Global Multiplex Testing Market
Market Size in USD Billion
CAGR :
% 
 USD
2.77 Billion
USD
5.32 Billion
2025
2033
USD
2.77 Billion
USD
5.32 Billion
2025
2033
| 2026 –2033 | |
| USD 2.77 Billion | |
| USD 5.32 Billion | |
|
|
|
|
Multiplex Testing Market Size
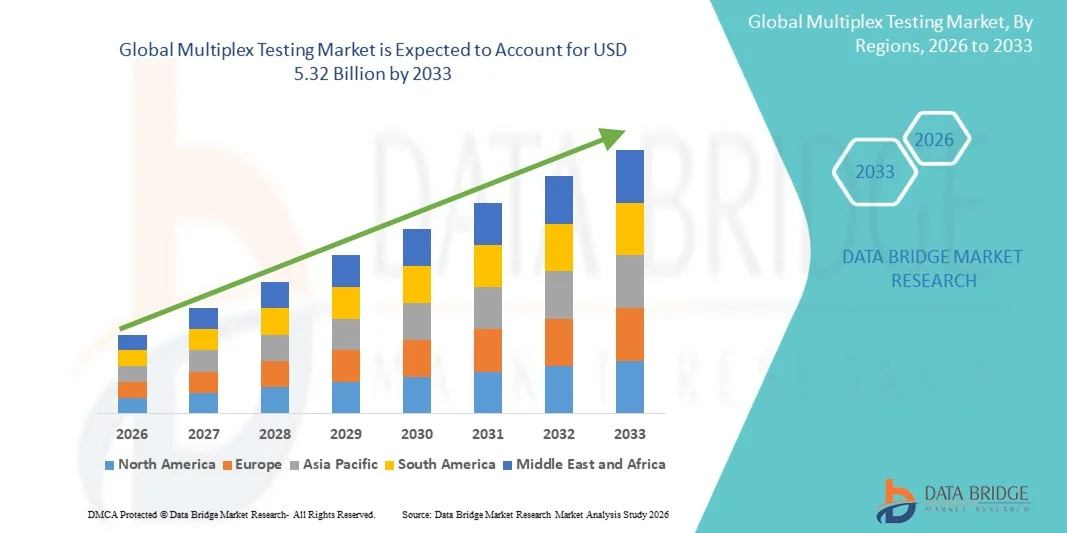
- The global multiplex testing market size was valued at USD 2.77 billion in 2025 and is expected to reach USD 5.32 billion by 2033, at a CAGR of 8.50% during the forecast period
- The market growth is primarily driven by the increasing demand for high-throughput diagnostic solutions, rising prevalence of infectious and chronic diseases, and the growing adoption of advanced molecular diagnostic technologies across clinical laboratories and research settings
- Furthermore, the shift toward cost-efficient, rapid, and accurate multi-analyte testing platforms is strengthening clinical decision-making capabilities. These factors, combined with continuous technological advancements in genomics and proteomics, are significantly accelerating the adoption of multiplex testing solutions, thereby boosting overall market expansion
Multiplex Testing Market Analysis
- Multiplex testing, enabling simultaneous detection of multiple biomarkers or pathogens in a single assay, is increasingly vital in modern diagnostics and biomedical research due to its enhanced efficiency, reduced sample volume requirements, and ability to deliver rapid, high-accuracy results across clinical and laboratory environments
- The escalating demand for multiplex testing is primarily fueled by the rising prevalence of infectious diseases, cancer, and genetic disorders, along with growing demand for cost-effective, high-throughput diagnostic solutions and continuous advancements in molecular diagnostic technologies
- North America dominated the multiplex testing market with the largest revenue share of 39.8% in 2025, characterized by advanced healthcare infrastructure, strong adoption of precision medicine, and high R&D investments, with the U.S. experiencing substantial growth in multiplex assay deployment across clinical diagnostics and research laboratories
- Asia-Pacific is expected to be the fastest growing region in the multiplex testing market during the forecast period due to increasing healthcare expenditure, expanding diagnostic laboratory networks, and rising awareness regarding early and accurate disease detection
- ELISA (Enzyme-Linked Immunosorbent Assay) segment dominated the multiplex testing market with a significant market share of 41.6% in 2025, driven by its high sensitivity, cost-effectiveness, and widespread clinical adoption for infectious disease screening, oncology diagnostics, and autoimmune disorder testing
Report Scope and Multiplex Testing Market Segmentation
|
Attributes |
Multiplex Testing Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
Multiplex Testing Market Trends
“Rising Shift Toward AI-Enabled High-Throughput Multi-Analyte Platforms”
- A significant and accelerating trend in the global multiplex testing market is the integration of artificial intelligence (AI) and advanced bioinformatics tools into multiplex assay platforms, improving biomarker interpretation, diagnostic speed, and clinical decision support in laboratory workflows
- For instance, Luminex-based multiplex platforms and similar systems are increasingly being paired with AI-driven analytics software to enhance multi-analyte disease profiling and improve accuracy in infectious disease and oncology testing applications
- AI integration in multiplex testing enables automated pattern recognition across large biomarker datasets, improved assay optimization, and more precise differentiation of disease subtypes in complex clinical conditions
- The seamless integration of multiplex testing systems with laboratory information management systems (LIMS) and digital health platforms is enabling centralized data handling, streamlined reporting, and improved workflow efficiency in diagnostic laboratories
- This trend toward highly automated, data-driven, and integrated diagnostic ecosystems is reshaping expectations in clinical diagnostics, with companies such as Bio-Rad and Thermo Fisher Scientific advancing multiplex platforms with enhanced software-driven capabilities
- The demand for AI-enabled multiplex testing solutions is growing rapidly across clinical diagnostics and research settings, as healthcare providers increasingly prioritize speed, accuracy, and multi-disease detection efficiency
- Furthermore, increasing use of multiplex testing in companion diagnostics is supporting targeted therapy development and strengthening personalized medicine adoption worldwide
Multiplex Testing Market Dynamics
Driver
“Rising Burden of Chronic and Infectious Diseases Driving Diagnostic Demand”
- The increasing prevalence of infectious diseases, cancer, and autoimmune disorders is a major driver for the global multiplex testing market, as healthcare systems require faster and more comprehensive diagnostic solutions
- For instance, in April 2025, major diagnostic laboratories expanded multiplex PCR-based respiratory panels to improve simultaneous detection of influenza, RSV, and COVID-19 infections in clinical settings
- As disease complexity increases, multiplex testing offers significant advantages by enabling simultaneous detection of multiple biomarkers, reducing diagnostic time and improving patient management outcomes
- Furthermore, the growing adoption of precision medicine is accelerating demand for multiplex assays, as clinicians increasingly rely on multi-marker profiling for personalized treatment strategies
- The convenience of high-throughput testing, reduced reagent consumption, and faster turnaround times is further propelling adoption in hospitals, diagnostic centers, and research laboratories
- In addition, expanding government-funded screening programs for early disease detection are further accelerating the deployment of multiplex diagnostic technologies in public healthcare systems
- Moreover, rising outbreak surveillance initiatives are increasing the use of multiplex testing for rapid pathogen identification and epidemiological monitoring
Restraint/Challenge
“High Assay Complexity and Standardization Challenges Limiting Scalability”
- Concerns surrounding assay complexity, cross-reactivity, and lack of standardized protocols pose a significant challenge to the widespread adoption of multiplex testing technologies across diverse clinical settings
- For instance, variations in multiplex assay performance across different platforms and laboratories have raised concerns regarding reproducibility and result comparability in multi-center clinical studies
- Addressing these challenges requires strict standardization of assay formats, improved validation frameworks, and robust quality control measures to ensure consistent diagnostic accuracy
- In addition, the relatively high initial investment required for advanced multiplex platforms and specialized instrumentation can limit adoption in cost-sensitive healthcare settings and developing regions
- While technological advancements are reducing operational complexity, concerns over assay optimization, regulatory approval timelines, and workflow integration continue to hinder rapid scalability of multiplex testing solutions
- Furthermore, limited skilled laboratory personnel trained in advanced multiplex technologies restrict efficient implementation in smaller diagnostic facilities
- In addition, variability in regulatory requirements across regions creates barriers to global standardization and slows down product commercialization timelines
Multiplex Testing Market Scope
The market is segmented on the basis of product, technology, application, and end user.
- By Product
On the basis of product, the multiplex testing market is segmented into reagents, consumables, and others. The reagents segment dominated the market with the largest revenue share of 46.3% in 2025, driven by its essential role in enabling multi-analyte detection across all diagnostic workflows and its recurring usage in every testing cycle. High test volumes in clinical diagnostics, infectious disease screening, and oncology applications significantly support continuous demand for reagents. Furthermore, increasing adoption of automated multiplex platforms is strengthening reagent consumption due to higher assay throughput requirements. Growing expansion of diagnostic laboratories globally is also contributing to sustained dominance of this segment.
The consumables segment is expected to witness the fastest growth rate of 18.9% from 2026 to 2033, fueled by rising automation in laboratories and increasing demand for standardized and ready-to-use testing components. Consumables such as microplates, beads, cartridges, and assay kits are gaining traction due to their ability to improve accuracy and reduce contamination risks. In addition, the expansion of high-throughput diagnostic facilities and increasing preference for integrated multiplex testing solutions are accelerating adoption. Growing investment in laboratory infrastructure across emerging economies further supports segment growth.
- By Technology
On the basis of technology, the market is segmented into xMAP (Multi-Analyte Profiling), RT-PCR (Real Time Polymerase Chain Reaction), ELISA (Enzyme-Linked Immunosorbent Assay), and others. The ELISA segment dominated the market with the largest revenue share of 41.6% in 2025, driven by its long-established clinical reliability, high sensitivity, and widespread use in detecting multiple biomarkers across infectious diseases, oncology, and autoimmune disorders. Its dominance is further supported by strong adoption in clinical laboratories and research institutes due to standardized protocols, cost-effectiveness, and ease of implementation. In addition, ELISA remains highly preferred in routine diagnostic workflows because it does not require highly complex instrumentation compared to other advanced molecular techniques. Continuous improvements in automated and high-throughput ELISA platforms are further enhancing efficiency and scalability in large-volume testing environments. Moreover, its broad clinical applicability and compatibility with existing laboratory infrastructure continue to reinforce its leading market position globally.
The RT-PCR segment is expected to witness the fastest growth rate of 20.4% from 2026 to 2033, driven by rising demand for highly sensitive molecular diagnostic techniques, particularly in infectious disease detection and respiratory pathogen identification. Its ability to detect low levels of genetic material with high accuracy makes it essential in outbreak surveillance and clinical diagnostics. Furthermore, increasing adoption of multiplex RT-PCR panels in point-of-care and decentralized testing settings is accelerating growth. Expanding government initiatives for early disease detection and pandemic preparedness are also boosting segment expansion.
- By Application
On the basis of application, the market is segmented into Influenza A, Influenza B, Parainfluenza Viruses, Adenovirus, Respiratory Syncytial Virus (RSV), Coronaviruses, Human Metapneumovirus (hMPV), and Group Rhino- and Enteroviruses. The Respiratory Syncytial Virus (RSV) segment dominated the market with the largest revenue share of 28.6% in 2025, driven by its high prevalence among infants, elderly populations, and immunocompromised patients, which increases demand for early and accurate diagnosis. RSV is also frequently included in multiplex respiratory panels due to its co-infection with other viruses. Seasonal outbreaks and rising hospitalization rates further strengthen its dominance. Increasing awareness of respiratory disease screening is also contributing to market growth.
The Coronaviruses segment is expected to witness the fastest growth rate of 22.1% from 2026 to 2033, driven by heightened global surveillance and preparedness following the COVID-19 pandemic. Multiplex panels capable of detecting SARS-CoV-2 alongside other respiratory pathogens are increasingly being adopted in hospitals and laboratories. Government-led infectious disease monitoring programs are further supporting demand. Continuous advancements in molecular diagnostic technologies are also accelerating segment growth.
- By End User
On the basis of end user, the market is segmented into hospitals, clinical laboratories, pharmaceutical and biotechnology companies, research institutes, and others. The clinical laboratories segment dominated the market with the largest revenue share of 44.5% in 2025, driven by high sample processing volumes, advanced diagnostic infrastructure, and strong adoption of multiplex testing for infectious and chronic disease screening. These laboratories offer efficient high-throughput capabilities, enabling large-scale diagnostic testing. Increasing outsourcing of diagnostic services from hospitals to specialized labs is further strengthening this dominance. Continuous expansion of laboratory networks across developed and emerging regions also supports growth.
The pharmaceutical and biotechnology companies segment is expected to witness the fastest growth rate of 19.7% from 2026 to 2033, driven by increasing use of multiplex testing in drug discovery, biomarker validation, and clinical trials. These companies rely on multiplex assays for precision medicine development and companion diagnostics. Rising investment in targeted therapy research is further boosting adoption. Expanding focus on personalized medicine and translational research is also accelerating segment growth globally.
Multiplex Testing Market Regional Analysis
- North America dominated the multiplex testing market with the largest revenue share of 39.8% in 2025, characterized by advanced healthcare infrastructure, strong adoption of precision medicine, and high R&D investments
- Healthcare providers and laboratories in the region highly value the accuracy, efficiency, and comprehensive disease profiling capabilities offered by multiplex testing platforms, particularly in infectious disease, oncology, and chronic disease diagnostics
- This widespread adoption is further supported by advanced healthcare infrastructure, high R&D investments, strong presence of key diagnostic companies, and increasing focus on precision medicine, establishing multiplex testing as a critical tool in modern clinical diagnostics and biomedical research
U.S. Multiplex Testing Market Insight
The U.S. multiplex testing market captured the largest revenue share in 2025 within North America, driven by the rapid adoption of advanced molecular diagnostics and the rising prevalence of infectious and chronic diseases. Healthcare providers are increasingly shifting toward multiplex assays to enable simultaneous detection of multiple pathogens, improving diagnostic efficiency and turnaround time. The growing emphasis on precision medicine and early disease detection is further supporting market expansion. In addition, strong R&D investments and the presence of leading diagnostic companies are accelerating product innovation. The integration of automated platforms and high-throughput technologies is also significantly boosting clinical adoption across hospitals and laboratories.
Europe Multiplex Testing Market Insight
The Europe multiplex testing market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by strong public healthcare systems and increasing demand for cost-effective diagnostic solutions. The rising burden of infectious diseases and cancer is encouraging the adoption of multiplex assays for accurate and rapid testing. European regulatory support for advanced in-vitro diagnostics is further fostering market growth. In addition, increasing investments in laboratory automation and personalized medicine initiatives are strengthening market penetration. The region is witnessing broad adoption across hospitals, research institutes, and diagnostic laboratories, enhancing overall clinical efficiency.
U.K. Multiplex Testing Market Insight
The U.K. multiplex testing market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by rising demand for rapid diagnostic solutions and expanding public health screening programs. Increasing concerns over antimicrobial resistance and viral outbreaks are pushing healthcare providers toward multiplex molecular testing platforms. The NHS’s focus on improving diagnostic capacity and reducing waiting times is further accelerating adoption. In addition, growing use of point-of-care testing and laboratory automation is supporting market expansion. The presence of strong academic and biotech research ecosystems is also contributing to continuous innovation in multiplex assay development.
Germany Multiplex Testing Market Insight
The Germany multiplex testing market is expected to expand at a considerable CAGR during the forecast period, fueled by increasing demand for high-precision diagnostics and strong healthcare infrastructure. Germany’s focus on early disease detection and preventive healthcare is driving the use of multiplex assays in clinical diagnostics. The country’s advanced laboratory networks and emphasis on technological innovation are further supporting adoption. In addition, growing investments in molecular diagnostics and personalized treatment approaches are strengthening market growth. Integration of multiplex platforms in hospital laboratories and diagnostic centers is becoming increasingly common across both urban and regional healthcare facilities.
Asia-Pacific Multiplex Testing Market Insight
The Asia-Pacific multiplex testing market is poised to grow at the fastest CAGR during the forecast period, driven by increasing disease burden, expanding healthcare infrastructure, and rising awareness of advanced diagnostics. Countries such as China, Japan, and India are witnessing strong adoption of multiplex technologies due to growing demand for rapid and accurate testing solutions. Government initiatives to strengthen public health systems and expand diagnostic accessibility are further supporting market growth. In addition, increasing investments from global diagnostic companies and local manufacturers are improving affordability and availability. The region is also experiencing rapid expansion of laboratory networks and point-of-care testing services.
Japan Multiplex Testing Market Insight
The Japan multiplex testing market is gaining momentum due to the country’s aging population, high healthcare standards, and strong focus on early disease detection. The demand for advanced diagnostic technologies is increasing, particularly for infectious disease screening and chronic condition monitoring. Japan’s well-developed laboratory infrastructure and strong adoption of automation are supporting market growth. In addition, integration of multiplex testing with digital health and AI-driven diagnostic platforms is enhancing clinical efficiency. The emphasis on precision medicine and preventive healthcare is further accelerating the use of multiplex assays across hospitals and diagnostic centers.
India Multiplex Testing Market Insight
The India multiplex testing market accounted for the largest market revenue share in Asia Pacific in 2025, attributed to rising healthcare expenditure, expanding diagnostic networks, and increasing prevalence of infectious diseases. The growing demand for affordable and rapid diagnostic solutions is driving adoption across hospitals, clinics, and diagnostic laboratories. Government initiatives focused on improving healthcare access and disease surveillance are further supporting market expansion. In addition, the rise of private diagnostic chains and point-of-care testing services is strengthening market penetration. India’s growing biotech ecosystem and domestic manufacturing capabilities are also contributing to improved accessibility and affordability of multiplex testing solutions.
Multiplex Testing Market Share
The Multiplex Testing industry is primarily led by well-established companies, including:
- Thermo Fisher Scientific Inc. (U.S.)
- F. Hoffmann-La Roche Ltd. (Switzerland)
- Abbott (U.S.)
- Danaher (U.S.)
- Bio-Rad Laboratories, Inc. (U.S.)
- QIAGEN (Germany)
- BD (U.S.)
- Seegene Inc. (South Korea)
- BIOMÉRIEUX (France)
- Siemens Healthineers AG (Germany)
- DiaSorin S.p.A. (Italy)
- Illumina, Inc. (U.S.)
- Agilent Technologies, Inc. (U.S.)
- PerkinElmer Inc. (U.S.)
- Bio-Techne Corporation (U.S.)
- Meso Scale Diagnostics, LLC (U.S.)
- Merck KGaA (Germany)
- Randox Laboratories Ltd (U.K.)
- Sysmex Corporation (Japan)
- Takara Bio Inc. (Japan)
What are the Recent Developments in Global Multiplex Testing Market?
- In February 2025, Luminex Corporation (DiaSorin) launched ARIES Group B Streptococcus multiplex PCR assay. Luminex Corporation (DiaSorin Molecular) launched the ARIES GBS Assay for its ARIES System, enabling fully automated multiplex PCR detection for intrapartum Group B Streptococcus screening
- In October 2024, Healgen Scientific, LLC received FDA De Novo authorization for its point-of-care multiplex antigen test capable of detecting COVID-19, Influenza A, and Influenza B in a single device. The test is designed for rapid diagnostics in clinical and decentralized settings
- In August 2024, Seegene Inc. announced the development of Novaplex™ MPXV/OPXV multiplex PCR assays capable of detecting multiple viral pathogens including mpox, herpes simplex virus (HSV-1/2), and varicella zoster virus in a single reaction
- In April 2024, Oncocyte and Bio-Rad announced the global launch of the GraftAssure multiplex droplet digital PCR assay for transplant monitoring. The test measures donor-derived cell-free DNA to assess organ rejection risk using multiplex molecular detection
- In June 2021, Bio-Rad Laboratories and Seegene formed a partnership to develop multiplex real-time PCR infectious disease assays. The collaboration focused on syndromic testing capable of detecting multiple pathogens in a single reaction. The assays were designed for use on Bio-Rad’s CFX96 Dx system in clinical laboratories
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.