Global Retinal Biologics Market
Market Size in USD Billion
CAGR :
% 
 USD
22.20 Billion
USD
53.42 Billion
2022
2030
USD
22.20 Billion
USD
53.42 Billion
2022
2030
| 2023 –2030 | |
| USD 22.20 Billion | |
| USD 53.42 Billion | |
|
|
|
|
Retinal Biologics Market Analysis and Size
Augmented frequency of retinal diseases such as age-related macular degeneration globally is driving the market expansion. As per the WHO statistics, approximately 2.2 billion individuals globally suffer from near or distant vision impairment. The most clinically prevalent risk factors include the duration of diabetes,hypertension, hyperglycemia. Additionally, lack of awareness among people associated with different dermatology treatment hamper the market growth.
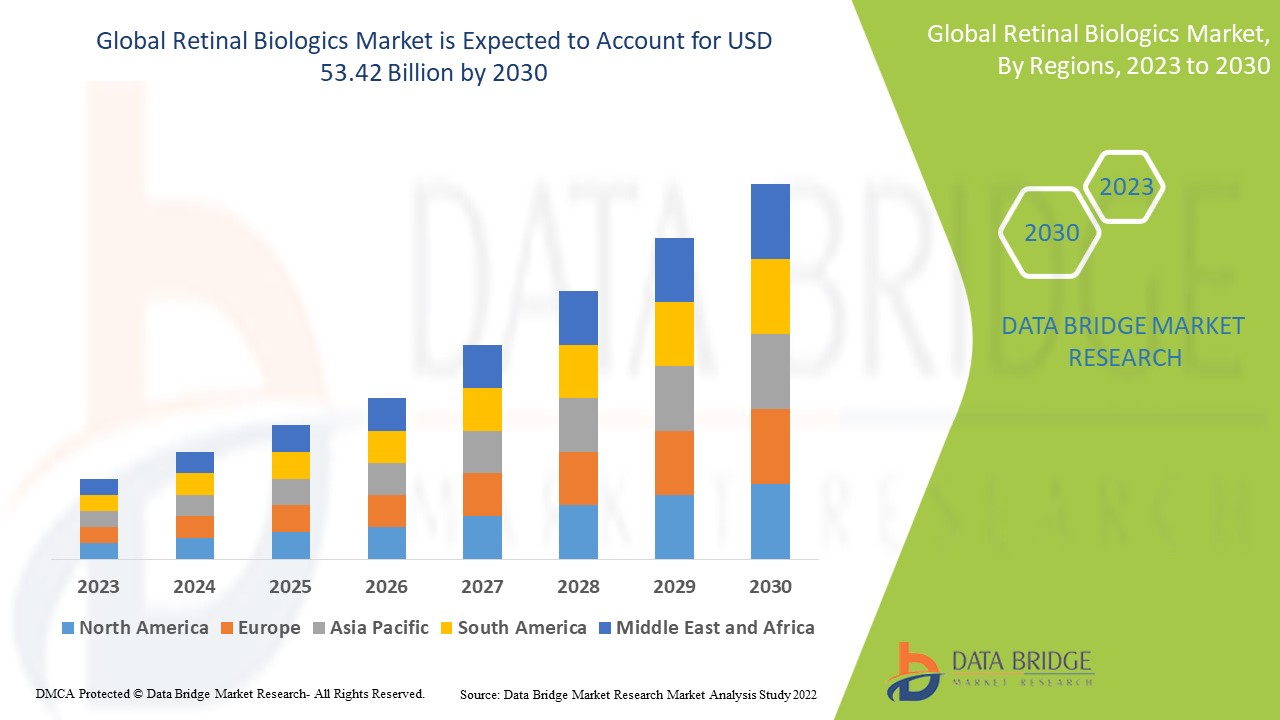
Data Bridge Market Research analyses a growth rate in the retinal biologics market in the forecast period 2023-2030. The expected CAGR of retinal biologics market tends to be around 11.6% in the mentioned forecast period. The market was valued at USD 22.2 billion in 2022 and would grow to USD 53.42 billion by 2030. In addition to the market insights such as market value, growth rate, market segments, geographical coverage, market players, and market scenario, the market report curated by the Data Bridge Market Research team also includes in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
Retinal Biologics Market Scope and Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015 - 2020) |
|
Quantitative Units |
Revenue in USD Billion, Volumes in Units, Pricing in USD |
|
Segments Covered |
Diseases Indication (Macular Degeneration, Diabetic Retinopathy, Retinal Vein, Occlusion), Drug Class (VEGF-A Antagonist, TNF-a Inhibitor), End-Users (Hospitals, Homecare, Speciality Centres, Others), Distribution Channel (Hospital Pharmacy, Online Pharmacy, Retail Pharmacy) |
|
Countries Covered |
U.S., Canada and Mexico in North America, Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe, China, Japan, India, South Korea, Singapore, Malaysia, Australia, Thailand, Indonesia, Philippines, Rest of Asia-Pacific (APAC) in the Asia-Pacific (APAC), Saudi Arabia, U.A.E, South Africa, Egypt, Israel, Rest of Middle East and Africa (MEA) as a part of Middle East and Africa (MEA), Brazil, Argentina and Rest of South America as part of South America |
|
Market Players Covered |
Pfizer Inc. (U.S.), Abbvie Inc. (U.S.), Novartis AG (Switzerland), Amgen Inc. (U.S.), Boehringer Ingelheim International GmbH. (Germany), Merck & Co Inc. (U.S.), Sun Pharmaceutical Industries Ltd. (India), Teva Pharmaceuticals Industries Ltd. (Israel), AstraZeneca (U.K.), Fresenius Kabi AG (Germany), Bayer AG (Germany), Aurobindo Pharma (India), AbbVie Inc. (U.S.), Santen Pharmaceutical Co., Ltd (Japan) |
|
Market Opportunities |
|
Market Definition
Retinal biologic are typically characterized as a large, complex molecules that is made from living cells or tissue. These products made treatment for numerous diseases simple, effective, and also inexpensive. They are technically advanced materials that can help in curing the disease. For instance, age-related macular degeneration (AMD) is the most common type of retinal disease that is related with aging and is a top cause of blindness among people above 50 years of age.
Retinal Biologics Market Dynamics
Drivers
- Increase in Vision Problems
As per the data published by the Centers for Disease Control and Prevention (CDC), in 2020, about 4.1 million U.S. Citizens were affected with diabetic retinopathy, and approximately 900,000 Americans were vulnerable to vision-damaging retinopathy. Diabetic retinopathy is one of the top disorders of posterior segment eye disorders. Growing incidence may increase the demand for treatment that may boost the market growth in the country.
- Increased Diabates Incidence
As per the article published by International Diabetes Federation in March 2020, the devastating consequences of diabetes mellitus are set to continue because of the predicted increase in occurrence from 463 million in 2019 to 700 million in 2045. Among this diabetic population, the global incidence of diabetic retinopathy (DR) and diabetic macular edema (DME) for 2019 was 27.0%.
Opportunities
- Launch of Drug Products
Several drug products are helpful in creating more opportunities for the market growth. For instance, in May 2019, Regeneron Pharmaceuticals, Inc. received U.S. FDA approval for EYLEA (aflibercept) injection to treat all stages of diabetic retinopathy. Such approvals are boosting the growth of the market during the forecast period.
- Increasing Strategic Initiatives by Several Organizations
There are several strategic initiatives taken by the major market players, such as product launches, partnerships, initiation of new programs, and mergers and acquisitions, may boost the market growth. For instance, in October 2021, Novartis received the U.S. FDA for the company’s supplemental Biologics License Application (sBLA) for the type-II variation application for Beovu 6 mg for the treatment of diabetic macular edema (DME). In July 2021, Genentech, a member of the Roche Group, announced that the US FDA accepted the company’s Biologics License Application (BLA) for faricimab used for the treatment of wet, or neovascular, age-related macular degeneration (AMD), diabetic macular edema (DME) and diabetic retinopathy.
Restraints/Challenges
- Lack of Skilled Professionals
A lack of trained healthcare professionals who are not aware of the appropriate treatment methods for this disease could decrease the growth of the global retinal biologics market during a forecast period.
- Adverse Side-Effects Associated with Retinal Biologics
Most retinal biologics are linked with adverse effects such as liver damage or type 2 diabetes. There are several drug classes that have been withdrawn or discontinued even in the later stages of trials due to the adverse effects or failure to achieve satisfactory results.
This retinal biologics market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the retinal biologics market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
Recent Developments:
- In 2022, the Food and Drug Administration (FDA) approved Roche for an eye disorder that could cause blindness in aged people, called Vabysmo, for wet age-related macular degeneration.
Global Retinal Biologics Market Scope
The retinal biologics market is segmented on the basis of disease indication, drug class, end user, distribution channel and end-user. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Disease Indication
- Macular Degeneration
- Diabetic Retinopathy
- Retinal Vein
- Occlusion
Drug Class
- VEGF-A Antagonist
- TNF-a Inhibitor
End-Users
- Hospitals
- Homecare
- Speciality Centres
- Others
Distribution Channel
- Hospital Pharmacy
- Online Pharmacy
- Retail Pharmacy
Retinal Biologics Market Regional Analysis/Insights
The retinal biologics market is analyzed and market size insights and trends are provided by disease indication, drug class, end user, distribution channel and end-user as referenced above.
The major countries covered in the retinal biologics market report are the U.S., Canada and Mexico in North America, Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe, China, Japan, India, South Korea, Singapore, Malaysia, Australia, Thailand, Indonesia, Philippines, Rest of Asia-Pacific (APAC) in the Asia-Pacific (APAC), Saudi Arabia, U.A.E, South Africa, Egypt, Israel, Rest of Middle East and Africa (MEA) as a part of Middle East and Africa (MEA), Brazil, Argentina and Rest of South America as part of South America.
North America is expected to have the highest market due to the presence of key manufacturers of the product is high and, increasing research and development activities
Asia Pacific dominates the market due to increased new research and developments on retinal biologics market.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Also, the presence and availability of global brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Competitive Landscape and Global Retinal Biologics Market Share Analysis
The retinal biologics market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, global presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to retinal biologics market.
Key players operating in the retinal biologics market include:
- Pfizer Inc. (U.S.)
- Novartis AG (Switzerland)
- Amgen Inc. (U.S.)
- Boehringer Ingelheim International GmbH. (Germany)
- Merck & Co Inc. (U.S.)
- Sun Pharmaceutical Industries Ltd. (India)
- Teva Pharmaceuticals Industries Ltd. (Israel)
- AstraZeneca (U.K.)
- Fresenius Kabi AG (Germany)
- Bayer AG (Germany)
- Aurobindo Pharma (India)
- AbbVie Inc. (U.S.)
- Santen Pharmaceutical Co., Ltd (Japan)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.












