market is segmented into empathogens, dissociatives, and others.
- On the basis drugs, the U.S. psychedelic drugs market is segmented into gamma-hydroxybutyric acid, ketamine, psilocybin, and others.
- On the basis of application, the U.S. psychedelic drugs market is segmented into narcolepsy, treatment resistant depression, major depressive disorder, opiate addiction, post-traumatic stress disorder, and others.
- On the basis of route of administration, the U.S. psychedelic drugs market is segmented into oral, inhalation, and injectable.
- On the basis of end user, the U.S. psychedelic drugs market is segmented into hospitals, specialty clinics, homecare, and others.
- On the basis of distribution channel, the U.S. psychedelic drugs market is segmented into hospitals pharmacy, retail pharmacy, and online pharmacy.
Major Players
Data Bridge Market Research recognizes the following companies as the major U.S. psychedelic drugs market players in U.S. psychedelic drugs market are Avadel (Ireland), Celon Pharma S.A. (Poland), Johnson & Johnson Private Limited (U.S.), Hikma Pharmaceuticals PLC (U.K.), Amneal Pharmaceuticals LLC. (U.S.), NeuroRx, Inc. (U.S.), Jazz Pharmaceuticals, Inc. (Ireland), COMPASS (U.K.).
Market Developments
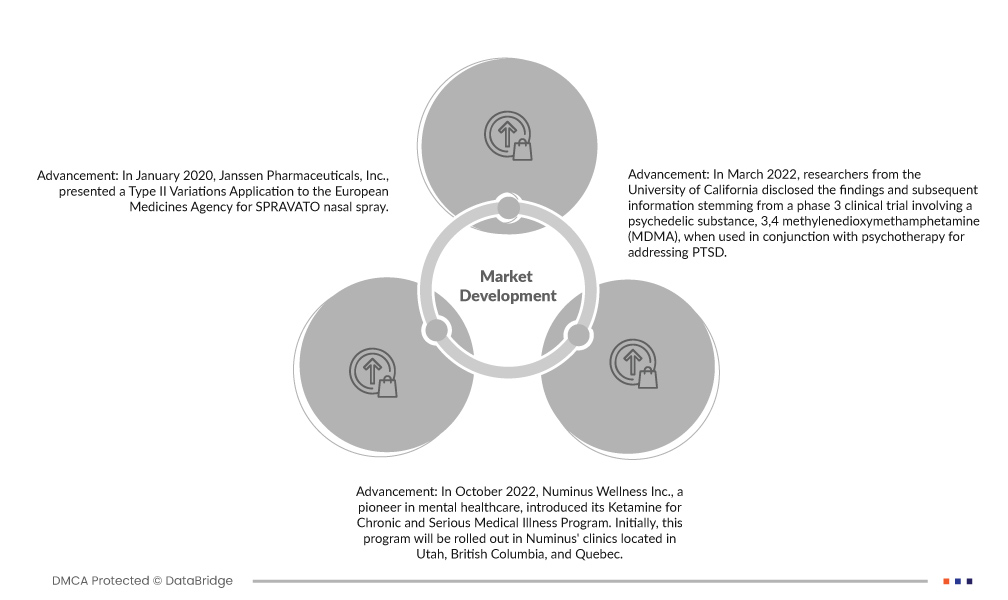
- In January 2020, Janssen Pharmaceuticals, Inc., presented a Type II Variations Application to the European Medicines Agency for SPRAVATO nasal spray. The application seeks to expand the utilization of the nasal spray as a short-term, acute therapy for managing depression symptoms across a broader range of patients. This strategic move aims to enhance the product's market reach and potentially boost revenue for the company.
- In October 2022, Numinus Wellness Inc., a pioneer in mental healthcare, introduced its Ketamine for Chronic and Serious Medical Illness Program. Initially, this program will be rolled out in Numinus' clinics located in Utah, British Columbia, and Quebec. The company has plans to progressively extend the program to additional clinics in the coming months, reflecting its commitment to advancing innovative, evidence-based psychedelic-assisted therapies for individuals with chronic and severe medical conditions.
- In March 2022, researchers from the University of California disclosed the findings and subsequent information stemming from a phase 3 clinical trial involving a psychedelic substance, 3,4 methylenedioxymethamphetamine (MDMA), when used in conjunction with psychotherapy for addressing PTSD. Their initial data indicated the therapy's efficacy, even in challenging cases, including patients with a history of drug or alcohol use disorders. This suggests promising potential for treating PTSD with this combined approach.
For more detailed information about the U.S. psychedelic drugs market report, click here – https://www.databridgemarketresearch.com/reports/us-psychedelic-drugs-market