Global Direct Lateral Interbody Fusion Dlif And Extreme Lateral Interbody Fusion Xlif Implants Market
Market Size in USD Billion
CAGR :
% 
 USD
2.60 Billion
USD
4.53 Billion
2025
2033
USD
2.60 Billion
USD
4.53 Billion
2025
2033
| 2026 –2033 | |
| USD 2.60 Billion | |
| USD 4.53 Billion | |
|
|
|
|
Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Size
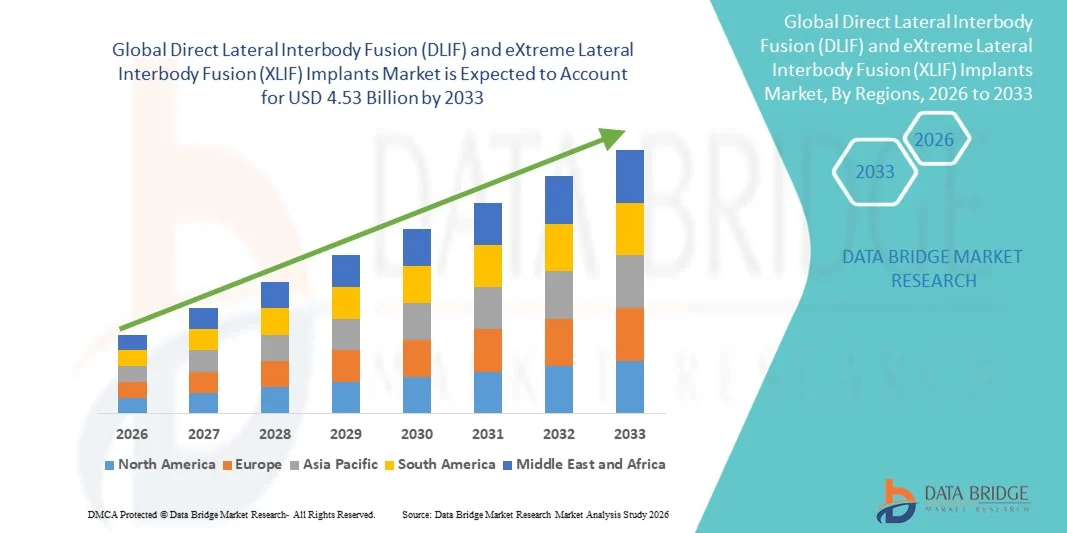
- The global Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) implants market size was valued at USD 2.60 billion in 2025and is expected to reach USD 4.53 billion by 2033, at a CAGR of 7.2% during the forecast period
- The market growth is largely fueled by the rising prevalence of spinal disorders such as degenerative disc disease and lumbar spine instability, along with increasing adoption of minimally invasive spine surgery techniques, growing geriatric population, and expanding preference for faster recovery procedures in both hospital and ambulatory surgical settings
- Furthermore, continuous advancements in lateral access spine surgery technologies, including improved implant designs, expandable cages, and image-guided surgical navigation systems, combined with increasing surgeon preference for reduced tissue disruption approaches, are significantly accelerating the adoption of DLIF and XLIF procedures, thereby driving steady industry growth
Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Analysis
- Lateral Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) implants, used in minimally invasive lateral spine surgery, are increasingly important in modern spinal care as they enable indirect decompression, restoration of disc height, and improved spinal alignment while reducing muscle disruption, blood loss, and recovery time compared to traditional open fusion procedures
- The escalating demand for Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) implants is primarily driven by the rising prevalence of degenerative spine disorders, growing geriatric population, increasing preference for minimally invasive surgical techniques, and continuous advancements in implant materials, cage design, and imaging-guided surgical systems improving procedural accuracy and outcomes
- North America dominated the Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) implants market with the largest revenue share of 40.2% in 2025, supported by advanced healthcare infrastructure, high adoption of minimally invasive spine surgeries, strong presence of key medical device manufacturers, and increasing procedural volumes in the U.S. driven by outpatient spine surgery growth and technological innovation
- Asia-Pacific is expected to be the fastest growing region in the Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) implants market during the forecast period due to improving healthcare access, rising healthcare expenditure, rapid urbanization, and increasing awareness and diagnosis of spinal disorders in countries such as China, India, and Japan
- Degenerative Disc Diseases segment dominated the Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) implants market with a market share of 45.9% in 2025, driven by its high global prevalence among aging populations and strong association with chronic back pain and spinal instability, making it the most frequently treated indication using lateral interbody fusion procedures
Report Scope and Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Segmentation
|
Attributes |
Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America · U.S. · Canada · Mexico Europe · Germany · France · U.K. · Netherlands · Switzerland · Belgium · Russia · Italy · Spain · Turkey · Rest of Europe Asia-Pacific · China · Japan · India · South Korea · Singapore · Malaysia · Australia · Thailand · Indonesia · Philippines · Rest of Asia-Pacific Middle East and Africa · Saudi Arabia · U.A.E. · South Africa · Egypt · Israel · Rest of Middle East and Africa South America · Brazil · Argentina · Rest of South America |
|
Key Market Players |
|
|
Market Opportunities |
· Expansion of ambulatory surgical centers (ASCs) adopting minimally invasive spine fusion procedures · Growing integration of navigation-assisted and robotics-guided spine surgery systems |
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Trends
“Rising Adoption of Minimally Invasive Lateral Spine Surgery Techniques”
- A significant and accelerating trend in the Global Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) implants market is the increasing shift toward minimally invasive lateral access spine surgery, as it enables reduced muscle disruption, lower blood loss, and faster patient recovery compared to traditional posterior fusion approaches
- For instance, the growing use of DLIF and XLIF procedures in treating degenerative lumbar conditions has expanded significantly across tertiary care hospitals adopting minimally invasive spine surgery programs
- Advancements in implant design such as expandable interbody cages and anatomically optimized spacers are improving spinal alignment correction and fusion success rates in DLIF and XLIF procedures, while reducing revision surgery risks and enhancing long-term clinical outcomes
- The integration of intraoperative imaging and navigation-assisted spine systems is enabling surgeons to perform DLIF and XLIF procedures with higher precision and reduced complication risks, supporting wider adoption in complex spinal deformity cases
- This trend toward less invasive, precision-driven spine surgery is reshaping clinical expectations in spinal care, leading companies such as Medtronic and NuVasive to continuously develop next-generation lateral fusion implant systems
- The demand for DLIF and XLIF implants is rising rapidly across hospitals and ambulatory surgical centers as healthcare providers increasingly prioritize shorter hospital stays, reduced complications, and cost-effective spinal treatment solutions
Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Dynamics
Driver
“Rising Burden of Degenerative Spine Disorders and Aging Population”
- The increasing prevalence of degenerative spine conditions such as degenerative disc disease, spondylolisthesis, and adult spinal deformities, coupled with the rapidly growing geriatric population, is a major driver for the Global Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) implants market
- For instance, in April 2025, leading spine device manufacturers expanded clinical adoption programs for lateral fusion systems to support higher procedural volumes in aging populations requiring spinal stabilization surgeries
- As patients increasingly seek durable surgical solutions for chronic back pain and spinal instability, DLIF and XLIF procedures offer clinically effective outcomes through indirect decompression and improved spinal alignment
- Furthermore, the rising preference for minimally invasive procedures among surgeons and patients is driving adoption of lateral interbody fusion techniques due to reduced postoperative complications and faster functional recovery
- The growing availability of advanced surgical training programs and increasing surgeon familiarity with lateral access techniques are further supporting procedural expansion in both developed and emerging healthcare markets
- Increasing healthcare expenditure and expanding insurance coverage for advanced spinal procedures are also enabling greater patient access to DLIF and XLIF implant-based treatments, particularly in urban tertiary care centers
- Continuous innovation by medical device companies in biomaterials and 3D-printed porous implant structures is further enhancing fusion rates and supporting wider clinical adoption
Restraint/Challenge
“High Procedure Complexity and Risk of Neurological Complications”
- Concerns related to the technical complexity of DLIF and XLIF procedures and the risk of neurological complications, particularly nerve injury during lateral access to the spine, pose a significant challenge to market expansion
- For instance, reported cases of transient thigh pain or lumbar plexus irritation associated with lateral approaches have made some surgeons cautious in adopting these techniques for high-risk patients
- Addressing these challenges requires advanced surgeon training, improved intraoperative neuromonitoring systems, and enhanced imaging guidance to reduce procedural risks and improve safety outcomes
- In addition, the relatively high cost of DLIF and XLIF implant systems compared to traditional spinal fusion devices can limit adoption in cost-sensitive healthcare systems, particularly in emerging economies
- While technological advancements are improving safety and outcomes, variability in surgical expertise and steep learning curves continue to restrict widespread adoption of lateral fusion techniques in some clinical settings
- Regulatory approval processes for new implant designs can also delay product commercialization, slowing down the introduction of advanced DLIF and XLIF solutions in certain regions
- Furthermore, post-surgical complications requiring revision surgeries in complex cases can increase overall treatment burden, reducing physician confidence in specific patient populations
Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Scope
The market is segmented on the basis of indication and end user.
- By Indication
On the basis of indication, the Global Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) implants market is segmented into degenerative disc diseases, spondylolisthesis, scoliosis, and others. Degenerative Disc Diseases segment dominated the market with the largest revenue share 45.9% in 2025, driven by its high global prevalence among aging populations and strong association with chronic lower back pain and spinal instability. This condition is one of the most common spinal disorders requiring surgical intervention, making it the primary indication for DLIF and XLIF procedures. The increasing incidence of age-related spinal degeneration, sedentary lifestyles, and obesity is further contributing to procedural volumes. In addition, DLIF and XLIF techniques are highly effective in restoring disc height and achieving indirect decompression in degenerative disc disease cases, which strengthens their clinical preference. Hospitals and spine surgeons widely adopt these procedures due to predictable outcomes and reduced surgical morbidity compared to traditional open fusion techniques. The strong reimbursement support in developed healthcare systems also reinforces the dominance of this segment.
Scoliosis segment is expected to witness the fastest growth rate from 2026 to 2033, driven by increasing adoption of minimally invasive correction techniques for adult spinal deformities. Rising prevalence of degenerative scoliosis in the aging population is significantly contributing to the demand for lateral interbody fusion procedures. DLIF and XLIF implants offer improved spinal alignment correction with reduced blood loss and shorter recovery times compared to conventional posterior fusion approaches, making them increasingly preferred in complex deformity cases. Advancements in surgical navigation and neuromonitoring systems are also improving the safety profile of scoliosis correction surgeries. Furthermore, growing surgeon expertise in lateral access techniques is expanding the use of DLIF and XLIF implants in deformity management. Increasing patient preference for less invasive procedures with faster rehabilitation is further accelerating growth in this segment.
- By End User
On the basis of end user, the Global Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) implants market is segmented into hospitals, ambulatory surgical units, clinics, and others. Hospitals segment dominated the market with the largest revenue share in 2025, driven by high patient inflow for complex spinal surgeries and availability of advanced surgical infrastructure. Hospitals are the primary centers for performing DLIF and XLIF procedures due to the requirement of specialized operating rooms, imaging systems, and skilled spine surgeons. The presence of multidisciplinary teams including orthopedic and neurosurgical specialists further supports hospital dominance in complex spinal fusion cases. In addition, hospitals handle a large proportion of degenerative disc disease and deformity-related surgeries, which are the key indications for these implants. Strong reimbursement frameworks and established surgical workflows also contribute to higher adoption in hospital settings. The ability to manage postoperative care and potential complications further reinforces hospitals as the leading end-user segment.
Ambulatory Surgical Units segment is expected to witness the fastest growth rate from 2026 to 2033, driven by the increasing shift toward minimally invasive and outpatient spine procedures. Advances in DLIF and XLIF techniques are enabling shorter hospital stays and faster patient recovery, making outpatient surgical centers more viable for selected cases. Rising healthcare cost pressures are encouraging payers and providers to adopt ambulatory surgical settings for appropriate spinal procedures. Improvements in surgical efficiency, anesthesia techniques, and postoperative care protocols are also supporting this shift. Patients increasingly prefer ambulatory centers due to reduced hospitalization time and lower overall treatment costs. Furthermore, expansion of specialized spine-focused surgical centers is accelerating the adoption of DLIF and XLIF implants in this segment.
Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Regional Analysis
- North America dominated the Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) implants market with the largest revenue share of 40.2% in 2025, supported by advanced healthcare infrastructure, high adoption of minimally invasive spine surgeries, strong presence of key medical device manufacturers
- The region benefits from advanced healthcare infrastructure and widespread availability of specialized spine surgery centers, which support high procedural volumes of DLIF and XLIF surgeries across hospitals and ambulatory surgical settings
- Surgeons in North America show strong preference for lateral interbody fusion techniques due to improved clinical outcomes, reduced recovery time, and lower complication rates compared to traditional open spinal fusion methods, further strengthening market dominance
U.S. Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Insight
The U.S. market captured the largest revenue share of 80% in North America in 2025, driven by high adoption of minimally invasive spine surgery and a rising burden of degenerative spinal disorders. The country benefits from advanced healthcare infrastructure, strong presence of leading spine implant manufacturers, and widespread availability of skilled spine surgeons performing DLIF and XLIF procedures. Increasing preference for outpatient and ambulatory spine surgeries, along with strong reimbursement support for spinal fusion procedures, is further boosting market growth. In addition, continuous technological innovation in expandable cages, navigation-assisted surgery, and biologics integration is significantly contributing to market expansion in the U.S.
Europe Global Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Insight
The Europe market is projected to expand at a steady CAGR throughout the forecast period, primarily driven by the growing geriatric population and increasing prevalence of degenerative spine conditions. Rising adoption of minimally invasive spinal fusion techniques across Germany, France, and Italy is supporting market growth. European healthcare systems are increasingly focusing on reducing hospital stays and improving surgical outcomes, which is encouraging the use of DLIF and XLIF implants. Furthermore, favorable regulatory frameworks and growing awareness among surgeons regarding lateral access spine surgery techniques are fostering gradual but consistent adoption across the region.
U.K. Global Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Insight
The U.K. market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by increasing demand for minimally invasive spinal procedures and rising incidence of chronic back disorders. Growing emphasis on improving patient recovery time and reducing surgical complications is encouraging hospitals to adopt DLIF and XLIF techniques. The country’s strong public and private healthcare infrastructure supports access to advanced spine surgery technologies. In addition, rising awareness among patients regarding modern spinal treatment options and increasing adoption of outpatient surgical models are further contributing to market growth in the U.K.
Germany Global Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Insight
The Germany market is expected to expand at a considerable CAGR during the forecast period, supported by strong healthcare infrastructure and high focus on technological innovation in surgical procedures. Increasing prevalence of degenerative disc diseases and spinal deformities is driving demand for advanced spinal fusion solutions. Germany’s emphasis on precision medicine and minimally invasive surgical techniques is promoting adoption of DLIF and XLIF implants. Furthermore, strong investment in hospital modernization and growing preference for motion-preserving and less invasive spine surgeries are contributing to steady market expansion.
Asia-Pacific Global Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Insight
The Asia-Pacific market is poised to grow at the fastest CAGR of around 8–9% during the forecast period, driven by rising healthcare expenditure, improving hospital infrastructure, and increasing burden of spinal disorders. Countries such as China, Japan, and India are witnessing rapid adoption of minimally invasive spine surgery techniques. Growing medical tourism, expanding access to advanced surgical technologies, and increasing awareness of modern spinal treatment options are further accelerating market growth. In addition, the presence of cost-competitive manufacturing and rising number of trained spine surgeons are supporting regional expansion.
Japan Global Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Insight
The Japan market is gaining momentum due to its rapidly aging population and high prevalence of degenerative spinal conditions requiring surgical intervention. Strong adoption of advanced medical technologies and minimally invasive surgical procedures is driving DLIF and XLIF implant usage. The country’s healthcare system emphasizes precision, safety, and faster recovery, which aligns well with lateral interbody fusion techniques. Furthermore, integration of robotic-assisted and navigation-guided spine surgery systems is enhancing procedural accuracy and supporting market growth in Japan.
India Global Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Insight
The India market accounted for the largest share in Asia-Pacific in 2025, attributed to rising prevalence of spinal disorders, expanding middle-class population, and increasing access to advanced healthcare services. Rapid urbanization and growing awareness of minimally invasive spine surgery techniques are driving adoption of DLIF and XLIF implants. The expansion of multispecialty hospitals and spine care centers, along with improving affordability of advanced surgical procedures, is further supporting market growth. In addition, government initiatives to strengthen healthcare infrastructure and rising medical tourism are contributing to increased procedural volumes in India.
Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market Share
The Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants industry is primarily led by well-established companies, including:
- Medtronic (Ireland)
- Stryker (U.S.)
- NuVasive, Inc. (U.S.)
- Globus Medical, Inc. (U.S.)
- Zimmer Biomet (U.S.)
- Orthofix Medical Inc. (U.S.)
- Alphatec Holdings, Inc. (U.S.)
- Spine Wave, Inc. (U.S.)
- Spineology, Inc. (U.S.)
- Aesculap Implant Systems (Germany)
- B. Braun SE (Germany)
- RTI Surgical Holdings, Inc. (U.S.)
- SeaSpine Holdings Corporation (U.S.)
- Life Spine, Inc. (U.S.)
- CoreLink LLC (U.S.)
- Aurora Spine Corporation (Canada)
- Xtant Medical Holdings, Inc. (U.S.)
- K2M Group Holdings, Inc. (U.S.)
- Captiva Spine, Inc. (U.S.)
What are the Recent Developments in Global Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants Market?
- In February 2026, Medtronic announced an expansion of FDA approvals supporting biologics used in spine fusion procedures, including applications that complement lateral interbody fusion systems such as Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants. The expanded indication enhances bone graft performance in lumbar interbody fusion surgeries, improving fusion rates and postoperative outcomes in degenerative spine conditions
- In June 2024, multiple regulatory approvals and product clearances were granted for next-generation spinal implant technologies, including expandable and 3D-printed interbody cages used in procedures such as Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants. These innovations focus on improving implant fixation, restoring disc height, and enhancing spinal alignment in minimally invasive surgery. The new generation of porous titanium and patient-specific cage designs improves osseointegration and long-term fusion outcomes
- In September 2023, Globus Medical completed its merger with NuVasive, a major developer of XLIF technology used in Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants procedures. The merger combined complementary minimally invasive spine portfolios, strengthening global leadership in lateral spine surgery technologies. It expanded access to advanced lateral interbody fusion systems, robotic-assisted spine platforms, and expandable cage technologies. The integration enhanced R&D capabilities and global commercialization reach in spine surgery solutions
- In May 2022, global spine surgery education programs expanded significantly, focusing on training surgeons in minimally invasive lateral approaches used in Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants procedures. These initiatives emphasized safe transpsoas access, neuromonitoring techniques, and complication management strategies. Increased hands-on training improved surgeon proficiency in complex spinal deformity correction cases. This contributed to higher adoption of lateral interbody fusion techniques across hospitals worldwide
- In March 2021, advancements in robotic-assisted and navigation-guided spine surgery systems were increasingly integrated with Direct Lateral Interbody Fusion (DLIF) and eXtreme Lateral Interbody Fusion (XLIF) Implants procedures. These technologies improved implant positioning accuracy and reduced intraoperative risks during lateral access spine surgeries. The integration of imaging guidance with surgical robotics enabled enhanced precision in complex spinal deformity correction
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.