Global Phenylketonuria Pku Treatment Market
Market Size in USD Billion
CAGR :
% 
 USD
1.18 Billion
USD
2.70 Billion
2025
2033
USD
1.18 Billion
USD
2.70 Billion
2025
2033
| 2026 –2033 | |
| USD 1.18 Billion | |
| USD 2.70 Billion | |
|
|
|
|
Phenylketonuria (PKU) Treatment Market Size
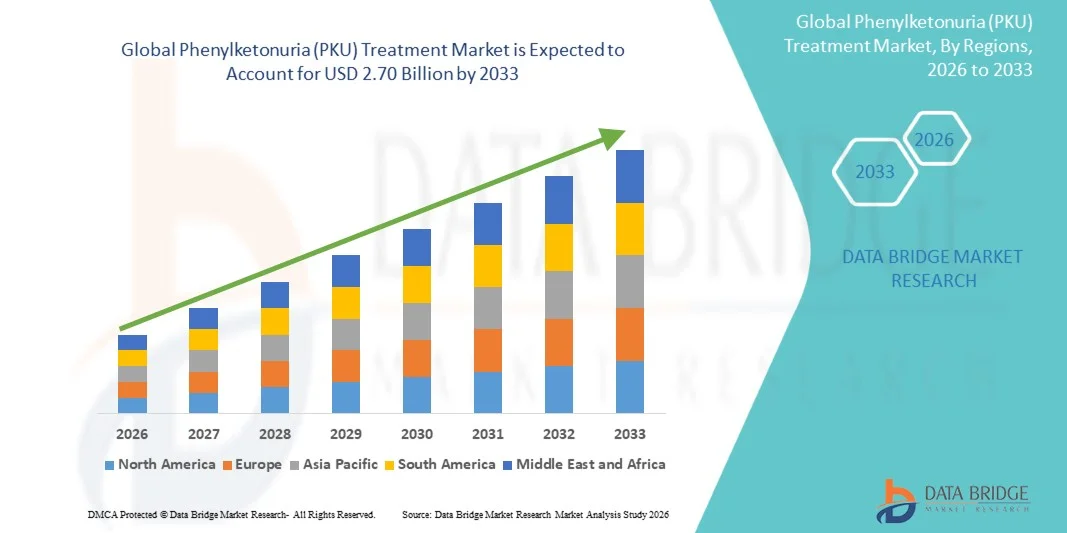
- The global Phenylketonuria (PKU) Treatment market size was valued at USD 1.18 billion in 2025 and is expected to reach USD 2.70 billion by 2033, at a CAGR of 10.95% during the forecast period
- The market growth is largely fueled by the increasing prevalence of rare metabolic disorders, rising newborn screening programs, and continuous advancements in genetic and nutritional therapies, leading to greater adoption of specialized treatment solutions across healthcare settings
- Furthermore, growing demand for effective long-term metabolic control, increasing awareness regarding early diagnosis of inherited disorders, and expanding availability of enzyme replacement, drug, and medical nutrition therapies are establishing Phenylketonuria (PKU) Treatment solutions as an essential component of modern rare disease management. These converging factors are accelerating the uptake of Phenylketonuria (PKU) Treatment solutions, thereby significantly boosting the industry's growth
Phenylketonuria (PKU) Treatment Market Analysis
- Phenylketonuria (PKU) treatment solutions, including medical foods, amino acid supplements, enzyme substitution therapies, and pharmacological treatments, are increasingly vital components of modern rare disease management due to their role in maintaining safe phenylalanine levels, preventing neurological complications, and improving long-term patient outcomes
- The escalating demand for Phenylketonuria (PKU) treatment solutions is primarily fueled by expanding newborn screening programs, rising awareness regarding inherited metabolic disorders, increasing access to orphan drugs, and continuous advancements in gene and enzyme-based therapeutic approaches
- North America dominated the Phenylketonuria (PKU) Treatment market with the largest revenue share of approximately 44.2% in 2025, characterized by advanced rare disease healthcare infrastructure, widespread newborn screening coverage, strong reimbursement systems, and early adoption of innovative PKU therapies across the U.S. and Canada
- Asia-Pacific is expected to be the fastest growing region in the Phenylketonuria (PKU) Treatment market during the forecast period due to improving diagnostic capabilities, rising healthcare expenditure, increasing awareness of metabolic disorders, and expanding access to specialized nutritional and pharmaceutical therapies across China, Japan, India, and Southeast Asia
- The Dietary Therapy segment accounted for the largest market revenue share of 68.2% in 2025, driven by its role as the standard first-line lifelong management approach for PKU patients
Report Scope and Phenylketonuria (PKU) Treatment Market Segmentation
|
Attributes |
Phenylketonuria (PKU) Treatment Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
Europe
Asia-Pacific
Middle East and Africa
South America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
Phenylketonuria (PKU) Treatment Market Trends
“Advancements in Enzyme Substitution Therapies and Personalized Dietary Management”
- A significant and accelerating trend in the global Phenylketonuria (PKU) Treatment market is the growing adoption of advanced therapeutic approaches, including enzyme substitution therapies, pharmacological treatments, and precision dietary management solutions. These developments are improving long-term metabolic control and enhancing quality of life for PKU patients across all age groups
- For instance, BioMarin Pharmaceutical Inc. has expanded the global use of Kuvan (sapropterin dihydrochloride) and Palynziq (pegvaliase), which help eligible PKU patients reduce blood phenylalanine levels and improve dietary flexibility
- Increased focus on personalized nutrition planning, supported by digital monitoring tools and specialized low-protein food products, is helping patients maintain treatment adherence more effectively. This is especially important for lifelong disease management
- Newborn screening programs in developed and emerging economies are enabling earlier diagnosis and treatment initiation, significantly reducing the risk of neurological impairment and developmental complications
- Ongoing research into gene therapy, microbiome-based therapies, and next-generation enzyme treatments is expanding the future treatment landscape and creating optimism for more durable solutions
- The trend toward comprehensive disease management, combining medication, nutrition therapy, and continuous monitoring, is reshaping the PKU Treatment market globally across hospitals, specialty clinics, and homecare settings
Phenylketonuria (PKU) Treatment Market Dynamics
Driver
“Growing Diagnosis Rates and Rising Demand for Long-Term Rare Disease Management”
- Increasing awareness of rare metabolic disorders, combined with expanding newborn screening coverage worldwide, is a major driver for the Phenylketonuria (PKU) Treatment market. Earlier detection allows faster intervention and improved long-term patient outcomes
- For instance, Centers for Disease Control and Prevention and several European national screening programs continue to support universal newborn screening initiatives that include PKU testing, leading to higher diagnosis and treatment rates
- Rising healthcare investments in rare disease management are encouraging broader access to specialized formulas, nutritional counseling, and pharmacological therapies
- Increased patient advocacy efforts and support organizations are improving awareness among families, clinicians, and policymakers regarding the importance of lifelong PKU management
- The growing adult PKU patient population requiring continued treatment beyond childhood is further expanding demand for maintenance therapies and nutritional products
- In addition, reimbursement support for orphan drugs in several countries is encouraging innovation and wider commercial adoption of advanced PKU treatments
Restraint/Challenge
“High Treatment Costs, Strict Dietary Compliance, and Limited Access in Developing Regions”
- High costs associated with specialty medications, medical foods, amino acid formulas, and long-term clinical monitoring remain a significant challenge for many patients and healthcare systems
- For instance, access to premium therapies such as pegvaliase remains limited in several low- and middle-income countries due to reimbursement constraints, regulatory delays, and affordability barriers
- Strict lifelong adherence to low-phenylalanine diets can be difficult for patients, especially adolescents and adults, leading to inconsistent metabolic control and treatment fatigue
- Limited availability of metabolic specialists, dietitians, and PKU-friendly food products in some regions further restricts effective disease management
- Variability in patient response to available therapies means not all individuals benefit equally from current treatment options, creating unmet clinical needs
- Overcoming these challenges through lower-cost treatment models, stronger reimbursement systems, expanded specialist access, and continued therapeutic innovation will be essential for sustained market growth
Phenylketonuria (PKU) Treatment Market Scope
The market is segmented on the basis of type, drug type, therapy type, route of administration, and end-user.
• By Type
On the basis of type, the Phenylketonuria (PKU) Treatment market is segmented into Hyperphenylalaninemia, Mild PKU, Moderate or Variant, and Classic PKU. The Classic PKU segment dominated the largest market revenue share of 44.8% in 2025, driven by its severe enzyme deficiency requiring lifelong treatment and strict phenylalanine management. Patients with classic PKU need continuous medical nutrition therapy, specialized formulas, and pharmacological interventions. Early newborn screening programs are increasing diagnosis rates globally. Rising awareness among healthcare professionals supports timely treatment initiation. Strong demand for long-term disease management products reinforces segment dominance. Increasing reimbursement support in developed markets further boosts adoption. Continuous monitoring requirements contribute to sustained market revenue.
The Moderate or Variant PKU segment is expected to witness the fastest growth rate of 23.1% from 2026 to 2033, driven by improved diagnostic classification and growing access to targeted therapies. These patients often respond better to newer pharmacological options such as BH4-responsive treatments. Increasing awareness regarding individualized treatment plans is supporting growth. Rising adoption of precision medicine approaches is accelerating demand. Expanded metabolic disorder screening programs further strengthen diagnosis rates. Growing physician focus on quality-of-life improvements supports treatment uptake. Advancements in personalized metabolic care continue to drive segment expansion.
• By Drugs Type
On the basis of drugs type, the Phenylketonuria (PKU) Treatment market is segmented into Kuvan, Sapropterin, Palynziq, Pagvaliase, and Biopten. The Kuvan segment held the largest market revenue share of 36.7% in 2025, driven by its established use in BH4-responsive PKU patients to lower blood phenylalanine levels. It is widely prescribed due to proven efficacy, oral administration convenience, and strong physician familiarity. Growing diagnosis of responsive PKU cases is supporting demand. Favorable regulatory approvals across multiple countries strengthen market presence. Long-standing commercial availability reinforces segment leadership. Rising treatment adherence due to ease of use further boosts revenue.
The Palynziq segment is expected to witness the fastest CAGR of 25.4% from 2026 to 2033, driven by increasing adoption for adult PKU patients with uncontrolled phenylalanine levels despite conventional management. It offers an advanced enzyme substitution approach for difficult-to-treat cases. Rising demand for effective long-term control is boosting uptake. Expanding specialist metabolic clinics are supporting access. Improved patient outcomes and reduced dietary burden strengthen adoption. Growing awareness of advanced PKU therapies accelerates demand. Continuous post-approval market expansion supports rapid growth.
• By Therapy Type
On the basis of therapy type, the Phenylketonuria (PKU) Treatment market is segmented into Gene Therapy and Dietary Therapy. The Dietary Therapy segment accounted for the largest market revenue share of 68.2% in 2025, driven by its role as the standard first-line lifelong management approach for PKU patients. Low-phenylalanine diets, medical foods, and amino acid supplements remain essential across all disease severities. Early intervention through dietary control prevents neurological complications. Strong physician recommendation and patient dependence support segment dominance. Growing availability of specialized nutritional products further boosts demand. Newborn screening programs continue to expand long-term patient enrollment.
The Gene Therapy segment is expected to witness the fastest growth rate of 27.3% from 2026 to 2033, driven by rising investment in curative treatment approaches targeting the root cause of PKU. Biotech companies are actively developing gene-editing and vector-based therapies. Increasing clinical trial success is boosting investor confidence. Demand for one-time treatments with long-term benefit supports growth. Growing burden of lifelong dietary restrictions encourages adoption interest. Regulatory support for rare disease innovation further accelerates development. Future commercialization prospects continue to strengthen segment momentum.
• By Route of Administration
On the basis of route of administration, the Phenylketonuria (PKU) Treatment market is segmented into Oral, Intravenous, Subcutaneous, and Others. The Oral segment dominated the largest market revenue share of 52.6% in 2025, driven by strong use of oral drugs, nutritional supplements, and medical foods in routine PKU management. Oral therapies are convenient, non-invasive, and suitable for long-term adherence across pediatric and adult patients. Widespread use of sapropterin-based treatments further supports dominance. Easy home administration enhances patient compliance. Growing availability of flavored and patient-friendly formulations boosts acceptance. Strong physician preference for first-line oral options reinforces leadership.
The Subcutaneous segment is expected to witness the fastest CAGR of 24.6% from 2026 to 2033, driven by increasing use of injectable enzyme therapies such as pegvaliase-based treatments. These therapies are particularly valuable for patients with uncontrolled phenylalanine levels. Rising demand for advanced biologic treatment options is accelerating growth. Improved training for self-administration supports adoption. Expanding metabolic disorder specialty centers further strengthen access. Growing acceptance of biologics in rare disease management boosts uptake. Continuous innovation in delivery devices supports expansion.
• By End-User
On the basis of end-user, the Phenylketonuria (PKU) Treatment market is segmented into Hospitals, Homecare, Specialty Clinics, and Others. The Homecare segment held the largest market revenue share of 39.4% in 2025, driven by the chronic nature of PKU management requiring daily nutritional therapy and routine medication outside clinical settings. Most patients manage diet plans, supplements, and oral drugs at home for long durations. Rising preference for convenient long-term care supports dominance. Increased parental involvement in pediatric PKU management further boosts demand. Availability of telehealth nutrition counseling strengthens homecare adoption. Cost-effectiveness versus repeated hospital visits supports leadership.
The Specialty Clinics segment is expected to witness the fastest CAGR of 23.8% from 2026 to 2033, driven by increasing establishment of metabolic disorder centers offering personalized PKU treatment. These clinics provide expert monitoring, dietary counseling, biomarker testing, and advanced therapy access. Growing awareness of specialized rare disease care is supporting demand. Expansion of multidisciplinary treatment models accelerates adoption. Improved patient outcomes through expert management strengthen growth. Rising referrals from general hospitals further boost volumes. Continuous development of rare disease infrastructure supports segment expansion.
Phenylketonuria (PKU) Treatment Market Regional Analysis
- North America dominated the phenylketonuria (PKU) treatment market with the largest revenue share of approximately 44.2% in 2025, characterized by advanced rare disease healthcare infrastructure, widespread newborn screening coverage, strong reimbursement systems, and early adoption of innovative PKU therapies across the U.S. and Canada. The region has also witnessed increasing use of medical foods, enzyme substitution therapies, and adjunct pharmacological treatments for long-term metabolic control
- Patients, caregivers, and healthcare providers in the region highly value early diagnosis, lifelong disease management support, and improved quality of life offered by advanced PKU treatment approaches. Growing preference for personalized nutrition programs and innovative therapeutic options continues to strengthen market demand
- This widespread adoption is further supported by high healthcare expenditure, strong patient advocacy networks, specialized metabolic clinics, and increasing awareness regarding inherited metabolic disorders, establishing North America as a leading market for PKU treatment solutions
U.S. Phenylketonuria (PKU) Treatment Market Insight
The U.S. phenylketonuria (PKU) treatment market captured the largest revenue share in 2025 within North America, fueled by comprehensive newborn screening programs, strong access to rare disease therapies, and high awareness among healthcare professionals. Physicians are increasingly prioritizing early dietary intervention, amino acid-based medical nutrition, and innovative therapies such as enzyme replacement and cofactor treatments. The growing preference for multidisciplinary care models, combined with robust demand across pediatric hospitals, metabolic clinics, and specialty treatment centers, further propels the market. Moreover, increasing support from insurance systems and patient assistance programs is significantly contributing to market expansion.
Europe Phenylketonuria (PKU) Treatment Market Insight
The Europe phenylketonuria (PKU) treatment market is projected to expand at a substantial CAGR throughout the forecast period, primarily driven by strong newborn screening systems, increasing awareness of inherited metabolic disorders, and favorable healthcare reimbursement frameworks. The region’s established rare disease treatment networks and structured clinical guidelines are fostering adoption of advanced PKU management strategies. European healthcare providers are also focused on lifelong nutritional therapy and improved patient adherence. The market is experiencing significant growth across hospitals, metabolic centers, and pediatric specialty clinics, with increasing availability of innovative pharmaceutical therapies.
U.K. Phenylketonuria (PKU) Treatment Market Insight
The U.K. phenylketonuria (PKU) treatment market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by strong NHS screening programs and growing emphasis on rare disease management. Increasing awareness regarding early intervention and dietary adherence is encouraging broader treatment uptake. The UK’s expanding metabolic disorder care infrastructure and access to specialist clinicians are expected to continue stimulating market growth.
Germany Phenylketonuria (PKU) Treatment Market Insight
The Germany phenylketonuria (PKU) treatment market is expected to expand at a considerable CAGR during the forecast period, fueled by strong healthcare infrastructure, advanced diagnostic systems, and increasing adoption of innovative metabolic disorder therapies. Germany’s emphasis on precision healthcare and long-term chronic disease management promotes the use of specialized PKU treatments. Integration of dietary counseling, monitoring programs, and pharmaceutical care pathways is also becoming increasingly prevalent.
Asia-Pacific Phenylketonuria (PKU) Treatment Market Insight
The Asia-Pacific phenylketonuria (PKU) treatment market is poised to grow at the fastest CAGR during the forecast period of 2026 to 2033, driven by improving diagnostic capabilities, rising healthcare expenditure, increasing awareness of metabolic disorders, and expanding access to specialized nutritional and pharmaceutical therapies across China, Japan, India, and Southeast Asia. The region’s expanding pediatric care networks and improving newborn screening coverage are accelerating diagnosis and treatment rates. Furthermore, growing government healthcare investments and increasing availability of imported rare disease therapies are making PKU treatment more accessible.
Japan Phenylketonuria (PKU) Treatment Market Insight
The Japan phenylketonuria (PKU) treatment market is gaining momentum due to the country’s advanced healthcare system, strong neonatal screening programs, and high focus on pediatric rare disease management. Japanese healthcare providers place significant emphasis on early diagnosis and long-term metabolic control, driving demand for medical nutrition products and pharmaceutical therapies. The integration of digital monitoring tools and structured follow-up care is fueling growth.
China Phenylketonuria (PKU) Treatment Market Insight
The China phenylketonuria (PKU) treatment market accounted for the largest market revenue share in Asia Pacific in 2025, attributed to the country’s expanding healthcare infrastructure, growing newborn screening initiatives, rising awareness of genetic disorders, and increasing access to specialty pediatric care. China is witnessing greater adoption of dietary management products and rare disease therapeutics in urban healthcare centers. Government support for rare disease treatment programs and improved diagnostic capabilities are key factors propelling market growth in China.
Phenylketonuria (PKU) Treatment Market Share
The Phenylketonuria (PKU) Treatment industry is primarily led by well-established companies, including:
- BioMarin Pharmaceutical Inc. (U.S.)
- PTC Therapeutics, Inc. (U.S.)
- Nestlé Health Science (Switzerland)
- Danone S.A. (France)
- Nutricia Ltd. (Netherlands)
- Ajinomoto Co., Inc. (Japan)
- Mead Johnson Nutrition Company (U.S.)
- Abbott (U.S.)
- Cambrooke Therapeutics, Inc. (U.S.)
- Vitaflo International Ltd. (U.K.)
- Synlogic, Inc. (U.S.)
- Homology Medicines, Inc. (U.S.)
- Codexis, Inc. (U.S.)
- Recordati Rare Diseases Inc. (Italy)
- Merck KGaA (Germany)
Latest Developments in Global Phenylketonuria (PKU) Treatment Market
- In January 2021, BioMarin continued global commercialization expansion of Palynziq (pegvaliase-pqpz), an enzyme substitution therapy for adults with phenylketonuria (PKU), supporting patients with uncontrolled blood phenylalanine levels and reinforcing its role as a key advanced PKU treatment option
- In April 2025, BioMarin announced that its Phase 3 PEGASUS trial for PALYNZIQ (pegvaliase-pqpz) in adolescents aged 12–17 with PKU met the primary efficacy endpoint, demonstrating statistically significant lowering of blood phenylalanine levels compared with diet alone. This milestone supported planned regulatory expansion into younger patients
- In June 2025, PTC Therapeutics announced that the European Commission granted marketing authorization for Sephience (sepiapterin) for children and adults living with PKU. The approval included a broad label covering all ages and disease severities, significantly expanding treatment options in Europe
- In July 2025, PTC Therapeutics announced that the U.S. FDA approved Sephience (sepiapterin) for treatment of children and adults with phenylketonuria. The oral therapy was approved for patients aged one month and older with sepiapterin-responsive PKU, creating a major new competitive entrant in the global PKU treatment market
- In September 2025, BioMarin presented new Phase 3 PEGASUS data at the International Congress of Inborn Errors of Metabolism showing that adolescents treated with PALYNZIQ achieved a 49.7% decrease in mean blood phenylalanine levels. The results strengthened prospects for expanded pediatric use
- In October 2025, BioMarin announced that the U.S. FDA accepted for Priority Review its supplemental Biologics License Application to expand PALYNZIQ use to adolescents aged 12–17 years with PKU, with a PDUFA target action date of February 2026
- In December 2025, PTC Therapeutics announced approval of Sephience (sepiapterin) in Japan for treatment of children and adults living with PKU. The indication covered all ages and the full spectrum of disease severity, marking the company’s first product approval in Japan and supporting global launch momentum
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.