Global Rare Protein Misfolding Disease Drug Market
Market Size in USD Million
CAGR :
% 
 USD
842.00 Million
USD
3,644.65 Million
2025
2033
USD
842.00 Million
USD
3,644.65 Million
2025
2033
| 2026 –2033 | |
| USD 842.00 Million | |
| USD 3,644.65 Million | |
|
|
|
|
Rare Protein Misfolding Disease Drug Market Size
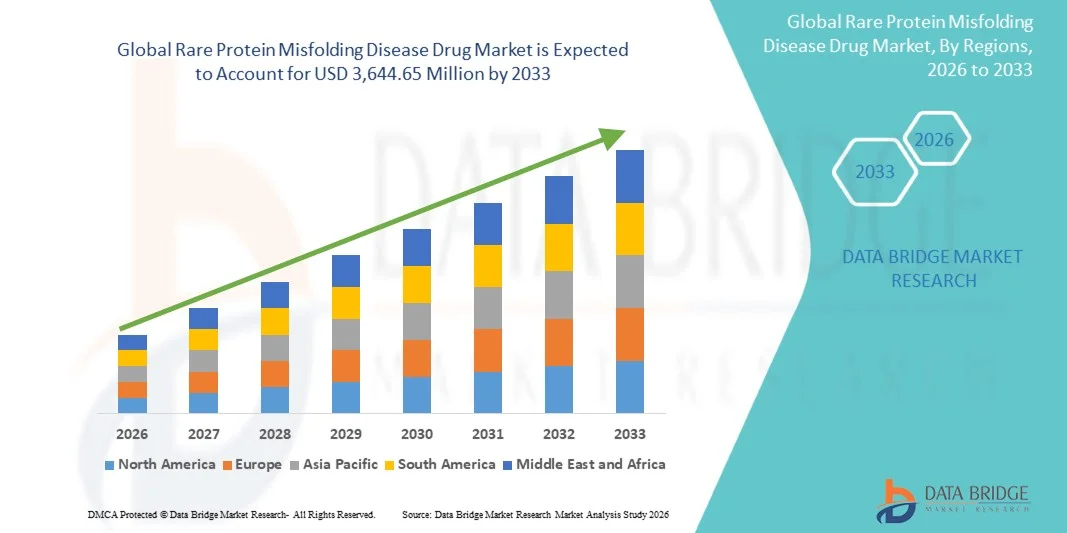
- The global rare protein misfolding disease drug market size was valued at USD 842.00 million in 2025and is expected to reach USD 3,644.65 million by 2033, at a CAGR of 20.10% during the forecast period
- The market growth is primarily driven by the rising prevalence of neurodegenerative disorders such as Alzheimer’s, Parkinson’s, ALS, and Huntington’s disease, which are strongly linked to abnormal protein folding and aggregation mechanisms
- Furthermore, increasing advancements in targeted biologics, gene therapies, and RNA-based therapeutics, along with growing R&D investments in neurodegenerative disease research, are accelerating drug development efforts
Rare Protein Misfolding Disease Drug Market Analysis
- Rare protein misfolding disease drug therapies, targeting abnormal protein aggregation in neurodegenerative disorders, are increasingly becoming a critical component of modern precision medicine due to their potential to modify disease progression in conditions such as Alzheimer’s, Parkinson’s, ALS, Huntington’s disease, and other rare proteinopathies
- The escalating demand for these therapies is primarily driven by the rising global prevalence of neurodegenerative diseases, improved understanding of protein misfolding pathways, and a growing shift toward disease-modifying biologics, gene therapies, and RNA-based treatment approaches
- North America dominated the rare protein misfolding disease drug market with the largest revenue share of 42.6% in 2025, characterized by strong biotech R&D infrastructure, high healthcare spending, and early adoption of advanced therapies, with the U.S. leading in clinical trials and regulatory approvals for novel biologics targeting amyloid and synuclein-related disorders
- Asia-Pacific is expected to be the fastest growing region in the rare protein misfolding disease drug market during the forecast period due to increasing aging population, expanding healthcare infrastructure, and rising investment in neuroscience-focused biotechnology research and drug development
- Monoclonal antibodies segment dominated the rare protein misfolding disease drug market with a market share of 38.9% in 2025, driven by strong clinical pipeline activity and increasing use in targeting misfolded proteins such as amyloid-beta and alpha-synuclein in Alzheimer’s and Parkinson’s disease management
Report Scope and Rare Protein Misfolding Disease Drug Market Segmentation
|
Attributes |
Rare Protein Misfolding Disease Drug Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America · U.S. · Canada · Mexico Europe · Germany · France · U.K. · Netherlands · Switzerland · Belgium · Russia · Italy · Spain · Turkey · Rest of Europe Asia-Pacific · China · Japan · India · South Korea · Singapore · Malaysia · Australia · Thailand · Indonesia · Philippines · Rest of Asia-Pacific Middle East and Africa · Saudi Arabia · U.A.E. · South Africa · Egypt · Israel · Rest of Middle East and Africa South America · Brazil · Argentina · Rest of South America |
|
Key Market Players |
|
|
Market Opportunities |
· Expansion of disease-modifying therapies targeting early-stage neurodegenerative biomarkers · Growing advancements in blood-based and CSF biomarker diagnostics |
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
Rare Protein Misfolding Disease Drug Market Trends
“Advancements in Targeted Biologics and Precision Neurology”
- A significant and accelerating trend in the global rare protein misfolding disease drug market is the growing shift toward targeted biologics and precision medicine approaches aimed at selectively addressing abnormal protein aggregation in neurodegenerative disorders such as Alzheimer’s, Parkinson’s, ALS, and Huntington’s disease
- For instance, monoclonal antibody therapies such as lecanemab and donanemab have demonstrated the ability to target amyloid-beta plaques, highlighting the growing focus on disease-modifying rather than symptomatic treatment strategies
- Advances in RNA-based therapies and antisense oligonucleotides are enabling gene-level intervention to reduce toxic protein production, improve disease control, and slow neurodegeneration progression in rare protein misfolding conditions
- The integration of biomarker-driven diagnostics, including CSF and blood-based assays, is enabling earlier and more accurate detection of protein aggregation disorders, thereby improving patient stratification for clinical trials and treatment response prediction.
- Increasing research focus on protein homeostasis restoration strategies such as molecular chaperones and proteostasis regulators is opening new therapeutic pathways for stabilizing misfolded proteins and preventing aggregation
- This trend toward precision-targeted, mechanism-based therapies is fundamentally reshaping neurodegenerative disease treatment expectations, with companies increasingly investing in highly specific biologics and gene modulation platforms to improve therapeutic outcomes
- The demand for advanced protein misfolding disease drugs is growing rapidly across global markets as healthcare systems increasingly prioritize early intervention, disease modification, and long-term neurological protection strategies
Rare Protein Misfolding Disease Drug Market Dynamics
Driver
“Rising Neurodegenerative Disease Burden and Strong R&D Investments”
- The increasing global burden of neurodegenerative diseases linked to protein misfolding, coupled with aging populations, is a major driver accelerating demand for advanced disease-modifying drug therapies
- For instance, in April 2025, major biopharmaceutical companies expanded late-stage clinical trials targeting amyloid and tau proteins, reflecting intensified efforts to develop effective treatments for Alzheimer’s disease
- Growing understanding of protein misfolding mechanisms and disease pathways is encouraging pharmaceutical companies to invest heavily in biologics, gene therapies, and RNA-based drug development programs
- Furthermore, rising funding from governments and research organizations is supporting accelerated drug discovery initiatives focused on rare neurodegenerative and protein aggregation disorders
- Increasing collaboration between biotech firms and academic research institutes is further accelerating innovation and enabling faster translation of laboratory discoveries into clinical applications
- Expanding application of artificial intelligence and computational drug discovery tools is improving target identification and reducing early-stage research timelines in neurodegenerative disease drug development
- Growing pharmaceutical partnerships and licensing agreements are strengthening pipeline development and expanding global access to innovative therapies
- Rising awareness and early diagnosis initiatives are increasing patient identification rates, thereby expanding the addressable treatment population for these therapies
Restraint/Challenge
“High Development Complexity and Clinical Trial Failures”
- The high complexity of targeting protein misfolding pathways and limited understanding of disease progression mechanisms pose significant challenges to successful drug development in this market
- For instance, multiple late-stage clinical failures in Alzheimer’s and Parkinson’s disease drug candidates have highlighted the difficulty in achieving consistent therapeutic efficacy in protein aggregation disorders
- Blood-brain barrier penetration limitations significantly restrict drug delivery efficiency, reducing the effectiveness of many biologics and small molecules targeting central nervous system proteins
- In addition, extremely high R&D costs and long development timelines create financial pressure on companies, especially for rare disease indications with smaller patient populations
- Lack of reliable early-stage clinical endpoints makes it difficult to measure treatment success, slowing regulatory approvals and increasing uncertainty in clinical validation processes
- Stringent regulatory requirements for demonstrating clear clinical benefit in neurodegenerative diseases further delay approvals and increase overall development risk for pharmaceutical firms
- Limited patient pool availability for rare conditions complicates clinical trial recruitment and prolongs study durations
- High treatment costs and reimbursement challenges can restrict patient access, particularly in low- and middle-income regions
Rare Protein Misfolding Disease Drug Market Scope
The market is segmented on the basis of drug type, target protein, disease indication, and end user.
- By Drug Type
On the basis of drug type, the market is segmented into small molecules, monoclonal antibodies, gene therapies, antisense oligonucleotide therapies, and protein stabilizers. The monoclonal antibodies segment dominated the market with the largest revenue share of 38.9% in 2025, driven by their strong clinical focus on targeting misfolded proteins such as amyloid-beta and alpha-synuclein. These therapies are increasingly being adopted due to their disease-modifying potential, particularly in Alzheimer’s and Parkinson’s disease. Pharmaceutical companies are heavily investing in antibody-based pipelines owing to their specificity and ability to clear toxic protein aggregates. In addition, regulatory approvals and late-stage clinical successes have strengthened confidence in biologics. Their integration with biomarker-based diagnostics further enhances treatment precision and patient outcomes.
The gene therapies segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by advancements in genetic engineering and viral vector delivery technologies. Gene therapies offer long-term or potentially curative solutions by directly modifying or silencing genes responsible for misfolded protein production. Increasing investment in rare disease research and favorable regulatory pathways are accelerating clinical development in this segment. Moreover, growing success in early-stage trials for neurodegenerative diseases is boosting market confidence. The ability to provide one-time treatment with sustained benefits is a key factor driving adoption. Expanding collaborations between biotech firms and research institutes further support rapid innovation in this space.
- By Target Protein
On the basis of target protein, the market is segmented into amyloid-beta (Aβ), tau protein, alpha-synuclein, huntingtin protein, TDP-43 protein, and others. The amyloid-beta (Aβ) segment dominated the market in 2025 due to its central role in Alzheimer’s disease pathology and the high volume of research and drug development targeting this protein. Aβ-targeting therapies, particularly monoclonal antibodies, have gained significant attention with multiple late-stage clinical trials and approvals. The high prevalence of Alzheimer’s disease globally further contributes to the dominance of this segment. Pharmaceutical companies continue to prioritize Aβ due to its well-established biomarker framework. In addition, advancements in imaging and diagnostic tools have improved patient identification for Aβ-targeted therapies.
The tau protein segment is expected to be the fastest growing during the forecast period, driven by increasing recognition of tau pathology as a critical factor in disease progression. Unlike amyloid-beta, tau accumulation correlates more directly with cognitive decline, making it an attractive therapeutic target. Growing research into tau-targeting antibodies and small molecules is expanding the development pipeline. Furthermore, combination therapies targeting both amyloid and tau are gaining traction. Increasing clinical evidence supporting tau-focused interventions is expected to accelerate market growth.
- By Disease Indication
On the basis of disease indication, the market is segmented into Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, Amyotrophic Lateral Sclerosis (ALS), prion diseases, and other protein misfolding disorders. The Alzheimer’s disease segment dominated the market with the largest share in 2025, driven by its high global prevalence and significant unmet medical need. Extensive research efforts and funding have resulted in a robust pipeline of therapies targeting amyloid-beta and tau proteins. The increasing aging population is further contributing to the rising patient pool. Regulatory approvals of novel biologics have strengthened treatment adoption in this segment. In addition, improved diagnostic techniques are enabling earlier intervention.
The ALS segment is projected to witness the fastest growth rate from 2026 to 2033, driven by advancements in genetic and molecular research. Increasing focus on rare neurodegenerative diseases and the availability of orphan drug incentives are accelerating drug development for ALS. Innovations in antisense oligonucleotide therapies targeting specific genetic mutations are showing promising results. Growing awareness and improved diagnosis rates are expanding the patient population. Furthermore, strong pipeline activity and clinical trial investments are expected to boost growth in this segment.
- By End User
On the basis of end user, the market is segmented into hospitals, specialty clinics, research & academic institutes, and homecare settings. The hospitals segment dominated the market in 2025 due to the availability of advanced diagnostic facilities and specialized treatment infrastructure required for managing complex neurodegenerative diseases. Hospitals are the primary centers for administering biologics and conducting clinical trials. The presence of skilled healthcare professionals and multidisciplinary care teams supports effective disease management. In addition, higher patient inflow and reimbursement support contribute to segment dominance. Increasing adoption of infusion-based therapies further strengthens hospital demand.
The research & academic institutes segment is expected to be the fastest growing during the forecast period, driven by increasing investment in neuroscience research and drug discovery programs. These institutes play a crucial role in early-stage research, biomarker development, and clinical trials. Growing collaborations with pharmaceutical and biotechnology companies are accelerating innovation. Government funding and grants for rare disease research are further supporting growth. In addition, advancements in molecular biology and genomics are enhancing research capabilities. The rising focus on translational research is expected to expand this segment significantly.
Rare Protein Misfolding Disease Drug Market Regional Analysis
- North America dominated the rare protein misfolding disease drug market with the largest revenue share of 42.6% in 2025, characterized by strong biotech R&D infrastructure, high healthcare spending, and early adoption of advanced therapies
- Healthcare providers and patients in the region highly value the availability of advanced biologics, precision therapies, and early diagnostic technologies that enable targeted treatment of protein aggregation disorders
- This widespread adoption is further supported by high healthcare expenditure, strong presence of leading biopharmaceutical companies, robust clinical trial activity, and favorable regulatory frameworks, establishing advanced protein misfolding therapies as a preferred treatment approach across specialized healthcare settings
U.S. Rare Protein Misfolding Disease Drug Market Insight
The U.S. rare protein misfolding disease drug market captured the largest revenue share of 82% in 2025 within North America, fueled by strong biopharmaceutical innovation and extensive clinical research in neurodegenerative diseases. Healthcare providers are increasingly prioritizing advanced biologics and gene-based therapies to address conditions such as Alzheimer’s, Parkinson’s, and ALS. The growing focus on early diagnosis and precision medicine, combined with high adoption of disease-modifying treatments, further propels the market. Moreover, the presence of leading biotech companies, robust regulatory support, and expanding clinical trial pipelines are significantly contributing to the market's expansion.
Europe Rare Protein Misfolding Disease Drug Market Insight
The Europe rare protein misfolding disease drug market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by increasing research funding and the rising burden of neurodegenerative disorders. The region’s strong regulatory framework and emphasis on rare disease treatment are fostering the adoption of advanced therapeutics. European healthcare systems are increasingly focusing on early diagnosis and long-term disease management strategies. The region is witnessing notable growth across hospitals and specialized neurology centers, with innovative therapies being incorporated into treatment protocols and clinical research initiatives.
U.K. Rare Protein Misfolding Disease Drug Market Insight
The U.K. rare protein misfolding disease drug market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by increasing awareness of neurodegenerative diseases and demand for innovative treatment options. In addition, strong government support for life sciences research and funding initiatives are encouraging the development of advanced biologics and gene therapies. The country’s focus on precision medicine and early diagnostic capabilities is expected to stimulate market growth. The expanding network of research institutions and clinical trial centers further supports innovation in this sector.
Germany Rare Protein Misfolding Disease Drug Market Insight
The Germany rare protein misfolding disease drug market is expected to expand at a considerable CAGR during the forecast period, fueled by increasing investment in biotechnology and neuroscience research. Germany’s advanced healthcare infrastructure and strong emphasis on innovation promote the adoption of novel therapeutic approaches. The integration of biomarker-based diagnostics with targeted treatments is becoming increasingly prevalent. In addition, the country’s focus on high-quality healthcare delivery and regulatory efficiency is supporting the uptake of advanced neurodegenerative disease therapies.
Asia-Pacific Rare Protein Misfolding Disease Drug Market Insight
The Asia-Pacific rare protein misfolding disease drug market is poised to grow at the fastest CAGR of 25% during the forecast period of 2026 to 2033, driven by rapidly aging populations and increasing prevalence of neurodegenerative disorders in countries such as China, Japan, and India. The region's growing healthcare investments and improving diagnostic capabilities are driving the adoption of advanced therapies. Furthermore, government initiatives promoting biotechnology innovation and rare disease treatment are accelerating market growth. As Asia-Pacific emerges as a key hub for clinical trials and pharmaceutical manufacturing, accessibility to these therapies is expanding significantly.
Japan Rare Protein Misfolding Disease Drug Market Insight
The Japan rare protein misfolding disease drug market is gaining momentum due to the country’s aging population, advanced healthcare system, and strong focus on neurological disease management. The Japanese market places significant emphasis on early diagnosis and effective treatment of neurodegenerative conditions. The adoption of advanced biologics and gene therapies is increasing, supported by ongoing research and innovation. Moreover, integration of precision medicine approaches and government support for rare disease treatment are fueling growth in both clinical and research settings.
India Rare Protein Misfolding Disease Drug Market Insight
The India rare protein misfolding disease drug market accounted for the largest market revenue share in Asia Pacific in 2025, attributed to the country’s large patient population, increasing awareness of neurological disorders, and improving healthcare infrastructure. India is emerging as a significant market for advanced therapeutics, with growing adoption across hospitals and specialty clinics. The push toward biotechnology development, along with increasing participation in global clinical trials, is driving market expansion. In addition, the availability of cost-effective treatment options and rising investment in research are key factors propelling the market in India.
Rare Protein Misfolding Disease Drug Market Share
The Rare Protein Misfolding Disease Drug industry is primarily led by well-established companies, including:
- Biogen Inc. (U.S.)
- Eisai Co., Ltd. (Japan)
- F. Hoffmann-La Roche Ltd. (Switzerland)
- Novartis AG (Switzerland)
- Pfizer Inc. (U.S.)
- Eli Lilly and Company (U.S.)
- AbbVie Inc. (U.S.)
- Bristol-Myers Squibb Company (U.S.)
- AstraZeneca PLC (U.K.)
- GSK plc (U.K.)
- Sanofi (France)
- Amgen Inc. (U.S.)
- Takeda Pharmaceutical Company Limited (Japan)
- UCB S.A. (Belgium)
- Ionis Pharmaceuticals, Inc. (U.S.)
- Denali Therapeutics Inc. (U.S.)
- Alnylam Pharmaceuticals, Inc. (U.S.)
- Wave Life Sciences Ltd. (Singapore)
- Prothena Corporation plc (Ireland)
What are the Recent Developments in Global Rare Protein Misfolding Disease Drug Market?
- In December 2025, Eisai Co., Ltd. and Biogen Inc. announced that their Alzheimer’s drug LEQEMBI (lecanemab) was included in China’s Commercial Insurance Innovative Drug List, improving patient access and reimbursement pathways for early Alzheimer’s treatment. This development highlights growing global efforts to expand accessibility of disease-modifying therapies targeting amyloid-beta protein aggregation
- In December 2025, South Korea-based ADEL entered a USD 1.04 billion partnership with Sanofi to co-develop ADEL-Y01, an experimental Alzheimer’s drug targeting abnormal tau protein acetylation. This collaboration reflects increasing investment in next-generation protein misfolding therapies with novel mechanisms beyond traditional amyloid targets
- In April 2025, regulatory progress continued for lecanemab across multiple countries, including approvals and expanded access initiatives in global markets, reinforcing its position as a leading monoclonal antibody therapy targeting amyloid-beta in Alzheimer’s disease. This marks a significant step toward broader commercialization of disease-modifying biologics worldwide
- In July 2023, the U.S. Food and Drug Administration granted full traditional approval to Leqembi (lecanemab) for Alzheimer’s disease after confirming clinical benefit, making it one of the first therapies to demonstrate disease-modifying potential by targeting amyloid-beta plaques in the brain
- In May 2023, Eli Lilly reported positive Phase 3 clinical trial results for donanemab, showing significant slowing of cognitive decline in early Alzheimer’s patients by targeting amyloid protein accumulation. This milestone strengthened confidence in monoclonal antibody-based approaches for protein misfolding diseases
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.