 Market Analysis and Insights : Europe Point-Of-Care Diagnostics Market
Market Analysis and Insights : Europe Point-Of-Care Diagnostics Market
The point-of-care diagnostics market is expected to witness market growth at a rate of 10.22% in the forecast period of 2022 to 2029. Data Bridge Market Research report on point-of-care diagnostics market provides analysis and insights regarding the various factors expected to be prevalent throughout the forecast period while providing their impacts on the market’s growth. The rise in the incidence of target diseases is escalating the growth of point-of-care diagnostics market.
Point-Of-Care diagnostics are the approaches that are utilized to identify infections early and quickly. Such procedures are extremely sensitive, cost-efficient and produce results within a short timeframe. These testing methods are utilized by schools, physicians, outpatients, clinical labs and home health.
Major factors that are expected to boost the growth of the point-of-care diagnostics market in the forecast period are the rise in the need for home healthcare has pushed the manufacturers. Furthermore, the growing importance of point-of-care diagnostics in the environmental monitoring and public health is further anticipated to propel the growth of the point-of-care diagnostics market. Moreover, the technological developments and new product releases is further estimated to cushion the growth of the point-of-care diagnostics market. On the other hand, the expenses pressure because of repayment cuts and budget restriction is further projected to impede the growth of the point-of-care diagnostics market in the timeline period.
In addition, the increase in the number of regulatory approvals for modern immunoassay techniques will further provide potential opportunities for the growth of the point-of-care diagnostics market in the coming years. However, the time-consuming agreement policies substantially upsurge the development period for product launches which might further challenge the growth of the point-of-care diagnostics market in the near future.
This point-of-care diagnostics market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, the impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the point-of-care diagnostics market, contact Data Bridge Market Research for an Analyst Brief. Our team will help you take an informed market decision to achieve market growth.
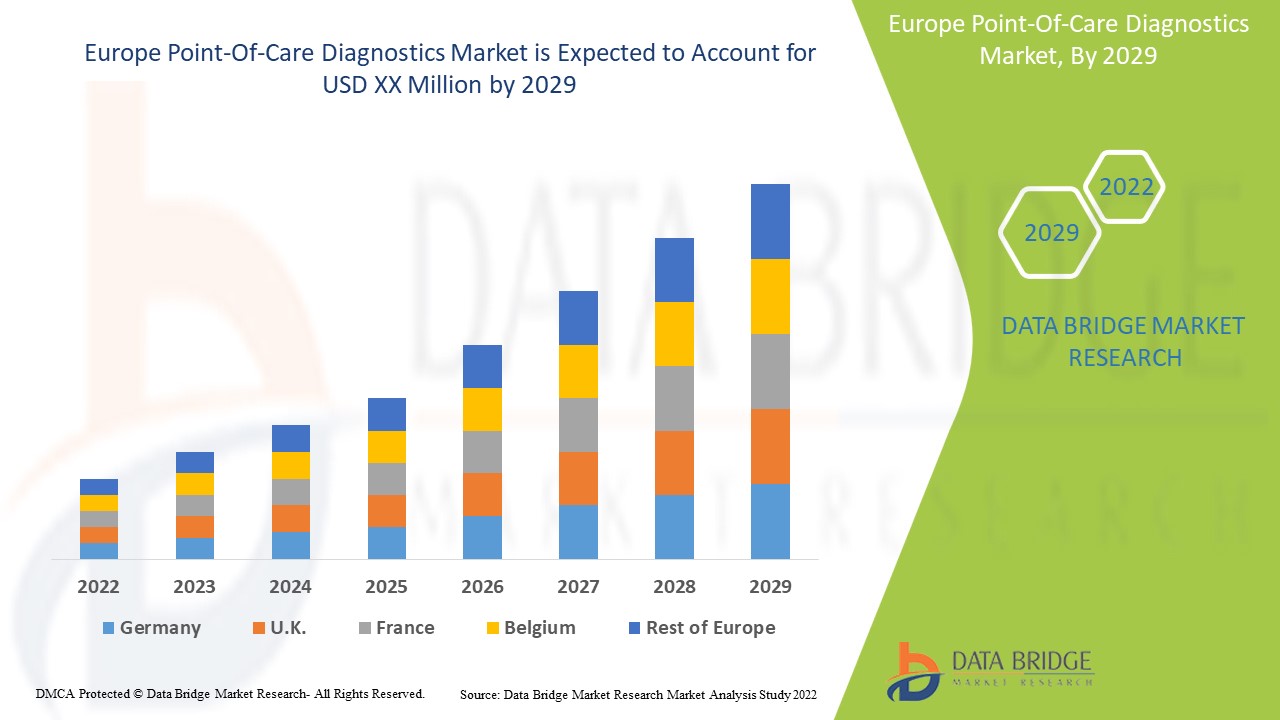
Europe Point-Of-Care Diagnostics Market Scope and Market Size
The point-of-care diagnostics market is segmented on the basis of product, platform, prescription and end user. The growth amongst these segments will help you analyse meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them in making strategic decisions for the identification of core market applications.
- On the basis of product, the point-of-care diagnostics market is segmented into glucose monitoring products, cardio metabolic, infectious disease, coagulation, pregnancy and fertility, tumor or cancer marker, urinalysis, cholesterol, haematology, drugs-of-abuse, fecal occult and others. Glucose monitoring products is further sub segmented into strips, meters and lancets and lancing device. Cardiometabolic is further sub segmented into cardiac marker, blood gas or electrolytes and HBA1C. Infectious disease is further sub segmented into influenza, HIV, hepatitis C, sexually transmitted disease (STD), healthcare-associated infection (HAI), respiratory infection, tropical disease and other infectious disease. Coagulation is further sub segmented into PT or INR and activated clotting time (ACT or APTT). Pregnancy and fertility is further sub segmented into pregnancy test and fertility test.
- On the basis of platform, the point-of-care diagnostics market is segmented into lateral flow assays (immunochromatography tests), dipsticks, microfluidics, molecular diagnostics and immunoassays.
- On the basis of prescription, the point-of-care diagnostics market is segmented into prescription-based testing and OTC testing.
- On the basis of end user, the point-of-care diagnostics market is segmented into clinical laboratories, outpatient healthcare and ambulatory care settings, hospitals or critical care centers, home care, research laboratories and other.
Point-Of-Care Diagnostics Market Country Level Analysis
The point-of-care diagnostics market is analysed, and market size insights and trends are provided by country, product, platform, prescription and end user as referenced above.
The countries covered in the point-of-care diagnostics market report are the Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe.
France is projected to observe significant amount of growth in the point-of-care diagnostics market due to the rise in the prevalence of unhealthy lifestyles and growing number of chronic diseases. Moreover, the augmented FDA approvals and government laws that are connected to healthcare advancement which is further anticipated to propel the growth of the point-of-care diagnostics market in the region in the coming years.
The country section of the point-of-care diagnostics market report also provides individual market impacting factors and changes in regulation in the market domestically that impact the current and future trends of the market. Data points such as consumption volumes, production sites, and volumes, import-export analysis, price trend analysis, cost of raw materials, downstream and upstream value chain analysis are some of the major pointers used to forecast the market scenario for individual countries. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, the impact of domestic tariffs, and trade routes are considered while providing forecast analysis of the country data.
Healthcare Infrastructure Growth Installed base and New Technology Penetration
The point-of-care diagnostics market also provides you with detailed market analysis for every country growth in healthcare expenditure for capital equipment, installed base of different kinds of products for the point-of-care diagnostics market, the impact of technology using lifeline curves, and changes in healthcare regulatory scenarios and their impact on the point-of-care diagnostics market. The data is available for the historic period 2010-2020.
Competitive Landscape and Point-Of-Care Diagnostics Market Share Analysis
The North America point-of-care diagnostics market competitive landscape provides details by a competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies’ focus related to the point-of-care diagnostics market.
The major players covered in the point-of-care diagnostics market report are Abbott, Danaher, BD, F. Hoffmann-La Roche Ltd, Johnson & Johnson Services, Inc., Instrumentation Laboratory S.p.A. – Werfen, bioMérieux SA, Trividia Health, Inc., QIAGEN, Sekisui Diagnostics, Siemens Healthcare GmbH, Quidel Corporation, Spectral Medical Inc., Abaxis, Nova Biomedical, Trinity Biotech, Alfa Scientific Designs, Inc., ?PTS Diagnostics, Bio-Rad Laboratories, Inc., and Medica Corporation among others.
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.













