Middle East And Africa Circulating Tumor Cells Ctc Liquid Biopsy Market
Market Size in USD Million
CAGR :
% 
 USD
45.44 Million
USD
156.31 Million
2025
2033
USD
45.44 Million
USD
156.31 Million
2025
2033
| 2026 –2033 | |
| USD 45.44 Million | |
| USD 156.31 Million | |
|
|
|
|
Middle East and Africa Circulating Tumor Cells (CTC) Liquid Biopsy Market Size
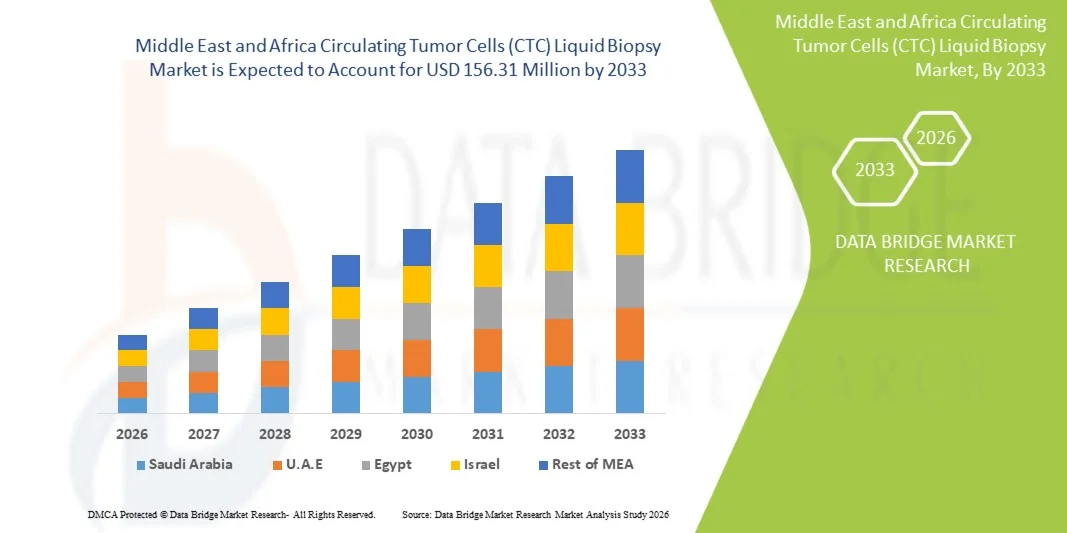
- The Middle East and Africa Circulating Tumor Cells (CTC) liquid biopsy market size was valued at USD 45.44 million in 2025and is expected to reach USD 156.31 million by 2033, at a CAGR of 16.70% during the forecast period
- The market growth is primarily driven by the increasing adoption of advanced cancer diagnostics and the gradual integration of liquid biopsy technologies into oncology workflows across key healthcare systems in the region, along with rising awareness of early cancer detection
- Furthermore, growing investments in precision medicine, expanding healthcare infrastructure, and increasing demand for minimally invasive diagnostic techniques are strengthening the adoption of CTC-based liquid biopsy solutions, thereby accelerating overall market expansion in the Middle East and Africa region
Middle East and Africa Circulating Tumor Cells (CTC) Liquid Biopsy Market Analysis
- Circulating Tumor Cells (CTC) liquid biopsy, a minimally invasive diagnostic approach that isolates and analyzes tumor cells from peripheral blood, is increasingly gaining importance in oncology across the Middle East and Africa due to its ability to enable early cancer detection, treatment monitoring, and personalized therapy selection, especially in healthcare systems gradually adopting precision medicine tools
- The growing cancer burden in the region, coupled with increasing awareness of non-invasive diagnostic techniques and expanding adoption of molecular oncology platforms in tertiary hospitals, is significantly driving demand for advanced CTC-based liquid biopsy solutions
- Saudi Arabia dominated the Middle East and Africa CTC liquid biopsy market with the largest revenue share of 38.6% in 2025, supported by strong government healthcare modernization programs, rising oncology research initiatives, and increasing deployment of advanced diagnostic technologies in leading cancer centers
- South Africa is expected to be the fastest growing country in the Middle East and Africa CTC liquid biopsy market during the forecast period due to improving diagnostic infrastructure, rising cancer screening programs, and increasing adoption of liquid biopsy technologies in private healthcare institutions
- CTC detection methods segment dominated the Middle East and Africa CTC liquid biopsy market with the largest market share of 44.8% in 2025, driven by high clinical adoption of enrichment-based and detection technologies such as immunoaffinity capture, microfluidics, and molecular assays, which are widely used for accurate tumor cell isolation and real-time cancer monitoring in oncology diagnostics
Report Scope and Middle East and Africa Circulating Tumor Cells (CTC) Liquid Biopsy Market Segmentation
|
Attributes |
Middle East and Africa Circulating Tumor Cells (CTC) Liquid Biopsy Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
Middle East and Africa · Saudi Arabia · U.A.E. · South Africa · Egypt · Israel · Rest of Middle East and Africa |
|
Key Market Players |
|
|
Market Opportunities |
· Expansion of CTC liquid biopsy applications in early cancer screening programs · Integration of CTC analysis with multi-omics platforms and AI-driven diagnostics |
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
Middle East and Africa Circulating Tumor Cells (CTC) Liquid Biopsy Market Trends
“Expanding Adoption of Advanced Detection and Precision Oncology Platforms”
- A significant and accelerating trend in the Middle East and Africa CTC liquid biopsy market is the increasing adoption of advanced detection technologies such as microfluidics, immunoaffinity-based enrichment, and molecular profiling platforms, improving sensitivity and clinical utility in oncology diagnostics
- For instance, CellSearch system-based CTC detection is increasingly being used in specialized oncology centers in Saudi Arabia and South Africa for monitoring metastatic breast and prostate cancer patients
- Integration of CTC analysis with next-generation sequencing (NGS) and multi-omics profiling is enhancing tumor characterization and enabling more precise treatment selection and disease progression tracking in advanced cancer cases
- Furthermore, growing collaboration between regional hospitals and global diagnostic companies is facilitating access to high-end liquid biopsy platforms, enabling centralized cancer testing and improved diagnostic accuracy across tertiary care centers
- This trend toward precision oncology integration is reshaping clinical workflows, with countries such as the United Arab Emirates and Egypt adopting CTC-based liquid biopsy in research-driven oncology programs and clinical trials
- The demand for CTC liquid biopsy solutions is growing rapidly across Saudi Arabia, South Africa, and the United Arab Emirates, as healthcare providers increasingly prioritize early cancer detection and minimally invasive monitoring approaches
- Furthermore, rising participation of Middle East countries in global oncology research networks is accelerating clinical validation and adoption of standardized CTC testing protocols
Middle East and Africa Circulating Tumor Cells (CTC) Liquid Biopsy Market Dynamics
Driver
“Rising Cancer Burden and Expanding Precision Oncology Adoption”
- The increasing prevalence of cancer across the Middle East and Africa, combined with growing awareness of early detection and precision medicine approaches, is a major driver for the rising demand for CTC liquid biopsy technologies
- For instance, in March 2025, King Faisal Specialist Hospital and Research Centre in Saudi Arabia expanded its liquid biopsy oncology program to enhance real-time monitoring of metastatic cancer patients using circulating tumor cell analysis
- As healthcare systems strengthen oncology infrastructure, CTC liquid biopsy offers significant advantages such as non-invasive sampling, dynamic disease monitoring, and improved treatment response evaluation compared to traditional tissue biopsies
- Furthermore, increasing investments in advanced laboratory diagnostics and molecular testing capabilities in countries such as South Africa and the United Arab Emirates are supporting broader clinical adoption of CTC-based technologies
- The growing need for personalized cancer treatment, coupled with rising hospital collaborations with global biotech firms, is making CTC liquid biopsy an integral part of modern oncology workflows in the region
- In addition, expanding government-led cancer screening initiatives and national cancer control programs are increasing early-stage diagnosis rates, further driving demand for CTC-based monitoring tools
- Furthermore, rising awareness among clinicians regarding minimal residual disease (MRD) monitoring using CTC assays is strengthening clinical acceptance across tertiary care hospitals
Restraint/Challenge
“High Cost and Limited Diagnostic Infrastructure Accessibility”
- Concerns regarding the high cost of CTC liquid biopsy systems and limited access to advanced diagnostic infrastructure remain a significant challenge for widespread adoption across several Middle East and Africa countries
- For instance, advanced platforms such as microfluidic-based CTC isolation systems are primarily concentrated in high-income healthcare systems such as Saudi Arabia and the United Arab Emirates, limiting accessibility in lower-income regions
- In addition, the requirement for highly specialized laboratory expertise and standardized protocols for CTC isolation and analysis creates operational challenges in many public healthcare institutions across Africa
- Furthermore, variability in regulatory frameworks and lack of standardized reimbursement policies for liquid biopsy procedures hinder large-scale clinical adoption in several countries
- While pilot programs and research collaborations are expanding, the high capital investment required for equipment and training continues to restrict penetration beyond major urban oncology centers
- Overcoming these challenges through cost reduction strategies, infrastructure development, and broader clinical validation studies will be essential for sustained market expansion in the MEA region
- In addition, limited availability of skilled molecular diagnostic professionals in several African countries further slows adoption and workflow standardization of CTC technologies
- Furthermore, lack of awareness among smaller healthcare providers about the clinical benefits of CTC-based monitoring reduces its utilization in routine oncology practice
Middle East and Africa Circulating Tumor Cells (CTC) Liquid Biopsy Market Scope
The market is segmented on the basis of technology, application, and end user.
- By Technology
On the basis of technology, the Middle East and Africa CTC liquid biopsy market is segmented into CTC Detection Methods, CTC Enrichment Methods, Ex Vivo Positive Selection, Molecular (RNA)-Based Technologies, Functional In vitro Cell Invasion Assay, Xenotransplantation Models, Microchips, Single Spiral Microchannel, and Negative Selection & Immunocytochemical Technologies. The CTC Detection Methods segment dominated the market with the largest market revenue share of 44.8% in 2025, driven by its strong clinical adoption in oncology diagnostics and routine cancer monitoring across hospitals and diagnostic laboratories. This segment is widely used in Saudi Arabia and South Africa for detecting circulating tumor cells in breast, prostate, and colorectal cancers with high accuracy. It is based on established techniques such as immunofluorescence, PCR-based detection, and imaging-based analysis, which ensure reliable results even at very low tumor cell counts. Growing integration with AI-powered image analysis and digital pathology systems is further improving diagnostic precision and workflow efficiency. Increasing adoption in tertiary care hospitals across the United Arab Emirates and Egypt is strengthening its clinical and research utilization.
The Microchips segment is expected to witness the fastest growth rate of 22.6% from 2026 to 2033, driven by increasing adoption of lab-on-chip and microfluidic technologies for automated and high-precision CTC isolation. These systems enable efficient separation of rare tumor cells with minimal sample volumes, making them highly suitable for advanced oncology applications. For instance, healthcare facilities in the United Arab Emirates are increasingly using microfluidic chip-based platforms for real-time cancer monitoring and research studies. Their automated operation reduces manual intervention and improves reproducibility in diagnostic workflows. Rising investments in precision medicine infrastructure in Saudi Arabia and Qatar are accelerating adoption of these advanced technologies.
- By Application
On the basis of application, the Middle East and Africa CTC liquid biopsy market is segmented into cancer stem cell research, multiple chromosome abnormalities, and others. The Cancer Stem Cell Research segment dominated the market with the largest market revenue share of 51.3% in 2025, driven by increasing focus on tumor initiation, metastasis, and cancer recurrence mechanisms. CTC-based technologies are widely used to identify circulating tumor stem cells, which are strongly associated with treatment resistance and disease progression. Research institutes in Saudi Arabia and South Africa are actively investing in oncology research programs supported by government funding and international collaborations. The segment benefits from growing use of CTC analysis in drug discovery and development of targeted cancer therapies. Integration of stem cell biology with liquid biopsy platforms is enhancing translational oncology research capabilities in the region.
The Multiple Chromosome Abnormalities segment is expected to witness the fastest growth rate of 23.1% from 2026 to 2033, driven by increasing adoption of genomic and cytogenetic profiling in cancer diagnostics. CTC-based detection of chromosomal instability is increasingly used to evaluate tumor aggressiveness and predict treatment outcomes. For instance, advanced oncology centers in the United Arab Emirates are integrating chromosomal analysis with liquid biopsy platforms for improved cancer characterization. Rising use of next-generation sequencing technologies is enhancing detection of complex genetic alterations in circulating tumor cells. Growing demand for non-invasive genomic testing in countries such as Egypt and Saudi Arabia is further supporting segment growth.
- By End User
On the basis of end user, the Middle East and Africa CTC liquid biopsy market is segmented into research & academic institutes, reference laboratories, and hospitals & physician laboratories. The Hospitals & Physician Laboratories segment dominated the market with the largest market revenue share of 56.4% in 2025, driven by high patient inflow and strong integration of advanced oncology diagnostics into clinical workflows. Hospitals in Saudi Arabia and the United Arab Emirates are leading adoption due to advanced healthcare infrastructure and strong government support for cancer care programs. The segment benefits from increasing use of CTC liquid biopsy for real-time cancer monitoring and treatment response assessment. Growing collaboration between hospitals and diagnostic companies is improving access to advanced molecular testing platforms. Rising demand for minimally invasive diagnostic tools among physicians is further strengthening clinical adoption across tertiary care centers.
The Research & Academic Institutes segment is expected to witness the fastest growth rate of 21.9% from 2026 to 2033, driven by increasing investment in cancer research and biomarker discovery initiatives. These institutes are increasingly utilizing CTC liquid biopsy technologies to study tumor biology, metastasis, and therapeutic resistance mechanisms. Universities in South Africa and Egypt are expanding molecular oncology research programs focused on circulating tumor cell analysis. Growing participation in international research collaborations is improving access to advanced sequencing and imaging technologies. Increasing government and private funding for precision medicine research is accelerating adoption in academic environments.
Middle East and Africa Circulating Tumor Cells (CTC) Liquid Biopsy Market Regional Analysis
- Saudi Arabia dominated the Middle East and Africa CTC liquid biopsy market with the largest revenue share of 38.6% in 2025, supported by strong government healthcare modernization programs, rising oncology research initiatives, and increasing deployment of advanced diagnostic technologies in leading cancer centers
- The country is witnessing rapid integration of precision medicine and liquid biopsy technologies under national healthcare modernization programs such as Vision 2030, which is significantly strengthening cancer diagnostics infrastructure
- High investment in oncology research, expansion of specialized cancer treatment facilities, and growing collaborations with global biotechnology companies are further accelerating the adoption of CTC-based liquid biopsy solutions
The Saudi Arabia Circulating Tumor Cells (CTC) Liquid Biopsy Market Insight
Saudi Arabia dominated the Middle East and Africa CTC liquid biopsy market with the largest revenue share in 2025, driven by strong government-led healthcare transformation programs and expanding oncology infrastructure. The country is increasingly adopting precision medicine and advanced liquid biopsy technologies under Vision 2030 initiatives, strengthening cancer diagnostics capabilities. Rising investments in molecular diagnostics, research centers, and specialized cancer hospitals are further accelerating adoption of CTC-based testing. Healthcare providers are prioritizing early cancer detection and real-time disease monitoring using minimally invasive techniques. Increasing collaboration with global biotechnology firms is enhancing access to advanced CTC platforms. In addition, growing awareness among clinicians regarding personalized oncology is reinforcing market leadership in the region.
South Africa Circulating Tumor Cells (CTC) Liquid Biopsy Market Insight
The South Africa CTC liquid biopsy market is expected to expand at a substantial CAGR throughout the forecast period, driven by rising cancer incidence and improving diagnostic infrastructure. Increasing focus on early cancer detection and expanding adoption of molecular diagnostic tools are supporting market growth. The country is witnessing gradual integration of liquid biopsy technologies in both public and private healthcare systems. Growing participation in oncology research and clinical trials is further strengthening adoption of CTC-based platforms. Rising collaborations with international diagnostic companies are improving access to advanced technologies. In addition, increasing awareness of precision medicine is accelerating clinical utilization across urban healthcare centers.
United Arab Emirates Circulating Tumor Cells (CTC) Liquid Biopsy Market Insight
The United Arab Emirates CTC liquid biopsy market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by strong healthcare infrastructure and rapid adoption of advanced diagnostic technologies. The country is focusing on precision oncology and personalized medicine, supporting increased use of CTC-based liquid biopsy solutions. Expanding investments in cancer research centers and smart healthcare systems are further driving market development. Hospitals are increasingly integrating liquid biopsy platforms for real-time cancer monitoring and treatment evaluation. Strong presence of international healthcare providers is enhancing technology adoption. In addition, government initiatives to position the UAE as a medical innovation hub are supporting market expansion.
Egypt Circulating Tumor Cells (CTC) Liquid Biopsy Market Insight
The Egypt CTC liquid biopsy market is expected to grow at a considerable CAGR during the forecast period, fueled by rising cancer burden and increasing demand for advanced diagnostic solutions. Expanding healthcare reforms and growing investment in oncology infrastructure are supporting adoption of liquid biopsy technologies. The country is witnessing gradual integration of molecular diagnostics in major hospitals and academic research institutions. Increasing collaborations with global healthcare organizations are improving access to advanced CTC platforms. Rising awareness of early cancer detection and minimally invasive diagnostics is further driving market penetration. In addition, growing focus on improving cancer care outcomes is strengthening adoption across clinical settings.
Middle East and Africa Circulating Tumor Cells (CTC) Liquid Biopsy Market Share
The Middle East and Africa Circulating Tumor Cells (CTC) Liquid Biopsy industry is primarily led by well-established companies, including:
- Menarini Silicon Biosystems SpA (Italy)
- Thermo Fisher Scientific Inc. (U.S.)
- F. Hoffmann-La Roche Ltd (Switzerland)
- QIAGEN Germany)
- Illumina, Inc. (U.S.)
- Bio-Rad Laboratories, Inc. (U.S.)
- Exact Sciences Corporation (U.S.)
- Myriad Genetics, Inc. (U.S.)
- Guardant Health, Inc. (U.S.)
- Bio-Techne Corporation (U.S.)
- Sysmex Corporation (Japan)
- NeoGenomics Laboratories, Inc. (U.S.)
- ANGLE plc (U.K.)
- Miltenyi Biotec B.V. & Co. KG (Germany)
- RareCyte, Inc. (U.S.)
- Fluxion Biosciences, Inc. (U.S.)
- Biocept, Inc. (U.S.)
- Biolidics Limited (Singapore)
- Vortex Biosciences, Inc. (U.S.)
- Lucence Health Inc. (Singapore)
What are the Recent Developments in Middle East and Africa Circulating Tumor Cells (CTC) Liquid Biopsy Market?
- In June 2025, M42 partnered with AstraZeneca and SOPHiA GENETICS to deploy advanced liquid biopsy testing powered by the SOPHiA DDM platform across the UAE healthcare network. The initiative focuses on enabling non-invasive genomic profiling using blood-based testing, including circulating tumor-related biomarkers relevant to oncology care
- In May 2025, Clinical validation programs across GCC countries showed growing use of liquid biopsy technologies for early-stage cancer detection and monitoring. Hospitals in Saudi Arabia and UAE are increasingly incorporating circulating tumor cell and ctDNA analysis into oncology workflows
- In December 2024, Abu Dhabi introduced a population-level blood-based colorectal cancer screening program aimed at improving early cancer detection rates. The program integrates non-invasive liquid biopsy testing to identify cancer biomarkers in blood samples. It is designed to improve patient participation compared to traditional invasive screening methods
- In November 2023, King Faisal Specialist Hospital & Research Centre (KFSHRC) implemented advanced liquid biopsy technology capable of analyzing 50+ cancer-related genes and thousands of mutations from blood samples. This advancement significantly improves circulating tumor detection and molecular profiling for precision oncology
- In July 2021, Research institutions in the United Arab Emirates expanded collaborations focused on circulating tumor cell analysis and liquid biopsy technologies. The programs aim to improve understanding of tumor metastasis and cancer progression using blood-based diagnostic approaches
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.