Global Analytical Standards Market
Market Size in USD Billion
CAGR :
% 
 USD
1.51 Billion
USD
2.46 Billion
2025
2033
USD
1.51 Billion
USD
2.46 Billion
2025
2033
| 2026 –2033 | |
| USD 1.51 Billion | |
| USD 2.46 Billion | |
|
|
|
|
Analytical Standards Market Size
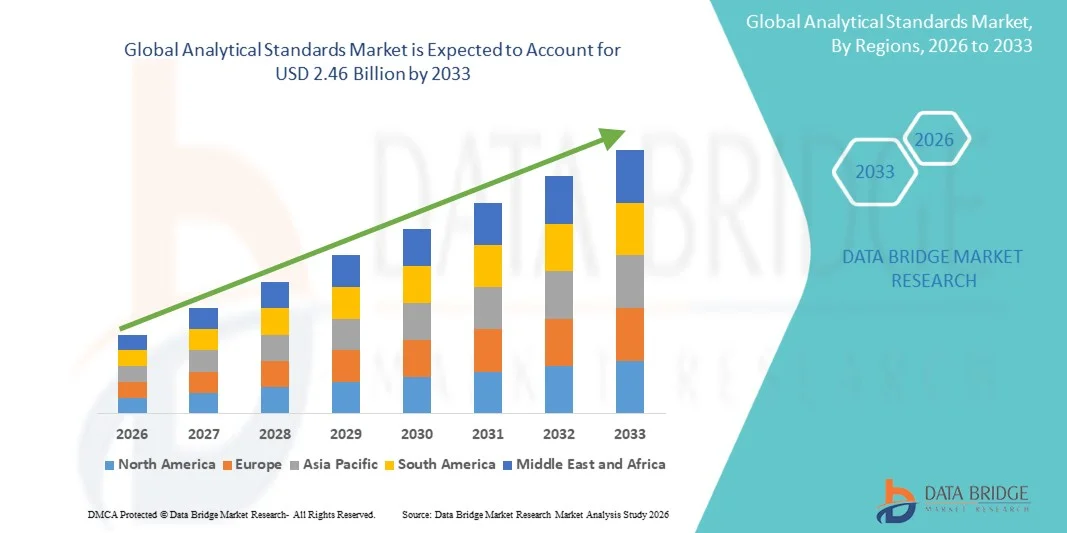
- The global analytical standards market size was valued at USD 1.51 billion in 2025 and is expected to reach USD 2.46 billion by 2033, at a CAGR of 6.30% during the forecast period
- The market growth is largely fuelled by the increasing demand for accurate quality control and regulatory compliance across pharmaceutical, biotechnology, and chemical industries, ensuring product safety and consistency
- Rising adoption of advanced analytical techniques in drug development, environmental testing, and food safety is further driving the need for high-quality reference standards across laboratories and research facilities
Analytical Standards Market Analysis
- The analytical standards market is witnessing steady growth due to increasing regulatory requirements and stringent quality assurance protocols across multiple end-use industries
- Expansion of pharmaceutical and biotechnology sectors is significantly boosting demand for certified reference materials used in drug formulation, testing, and validation processes
- North America dominated the analytical standards market with the largest revenue share in 2025, driven by strong pharmaceutical and biotechnology industries, increasing regulatory compliance requirements, and high adoption of advanced analytical testing methods
- The Asia-Pacific region is expected to witness the highest growth rate in the global analytical standards market, driven by rapid industrialization, rising demand for food and environmental testing, and increasing adoption of quality control and analytical testing standards across emerging economies
- The organic standard segment held the largest market revenue share in 2025 driven by its extensive use in pharmaceutical development, bioanalytical testing, and quality control processes. Organic standards are widely preferred due to their critical role in ensuring accuracy, consistency, and regulatory compliance in complex biological and chemical analyses. These standards are essential in drug discovery, formulation development, and impurity profiling across laboratories. Increasing demand for high-purity compounds in life sciences research further supports segment dominance. Continuous innovation in reference material development is also enhancing reliability and precision in testing workflows
Report Scope and Analytical Standards Market Segmentation
|
Attributes |
Analytical Standards Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
• Merck KGaA (Germany) |
|
Market Opportunities |
• Expanding Pharmaceutical And Biotechnology Research |
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Analytical Standards Market Trends
“Rising Demand for High-Precision Testing and Regulatory Compliance”
• The increasing emphasis on accurate analytical measurement and stringent regulatory frameworks is significantly shaping the analytical standards market, as industries increasingly rely on certified reference materials to ensure data reliability and compliance. Analytical standards are gaining importance due to their critical role in calibration, validation, and quality assurance across laboratories. This trend is strengthening their adoption across pharmaceutical, biotechnology, environmental, and food testing industries, encouraging continuous innovation in reference material development
• Increasing focus on drug safety, environmental protection, and food quality has accelerated the demand for analytical standards in bioanalytical testing, stability studies, and contamination detection applications. Regulatory agencies and quality control laboratories are actively mandating the use of certified standards, prompting organizations to prioritize accuracy and traceability in analytical workflows. This has also led to stronger collaboration between reference material manufacturers and research institutions to enhance testing precision and reliability
• Quality assurance and compliance requirements are influencing laboratory practices, with manufacturers and testing facilities emphasizing certified reference materials, accreditation standards, and validated analytical methods. These factors are helping laboratories improve credibility, ensure reproducibility of results, and meet international regulatory expectations. Companies are increasingly investing in advanced standardization protocols to support global regulatory harmonization and laboratory accreditation
• For instance, in 2024, Merck Group in Germany and Thermo Fisher Scientific in the U.S. expanded their analytical standards portfolios by introducing new certified reference materials for pharmaceutical impurity testing and environmental monitoring applications. These launches were introduced in response to increasing demand for high-accuracy testing solutions, with distribution across pharmaceutical, research, and industrial laboratories. The products were also positioned to support regulatory compliance and improve analytical efficiency across quality control workflows
• While demand for analytical standards is growing, sustained market expansion depends on continuous advancements in material purity, cost-efficient production, and global standard harmonization. Manufacturers are also focusing on improving traceability, expanding certified product portfolios, and developing innovative solutions that meet evolving regulatory and industry-specific testing requirements
Analytical Standards Market Dynamics
Driver
“Growing Need for Accurate Quality Control and Regulatory Compliance”
• Rising demand for precise analytical testing is a major driver for the analytical standards market. Industries are increasingly relying on certified reference materials to ensure accuracy, reproducibility, and compliance with global regulatory frameworks. This trend is particularly strong in pharmaceutical and biotechnology sectors, where product safety and efficacy are critical
• Expanding applications in drug development, environmental monitoring, food safety testing, and chemical analysis are significantly influencing market growth. Analytical standards help maintain consistency in testing procedures, enabling laboratories to meet stringent quality control requirements and regulatory guidelines. The growing complexity of analytical methods further reinforces their importance
• Pharmaceutical and life sciences companies are actively integrating analytical standards into research, development, and manufacturing processes through advanced quality assurance systems and regulatory compliance programs. These efforts are supported by increasing global harmonization of testing standards, encouraging collaboration between manufacturers, laboratories, and regulatory authorities to improve data reliability and traceability
• For instance, in 2023, Agilent Technologies in the U.S. and Waters Corporation in the U.S. reported increased adoption of certified analytical standards in pharmaceutical testing and environmental analysis workflows. This expansion followed stricter regulatory requirements and rising demand for validated analytical methods, improving accuracy and strengthening product compliance across industries
• Although regulatory-driven adoption supports growth, wider market expansion depends on cost optimization, availability of high-purity reference materials, and continuous innovation in analytical technologies. Investment in advanced manufacturing processes and global standard harmonization will be critical for meeting increasing demand and maintaining analytical accuracy
Restraint/Challenge
“High Cost of Certified Standards and Limited Accessibility in Emerging Markets”
• The relatively high cost of certified analytical standards compared to general laboratory reagents remains a key challenge, limiting adoption among small and medium-scale laboratories. Complex production processes, stringent certification requirements, and high purity levels contribute to elevated pricing and restrict affordability in cost-sensitive markets
• Limited awareness and technical expertise in developing regions also hinder market growth, particularly where advanced analytical testing infrastructure is still emerging. Many laboratories lack sufficient training in the use of certified reference materials, leading to slower adoption in routine testing applications
• Supply chain complexities and stringent regulatory certification processes further impact market availability, as analytical standards require controlled production, storage, and distribution conditions to maintain accuracy and stability. These requirements increase operational costs and create logistical challenges for global distribution
• For instance, in 2024, laboratories in Southeast Asia and parts of Africa reported slower adoption of certified analytical standards in food safety and environmental testing due to high costs and limited access to certified suppliers. Delays in procurement and reliance on imported reference materials also affected testing efficiency and compliance capabilities in several facilities
• Overcoming these challenges will require cost-effective production techniques, expanded distribution networks, and increased awareness programs for end users. Collaboration between regulatory agencies, manufacturers, and research institutions can help improve accessibility and promote standardized testing practices globally. Furthermore, development of affordable and region-specific reference materials will be essential for long-term market expansion
Analytical Standards Market Scope
The market is segmented on the basis of category, technique, application, and end users.
• By Category
On the basis of category, the analytical standards market is segmented into organic standard and inorganic standard. The organic standard segment held the largest market revenue share in 2025 driven by its extensive use in pharmaceutical development, bioanalytical testing, and quality control processes. Organic standards are widely preferred due to their critical role in ensuring accuracy, consistency, and regulatory compliance in complex biological and chemical analyses. These standards are essential in drug discovery, formulation development, and impurity profiling across laboratories. Increasing demand for high-purity compounds in life sciences research further supports segment dominance. Continuous innovation in reference material development is also enhancing reliability and precision in testing workflows.
The inorganic standard segment is expected to witness the fastest growth rate from 2026 to 2033, driven by increasing demand in environmental testing, petrochemical analysis, and industrial quality assurance applications. Inorganic standards are gaining traction due to their importance in detecting metals, minerals, and elemental impurities across various end-use industries. Rising concerns regarding heavy metal contamination in water, soil, and air are further accelerating demand. These standards are widely used in regulatory compliance testing and industrial monitoring processes. Expanding mining, energy, and manufacturing activities globally are also contributing to segment growth.
• By Technique
On the basis of technique, the analytical standards market is segmented into chromatography standards, spectroscopy standards, titrimetry standards, and physical properties testing standards. The chromatography standards segment held the largest market revenue share in 2025 driven by widespread adoption in pharmaceutical and life sciences research for compound separation, identification, and purity testing. These standards are essential for ensuring reliable analytical results in drug development and quality control. Increasing use in clinical research and biotechnology applications further strengthens segment dominance. High accuracy and reproducibility make chromatography techniques highly preferred in regulated industries. Continuous advancements in HPLC and GC technologies are also boosting demand for associated standards.
The spectroscopy standards segment is expected to witness the fastest growth rate from 2026 to 2033, driven by increasing usage in environmental monitoring, chemical analysis, and advanced research applications. Spectroscopy standards are widely used for precise molecular characterization and are gaining importance due to technological advancements in analytical instrumentation. Growing adoption of UV, IR, and mass spectroscopy in research laboratories is further supporting growth. Expanding applications in food safety testing and forensic analysis are also contributing to rising demand. Increasing investment in advanced analytical instruments is accelerating market penetration.
• By Application
On the basis of application, the analytical standards market is segmented into bioanalytical testing, stability testing, raw material testing, microbial testing, and other applications. The bioanalytical testing segment held the largest market revenue share in 2025 driven by strong demand from pharmaceutical and biotechnology industries for drug development and clinical research. Analytical standards are widely used to ensure accuracy, safety, and regulatory compliance in biological sample analysis. Growing clinical trial activities and increasing biologics development are further supporting segment dominance. Rising demand for personalized medicine is also boosting usage in bioanalysis workflows. Continuous expansion of pharmaceutical R&D pipelines strengthens long-term demand.
The stability testing segment is expected to witness the fastest growth rate from 2026 to 2033, driven by increasing regulatory requirements for drug shelf-life validation and product quality assurance. Growing emphasis on product safety and long-term efficacy is further supporting demand for reliable analytical standards in stability assessment studies. Pharmaceutical companies are increasingly focusing on compliance with international regulatory guidelines. Expansion of generic drug manufacturing is also contributing to higher testing requirements. Rising investments in quality assurance laboratories are further accelerating segment growth.
• By End Users
On the basis of end users, the analytical standards market is segmented into food and beverages, forensics, veterinary, petrochemistry, environmental, and pharmaceutical and life sciences standards. The pharmaceutical and life sciences standards segment held the largest market revenue share in 2025 driven by strong demand for drug testing, quality control, and regulatory compliance in pharmaceutical manufacturing and research activities. Analytical standards are widely used in formulation development and validation processes. Increasing global healthcare expenditure and drug development activities further strengthen segment leadership. Expanding biologics and biosimilars market is also boosting demand. Strong regulatory frameworks ensure continuous adoption across laboratories.
The environmental segment is expected to witness the fastest growth rate from 2026 to 2033, driven by increasing focus on pollution monitoring, water quality testing, and environmental safety regulations. Rising government initiatives for environmental protection and sustainable development are further accelerating the adoption of analytical standards in environmental testing laboratories. Growing industrial emissions monitoring requirements are also driving demand. Increasing public awareness regarding environmental safety supports long-term growth. Expansion of climate change monitoring programs further strengthens segment adoption.
Analytical Standards Market Regional Analysis
• North America dominated the analytical standards market with the largest revenue share in 2025, driven by strong pharmaceutical and biotechnology industries, increasing regulatory compliance requirements, and high adoption of advanced analytical testing methods
• Laboratories and manufacturers in the region highly value accuracy, traceability, and consistency offered by certified reference materials used across drug development, environmental testing, and food safety applications
• This widespread adoption is further supported by strong R&D investments, advanced laboratory infrastructure, and the presence of leading pharmaceutical and life sciences companies, establishing analytical standards as essential tools for quality assurance and regulatory validation
U.S. Analytical Standards Market Insight
The U.S. analytical standards market captured the largest revenue share in North America in 2025, fueled by rapid growth in pharmaceutical research, clinical trials, and stringent FDA regulatory requirements. Laboratories are increasingly adopting certified reference materials to ensure data accuracy and compliance with global standards. The growing focus on biologics, biosimilars, and personalized medicine is further driving demand for high-precision analytical standards. In addition, strong investment in research infrastructure and advanced analytical instrumentation is supporting market expansion across academic, industrial, and regulatory laboratories.
Europe Analytical Standards Market Insight
The Europe analytical standards market is expected to witness strong growth from 2026 to 2033, primarily driven by strict regulatory frameworks such as EMA guidelines and increasing focus on environmental safety and food quality testing. The region’s emphasis on sustainable development and public health is fostering higher adoption of certified analytical materials. Growing pharmaceutical manufacturing activities and expanding research collaborations are also supporting market growth across laboratory and industrial applications.
U.K. Analytical Standards Market Insight
The U.K. analytical standards market is expected to witness steady growth from 2026 to 2033, driven by increasing demand from pharmaceutical research, forensic testing, and environmental monitoring sectors. Strong regulatory compliance requirements and rising investments in life sciences research are supporting the adoption of certified reference materials. The presence of advanced laboratory infrastructure and growing clinical research activities further contribute to market expansion.
Germany Analytical Standards Market Insight
The Germany analytical standards market is expected to witness strong growth from 2026 to 2033, fueled by the country’s advanced chemical and pharmaceutical industries and strong focus on precision engineering and quality assurance. Increasing environmental monitoring requirements and strict EU regulatory standards are driving demand for certified analytical materials. Germany’s emphasis on innovation and high-quality manufacturing is further strengthening market adoption across industrial and research applications.
Asia-Pacific Analytical Standards Market Insight
The Asia-Pacific analytical standards market is expected to witness the fastest growth rate from 2026 to 2033, driven by rapid expansion of pharmaceutical manufacturing, increasing environmental testing requirements, and growing food safety concerns. Rising investments in healthcare infrastructure and laboratory development are further boosting demand for analytical standards. Government initiatives supporting quality control and regulatory compliance are also accelerating adoption across emerging economies.
Japan Analytical Standards Market Insight
The Japan analytical standards market is expected to witness steady growth from 2026 to 2033, driven by the country’s advanced pharmaceutical sector, strong focus on precision testing, and high-quality manufacturing standards. Increasing adoption of analytical techniques in environmental monitoring, food safety, and clinical research is supporting market expansion. Japan’s strong regulatory environment and emphasis on technological innovation further enhance demand for certified reference materials.
China Analytical Standards Market Insight
The China analytical standards market accounted for the largest market revenue share in Asia Pacific in 2025, attributed to rapid industrialization, expanding pharmaceutical production, and increasing regulatory enforcement. Growing demand for food safety testing, environmental monitoring, and drug quality control is driving widespread adoption of analytical standards. Strong domestic manufacturing capabilities and government support for quality assurance systems are further propelling market growth across multiple end-use industries.
Analytical Standards Market Share
The Analytical Standards industry is primarily led by well-established companies, including:
• Merck KGaA (Germany)
• LGC Limited (U.K.)
• SPEX CertiPrep (U.S.)
• Restek Corporation (U.S.)
• Agilent Technologies, Inc (U.S.)
• AccuStandard (U.S.)
• PerkinElmer Inc (U.S.)
• Thermo Fisher Scientific (U.S.)
• GENERAL ELECTRIC (U.S.)
• Mallinckrodt (U.S.)
• Cayman Chemical (U.S.)
• GFS Chemicals, Inc. (U.S.)
• Campro Scientific GmbH (Germany)
• Waters Corporation (U.S.)
• CPI International (U.S.)
• Crescent. (U.S.)
• Inorganic Ventures, Inc. (U.S.)
• John Barron_Reagecon (Ireland)
Latest Developments in Global Analytical Standards Market
- In July 2025, Waters Corporation and Becton Dickinson, merger announcement, unveiled a USD 17.5 billion merger aimed at forming the largest analytical instrument group, intended to expand product portfolio, strengthen global market leadership, and enhance innovation capacity across life sciences and analytical testing markets. The merger is also expected to combine advanced chromatography, diagnostics, and laboratory technologies under one platform. It will improve R&D synergies and accelerate development of next-generation analytical solutions. In addition, the consolidation is likely to enhance global distribution networks and customer reach. This development is expected to significantly reshape competitive dynamics in the analytical instruments and standards ecosystem
- In June 2025, Thermo Fisher Scientific, product launch, introduced Orbitrap Astral Zoom and Orbitrap Excedion Pro systems designed to improve scan speed by 35% and throughput by 40%, aimed at enhancing analytical efficiency, accelerating research workflows, and strengthening its position in high-performance mass spectrometry solutions. These systems are designed for advanced proteomics and complex molecular analysis applications. The launch supports faster data acquisition and improved resolution in research environments. It also enhances laboratory productivity by reducing analysis time. Overall, it strengthens Thermo Fisher’s dominance in high-end analytical instrumentation
- In May 2025, Bruker Corporation, product innovation, launched timsMetabo for 4D-metabolomics with Mobility Range Enhancement, aimed at improving molecular analysis precision, expanding metabolomics research capabilities, and supporting advanced biomedical and pharmaceutical research applications. The solution enables deeper insights into metabolic pathways and biomarker discovery. It enhances sensitivity and resolution in complex biological samples. The technology supports drug discovery and precision medicine research. It also strengthens Bruker’s position in advanced life science analytical tools
- In October 2024, Agilent Technologies, Inc., product launch, introduced the Infinity III LC Series featuring InfinityLab Assist Technology, aimed at improving liquid chromatography performance, increasing laboratory productivity, and enhancing accuracy and automation in analytical testing workflows. The system integrates advanced automation to reduce manual errors and improve reproducibility. It supports high-throughput testing in pharmaceutical and chemical laboratories. The platform is designed to optimize workflow efficiency and data quality. This launch reinforces Agilent’s leadership in chromatography solutions
- In June 2023, Agilent Technologies, Inc., product expansion, launched the 6495D LC/TQ and Revident LC/Q-TOF systems along with MassHunter Explorer and ChemVista software, aimed at improving mass spectrometry performance, enhancing data analysis capabilities, and strengthening its leadership in analytical instrumentation solutions. These systems deliver higher sensitivity and improved quantitative accuracy. The software integration enhances data visualization and compound identification. It supports advanced research in pharmaceuticals and environmental testing. This expansion significantly broadens Agilent’s analytical portfolio and market reach
- In March 2023, LGC Limited, acquisition, acquired Lipomed AG’s reference materials business focused on drugs of abuse and toxicology standards, aimed at expanding its analytical standards portfolio, strengthening its position in forensic and pharmaceutical testing markets, and improving availability of specialized reference materials globally. The acquisition enhances LGC’s capabilities in forensic toxicology and controlled substance analysis. It broadens access to certified reference materials for regulatory and clinical laboratories. The move also improves its product diversity in high-demand niche applications. Overall, it strengthens LGC’s global leadership in analytical reference standards
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.