Global Antisense And Rnai Therapeutics Market
Market Size in USD Billion
CAGR :
% 
 USD
7.59 Billion
USD
24.03 Billion
2025
2033
USD
7.59 Billion
USD
24.03 Billion
2025
2033
| 2026 –2033 | |
| USD 7.59 Billion | |
| USD 24.03 Billion | |
|
|
|
|
Antisense and RNAi Therapeutics Treatment Market Size
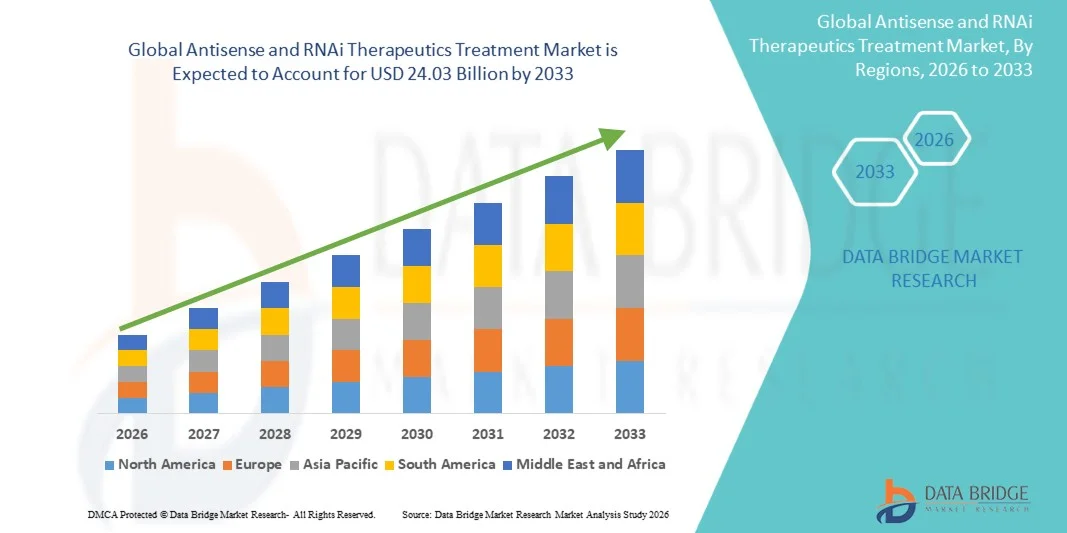
- The global antisense and RNAi therapeutics treatment market size was valued at USD 7.59 billion in 2025 and is expected to reach USD 24.03 billion by 2033, at a CAGR of 15.50% during the forecast period
- The market growth is largely fueled by the increasing prevalence of genetic disorders, rare diseases, and chronic conditions, which is driving the demand for highly targeted and precision-based therapeutic approaches such as antisense and RNA interference (RNAi) technologies
- Furthermore, continuous advancements in gene-silencing technologies, growing investment in biotechnology research, and increasing regulatory approvals for innovative RNA-based therapies are establishing Antisense and RNAi therapeutics as promising treatment options. These converging factors are accelerating the uptake of Antisense and RNAi Therapeutics Treatment solutions, thereby significantly boosting the industry's growth
Antisense and RNAi Therapeutics Treatment Market Analysis
- Antisense and RNAi therapeutics, which utilize gene-silencing mechanisms to inhibit disease-causing gene expression, are becoming increasingly important in the treatment of genetic disorders, rare diseases, and certain cancers due to their high specificity, precision targeting, and potential to address previously untreatable conditions
- The growing demand for Antisense and RNAi therapeutics is primarily driven by the rising prevalence of rare genetic disorders, increasing investments in biotechnology research, and expanding clinical pipelines focused on RNA-based medicines, along with supportive regulatory frameworks encouraging advanced therapeutic development
- North America dominated the antisense and RNAi therapeutics treatment market with the largest revenue share of 45.6% in 2025, supported by strong biotechnology infrastructure, significant R&D investments, and the presence of leading pharmaceutical and biotech companies, with the U.S. witnessing substantial growth in RNA-based drug development and approvals
- Asia-Pacific is expected to be the fastest growing region in the Antisense and RNAi Therapeutics Treatment market during the forecast period due to expanding biotechnology sectors, increasing healthcare investments, and growing clinical research activities in countries such as China, Japan, and South Korea
- The RNA interference segment dominated the largest market revenue share of 56.2% in 2025, driven by its strong capability to selectively silence disease-causing genes at the molecular level
Report Scope and Antisense and RNAi Therapeutics Treatment Market Segmentation
|
Attributes |
Antisense and RNAi Therapeutics Treatment Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
• F. Hoffmann-La Roche Ltd. (Switzerland) |
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Antisense and RNAi Therapeutics Treatment Market Trends
“Advancements in Precision Medicine and Targeted Gene Silencing Technologies”
- A significant and accelerating trend in the global Antisense and RNAi Therapeutics Treatment market is the growing advancement of precision medicine approaches that utilize gene-silencing technologies to target the root causes of genetic and rare diseases. These therapies work by selectively modulating or inhibiting the expression of disease-causing genes, offering a highly targeted treatment strategy compared to traditional pharmaceuticals
- For instance, RNA interference (RNAi) and antisense oligonucleotide technologies are increasingly being used to treat rare genetic disorders by blocking the production of harmful proteins at the molecular level. Therapies such as RNAi-based treatments have demonstrated promising outcomes in conditions including hereditary transthyretin-mediated amyloidosis and certain liver-related genetic diseases
- Continuous progress in nucleic acid chemistry, delivery systems, and lipid nanoparticle technologies is significantly improving the stability, specificity, and therapeutic efficiency of antisense and RNAi drugs. These advancements enable improved delivery of genetic medicines to targeted tissues such as the liver, central nervous system, and muscles
- In addition, pharmaceutical and biotechnology companies are investing heavily in research to develop next-generation antisense and RNAi therapeutics with enhanced potency, longer duration of action, and reduced side effects. This includes the development of chemically modified oligonucleotides designed to improve treatment outcomes and patient safety
- The expansion of genomic research, coupled with advances in molecular diagnostics and personalized medicine, is further accelerating the development of gene-silencing therapies tailored to specific patient populations. Such innovations are helping clinicians design targeted treatment strategies for previously untreatable genetic conditions
- As a result, the Antisense and RNAi Therapeutics Treatment market is witnessing increasing clinical trials, regulatory approvals, and collaborations between pharmaceutical companies and research institutions aimed at developing novel gene-targeting therapies for a wide range of rare and chronic diseases
Antisense and RNAi Therapeutics Treatment Market Dynamics
Driver
“Rising Prevalence of Genetic Disorders and Increasing Investment in RNA-Based Drug Development”
- The growing prevalence of rare genetic diseases and chronic disorders is a major factor driving demand for Antisense and RNAi Therapeutics Treatment solutions worldwide. These therapies offer a targeted mechanism for addressing diseases at the genetic level, making them particularly valuable for conditions where conventional treatments are ineffective or unavailable
- For instance, in September 2022, Alnylam Pharmaceuticals received regulatory approval for AMVUTTRA (vutrisiran), an RNAi therapeutic designed for the treatment of hereditary transthyretin-mediated amyloidosis with polyneuropathy in adults. Such developments demonstrate the increasing clinical adoption of RNA-based therapeutics
- Growing investments from pharmaceutical companies, biotechnology firms, and government research organizations are accelerating the development of innovative antisense and RNAi therapies. Funding initiatives aimed at advancing genomic medicine and rare disease treatments are further strengthening research and commercialization efforts in this field
- In addition, advancements in gene sequencing technologies and improved understanding of disease-causing genetic mutations are enabling researchers to design more precise therapeutic molecules capable of selectively targeting specific genes responsible for disease progression
- The expansion of clinical pipelines for RNA-based therapies, along with increasing regulatory support for innovative treatments addressing unmet medical needs, is further contributing to market growth. Several pharmaceutical companies are actively conducting clinical trials for antisense and RNAi drugs targeting neurological disorders, metabolic diseases, and rare genetic conditions
- Furthermore, the rising adoption of precision medicine and personalized healthcare approaches is encouraging healthcare providers to integrate gene-targeting therapies into treatment strategies, thereby strengthening the overall growth of the Antisense and RNAi Therapeutics Treatment market
Restraint/Challenge
“High Treatment Costs and Complex Drug Delivery Mechanisms”
- One of the major challenges affecting the growth of the Antisense and RNAi Therapeutics Treatment market is the high cost associated with the development and administration of these advanced genetic therapies. The complex research processes, specialized manufacturing requirements, and lengthy clinical trials involved in developing RNA-based drugs significantly increase overall treatment cost
- For instance, several approved antisense and RNAi therapeutics for rare genetic diseases are priced at premium levels due to their highly specialized nature and limited patient populations, which can restrict accessibility for patients in low- and middle-income regions
- Another major challenge lies in the complexity of delivering nucleic acid-based drugs effectively to targeted tissues within the human body. Ensuring stability, avoiding immune system reactions, and achieving precise cellular uptake remain significant technical hurdles for researchers and drug developers
- In addition, strict regulatory requirements and extensive safety evaluations are necessary for genetic therapies, as long-term effects and potential off-target gene interactions must be carefully assessed before regulatory approval can be granted. These regulatory complexities can lengthen the drug development timeline
- Limited awareness and availability of specialized treatment centers in certain regions may also restrict patient access to advanced RNA-based therapies. Many of these treatments require expert medical supervision and specialized healthcare infrastructure for safe administration and monitoring
- Addressing these challenges through improved drug delivery technologies, cost-effective manufacturing processes, expanded clinical research, and supportive regulatory frameworks will be essential for ensuring broader adoption and long-term growth of the Antisense and RNAi Therapeutics Treatment market.
Antisense and RNAi Therapeutics Treatment Market Scope
The market is segmented on the basis of indication, technology, route of administration, end users, and distribution channel.
• By Indication
On the basis of indication, the Antisense and RNAi Therapeutics Treatment market is segmented into oncology, cardiovascular diseases (CVDs), respiratory disorders, neurological disorders, infectious diseases, and others. The oncology segment dominated the largest market revenue share of 34.6% in 2025, driven by the increasing global burden of cancer and the growing need for targeted therapies. Antisense and RNAi therapeutics are widely used in oncology research to silence specific cancer-causing genes and inhibit tumor progression. Pharmaceutical companies are investing heavily in RNA-based therapies to develop precision medicines targeting genetic mutations responsible for cancer development. The growing number of clinical trials investigating RNA-targeted therapies for solid tumors and hematological cancers further strengthens segment growth. These therapies offer improved specificity and reduced off-target effects compared with conventional chemotherapy treatments. Increasing regulatory approvals for RNA-based drugs and strong investment in oncology drug pipelines also contribute to the segment’s dominance. In addition, advancements in gene-silencing technologies and delivery systems are improving treatment efficacy and safety profiles. The rising prevalence of lung, breast, and colorectal cancers globally continues to increase the demand for innovative RNA-based therapeutics. As precision medicine gains momentum, oncology remains the leading therapeutic area in the antisense and RNAi therapeutics market.
The neurological disorders segment is expected to witness the fastest CAGR of 13.1% from 2026 to 2033, driven by increasing research into gene-based therapies for rare and genetic neurological diseases. Antisense therapies have shown strong potential in treating conditions such as spinal muscular atrophy, Huntington’s disease, and amyotrophic lateral sclerosis. These therapies target the underlying genetic mechanisms responsible for neurological degeneration, offering disease-modifying treatment approaches. The growing prevalence of neurodegenerative disorders and the lack of effective treatment options further support the development of RNA-based therapeutics in this field. Pharmaceutical companies and biotechnology firms are investing heavily in neurological RNA therapy pipelines. Advancements in gene delivery technologies and RNA stabilization techniques are improving therapeutic outcomes. In addition, increasing regulatory incentives for rare disease treatments are accelerating drug development programs. Research institutions and biotechnology companies are actively conducting clinical trials to expand the application of RNA interference technologies in neurological diseases. The rising focus on precision medicine and personalized treatment approaches is expected to further drive the rapid growth of this segment.
• By Technology
On the basis of technology, the market is segmented into RNA interference and antisense RNA. The RNA interference segment dominated the largest market revenue share of 56.2% in 2025, driven by its strong capability to selectively silence disease-causing genes at the molecular level. RNA interference technology uses small interfering RNA molecules to inhibit the expression of specific genes responsible for disease progression. The increasing number of approved RNAi-based therapeutics and growing research investments from pharmaceutical companies contribute to segment growth. RNAi technology is widely applied in treating genetic disorders, metabolic diseases, and viral infections. Advances in lipid nanoparticle delivery systems have significantly improved the stability and targeted delivery of RNAi therapeutics. Pharmaceutical companies are focusing on expanding RNAi drug pipelines to address a wide range of chronic and rare diseases. The technology’s ability to target previously “undruggable” genes further strengthens its adoption in drug discovery. Increasing collaboration between biotechnology companies and research institutions also supports technological advancement. As a result, RNA interference continues to dominate the global antisense and RNAi therapeutics market.
The antisense RNA segment is projected to witness the fastest CAGR of 12.4% from 2026 to 2033, driven by advancements in antisense oligonucleotide drug development. Antisense RNA therapeutics work by binding to messenger RNA and blocking the production of harmful proteins responsible for disease progression. The technology has demonstrated significant success in treating rare genetic disorders and neurological diseases. Increasing clinical trial activity and regulatory approvals are supporting the expansion of antisense therapeutics globally. Pharmaceutical companies are investing in next-generation antisense technologies with improved stability and targeted delivery capabilities. These therapies offer high specificity and the ability to modulate gene expression effectively. Growing research in personalized medicine and gene-targeted treatments is further accelerating segment growth. In addition, antisense therapies are gaining attention for their potential in treating cardiovascular and metabolic diseases. The rising demand for innovative gene-based treatment approaches is expected to drive the rapid growth of antisense RNA technologies during the forecast period.
• By Route of Administration
On the basis of route of administration, the market is segmented into pulmonary delivery, intravenous injections, intra-dermal, intraperitoneal, topical, and others. The intravenous injections segment dominated the largest market revenue share of 41.8% in 2025, supported by the need for precise and rapid systemic delivery of RNA-based therapeutics. Intravenous administration ensures efficient distribution of antisense and RNAi molecules throughout the bloodstream, allowing effective targeting of diseased cells. Healthcare providers frequently use this route in hospital settings for the treatment of severe genetic and metabolic disorders. The availability of advanced infusion technologies further enhances treatment efficiency and patient safety. Intravenous delivery also supports controlled dosing and improved therapeutic outcomes in clinical settings. Pharmaceutical companies are developing advanced formulations designed specifically for intravenous administration to enhance drug stability and delivery efficiency. Increasing hospital-based treatments and the expansion of specialized genetic therapy programs further contribute to segment growth. In addition, intravenous administration allows for better monitoring of patient response during therapy. These factors collectively support the dominance of this route of administration.
The pulmonary delivery segment is expected to witness the fastest CAGR of 11.7% from 2026 to 2033, driven by increasing research into non-invasive delivery methods for RNA therapeutics. Pulmonary delivery allows drugs to be administered directly to the lungs using inhalation systems, improving localized treatment for respiratory diseases. This route offers advantages such as improved patient compliance and reduced systemic side effects compared with injectable therapies. Pharmaceutical companies are developing inhalable RNA formulations designed to target respiratory infections and pulmonary disorders. Advancements in nanoparticle delivery systems and aerosol technologies are enhancing drug stability and absorption in lung tissues. The growing prevalence of respiratory diseases such as asthma and chronic obstructive pulmonary disease further supports segment growth. Researchers are also exploring pulmonary delivery for systemic gene therapy applications due to the large surface area of lung tissues. Increasing clinical research and technological advancements are expected to accelerate the adoption of pulmonary RNA therapeutics. As a result, pulmonary delivery is projected to emerge as the fastest-growing administration route.
• By End Users
On the basis of end users, the market is segmented into hospitals, specialty clinics, and others. The hospitals segment dominated the largest market revenue share of 61.5% in 2025, supported by the availability of advanced diagnostic infrastructure and specialized treatment facilities. Hospitals are primary centers for administering RNA-based therapies, particularly for patients with severe genetic and rare diseases. The presence of trained healthcare professionals and advanced laboratory facilities enables accurate diagnosis and targeted treatment approaches. Hospitals also play a key role in conducting clinical trials for antisense and RNAi therapeutics. The growing number of patients requiring advanced genetic therapies further increases demand within hospital settings. In addition, hospitals maintain specialized departments for oncology, neurology, and rare diseases where RNA therapeutics are frequently used. Government funding and healthcare investments are improving hospital infrastructure worldwide. The presence of integrated healthcare services ensures comprehensive patient care and treatment monitoring. These factors collectively strengthen the dominance of hospitals as the leading end users of RNA-based therapies.
The specialty clinics segment is projected to witness the fastest CAGR of 10.9% from 2026 to 2033, driven by the increasing number of specialized treatment centers for rare diseases and genetic disorders. Specialty clinics focus on providing targeted therapies and personalized treatment solutions for complex medical conditions. The expansion of precision medicine practices and genetic testing services supports the growth of these clinics. Patients increasingly prefer specialized healthcare facilities that offer focused expertise in managing rare and chronic diseases. Specialty clinics also provide outpatient administration of RNA-based therapies, improving treatment accessibility. Pharmaceutical companies frequently collaborate with specialty clinics to conduct clinical trials and treatment programs. The rising demand for personalized and gene-based treatment approaches further drives segment expansion. In addition, specialty clinics offer shorter waiting times and more personalized care compared with large hospitals. These advantages are expected to accelerate the adoption of antisense and RNAi therapeutics in specialty clinic settings.
• By Distribution Channel
On the basis of distribution channel, the market is segmented into hospital pharmacy, retail pharmacy, and others. The hospital pharmacy segment dominated the largest market revenue share of 47.8% in 2025, primarily due to the specialized nature of antisense and RNAi therapeutics. These therapies often require physician supervision and controlled dispensing, making hospital pharmacies the primary distribution point. Hospital pharmacies maintain strict storage and handling protocols for advanced biologic therapies. Healthcare professionals rely on hospital pharmacies to ensure accurate dosing and patient safety. The increasing number of hospital-based treatments and clinical trials further supports demand in this segment. In addition, hospitals maintain strong procurement systems and supply chains for specialized medications. The presence of trained pharmacists ensures proper medication counseling and monitoring. Hospital pharmacies also play a critical role in managing complex treatment regimens for genetic disorders. These factors collectively contribute to the dominance of hospital pharmacies in the global market.
The retail pharmacy segment is expected to witness the fastest CAGR of 9.8% from 2026 to 2033, driven by improving accessibility to advanced therapeutics and expanding pharmaceutical distribution networks. Retail pharmacies are increasingly involved in dispensing specialty medications prescribed for chronic and rare diseases. The expansion of pharmacy chains and improved healthcare infrastructure supports greater access to RNA-based treatments. Retail pharmacies also offer convenient medication access for patients undergoing long-term therapy. Pharmaceutical companies are partnering with pharmacy networks to expand distribution channels for specialty drugs. Increasing awareness about gene-based therapies among patients and healthcare professionals further drives segment growth. Retail pharmacies also support medication adherence through patient counseling and monitoring services. The integration of digital pharmacy platforms and prescription management systems further enhances service efficiency. As RNA-based therapies become more widely adopted, retail pharmacies are expected to play a growing role in global distribution networks.
Antisense and RNAi Therapeutics Treatment Market Regional Analysis
- North America dominated the antisense and RNAi therapeutics treatment market with the largest revenue share of 45.6% in 2025, supported by strong biotechnology infrastructure, substantial research and development investments, and the presence of leading pharmaceutical and biotechnology companies actively involved in RNA-based drug discovery
- The region benefits from a highly advanced healthcare system, strong regulatory support for innovative therapies, and increasing adoption of precision medicine approaches that focus on treating diseases at the genetic and molecular level
- In addition, growing clinical research activities, strong academic–industry collaborations, and the increasing number of FDA approvals for RNA-based therapeutics are accelerating the commercialization and adoption of antisense and RNAi treatments across the region, particularly for rare genetic and neurological disorders
U.S. Antisense and RNAi Therapeutics Treatment Market Insight
The U.S. antisense and RNAi therapeutics treatment market captured the largest revenue share in 2025 within North America, driven by robust biotechnology innovation, extensive research funding, and the strong presence of global pharmaceutical companies developing RNA-based therapies. The country is home to several pioneering biotechnology firms focused on antisense and RNA interference technologies, which significantly contributes to drug development and commercialization. Moreover, the increasing number of clinical trials targeting rare genetic diseases, neurological disorders, and metabolic conditions is accelerating market growth. Government support for precision medicine initiatives, well-established regulatory pathways for novel therapies, and strong pharmaceutical distribution networks further strengthen the expansion of the Antisense and RNAi Therapeutics Treatment industry in the United States.
Europe Antisense and RNAi Therapeutics Treatment Market Insight
The Europe antisense and RNAi therapeutics treatment market is projected to expand at a substantial CAGR throughout the forecast period, driven by increasing investments in biotechnology research, strong academic collaborations, and supportive regulatory frameworks for advanced therapies. Countries across Europe are actively promoting the development of innovative genetic medicines to address rare diseases and complex chronic conditions. Furthermore, the presence of well-established pharmaceutical companies, growing clinical trial activities, and increased government funding for genomic research are encouraging the adoption of antisense and RNAi therapies across the region. The expansion of precision medicine programs and growing awareness of gene-based therapies among healthcare providers are also supporting market growth.
U.K. Antisense and RNAi Therapeutics Treatment Market Insight
The U.K. antisense and RNAi therapeutics treatment market is anticipated to grow at a noteworthy CAGR during the forecast period, supported by the country’s strong life sciences ecosystem and increasing investment in genomic research and biotechnology innovation. The United Kingdom has established itself as a leading hub for advanced therapeutic development, with numerous academic institutions and biotechnology firms actively engaged in RNA-based drug research. In addition, government initiatives aimed at strengthening precision medicine and rare disease research are accelerating clinical trials and the development of targeted genetic therapies. These factors are expected to drive continued growth of the Antisense and RNAi Therapeutics Treatment market in the country.
Germany Antisense and RNAi Therapeutics Treatment Market Insight
The Germany antisense and RNAi therapeutics treatment market is expected to expand at a considerable CAGR during the forecast period, fueled by strong pharmaceutical manufacturing capabilities, advanced healthcare infrastructure, and increasing research initiatives in molecular medicine. Germany’s emphasis on biomedical innovation and translational research is encouraging the development of next-generation RNA-based therapies aimed at treating rare and complex diseases. In addition, collaborations between biotechnology firms, academic research centers, and pharmaceutical companies are fostering the advancement of antisense and RNAi therapeutic technologies. The country’s focus on personalized medicine and innovative drug development is further contributing to the growth of the market.
Asia-Pacific Antisense and RNAi Therapeutics Treatment Market Insight
The Asia-Pacific antisense and RNAi therapeutics treatment market is expected to be the fastest-growing region during the forecast period, driven by expanding biotechnology sectors, increasing healthcare investments, and growing clinical research activities in countries such as China, Japan, and South Korea. Governments across the region are prioritizing the development of advanced healthcare technologies and supporting research in genomics and molecular medicine. In addition, the rising prevalence of genetic disorders, increasing awareness of advanced therapies, and growing collaborations between international pharmaceutical companies and regional biotech firms are contributing to market growth. The expanding healthcare infrastructure and increasing investment in biomedical innovation are also accelerating the adoption of antisense and RNAi therapies across Asia-Pacific.
Japan Antisense and RNAi Therapeutics Treatment Market Insight
The Japan antisense and RNAi therapeutics treatment market is gaining momentum due to the country’s strong focus on advanced biomedical research, high healthcare expenditure, and growing investment in innovative therapeutic technologies. Japan has a well-established pharmaceutical industry that actively supports the development of RNA-based therapies through research collaborations and government-backed initiatives. In addition, the country’s increasing emphasis on precision medicine and targeted treatments for rare diseases is encouraging the adoption of antisense and RNAi therapeutics. The presence of advanced clinical research facilities and supportive regulatory policies for novel therapies further strengthens the market outlook in Japan.
China Antisense and RNAi Therapeutics Treatment Market Insight
The China antisense and RNAi therapeutics treatment market accounted for the largest market revenue share in Asia-Pacific in 2025, supported by the country’s rapidly expanding biotechnology sector, strong government funding for pharmaceutical innovation, and growing clinical research capabilities. China has emerged as a major hub for biotechnology development, with increasing investments in gene-based therapeutics and advanced drug discovery platforms. In addition, the rising prevalence of chronic and genetic diseases, coupled with the expansion of domestic pharmaceutical companies engaged in RNA-based therapy development, is accelerating market growth. Government initiatives aimed at strengthening biomedical research and promoting innovative drug development are also playing a crucial role in expanding the Antisense and RNAi Therapeutics Treatment market in China.
Antisense and RNAi Therapeutics Treatment Market Share
The Antisense and RNAi Therapeutics Treatment industry is primarily led by well-established companies, including:
• F. Hoffmann-La Roche Ltd. (Switzerland)
• Alnylam Pharmaceuticals, Inc. (U.S.)
• Ionis Pharmaceuticals, Inc. (U.S.)
• Novartis AG (Switzerland)
• Pfizer Inc. (U.S.)
• Arrowhead Pharmaceuticals, Inc. (U.S.)
• Silence Therapeutics plc (U.K.)
• Dicerna Pharmaceuticals, Inc. (U.S.)
• Benitec Biopharma Inc. (Australia)
• Sarepta Therapeutics, Inc. (U.S.)
• Wave Life Sciences Ltd. (Singapore)
• Arbutus Biopharma Corporation (Canada)
• Sanofi (France)
• AstraZeneca plc (U.K.)
• Biogen Inc. (U.S.)
• GSK plc (U.K.)
• RxRx, Inc. (U.S.)
• Regulus Therapeutics Inc. (U.S.)
• CureVac N.V. (Germany)
• Moderna, Inc. (U.S.)
Latest Developments in Global Antisense and RNAi Therapeutics Treatment Market
- In March 2022, Biogen Inc. and Ionis Pharmaceuticals announced positive results from a Phase 1 clinical study of an investigational antisense oligonucleotide therapy for amyotrophic lateral sclerosis (ALS), highlighting progress in RNA-targeted treatments for neurodegenerative diseases and strengthening the pipeline of antisense therapeutics
- In January 2024, Silence Therapeutics announced a strategic collaboration with AstraZeneca to co-develop antisense therapies across multiple disease indications, combining Silence’s RNA-targeting platform with AstraZeneca’s global development and commercialization capabilities to accelerate RNA-based drug innovation
- In March 2024, Ionis Pharmaceuticals expanded its collaboration with Sanofi to accelerate the development of RNA-targeting antisense medicines, adding several new therapeutic targets and strengthening their joint portfolio of RNA-based therapies for multiple diseases
- In August 2025, Ionis Pharmaceuticals announced that donidalorsen, a prekallikrein-directed antisense oligonucleotide therapy, received approval in the United States for the prevention of hereditary angioedema attacks, marking a significant advancement in antisense-based treatments for rare genetic disorders
- In July 2025, Alnylam Pharmaceuticals launched a new RNA interference (RNAi) therapeutic for a hepatic rare disease, expanding its commercial RNAi medicine portfolio and reinforcing the growing role of gene-silencing technologies in treating genetic disorders
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.