Global Immune Checkpoint Inhibitors Market
Market Size in USD Billion
CAGR :
% 
 USD
68.47 Billion
USD
260.89 Billion
2025
2033
USD
68.47 Billion
USD
260.89 Billion
2025
2033
| 2026 –2033 | |
| USD 68.47 Billion | |
| USD 260.89 Billion | |
|
|
|
|
Immune Checkpoint Inhibitors Market Size
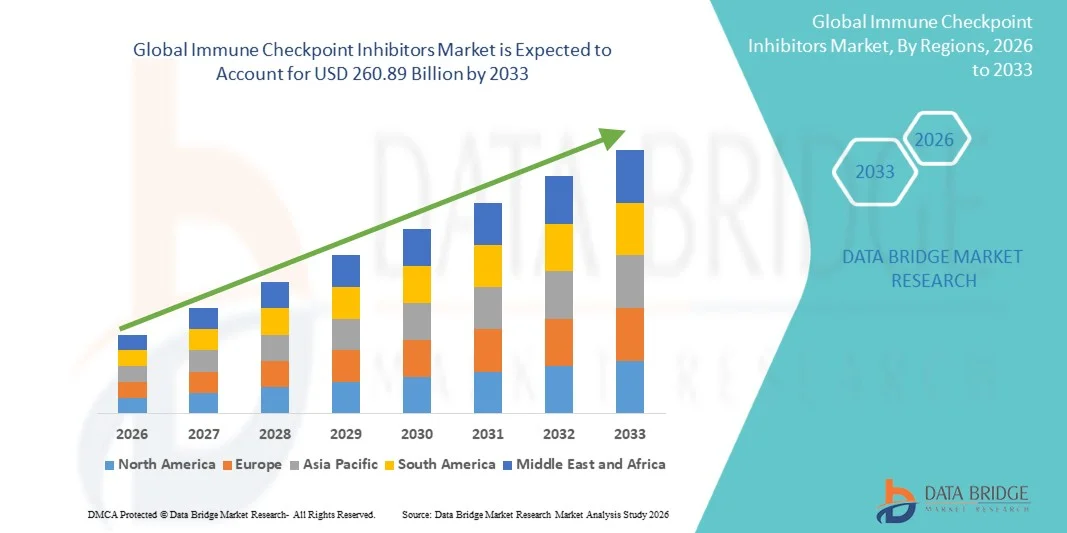
- The global immune checkpoint inhibitors market size was valued at USD 68.47 billion in 2025 and is expected to reach USD 260.89 billion by 2033, at a CAGR of 18.2% during the forecast period
- The market growth is largely fueled by the increasing global burden of cancer and the rising demand for advanced immunotherapy treatments, leading to significant adoption of immune checkpoint inhibitors across oncology care settings
- Furthermore, growing advancements in precision medicine, biomarker-based patient selection, and combination therapy approaches are positioning checkpoint inhibitors as a preferred treatment modality. These converging factors are accelerating the clinical adoption of immunotherapies, thereby significantly boosting the industry’s growth
Immune Checkpoint Inhibitors Market Analysis
- Immune checkpoint inhibitors are a class of immunotherapy drugs that enhance the body’s immune response against cancer cells by targeting regulatory pathways such as PD-1, PD-L1, and CTLA-4. These therapies are widely used across multiple cancer types including lung cancer, melanoma, and renal cell carcinoma, improving survival outcomes and treatment efficacy
- The escalating demand for immune checkpoint inhibitors is primarily driven by increasing cancer prevalence, continuous regulatory approvals, expanding clinical indications, and strong investment in oncology research, which is accelerating innovation and broadening the application scope of immuno-oncology treatments
- North America dominated the immune checkpoint inhibitors market with a share of 63.31% due to the strong presence of leading biopharmaceutical companies and high adoption of advanced immunotherapy treatments across oncology care
- Asia-Pacific is expected to be the fastest growing region in the immune checkpoint inhibitors market during the forecast period due to rising cancer burden, improving healthcare infrastructure, and increasing access to advanced treatment options in emerging economies
- PD-1 segment dominated the market with a market share of 73.8% due to its widespread clinical usage across multiple cancer types and strong therapeutic outcomes. PD-1 inhibitors demonstrate higher efficacy with relatively manageable safety profiles, making them a preferred choice among oncologists
Report Scope and Immune Checkpoint Inhibitors Market Segmentation
|
Attributes |
Immune Checkpoint Inhibitors Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Immune Checkpoint Inhibitors Market Trends
“Rising Adoption of Combination Immunotherapy Regimens”
- A significant trend in the immune checkpoint inhibitors market is the increasing adoption of combination immunotherapy regimens, driven by the need to improve treatment efficacy and overcome resistance observed in monotherapy approaches across multiple cancer types. This trend is strengthening the role of checkpoint inhibitors as core components in advanced oncology treatment protocols and expanding their clinical applicability
- For instance, Bristol Myers Squibb has widely advanced the combination use of Opdivo and Yervoy across melanoma and lung cancer indications, demonstrating improved survival outcomes compared to single-agent therapies. Such developments are reinforcing the clinical preference for dual checkpoint blockade strategies and supporting broader market adoption
- The integration of checkpoint inhibitors with chemotherapy and targeted therapies is gaining traction as it enhances immune response activation and increases patient response rates. This shift is driving pharmaceutical companies to focus on multi-drug regimens that address tumor heterogeneity and improve long-term outcomes
- Ongoing clinical trials evaluating novel combinations are expanding treatment possibilities across solid tumors and hematologic malignancies. These efforts are accelerating innovation pipelines and contributing to a more diversified therapeutic landscape within immuno-oncology
- Healthcare providers are increasingly adopting personalized treatment approaches where combination therapies are selected based on biomarker profiles and disease characteristics. This is enhancing precision in treatment delivery and improving overall clinical success rates
- The growing body of clinical evidence supporting combination therapies continues to strengthen their adoption across global oncology practices. This trend is shaping the future direction of cancer treatment and reinforcing the expansion of the immune checkpoint inhibitors market
Immune Checkpoint Inhibitors Market Dynamics
Driver
“Increasing Global Cancer Burden Driving Demand for Immunotherapy”
- The rising global incidence of cancer is significantly driving demand for immune checkpoint inhibitors as healthcare systems increasingly prioritize advanced treatment solutions that improve survival outcomes and quality of life for patients. The growing prevalence of cancers such as lung, breast, and colorectal cancer is expanding the patient pool eligible for immunotherapy
- For instance, World Health Organization has reported a continuous increase in global cancer cases, prompting pharmaceutical leaders such as Merck & Co. to expand the indications of KEYTRUDA across multiple tumor types. Such initiatives are enhancing treatment accessibility and driving widespread adoption of checkpoint inhibitors
- The growing awareness among patients and healthcare providers regarding the benefits of immunotherapy is accelerating its integration into standard cancer treatment protocols. This awareness is contributing to earlier diagnosis and timely intervention using advanced therapies
- Strong investments in oncology research and development are enabling continuous innovation in checkpoint inhibitor drugs, improving their efficacy and safety profiles. These advancements are further strengthening their role as a preferred treatment option
- The sustained increase in cancer burden combined with advancements in immuno-oncology is reinforcing the long-term demand for checkpoint inhibitors. This driver continues to play a critical role in shaping market expansion and innovation
Restraint/Challenge
“High Cost of Treatment and Immune-Related Adverse Effects”
- The immune checkpoint inhibitors market faces significant challenges due to the high cost of treatment, which limits accessibility for patients, particularly in low- and middle-income regions. These therapies often require prolonged treatment durations, further increasing the overall financial burden on healthcare systems and patients
- For instance, therapies such as KEYTRUDA by Merck & Co. and Opdivo by Bristol Myers Squibb are associated with high treatment costs, which can restrict widespread adoption despite their clinical benefits. This cost barrier is creating disparities in access to advanced cancer therapies
- Immune-related adverse effects such as inflammation of organs and autoimmune reactions present additional challenges in treatment management. These side effects require careful monitoring and may lead to discontinuation in certain patients
- The complexity of managing adverse reactions increases the burden on healthcare providers and necessitates specialized clinical expertise. This can limit the use of checkpoint inhibitors in regions with limited healthcare infrastructure
- The combination of high costs and safety concerns continues to pose a challenge for market expansion. Addressing these issues through pricing strategies and improved safety profiles remains critical for sustaining long-term growth in the immune checkpoint inhibitors market
Immune Checkpoint Inhibitors Market Scope
The market is segmented on the basis of indication, target, route of administration, end-users, and distribution channel.
• By Indication
On the basis of indication, the immune checkpoint inhibitors market is segmented into breast cancer, bladder cancer, cervical cancer, Hodgkin lymphoma, liver cancer, lung cancer, kidney cancer, solid cancer, and others. The lung cancer segment dominated the largest market revenue share in 2025, driven by the high global prevalence of non-small cell lung cancer and the strong clinical efficacy of checkpoint inhibitors in improving survival outcomes. The widespread adoption of PD-1 and PD-L1 inhibitors as first-line and second-line therapies has significantly strengthened segment growth. Increasing approvals of combination therapies and rising biomarker-based patient selection have further supported demand. Continuous advancements in immuno-oncology research and expanding clinical trial pipelines also contribute to sustained dominance of this segment.
The breast cancer segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by increasing clinical evidence supporting the use of checkpoint inhibitors in triple-negative breast cancer. Rising awareness regarding immunotherapy benefits and expanding regulatory approvals for combination regimens are accelerating adoption. Pharmaceutical companies are actively investing in trials to enhance response rates in broader breast cancer subtypes. Improved diagnostic techniques for identifying eligible patients and growing oncology treatment infrastructure are further supporting segment expansion.
• By Target
On the basis of target, the immune checkpoint inhibitors market is segmented into PD-1, PD-L1, CTLA-4, and others. The PD-1 segment held the largest market revenue share of 73.8% in 2025, driven by its widespread clinical usage across multiple cancer types and strong therapeutic outcomes. PD-1 inhibitors demonstrate higher efficacy with relatively manageable safety profiles, making them a preferred choice among oncologists. Extensive approvals across indications such as lung, melanoma, and kidney cancers have strengthened their market position. Continuous innovation in combination therapies and biomarker-driven treatments further reinforces dominance.
The PD-L1 segment is expected to witness the fastest CAGR from 2026 to 2033, driven by increasing adoption in combination with chemotherapy and targeted therapies. Growing focus on precision medicine and patient stratification is enhancing the clinical relevance of PD-L1 inhibitors. Pharmaceutical companies are expanding research pipelines to improve response rates and broaden indication coverage. Rising approvals and favorable reimbursement scenarios are also contributing to accelerated growth in this segment.
• By Route of Administration
On the basis of route of administration, the immune checkpoint inhibitors market is segmented into oral, parenteral, and others. The parenteral segment dominated the largest market revenue share in 2025, driven by the intravenous administration of most approved checkpoint inhibitors ensuring controlled dosing and rapid therapeutic action. Hospitals and oncology centers prefer parenteral routes due to their established clinical protocols and higher bioavailability. The ability to monitor patients closely during infusion enhances safety and treatment outcomes. Strong regulatory approvals for injectable biologics further support this segment’s dominance.
The oral segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by ongoing research focused on improving patient convenience and adherence. Oral formulations offer ease of administration and reduce the need for frequent hospital visits. Advancements in drug delivery technologies and increasing investment in oral immunotherapy development are driving innovation. Growing preference for home-based treatment options is also supporting the expansion of this segment.
• By End-Users
On the basis of end-users, the immune checkpoint inhibitors market is segmented into hospitals, specialty clinics, and others. The hospitals segment held the largest market revenue share in 2025, driven by the availability of advanced oncology infrastructure and skilled healthcare professionals. Hospitals serve as primary centers for administering immunotherapies due to the need for continuous monitoring and management of adverse effects. High patient inflow and access to multidisciplinary care further strengthen their dominance. Increasing investments in cancer treatment facilities also support growth.
The specialty clinics segment is expected to witness the fastest CAGR from 2026 to 2033, driven by the rising trend of decentralized cancer care and personalized treatment approaches. Specialty clinics offer focused oncology services with shorter waiting times and improved patient experience. Growing demand for outpatient immunotherapy services and advancements in clinical capabilities are accelerating their adoption. Expansion of private oncology networks is further contributing to segment growth.
• By Distribution Channel
On the basis of distribution channel, the immune checkpoint inhibitors market is segmented into hospital pharmacy, retail pharmacy, and others. The hospital pharmacy segment dominated the largest market revenue share in 2025, driven by the high volume of immunotherapy drugs administered within hospital settings. Strict regulatory requirements and the need for controlled storage and handling of biologics support the dominance of hospital pharmacies. Direct procurement and distribution within hospitals ensure timely availability of therapies. Increasing hospitalization rates for cancer treatment further strengthen this segment.
The retail pharmacy segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by the gradual expansion of specialty drug distribution outside hospital settings. Increasing availability of certain immunotherapy drugs through retail channels and growing patient preference for convenient access are supporting growth. Advancements in cold chain logistics and pharmacy capabilities are enabling wider distribution. Rising focus on outpatient care models is also contributing to the expansion of this segment.
Immune Checkpoint Inhibitors Market Regional Analysis
- North America dominated the immune checkpoint inhibitors market with the largest revenue share of 63.31% in 2025, driven by the strong presence of leading biopharmaceutical companies and high adoption of advanced immunotherapy treatments across oncology care
- Patients in the region benefit from early access to innovative therapies supported by favorable reimbursement frameworks and extensive clinical research activities focused on cancer treatment advancements
- This widespread adoption is further supported by increasing cancer prevalence, robust healthcare infrastructure, and strong investment in precision medicine, establishing immune checkpoint inhibitors as a critical component of modern cancer therapeutics
U.S. Immune Checkpoint Inhibitors Market Insight
The U.S. immune checkpoint inhibitors market captured the largest revenue share within North America in 2025, fueled by rapid regulatory approvals and high healthcare spending on advanced oncology treatments. The country benefits from a well-established clinical trial ecosystem and strong presence of key market players driving innovation in immunotherapy. Increasing adoption of combination therapies and biomarker-driven treatment approaches is further enhancing treatment outcomes. Moreover, growing awareness among patients and healthcare providers regarding immunotherapy benefits continues to support market expansion.
Europe Immune Checkpoint Inhibitors Market Insight
The Europe immune checkpoint inhibitors market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by increasing cancer incidence and supportive government initiatives for oncology research. The region is witnessing rising adoption of immunotherapy as a preferred treatment option across multiple cancer indications. Strong regulatory frameworks and reimbursement policies are facilitating access to advanced therapies. Growth in clinical collaborations and research funding is further strengthening the regional market landscape.
U.K. Immune Checkpoint Inhibitors Market Insight
The U.K. immune checkpoint inhibitors market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by increasing focus on early cancer diagnosis and access to innovative treatment options. The presence of national healthcare programs supporting cancer care enhances patient access to immunotherapies. Rising investment in oncology research and collaborations between public and private sectors are accelerating adoption. Growing awareness regarding personalized medicine is also contributing to market growth.
Germany Immune Checkpoint Inhibitors Market Insight
The Germany immune checkpoint inhibitors market is expected to expand at a considerable CAGR during the forecast period, fueled by advanced healthcare infrastructure and strong emphasis on research and innovation in oncology. The country’s focus on precision medicine and adoption of cutting-edge therapies is driving demand for checkpoint inhibitors. Increasing clinical trials and collaborations between research institutions and pharmaceutical companies further support growth. High healthcare expenditure and patient awareness also contribute to expanding adoption.
Asia-Pacific Immune Checkpoint Inhibitors Market Insight
The Asia-Pacific immune checkpoint inhibitors market is poised to grow at the fastest CAGR during the forecast period of 2026 to 2033, driven by rising cancer burden, improving healthcare infrastructure, and increasing access to advanced treatment options in emerging economies. China is anticipated to be the fastest growing country in this region, driven by rapid expansion of oncology treatment facilities, strong government support for cancer care, and increasing domestic production of immunotherapy drugs. The region is also benefiting from growing medical tourism, increasing awareness regarding immunotherapy, and rising investments by global pharmaceutical companies, supporting wider adoption across diverse patient populations.
Japan Immune Checkpoint Inhibitors Market Insight
The Japan immune checkpoint inhibitors market is gaining momentum due to the country’s strong focus on advanced healthcare technologies and increasing adoption of innovative cancer treatments. The aging population and rising cancer incidence are significantly driving demand for effective immunotherapies. Integration of precision medicine approaches and biomarker testing is enhancing treatment outcomes. Continuous research and development efforts are further supporting market growth.
China Immune Checkpoint Inhibitors Market Insight
The China immune checkpoint inhibitors market accounted for the largest market revenue share in Asia Pacific in 2025, attributed to the country’s large patient population and rapidly evolving healthcare infrastructure. Increasing government initiatives to improve cancer diagnosis and treatment accessibility are driving adoption. Strong presence of domestic pharmaceutical companies and growing investment in immuno-oncology research are accelerating market expansion. Rising affordability and wider availability of checkpoint inhibitors are also contributing to growth.
Immune Checkpoint Inhibitors Market Share
The immune checkpoint inhibitors industry is primarily led by well-established companies, including:
- Merck & Co., Inc. (U.S.)
- Bristol-Myers Squibb Company (U.S.)
- Novartis AG (Switzerland)
- AbbVie Inc. (U.S.)
- Eli Lilly and Company (U.S.)
- F. Hoffmann-La Roche Ltd. (Switzerland)
- Regeneron Pharmaceuticals Inc. (U.S.)
- Johnson & Johnson Services, Inc. (U.S.)
- AstraZeneca (U.K.)
- Bausch Health Companies Inc. (Canada)
- GlaxoSmithKline plc (U.K.)
- Teva Pharmaceutical Industries Ltd. (Israel)
- Sanofi (France)
- Sun Pharmaceutical Industries Ltd. (India)
- EMD Serono, Inc. (U.S.)
- Pfizer Inc. (U.S.)
Latest Developments in Global Immune Checkpoint Inhibitors Market
- In March 2025, Bristol Myers Squibb reported expanded clinical success of Opdivo in combination immunotherapy trials across multiple solid tumors, with late-stage data indicating significantly improved progression-free and overall survival rates compared to monotherapy approaches. This advancement is accelerating the clinical shift toward combination-based immunotherapy regimens, strengthening physician confidence in multi-drug protocols and increasing treatment adoption across broader oncology settings. As a result, the development is enhancing revenue generation potential, extending product lifecycle value, and intensifying competition among key players focusing on combination checkpoint inhibitor strategies
- In November 2024, Merck & Co. announced positive Phase III trial outcomes for KEYTRUDA in earlier-stage cancer indications, supporting its integration into adjuvant and neoadjuvant treatment settings beyond advanced metastatic disease. This expansion into earlier lines of therapy is significantly increasing the eligible patient population and extending treatment duration, which directly contributes to higher drug utilization rates. The development is also reinforcing the dominance of PD-1 inhibitors in clinical protocols while driving long-term market growth through earlier intervention strategies and improved survival outcomes
- In October 2023, Bristol Myers Squibb received U.S. FDA approval for Opdivo for the treatment of completely resected stage IIB or IIC melanoma in adult and pediatric patients aged 12 years and above. This approval marked a critical expansion into earlier-stage melanoma treatment, enabling the use of checkpoint inhibitors in adjuvant settings to reduce recurrence risk. The development is increasing treatment accessibility, driving higher adoption among oncologists, and strengthening the drug’s market share by capturing a larger segment of melanoma patients at earlier disease stages
- In September 2023, Merck & Co. achieved priority review acceptance from the U.S. FDA for a supplemental application of WELIREG targeting previously treated renal cell carcinoma patients. This regulatory milestone is expected to accelerate the approval timeline and facilitate faster commercialization, enabling earlier patient access to innovative targeted therapies. The development supports portfolio expansion within oncology and enhances competitive positioning by introducing differentiated treatment options, ultimately contributing to revenue diversification and market growth
- In January 2023, Merck & Co. entered into a clinical collaboration with Teon Therapeutics to evaluate TT-816 in combination with KEYTRUDA for patients with advanced solid tumors. This collaboration is advancing the exploration of novel combination therapies aimed at overcoming resistance mechanisms and improving patient response rates. The initiative is fostering innovation in immuno-oncology pipelines, encouraging strategic partnerships across the industry, and expanding future growth opportunities by unlocking new therapeutic pathways within the immune checkpoint inhibitors market
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.