Global Amyotrophic Lateral Sclerosis Als Market
Market Size in USD Million
CAGR :
% 
 USD
850.00 Million
USD
1,368.12 Million
2025
2033
USD
850.00 Million
USD
1,368.12 Million
2025
2033
| 2026 –2033 | |
| USD 850.00 Million | |
| USD 1,368.12 Million | |
|
|
|
|
Amyotrophic Lateral Sclerosis (ALS) Market Size
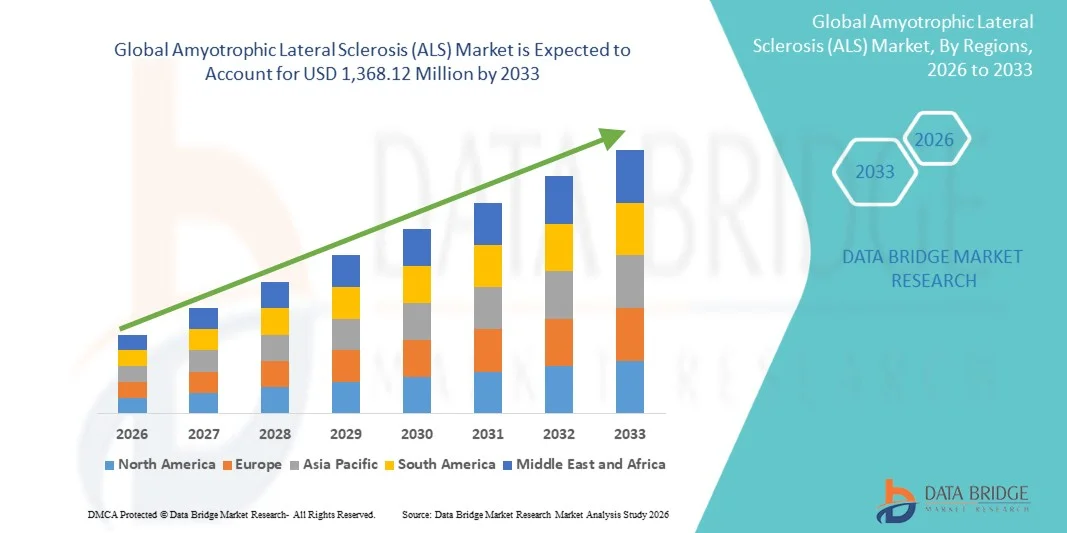
- The global Amyotrophic Lateral Sclerosis (ALS) market size was valued at USD 850.00 million in 2025 and is expected to reach USD 1,368.12 million by 2033, at a CAGR of 6.13% during the forecast period
- The market growth is largely fueled by the increasing prevalence of Amyotrophic Lateral Sclerosis and continuous advancements in therapeutic development, including novel drug formulations, gene therapies, and disease-modifying treatments, leading to improved diagnosis and management in both clinical and homecare settings
- Furthermore, rising patient awareness, growing healthcare expenditure, and strong demand for effective and targeted treatment options are establishing ALS therapies as a critical focus area in rare disease management. These converging factors are accelerating the adoption of advanced treatment solutions, thereby significantly boosting the industry's growth
Amyotrophic Lateral Sclerosis (ALS) Market Analysis
- Amyotrophic Lateral Sclerosis (ALS) market, centered on a progressive neurodegenerative disorder affecting nerve cells responsible for voluntary muscle movement, is becoming an increasingly critical focus area within rare disease therapeutics due to its high unmet medical need, limited treatment options, and significant impact on patient quality of life across both clinical and specialized care settings
- The escalating demand in the Amyotrophic Lateral Sclerosis (ALS) market is primarily fueled by the rising incidence and improved diagnosis of Amyotrophic Lateral Sclerosis, increasing investments in drug development, and a growing emphasis on disease-modifying therapies and supportive care approaches
- North America dominated the Amyotrophic Lateral Sclerosis (ALS) market with the largest revenue share of 42.9% in 2025, characterized by advanced healthcare infrastructure, strong research funding, and the presence of leading biopharmaceutical companies, with the United States experiencing significant growth in ALS treatment adoption, driven by regulatory approvals and ongoing clinical trials
- Asia-Pacific is expected to be the fastest growing region in the Amyotrophic Lateral Sclerosis (ALS) market during the forecast period due to improving healthcare access, increasing awareness of rare diseases, and rising healthcare expenditure across emerging economies
- Medication segment dominated the Amyotrophic Lateral Sclerosis (ALS) market with a market share of 50.2% in 2025, driven by the widespread use of approved pharmacological treatments and increasing focus on therapies aimed at slowing disease progression and managing symptoms effectively
Report Scope and Amyotrophic Lateral Sclerosis (ALS) Market Segmentation
|
Attributes |
Amyotrophic Lateral Sclerosis (ALS) Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
Amyotrophic Lateral Sclerosis (ALS) Market Trends
“Advancements in Disease-Modifying Therapies and Precision Medicine”
- A significant and accelerating trend in the global Amyotrophic Lateral Sclerosis (ALS) market is the growing focus on disease-modifying therapies, gene-based treatments, and precision medicine approaches targeting specific patient subgroups. This evolution in therapeutic strategies is significantly enhancing treatment effectiveness and long-term disease management
- For instance, emerging therapies targeting genetic mutations such as SOD1 and C9orf72 are being actively developed and tested, offering more personalized treatment pathways. Similarly, advances in antisense oligonucleotide therapies are gaining traction as promising interventions in ALS care
- Integration of precision medicine in the Amyotrophic Lateral Sclerosis (ALS) market enables tailored treatment approaches based on genetic profiling and disease progression patterns. For instance, certain therapies are designed to slow neuronal degeneration by targeting specific molecular pathways, while others focus on reducing toxic protein accumulation. Furthermore, innovative drug delivery systems are improving treatment accessibility and patient adherence
- The increasing incorporation of advanced diagnostics and biomarkers facilitates early detection and better monitoring of disease progression. Through integrated healthcare platforms, clinicians can optimize treatment strategies and improve patient outcomes by combining pharmacological and supportive therapies
- This trend towards more targeted, personalized, and technologically advanced treatment solutions is fundamentally reshaping expectations in ALS care. Consequently, companies such as Biogen and Ionis Pharmaceuticals are developing next-generation therapies focusing on genetic targeting and neuroprotection mechanisms
- The demand for advanced and personalized treatment options is growing rapidly across healthcare systems, as stakeholders increasingly prioritize improved survival rates and enhanced quality of life for ALS patients
- Growing adoption of digital health tools and remote patient monitoring solutions is enhancing disease tracking and enabling more efficient long-term management of ALS patients
Amyotrophic Lateral Sclerosis (ALS) Market Dynamics
Driver
“Rising Disease Burden and Increasing Investment in Therapeutic Development”
- The increasing prevalence of neurodegenerative disorders, coupled with rising investments in rare disease research, is a significant driver for the heightened demand in the Amyotrophic Lateral Sclerosis (ALS) market
- For instance, in recent years, multiple pharmaceutical companies have expanded their ALS pipelines, focusing on innovative drug candidates and combination therapies to address unmet medical needs. Such strategies by key companies are expected to drive the Amyotrophic Lateral Sclerosis (ALS) market growth in the forecast period
- As awareness of Amyotrophic Lateral Sclerosis increases and diagnostic capabilities improve, healthcare providers are identifying more cases at earlier stages, leading to greater demand for effective treatment and care solutions
- Furthermore, the growing emphasis on multidisciplinary care approaches, including respiratory and physical therapy, is making ALS management more comprehensive, improving patient survival and quality of life outcomes
- The availability of approved medications, ongoing clinical trials, and supportive government initiatives are key factors propelling the adoption of ALS therapies across major healthcare markets. The expansion of healthcare infrastructure and reimbursement frameworks further contributes to market growth
- Increasing regulatory support and orphan drug designations are encouraging pharmaceutical companies to invest in ALS research and accelerate product approvals
- Rising healthcare expenditure in emerging economies is improving access to ALS diagnosis and treatment, thereby expanding the overall patient treatment base
Restraint/Challenge
“High Treatment Costs and Limited Curative Options”
- Concerns surrounding the high cost of treatment and limited availability of curative therapies pose a significant challenge to broader adoption in the Amyotrophic Lateral Sclerosis (ALS) market. As ALS treatments often involve long-term care and specialized interventions, they create substantial financial burdens for patients and healthcare systems
- For instance, advanced therapies such as gene-based treatments and long-term supportive care can be expensive, limiting accessibility in low- and middle-income regions. High treatment costs may discourage widespread adoption despite clinical benefits
- Addressing these cost-related challenges through improved reimbursement policies, expanded insurance coverage, and pricing strategies is crucial for improving patient access. Companies are increasingly focusing on cost-effective treatment solutions and value-based care models to enhance affordability. In addition, the complexity of ALS diagnosis and variability in disease progression can delay treatment initiation and impact clinical outcomes
- While research efforts are ongoing, the absence of a definitive cure and limited efficacy of existing therapies can hinder patient confidence and treatment uptake. Variability in patient response to therapies further complicates disease management
- Overcoming these challenges through continuous innovation, increased funding for rare disease research, and enhanced patient support programs will be vital for sustained growth in the Amyotrophic Lateral Sclerosis (ALS) market
- Limited patient population and disease rarity restrict large-scale clinical trials, creating challenges in generating robust clinical evidence for new therapies
- Lack of standardized treatment protocols across regions can lead to inconsistencies in patient care and hinder optimal disease management
Amyotrophic Lateral Sclerosis (ALS) Market Scope
The market is segmented on the basis of type, treatment type, diagnosis, symptoms, end-users, and distribution channel.
- By Type
On the basis of type, the Amyotrophic Lateral Sclerosis (ALS) market is segmented into Sporadic ALS and Familial ALS. The Sporadic ALS segment dominated the market with the largest revenue share in 2025, driven by its significantly higher prevalence, accounting for 90% of all Amyotrophic Lateral Sclerosis cases globally. This segment benefits from broader patient pools, leading to higher demand for diagnosis and treatment solutions. Increased awareness, improved diagnostic techniques, and greater clinical focus further support its dominance. In addition, most ongoing clinical trials and approved therapies are primarily targeted toward sporadic ALS, reinforcing its market leadership. The widespread occurrence across diverse populations also contributes to continuous demand for therapeutics and care services.
The Familial ALS segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by increasing advancements in genetic testing and precision medicine. Familial ALS, though rare, is gaining attention due to its strong genetic basis and suitability for targeted therapies. Innovations in gene-editing technologies and mutation-specific treatments are accelerating research in this segment. Growing adoption of genetic counseling and early diagnosis is also supporting market expansion. Furthermore, increasing investment in personalized medicine is expected to drive significant growth in this niche yet rapidly evolving segment.
- By Treatment Type
On the basis of treatment type, the market is segmented into chemotherapy, medication, stem cell therapy, physical therapy, respiratory therapy, speech therapy, and others. The Medication segment dominated the market with the largest revenue share of 50.2% in 2025, driven by the widespread use of approved drugs such as riluzole and edaravone. These pharmacological treatments remain the primary line of management for ALS patients. Continuous advancements in drug formulations and the development of disease-modifying therapies further support this segment’s dominance. The accessibility and regulatory approvals of medications across major markets contribute to their high adoption. In addition, ongoing clinical trials focused on improving drug efficacy continue to strengthen this segment’s position.
The Stem Cell Therapy segment is expected to witness the fastest growth rate from 2026 to 2033, driven by increasing research into regenerative medicine and its potential to repair damaged neurons. Stem cell-based approaches are gaining traction as promising alternatives to conventional therapies. Growing clinical trials and supportive regulatory frameworks are accelerating development in this area. In addition, rising investments from biotechnology firms and research institutions are enhancing innovation. The potential to slow or reverse disease progression makes this segment highly attractive for future growth.
- By Diagnosis
On the basis of diagnosis, the market is segmented into Electromyogram, MRI, Blood and Urine Tests, Spinal Tap, and Muscle Biopsy. The Electromyogram segment dominated the market with the largest revenue share in 2025, driven by its critical role in confirming ALS diagnosis by detecting electrical activity in muscles. It is widely used as a standard diagnostic tool due to its accuracy and reliability. Physicians rely heavily on electromyography to differentiate ALS from other neuromuscular disorders. Its integration into routine diagnostic protocols further strengthens its dominance. In addition, technological advancements in diagnostic equipment are improving efficiency and patient outcomes.
The MRI segment is expected to witness the fastest growth rate from 2026 to 2033, driven by advancements in imaging technologies and increasing use for early and differential diagnosis. MRI helps rule out other neurological conditions, making it a valuable complementary tool. Improved imaging resolution and accessibility are enhancing its adoption across healthcare settings. Growing investments in diagnostic infrastructure are also contributing to its rapid growth. Furthermore, the rising focus on early detection is expected to drive demand for advanced imaging techniques.
- By Symptoms
On the basis of symptoms, the market is segmented into cognitive changes, pain, slurring of speech, fatigue, difficulty breathing and swallowing problems with saliva and mucus, increased clumsiness, cramping and twitching, weakness, difficulty maintaining good posture, and others. The Weakness segment dominated the market with the largest revenue share in 2025, as it is one of the earliest and most common symptoms experienced by ALS patients. This symptom significantly impacts mobility and daily functioning, driving demand for early diagnosis and treatment. Healthcare providers prioritize addressing muscle weakness through therapeutic interventions and supportive care. Its high prevalence across ALS cases reinforces its leading position. In addition, increasing awareness about early symptoms contributes to higher diagnosis rates.
The Difficulty Breathing and Swallowing Problems segment is expected to witness the fastest growth rate from 2026 to 2033, driven by disease progression and the need for advanced supportive care. As ALS advances, respiratory complications become a critical concern requiring specialized interventions. Increasing use of ventilatory support and feeding assistance devices is driving this segment. Growing focus on improving patient quality of life further supports its growth. In addition, advancements in respiratory care technologies are enhancing treatment outcomes.
- By End-Users
On the basis of end-users, the market is segmented into clinic, hospital, and others. The Hospital segment dominated the market with the largest revenue share in 2025, driven by the availability of advanced diagnostic tools, specialized care units, and multidisciplinary treatment approaches. Hospitals serve as primary centers for ALS diagnosis, treatment, and long-term management. The presence of skilled healthcare professionals and access to clinical trials further strengthen this segment. In addition, hospitals are better equipped to handle complex cases and provide comprehensive care. Increasing patient admissions for neurological disorders also contribute to its dominance.
The Clinic segment is expected to witness the fastest growth rate from 2026 to 2033, driven by the rising preference for outpatient care and early-stage diagnosis. Clinics offer accessible and cost-effective treatment options for patients. Increasing establishment of specialized neurology clinics is supporting this growth. In addition, improved referral systems and patient awareness are boosting clinic visits. The shift toward decentralized healthcare delivery models further enhances this segment’s expansion.
- By Distribution Channel
On the basis of distribution channel, the market is segmented into hospital pharmacy, retail pharmacy, and online pharmacy. The Hospital Pharmacy segment dominated the market with the largest revenue share in 2025, driven by the direct dispensing of prescribed ALS medications within hospital settings. Patients undergoing treatment in hospitals rely heavily on in-house pharmacies for medication access. The availability of specialized drugs and professional guidance supports this segment’s dominance. In addition, hospital pharmacies ensure proper storage and handling of sensitive medications. Increasing hospital-based treatments further contribute to its leading position.
The Online Pharmacy segment is expected to witness the fastest growth rate from 2026 to 2033, driven by increasing digitalization and the convenience of home delivery services. Patients are increasingly opting for online platforms to access medications easily. The expansion of e-commerce in healthcare is supporting this trend. In addition, competitive pricing and subscription-based models are attracting more users. Growing internet penetration and digital health adoption are expected to further accelerate this segment’s growth.
Amyotrophic Lateral Sclerosis (ALS) Market Regional Analysis
- North America dominated the Amyotrophic Lateral Sclerosis (ALS) market with the largest revenue share of 42.9% in 2025, characterized by advanced healthcare infrastructure, strong research funding, and the presence of leading biopharmaceutical companies
- Patients and healthcare providers in the region highly prioritize access to advanced treatment options, specialized care centers, and innovative therapies, along with the availability of supportive services such as respiratory and physical therapy
- This widespread adoption is further supported by advanced healthcare infrastructure, favorable reimbursement policies, a strong presence of leading biopharmaceutical companies, and the growing focus on clinical trials and drug development, establishing ALS treatment solutions as a critical component of neurological care across the region
U.S. Amyotrophic Lateral Sclerosis (ALS) Market Insight
The United States Amyotrophic Lateral Sclerosis (ALS) market captured the largest revenue share of 82% in 2025 within North America, fueled by advanced healthcare infrastructure and strong investment in rare disease research. Patients and healthcare providers are increasingly prioritizing early diagnosis and access to innovative treatment options for Amyotrophic Lateral Sclerosis. The growing presence of leading biopharmaceutical companies, combined with robust clinical trial activity and regulatory support, further propels the market. Moreover, increasing adoption of multidisciplinary care approaches and availability of approved therapies are significantly contributing to the market's expansion.
Europe Amyotrophic Lateral Sclerosis (ALS) Market Insight
The Europe Amyotrophic Lateral Sclerosis (ALS) market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by rising awareness of neurodegenerative diseases and increasing healthcare investments. The growth in aging population, coupled with advancements in diagnostic technologies, is fostering the adoption of ALS treatments. European healthcare systems are also focused on improving access to specialized care and rare disease therapies. The region is experiencing steady growth across clinical and homecare settings, with ALS management being integrated into broader neurological care frameworks.
U.K. Amyotrophic Lateral Sclerosis (ALS) Market Insight
The United Kingdom Amyotrophic Lateral Sclerosis (ALS) market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by increasing awareness and improved access to neurological care services. In addition, government support and funding for rare disease research are encouraging early diagnosis and treatment adoption. The country’s strong healthcare infrastructure, alongside active participation in clinical trials, is expected to continue to stimulate market growth.
Germany Amyotrophic Lateral Sclerosis (ALS) Market Insight
The Germany Amyotrophic Lateral Sclerosis (ALS) market is expected to expand at a considerable CAGR during the forecast period, fueled by increasing focus on advanced diagnostics and innovative therapeutic solutions. Germany’s well-developed healthcare system, combined with its emphasis on research and development, promotes the adoption of ALS treatments. The integration of specialized care programs and rehabilitation services is also becoming increasingly prevalent, with a strong preference for high-quality, patient-centric care aligning with local healthcare standards.
Asia-Pacific Amyotrophic Lateral Sclerosis (ALS) Market Insight
The Asia-Pacific Amyotrophic Lateral Sclerosis (ALS) market is poised to grow at the fastest CAGR during the forecast period of 2026 to 2033, driven by improving healthcare infrastructure, rising disposable incomes, and increasing awareness of rare diseases in countries such as China, Japan, and India. The region's growing focus on expanding access to neurological care, supported by government initiatives, is driving the adoption of ALS diagnosis and treatment solutions. Furthermore, as APAC strengthens its clinical research capabilities, the availability and accessibility of ALS therapies are expanding to a wider patient base.
Japan Amyotrophic Lateral Sclerosis (ALS) Market Insight
The Japan Amyotrophic Lateral Sclerosis (ALS) market is gaining momentum due to the country’s advanced healthcare system, aging population, and increasing demand for effective neurological care. The Japanese market places a significant emphasis on early diagnosis and long-term disease management, and the adoption of ALS treatments is driven by the increasing number of diagnosed cases. The integration of advanced diagnostic tools and supportive care solutions is fueling growth. Moreover, Japan's aging demographic is likely to spur demand for comprehensive and accessible ALS treatment options in both clinical and homecare settings.
India Amyotrophic Lateral Sclerosis (ALS) Market Insight
The India Amyotrophic Lateral Sclerosis (ALS) market accounted for a notable market share in Asia Pacific in 2025, attributed to the country's improving healthcare infrastructure, increasing awareness of neurological disorders, and growing access to diagnostic services. India is emerging as a key market for rare disease management, and ALS treatment adoption is gradually increasing across urban healthcare centers. The push towards strengthening healthcare systems and the availability of cost-effective treatment options, alongside rising investments in research, are key factors propelling the market in India.
Amyotrophic Lateral Sclerosis (ALS) Market Share
The Amyotrophic Lateral Sclerosis (ALS) industry is primarily led by well-established companies, including:
- Biogen Inc. (U.S.)
- Ionis Pharmaceuticals, Inc. (U.S.)
- Mitsubishi Tanabe Pharma Corporation (Japan)
- Sanofi (France)
- Novartis AG (Switzerland)
- Amylyx Pharmaceuticals, Inc. (U.S.)
- BrainStorm Cell Therapeutics Inc. (U.S.)
- Aquestive Therapeutics, Inc. (U.S.)
- Otsuka Pharmaceutical Co., Ltd. (Japan)
- Clene Nanomedicine, Inc. (U.S.)
- Genervon Biopharmaceuticals, LLC (U.S.)
- Cerevel Therapeutics Holdings, Inc. (U.S.)
- uniQure N.V. (Netherlands)
- NeuroSense Therapeutics Ltd. (Israel)
- Alchemab Therapeutics Ltd (U.K.)
- Spinogenix, Inc. (U.S.)
- Asha Therapeutics, Inc. (U.S.)
- Cellenkos, Inc. (U.S.)
- Apellis Pharmaceuticals, Inc. (U.S.)
- Annexon Biosciences, Inc. (U.S.)
What are the Recent Developments in Global Amyotrophic Lateral Sclerosis (ALS) Market?
- In May 2024, multiple pharmaceutical manufacturers including Dr. Reddy’s Laboratories and others received U.S. FDA approvals for generic versions of edaravone injection for the treatment of Amyotrophic Lateral Sclerosis (ALS). The introduction of generics is expected to improve affordability and expand patient access to established ALS therapies. This development reflects increasing competition and maturation within the ALS treatment landscape
- In April 2024, Amylyx Pharmaceuticals announced the voluntary withdrawal of Relyvrio from the U.S. and Canadian markets after a Phase 3 clinical trial failed to confirm its efficacy in ALS patients. The study showed no significant improvement in survival or disease progression, prompting the company to halt sales despite earlier approval. This development highlights the challenges in ALS drug development and the importance of robust clinical validation
- In April 2023, Biogen, in collaboration with Ionis Pharmaceuticals, received U.S. FDA approval for Qalsody (tofersen), a novel antisense oligonucleotide therapy for the treatment of SOD1-related Amyotrophic Lateral Sclerosis (ALS). This marked the first gene-targeting therapy for ALS, designed to reduce production of the toxic SOD1 protein. The approval was granted under the accelerated pathway based on biomarker reduction, representing a major advancement in precision medicine for ALS treatment. This development underscores a shift toward genetic and disease-modifying therapies in the ALS market
- In September 2022, Amylyx Pharmaceuticals received FDA approval for Relyvrio (AMX0035), a combination therapy aimed at slowing disease progression in Amyotrophic Lateral Sclerosis (ALS). The drug demonstrated modest clinical benefits in slowing functional decline and extending survival in early trials. This approval was notable as it introduced a new mechanism targeting neuronal cell death pathways, addressing a critical unmet need in ALS therapeutics
- In May 2022, U.S. Food and Drug Administration approved an oral suspension formulation of edaravone (Radicava ORS) for the treatment of Amyotrophic Lateral Sclerosis (ALS), providing a more convenient alternative to intravenous administration. This development significantly improved patient accessibility and adherence to treatment, reducing the burden associated with hospital-based infusions. The approval was based on clinical studies demonstrating comparable safety and efficacy to the IV formulation, enhancing treatment flexibility in ALS care
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.