Global Bio Decontamination Market
Market Size in USD Million
CAGR :
% 
 USD
256.82 Million
USD
413.98 Million
2025
2033
USD
256.82 Million
USD
413.98 Million
2025
2033
| 2026 –2033 | |
| USD 256.82 Million | |
| USD 413.98 Million | |
|
|
|
|
Bio Decontamination Market Size
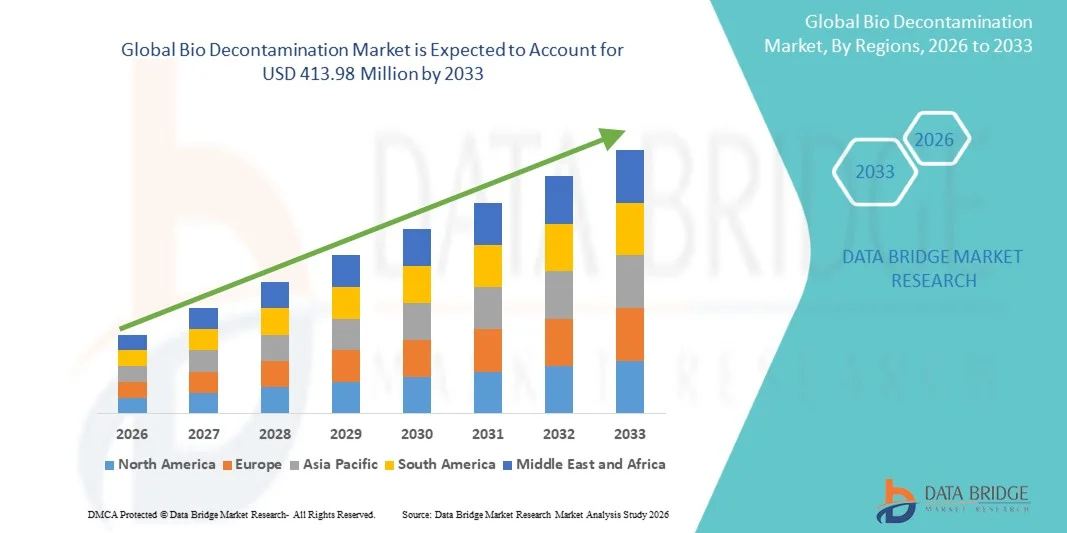
- The global Bio Decontamination market size was valued at USD 256.82 Million in 2025 and is expected to reach USD 413.98 Million by 2033, at a CAGR of 6.15% during the forecast period
- The market growth is largely fueled by the increasing need for effective infection control measures and rising awareness regarding contamination prevention in healthcare facilities, pharmaceutical manufacturing units, and research laboratories, leading to greater adoption of advanced bio decontamination technologies
- Furthermore, the growing demand for safe, efficient, and automated sterilization solutions across hospitals, biotechnology companies, and life science laboratories is establishing bio decontamination systems as essential tools for maintaining sterile environments. These converging factors are accelerating the uptake of bio decontamination solutions, thereby significantly boosting the industry's growth
Bio Decontamination Market Analysis
- Bio decontamination systems, used for eliminating harmful microorganisms such as bacteria, viruses, and spores from surfaces, equipment, and enclosed environments, are becoming increasingly vital across healthcare facilities, pharmaceutical manufacturing units, and life science laboratories due to their ability to maintain sterile environments and ensure regulatory compliance
- The escalating demand for bio decontamination solutions is primarily fueled by the growing focus on infection prevention, increasing pharmaceutical and biotechnology research activities, and the rising need for contamination control in hospitals, cleanrooms, and research laboratories
- North America dominated the bio decontamination market with the largest revenue share of 39.4% in 2025, characterized by advanced healthcare infrastructure, strong presence of pharmaceutical and biotechnology companies, and stringent regulatory standards for sterilization and contamination control, with the U.S. experiencing significant growth in bio decontamination adoption across hospitals, research laboratories, and pharmaceutical manufacturing facilities
- Asia-Pacific is expected to be the fastest growing region in the bio decontamination market during the forecast period due to expanding pharmaceutical manufacturing, growing healthcare infrastructure, increasing investments in biotechnology research, and rising awareness regarding infection control and sterilization practices
- The chamber decontamination segment dominated the largest market revenue share of 55.2% in 2025, driven by its extensive use in pharmaceutical and biotechnology manufacturing facilities
Report Scope and Bio Decontamination Market Segmentation
|
Attributes |
Bio Decontamination Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
• STERIS plc (U.S.) |
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Bio Decontamination Market Trends
“Increasing Adoption of Advanced Sterilization and Decontamination Technologies”
- A significant and accelerating trend in the global bio decontamination market is the growing adoption of advanced sterilization and decontamination technologies across healthcare facilities, pharmaceutical manufacturing units, and biotechnology laboratories. The increasing need to maintain sterile environments and prevent microbial contamination in critical areas such as cleanrooms, operating rooms, and production facilities is significantly encouraging the deployment of bio decontamination systems
- For instance, pharmaceutical manufacturers and research laboratories are increasingly utilizing hydrogen peroxide vapor (HPV) and chlorine dioxide–based bio decontamination systems to effectively eliminate harmful microorganisms and maintain strict contamination control standards within controlled environments
- The advancement of automated and portable bio decontamination systems is enabling healthcare facilities and pharmaceutical companies to perform rapid and highly effective sterilization procedures. These systems are designed to ensure uniform distribution of decontamination agents, improving efficiency while minimizing manual intervention. As a result, many hospitals and research institutions are adopting automated bio decontamination technologies to maintain consistent infection control practices
- The increasing implementation of stringent regulatory guidelines related to sterilization, contamination prevention, and cleanroom maintenance is further supporting the demand for bio decontamination solutions. Regulatory bodies and healthcare authorities are emphasizing the importance of effective microbial control to ensure patient safety, product quality, and regulatory compliance within healthcare and pharmaceutical environments
- This trend toward advanced contamination control solutions is significantly reshaping infection prevention strategies in healthcare and life sciences industries. Consequently, manufacturers are focusing on developing more efficient, faster, and environmentally safe bio decontamination technologies to meet the evolving requirements of hospitals, laboratories, and pharmaceutical production facilities
- The demand for reliable bio decontamination systems is increasing rapidly across healthcare, biotechnology, and pharmaceutical sectors as organizations prioritize stringent hygiene standards, contamination prevention, and regulatory compliance
Bio Decontamination Market Dynamics
Driver
“Growing Demand for Infection Control and Sterile Healthcare Environments”
- The increasing prevalence of hospital-acquired infections (HAIs), along with the growing emphasis on maintaining sterile healthcare environments, is a significant driver for the rising demand for bio decontamination systems
- Hospitals, research laboratories, and pharmaceutical manufacturing facilities are increasingly implementing advanced sterilization technologies to reduce microbial contamination and ensure patient safety
- For instance, several hospitals and pharmaceutical production facilities have been investing in hydrogen peroxide vapor–based bio decontamination systems to effectively sterilize operating rooms, isolation wards, and cleanroom environments in order to maintain strict hygiene and infection control standards
- As healthcare providers become more focused on preventing infections and improving patient safety, bio decontamination systems offer an effective solution for eliminating bacteria, viruses, fungi, and spores from critical environments. These systems help healthcare facilities maintain sterile conditions, thereby reducing the risk of contamination and improving overall clinical outcomes
- Furthermore, the rapid expansion of pharmaceutical and biotechnology industries is increasing the need for contamination-free manufacturing environments. Bio decontamination technologies play a crucial role in ensuring sterile production processes, protecting sensitive pharmaceutical products from microbial contamination, and maintaining compliance with regulatory guidelines
- The growing number of surgical procedures, expanding healthcare infrastructure, and increasing investments in infection control programs are key factors driving the adoption of bio decontamination solutions across hospitals, laboratories, and pharmaceutical manufacturing facilities worldwide
Restraint/Challenge
“High Equipment Costs and Operational Complexity”
- The relatively high cost associated with advanced bio decontamination systems presents a significant challenge to widespread market adoption
- Many healthcare facilities, particularly in developing regions, face budget constraints that limit their ability to invest in sophisticated sterilization technologies and automated decontamination equipment
- For instance, the installation of large-scale hydrogen peroxide vapor decontamination systems or integrated cleanroom sterilization solutions often requires substantial initial investment, specialized infrastructure, and trained personnel, which may discourage adoption among smaller healthcare facilities and laboratories
- Addressing these cost-related challenges requires manufacturers to focus on developing cost-effective and easy-to-operate bio decontamination solutions. Simplified system designs, portable equipment, and improved automation can help reduce operational complexity and make these technologies more accessible to a broader range of healthcare providers
- In addition, the operational requirements associated with certain bio decontamination technologies, such as specialized training, maintenance procedures, and strict safety protocols, can increase the overall complexity of system deployment and management. These factors may limit adoption in facilities that lack adequate technical expertise or operational resources
- Overcoming these challenges through technological innovation, improved cost efficiency, and enhanced user training programs will be essential for ensuring the long-term growth and broader adoption of bio decontamination solutions across global healthcare and life sciences industries
Bio Decontamination Market Scope
The market is segmented on the basis of product and service, agent type, type, and end user.
• By Product and Service
On the basis of product and service, the Bio Decontamination market is segmented into equipment, services, and consumables. The equipment segment dominated the largest market revenue share of 46.8% in 2025, driven by the growing adoption of automated bio decontamination systems in pharmaceutical manufacturing, hospitals, and research laboratories. Bio decontamination equipment such as vaporized hydrogen peroxide generators and integrated room decontamination systems play a critical role in maintaining sterile environments. These systems are widely used to eliminate microbial contamination in controlled environments including cleanrooms and laboratories. Increasing regulatory requirements for contamination control in pharmaceutical and biotechnology industries are significantly supporting the demand for bio decontamination equipment. Hospitals and healthcare facilities also rely heavily on these systems to ensure infection prevention and patient safety. Technological advancements have resulted in highly efficient and automated decontamination equipment with improved safety features. The growing emphasis on sterile production environments in drug manufacturing further supports segment expansion. Equipment solutions are also preferred for their reliability and ability to provide consistent decontamination results. Rising investments in healthcare infrastructure and pharmaceutical manufacturing facilities further strengthen this segment’s growth. Continuous product innovation by manufacturers is also enhancing system efficiency and performance. These factors collectively contribute to the dominance of the equipment segment in the global bio decontamination market.
The services segment is expected to witness the fastest growth rate of 14.7% CAGR from 2026 to 2033, driven by the increasing outsourcing of decontamination services by healthcare institutions and pharmaceutical companies. Many organizations prefer specialized service providers for bio decontamination procedures due to their expertise and regulatory compliance capabilities. Professional service providers offer scheduled decontamination, emergency response, and validation services. The growing number of pharmaceutical manufacturing facilities and biotechnology laboratories is increasing the demand for contract decontamination services. Hospitals also rely on professional service providers to ensure high-level sterilization in critical areas such as operating rooms and isolation wards. Outsourced services reduce operational complexity and allow healthcare facilities to focus on core medical activities. Technological advancements in decontamination processes have also improved service efficiency. Increasing awareness regarding infection prevention and contamination control further supports service demand. Regulatory bodies emphasize strict sanitation and sterilization standards, encouraging organizations to adopt professional services. The rise of contract research organizations and biopharmaceutical laboratories further contributes to service market growth. These factors are expected to accelerate the expansion of the services segment during the forecast period.
• By Agent Type
On the basis of agent type, the Bio Decontamination market is segmented into hydrogen peroxide, chlorine dioxide, peracetic acid, and nitrogen dioxide. The hydrogen peroxide segment dominated the largest market revenue share of 52.6% in 2025, driven by its high antimicrobial effectiveness and widespread adoption in sterilization processes. Hydrogen peroxide vapor systems are widely used in pharmaceutical cleanrooms, research laboratories, and hospital environments. The agent is highly effective in eliminating bacteria, viruses, spores, and fungi from contaminated environments. Hydrogen peroxide-based systems are preferred due to their rapid action and residue-free decontamination properties. They are also compatible with various medical equipment and sensitive laboratory instruments. The increasing need for contamination-free manufacturing environments in pharmaceutical industries significantly supports segment growth. Healthcare facilities also utilize hydrogen peroxide decontamination for infection prevention. Technological advancements have improved the efficiency and safety of hydrogen peroxide-based systems. Growing demand for automated sterilization technologies further drives adoption. Regulatory guidelines emphasizing contamination control in healthcare and pharmaceutical sectors also support the segment. Continuous innovation in vaporized hydrogen peroxide systems enhances their effectiveness in controlled environments. These factors collectively ensure the dominance of the hydrogen peroxide segment in the bio decontamination market.
The chlorine dioxide segment is anticipated to witness the fastest CAGR of 13.8% from 2026 to 2033, driven by its strong oxidizing properties and high efficiency in large-area decontamination. Chlorine dioxide gas is widely used for room and facility sterilization due to its ability to penetrate hard-to-reach areas. It is particularly effective in eliminating microbial contamination in large pharmaceutical production areas. Chlorine dioxide is also capable of destroying resistant bacterial spores and viruses. Increasing use of chlorine dioxide in industrial and healthcare sterilization applications supports market expansion. Its ability to decontaminate enclosed spaces without leaving toxic residues is a key advantage. Many healthcare and life sciences organizations are adopting chlorine dioxide systems for large-scale sterilization processes. The technology is also gaining traction in biosafety laboratories and research institutions. Growing concerns about healthcare-associated infections further encourage the use of advanced decontamination agents. Continuous research is improving chlorine dioxide generation technologies and safety mechanisms. These factors are expected to drive significant growth in the chlorine dioxide segment during the forecast period.
• By Type
On the basis of type, the Bio Decontamination market is segmented into chamber decontamination and room decontamination. The chamber decontamination segment dominated the largest market revenue share of 55.2% in 2025, driven by its extensive use in pharmaceutical and biotechnology manufacturing facilities. Chamber decontamination systems are designed to sterilize equipment, tools, and materials before entering controlled environments such as cleanrooms. These systems ensure that contaminants are not introduced into sterile manufacturing areas. Pharmaceutical companies widely rely on chamber decontamination to maintain strict quality and safety standards. The rising demand for biologics and sterile drug manufacturing further drives the adoption of chamber decontamination systems. These systems provide controlled and validated sterilization processes, ensuring consistent results. Regulatory authorities require strict contamination control measures during pharmaceutical production. Hospitals and laboratories also use chamber systems for sterilizing medical instruments. Technological advancements have improved automation and efficiency in chamber decontamination systems. Increasing investments in pharmaceutical manufacturing infrastructure further support the segment’s growth. The expansion of biologics and vaccine production facilities also contributes to rising demand. These factors collectively contribute to the dominance of the chamber decontamination segment in the global market.
The room decontamination segment is expected to witness the fastest CAGR of 15.3% from 2026 to 2033, driven by the increasing need for large-area sterilization in hospitals and research laboratories. Room decontamination systems are widely used to eliminate pathogens from hospital wards, isolation rooms, and intensive care units. These systems help reduce the risk of healthcare-associated infections and ensure patient safety. The rising prevalence of infectious diseases has increased the demand for efficient room sterilization technologies. Healthcare facilities are investing in automated decontamination systems to maintain high hygiene standards. Pharmaceutical and biotechnology laboratories also rely on room decontamination for maintaining sterile research environments. Technological advancements have enabled faster and more efficient decontamination processes. Portable and automated room sterilization systems are gaining popularity in healthcare settings. Government initiatives promoting infection control practices further support the adoption of these systems. Increasing awareness regarding environmental sterilization also drives market growth. These factors are expected to significantly accelerate the growth of the room decontamination segment during the forecast period.
• By End User
On the basis of end user, the Bio Decontamination market is segmented into pharmaceutical and medical device manufacturing companies, life sciences and biotechnology research organizations, and hospitals and healthcare facilities. The pharmaceutical and medical device manufacturing companies segment dominated the largest market revenue share of 48.3% in 2025, driven by stringent regulatory requirements for sterile manufacturing environments. Pharmaceutical companies must maintain contamination-free facilities to ensure the safety and quality of drugs and medical devices. Bio decontamination systems are widely used in cleanrooms and production facilities to eliminate microbial contamination. The increasing production of biologics, vaccines, and sterile injectable drugs further supports the demand for advanced decontamination technologies. Regulatory agencies such as global health authorities require strict adherence to contamination control standards. Pharmaceutical manufacturers invest heavily in sterilization systems to maintain product integrity and regulatory compliance. The expansion of pharmaceutical production facilities worldwide further contributes to market growth. Continuous innovation in sterilization technologies enhances efficiency and safety in manufacturing environments. The rising demand for high-quality pharmaceutical products also encourages adoption of bio decontamination solutions. These factors collectively contribute to the dominance of pharmaceutical and medical device manufacturing companies in the market.
The hospitals and healthcare facilities segment is anticipated to witness the fastest growth rate of 14.9% CAGR from 2026 to 2033, driven by the increasing focus on infection prevention and hospital hygiene. Hospitals require advanced sterilization solutions to control the spread of infectious diseases and healthcare-associated infections. Bio decontamination systems are widely used in operating rooms, patient wards, and intensive care units. The rising number of hospital admissions and surgical procedures increases the need for efficient sterilization technologies. Healthcare institutions are investing in automated decontamination systems to improve patient safety and operational efficiency. Technological advancements have introduced portable and user-friendly sterilization solutions suitable for hospital environments. Government initiatives promoting hospital hygiene and infection control further support adoption. Increasing awareness among healthcare professionals regarding sterilization practices also contributes to market growth. Hospitals are continuously upgrading their infrastructure to include advanced infection control technologies. These factors are expected to significantly accelerate the growth of the hospitals and healthcare facilities segment during the forecast period.
Bio Decontamination Market Regional Analysis
- North America dominated the bio decontamination market with the largest revenue share of 39.4% in 2025, characterized by advanced healthcare infrastructure, a strong presence of pharmaceutical and biotechnology companies, and stringent regulatory standards for sterilization and contamination control
- The demand for reliable contamination control solutions is rising across hospitals, pharmaceutical manufacturing plants, and research laboratories where maintaining sterile environments is critical
- Growing investments in life sciences research, increasing biopharmaceutical production, and the need to prevent hospital-acquired infections are further supporting the adoption of bio decontamination technologies across the region
U.S. Bio Decontamination Market Insight
The U.S. bio decontamination market captured the largest revenue share in 2025 within North America, driven by the strong presence of pharmaceutical manufacturers, biotechnology firms, and advanced healthcare facilities. Increasing regulatory focus on contamination prevention and sterilization in drug manufacturing and laboratory environments is significantly supporting market growth. Moreover, the rising demand for aseptic production environments, expansion of research laboratories, and increasing focus on infection prevention in hospitals are encouraging the adoption of advanced bio decontamination systems such as hydrogen peroxide vapor and ultraviolet technologies.
Europe Bio Decontamination Market Insight
The Europe bio decontamination market is projected to expand at a substantial CAGR during the forecast period, primarily driven by strict regulatory frameworks governing sterilization, biosafety, and contamination control in healthcare and pharmaceutical sectors. The increasing expansion of biotechnology research, rising investments in pharmaceutical production, and the presence of well-established healthcare systems are supporting the demand for bio decontamination solutions. In addition, growing awareness regarding infection prevention and biosafety standards in laboratories and cleanroom environments is further accelerating market growth across the region.
U.K. Bio Decontamination Market Insight
The U.K. bio decontamination market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by the increasing focus on infection control in healthcare facilities and research laboratories. The expansion of pharmaceutical manufacturing and biotechnology research activities in the country is also contributing to the demand for bio decontamination technologies. In addition, strong regulatory standards and government initiatives aimed at improving biosafety and contamination control practices are expected to further stimulate market growth.
Germany Bio Decontamination Market Insight
The Germany bio decontamination market is expected to expand at a considerable CAGR during the forecast period, supported by the country’s well-established pharmaceutical and biotechnology industries. Germany’s strong emphasis on research, innovation, and advanced healthcare infrastructure is encouraging the adoption of efficient contamination control solutions. Furthermore, the increasing number of research laboratories, cleanrooms, and pharmaceutical production facilities is boosting the demand for bio decontamination systems to maintain sterile and compliant environments.
Asia-Pacific Bio Decontamination Market Insight
The Asia-Pacific bio decontamination market is expected to be the fastest growing region during the forecast period, driven by expanding pharmaceutical manufacturing, growing healthcare infrastructure, increasing investments in biotechnology research, and rising awareness regarding infection control and sterilization practices. Rapid industrialization in the life sciences sector, particularly in countries such as China, Japan, and India, is significantly increasing the demand for advanced decontamination technologies in laboratories, hospitals, and pharmaceutical production facilities. Government initiatives supporting biotechnology development and the expansion of cleanroom manufacturing facilities are further strengthening the adoption of bio decontamination systems across the region.
Japan Bio Decontamination Market Insight
The Japan bio decontamination market is gaining traction due to the country’s strong biotechnology research ecosystem and advanced healthcare infrastructure. Increasing investments in pharmaceutical innovation and the expansion of cleanroom facilities in pharmaceutical and semiconductor manufacturing are contributing to the demand for reliable bio decontamination solutions. In addition, the growing emphasis on biosafety standards and infection prevention in hospitals and laboratories is further supporting market growth.
China Bio Decontamination Market Insight
The China bio decontamination market accounted for the largest revenue share in Asia Pacific in 2025, attributed to the rapid expansion of pharmaceutical manufacturing, biotechnology research, and healthcare infrastructure in the country. China’s growing role as a global pharmaceutical production hub is driving the demand for contamination control technologies in manufacturing facilities and laboratories. Moreover, increasing government investments in healthcare modernization, rising research activities, and strict quality standards in pharmaceutical production are key factors propelling the adoption of bio decontamination systems in the country.
Bio Decontamination Market Share
The Bio Decontamination industry is primarily led by well-established companies, including:
• STERIS plc (U.S.)
• Ecolab Inc. (U.S.)
• TOMI Environmental Solutions, Inc. (U.S.)
• Bioquell (Ecolab) (U.K.)
• Fedegari Autoclavi S.p.A. (Italy)
• Getinge AB (Sweden)
• Stryker Corporation (U.S.)
• Amira S.r.l. (Italy)
• ClorDiSys Solutions, Inc. (U.S.)
• Noxilizer, Inc. (U.S.)
• Curtiss-Wright Corporation (U.S.)
• Howorth Air Technology Ltd. (U.K.)
• JCE Biotechnology (France)
• Ecolab Life Sciences (U.S.)
• Shibuya Corporation (Japan)
• SOLIDFOG Technologies (U.S.)
• STERIS AST (U.S.)
• Panasonic Healthcare Holdings Co., Ltd. (Japan)
• HART BioMedical, Inc. (U.S.)
• Lindner Group SE (Germany)
Latest Developments in Global Bio Decontamination Market
- In April 2021, Amira Srl, a provider of advanced sterilization and contamination control technologies, announced the launch of Bioreset Max, a compact vapor generator designed for automated hydrogen peroxide bio-decontamination. The system automatically adjusts hydrogen peroxide levels to ensure optimal sterilization efficiency and improved safety in pharmaceutical, laboratory, and healthcare environments, strengthening Amira’s portfolio of advanced decontamination equipment
- In September 2021, STERIS plc, a global leader in infection prevention and sterilization solutions, launched the VHP 100i and VHP 1000i vaporized hydrogen peroxide bio-decontamination systems. These systems were developed for pharmaceutical and medical device manufacturing facilities and are designed to deliver up to a 6-log reduction in microbial contamination while providing enhanced automation, monitoring, and user-friendly operational controls
- In June 2023, Getinge Group, a global medical technology company specializing in infection control and sterilization solutions, introduced an integrated hydrogen peroxide vapor (HPV) decontamination system compatible with HVAC environments. The system was designed to enhance sterility levels in pharmaceutical cleanrooms and laboratory facilities by enabling automated large-area decontamination with improved cycle control and operational efficiency
- In September 2024, Diversey Holdings, a provider of hygiene and infection-prevention technologies, announced a strategic partnership with Palmer Scientific to co-develop advanced surface disinfectants and automated bio-decontamination workflows for large healthcare facilities. The collaboration focuses on developing next-generation infection control solutions that integrate advanced disinfectant chemistry with automated room-decontamination systems to improve safety and operational efficiency
- In March 2025, STERIS plc, a global provider of infection prevention products and services, announced the acquisition of CleanSafe Solutions, a company specializing in on-site sterilization and decontamination services. This acquisition aims to expand STERIS’s global bio-decontamination service capabilities and integrate CleanSafe’s advanced sterilization technologies with its existing infection prevention portfolio, strengthening its position in the global bio-decontamination market
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.