Europe Fecal Occult Blood Tests Market Analysis and Size
One of the primary factors driving the growth of the fecal occult blood tests market is the rising cases of colorectal cancer in Europe. The continuing clinical trial research being conducted by several companies for better diagnosis leads to market expansion. The market is also influenced by the development of direct-to-consumer laboratory testing and the easy availability of fecal occult immunological test devices in online stores.
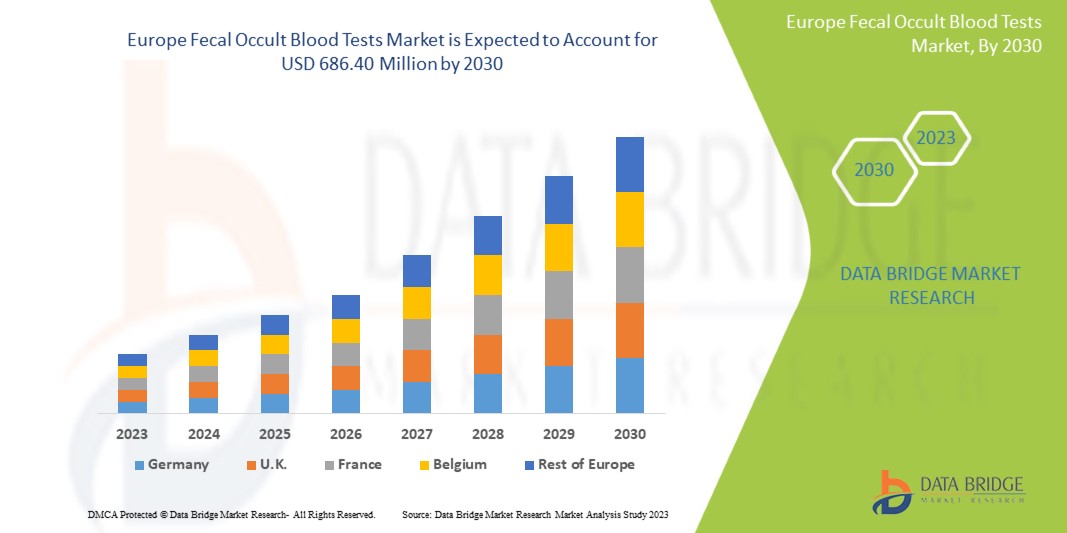
Europe fecal occult blood tests market is expected to gain market growth in the forecast period of 2023 to 2030. Data Bridge Market Research analyses that the market is growing with a CAGR of 5.1% in the forecast period of 2023 to 2030 and is expected to reach USD 686.40 million by 2030.
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customisable to 2015-2020) |
|
Quantitative Units |
Revenue in USD Million |
|
Segments Covered |
By Test Type (Lateral Flow Immuno-Fob Test Kits, Immuno-FOB Agglutination Test Kits, Immuno-FOB ELISA Test Kits, Guaiac FOB Stool Test, Flushable Reagent Pad or Tissue, and Others), Application (Colorectal Cancer Screening, Concern for Gastrointestinal Bleeding, Polyps, Diverticulosis, Ulcers, Haemorrhoids, Colitis, Anemia, and Others), End User (Diagnostic Laboratories, Hospitals, Specialty Clinics, and Others), Distribution Channel (Direct Sales, Retail Sales, E-Commerce, and Others) |
|
Countries Covered |
Russia, Spain, Turkey, Germany, Italy, U.K., France, Belgium, Switzerland, Netherlands, Rest of Europe |
|
Market Players Covered |
Biopanda Reagents Ltd, HUMASIS.COM, CTK Biotech, Inc., Biohit Oyj, CERTEST BIOTEC, Alfa Scientific Designs, Inc., Cenogenics Corporation, and Jant Pharmacal Corporation., Quidel Corporation, Wondfo., Siemens Healthcare GmbH among others |
Market Definition
A fecal occult blood test (FOBT) is a diagnostic screening test performed to analyze hidden (occult) blood in stool samples. The fecal occult blood test is also known as stool occult blood, immunoassay FOBT, hemoccult, and IFOBT and guaiac smear test commonly. The test is majorly utilized to screen colon cancer and analyze possible causes of unexplained anemia.
Fecal Occult Blood Tests Market Dynamics
This section deals with understanding the market drivers, opportunities, restraints, and challenges. All of this is discussed in detail below:
Drivers
- Rising Cases Of Colorectal Cancer in Europe
Colorectal cancer is a disease of the colon or rectum, which are parts of the digestive system. Unlike most cancers, colorectal cancer is often preventable with screening & can be treated when detected early. Most cases of colorectal cancer occur in people ages 45 & older. The disease is increasingly affecting younger people due to unhygienic lifestyles & smoking habits. Colorectal cancers may develop without symptoms. The common symptoms may include blood in or on the stool, persistent unusual bowel movements like constipation or diarrhea, stomach pain, aches or cramps that last longer and losing weight for no reason.
Most colorectal cancers start as abnormal tissue growth, called a polyp, inside the colon or rectum. Doctors can detect polyps with the help of screening tests & can remove them from developing into colorectal cancer. Even if it spreads into nearby lymph nodes, chemotherapy & surgical treatments are effective ways to treat colorectal cancer. Research is going on to learn more about this disease and provide more hope to people with all stages of colorectal cancer.
- Direct-to-consumer Laboratory Testing
Direct-to-consumer laboratory testing is an effective way for users where they can order their corresponding laboratory tests directly from a lab without any healthcare providers. Nowadays, the healthcare industry is shifting towards direct-to-consumer laboratory testing from physician-based lab testing & hence the demand has increased drastically. Most of the patients are focusing on direct-to-consumer based laboratory tests without going through a lengthy in-clinic process, which is fueling the growth of the fecal occult blood tests market.
For the identification of previously unknown medical disorders, the test results are used. The advancements in the field of direct-to-consumer laboratory testing have become crucial to increase patient engagement for better monitoring & screening of existing health conditions. Hence, it is very important for laboratory professionals to upgrade the devices & instruments used in testing laboratories.
-
Easy Availability Of Fecal Occult Immunological Test Devices In Online Stores
A fecal occult blood test (FOBT) generally focuses on a stool sample to check for blood. The presence of blood in stool refers that there is bleeding in the digestive tract. The bleeding can be caused by a variety of conditions like Polyps (abnormal growths on the lining of the colon or rectum), Hemorrhoids (swollen veins in the anus or rectum), Diverticulosis (a condition with small pouches in the inside wall of the colon), Ulcers (sores in the lining of the digestive tract), Colitis (a type of inflammatory bowel disease) and Colorectal cancer.
There are a huge number of fecal occult immunological test devices available in the market, and people can buy the kits & devices from various online sites. Hence, it can be a driving factor for the growth of the fecal occult blood tests market.
Opportunities
-
Increase in screening procedures
Most government agencies and scientific societies recommend cervical, breast, and colorectal screening because of the burden of these cancers, the availability of screening tests, and the proven efficacy of screening in reducing mortality, as well as incidence, for cervical and colorectal cancers.
The European Community recommends the implementation of population-based screening programs for colorectal cancers. This recommendation is supported by many observational studies showing that organized programs effectively reduce mortality and control the inappropriate use of screening tests.
-
Rising technical advancements
A blood test has always been a technology- and data-dependent field. As data and technology potentially revolutionized diagnostic services and had positioned to partner with patients, families, practitioners, researchers, industry leaders, policymakers, and administrators to ensure that humanistic, high-value, ever-improving precise diagnosis remains the central goal for the future of blood test diagnosis.
The use of fecal occult blood testing has expanded to several indications, even though its primary use is for evaluating an occult source of GI blood loss and for home-based kits in colorectal carcinoma screening. One of the most common clinical uses for the hemoccult is for checking a stool specimen for occult bleeding prior to anticoagulation.
Restraints/Challenges
- Change In Consumer Preference Towards Cost-Effective Alternatives
The fecal occult blood test products bear a high cost and usually cannot be afforded by everyone in general. As technology is getting more advanced day by day & there are a number of test alternatives available in the market. Hence, people prefer cost-effective alternatives for fecal occult blood tests. There are various screening programs that often target people with apparently healthy conditions & the number of people using screening services is usually higher than the true number of patients. The FOBT has advantages over colonoscopy in terms of being less costly and easy to perform.
As most of the fecal occult blood test alternatives are available in the market. Hence, people prefer substitutes with lesser cost & more accurate results. Hence, the change in consumer preference toward cost-effective alternatives may restrain the market growth in the forecast period.
- Low efficiency
The fecal occult blood test is not always accurate and could show a negative test result when cancer is present and have a false-negative result if cancer or polyps do not bleed.
The test could show a positive result when the person has no cancer (false-positive result) or have bleeding from other sources, such as a stomach ulcer, hemorrhoid, or even blood swallowed from the mouth or nose.
Post-COVID-19 Impact on the Fecal Occult Blood Tests Market
The COVID-19 pandemic had a great impact on the fecal occult blood test industries. Trade groups in colorectal cancer and gastrointestinal bleeding diagnostic products sector claim that the Europe supply chain for diagnostic products has been significantly damaged, impacting end-user consumption of the fecal occult blood tests market. Sales of test products in the first quarter of 2020 were significantly delayed due to logistical and transportation issues. On the demand side, the market is increasing as people have to get diagnosed after the lockdown scenario. The situation needs to be taken into consideration, and emergency diagnostics need to be done. Furthermore, on the supply side, the growth of the market is on a negative scale. This is due to the lockdown situations in many countries manufacturing diagnostic, operating and cancer care and fecal occult blood test instruments.
Recent Developments
- In November 2022, Eiken Chemical Co, Ltd. had launched the fully automated fecal immunochemical test/ Faecal Calprotectin Analyzer “OC-SENSOR Ceres.” This has helped the company to establish its product Europely
- In January 2022, Boditech Med Inc. announced that it had signed a USD19.2 Million supply agreement with A. Menarini Diagnostics to accelerate its product portfolio. This helps the organization in generating revenue
Europe Fecal Occult Blood Tests Market Scope
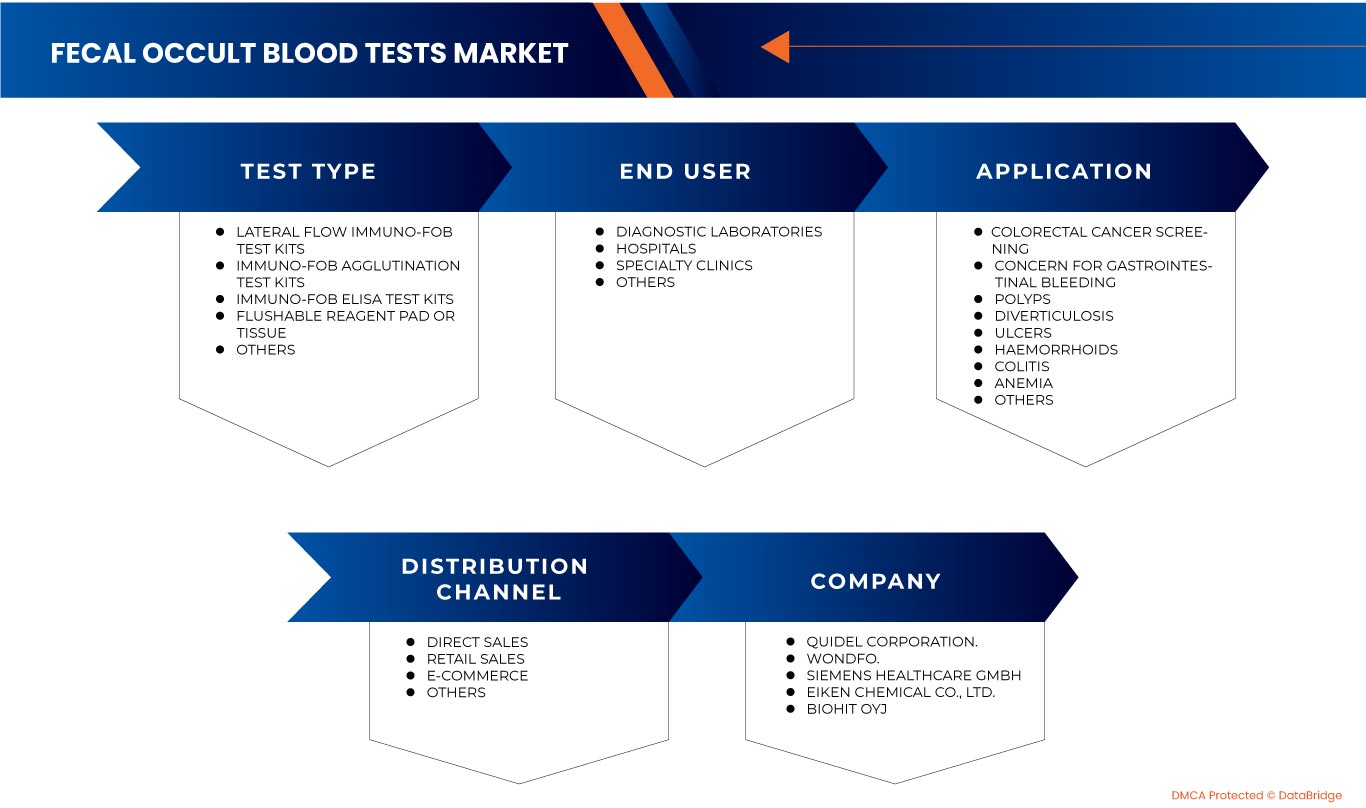
The Europe fecal occult blood tests market is segmented into four notable segments based on test type, application, end user and distribution channel. The growth amongst these segments will help you analyze meager growth segments in the industries and provide the users with a valuable market overview and market insights to make strategic decisions to identify core market applications.
Test Type
- Lateral Flow Immuno-FOB Test Kits
- Immuno-FOB Agglutination Test Kits
- Immuno-FOB ELISA Test Kits
- Guaiac FOB Stool Tests
- Flushable Reagent Pad Or Tissue
- Others
On the basis of test type, the Europe fecal occult blood tests market is segmented into lateral flow immuno-FOB test kits, immuno-FOB agglutination test kits, immuno-FOB ELISA test kits, guaiac FOB stool tests, flushable reagent pad or tissue, and others.
Application
- Colorectal Cancer Screening
- Concern For Gastrointestinal Bleeding
- Polyps
- Diverticulosis
- Ulcers
- Haemorrhoids
- Colitis
- Anemia
- Others
On the basis of application, the Europe fecal occult blood tests market is segmented into Colorectal Cancer Screening, Concern for gastrointestinal bleeding, Polyps, Diverticulosis, Ulcers, Haemorrhoids, Colitis, Anemia, and others.
End User
- Diagnostic Laboratories
- Hospitals
- Specialty Clinics
- Others
On the basis of end user, the Europe fecal occult blood tests market is segmented into diagnostic laboratories, hospitals, specialty clinics, and others.
Distribution Channel
- Direct Tender
- Retail Sales
- E-Commerce
- Others
On the basis of distribution channel, the Europe fecal occult blood tests market is segmented into direct tender, retail sales, e-commerce, and others.
Fecal Occult Blood Tests Market Regional Analysis/Insights
The fecal occult blood tests market is analysed, and market size insights and trends are provided by country, test type, application, end user, distribution channel, as referenced above.
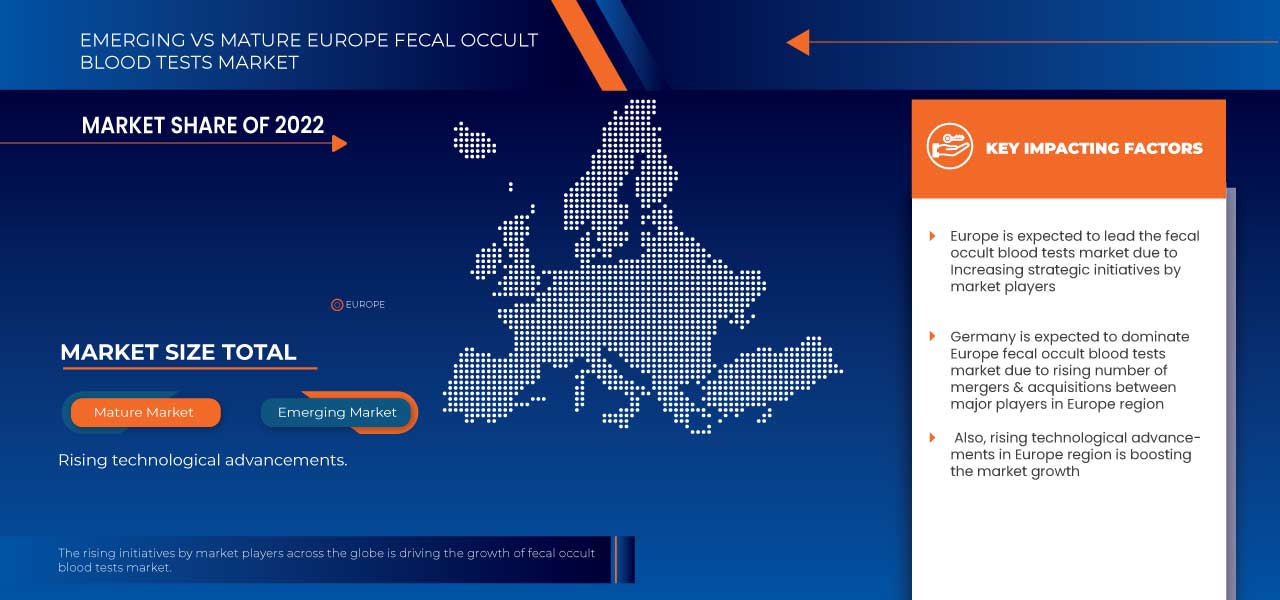
Europe fecal occult blood tests market comprises of the countries Russia, Spain, Turkey, Germany, Italy, U.K., France, Belgium, Switzerland, Netherlands and Rest of Europe.
Germany is expected to dominate the Europe fecal occult blood tests market due to an increasing inclination towards point-of-care diagnostics. An increase in demand for fecal occult blood test products and an increase in research and development activities in the industry are also expected to drive the market in the forecasted period.
The country section of the report also provides individual market impacting factors and changes in regulations in the market that impact the current and future trends of the market. Data points, such as new and replacement sales, country demographics, disease epidemiology, and import-export tariffs, are some of the major pointers used to forecast the market scenario for individual countries. In addition, the presence and availability of Europe brands and their challenges faced due to high competition from local and domestic brands, and impact of sales channels are considered while providing forecast analysis of the country data.
Competitive Landscape and Europe Fecal Occult Blood Tests Market Share Analysis
The fecal occult blood tests market competitive landscape provides details by the competitors. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, Europe presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, and application dominance. The above data points provided are only related to the companies focus on the Europe Fecal occult blood tests market.
Some of the major players operating in this market are Biopanda Reagents Ltd., ulti med Products (Deutschland) GmbH, CERTEST BIOTEC., Alfa Scientific Designs, Inc., Biohit Oyj, Hangzhou Clongene Biotech Co., Ltd., Wondfo., PRIMA Lab SA, Boditech Med Inc., Epitope Diagnostics, Inc., Accuquik Test Kits, Aidian, Apacor Limited, Quidel Corporation., Eurolyser Diagnostica GmbH, Siemens Healthcare GmbH, and Eiken Chemical Co., Ltd., among others.
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.












