North America Angioplasty Balloons Market
Market Size in USD Billion
CAGR :
% 
 USD
1.07 Billion
USD
1.19 Billion
2025
2033
USD
1.07 Billion
USD
1.19 Billion
2025
2033
| 2026 –2033 | |
| USD 1.07 Billion | |
| USD 1.19 Billion | |
|
|
|
|
North America Angioplasty Balloons Market Size
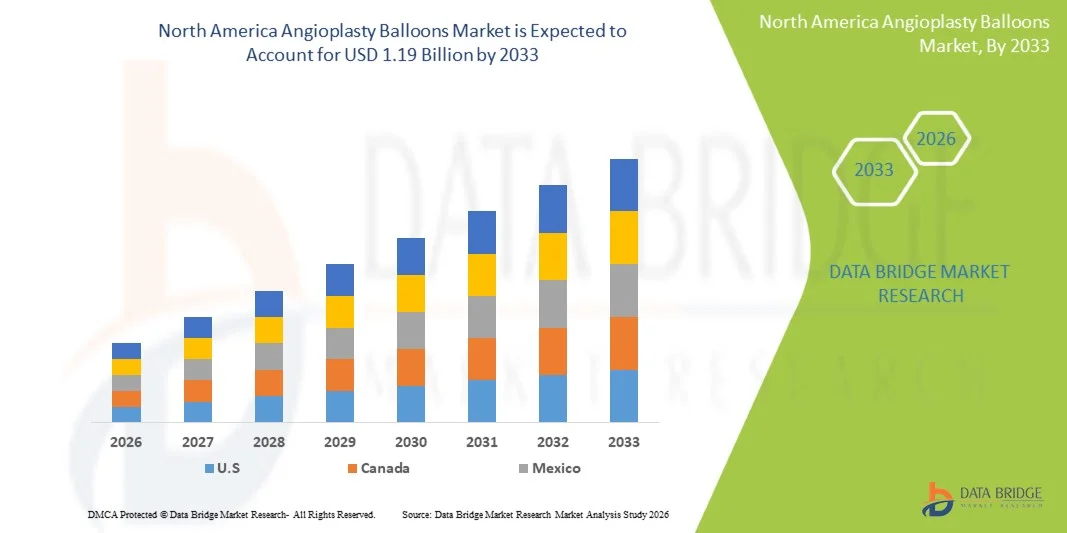
- The North America angioplasty balloons market size was valued at USD 1.07 billion in 2025 and is expected to reach USD 1.19 billion by 2033, at a CAGR of 1.4% during the forecast period
- The market growth is largely fueled by the rising prevalence of cardiovascular diseases, increasing number of percutaneous coronary intervention (PCI) procedures, and continuous advancements in balloon catheter technologies, leading to improved clinical outcomes and procedural efficiency across healthcare settings
- Furthermore, growing demand for minimally invasive treatment options, increasing geriatric population, and the adoption of advanced products such as drug-coated balloons and specialty balloons are establishing angioplasty balloons as a critical component in interventional cardiology. These converging factors are accelerating the adoption of angioplasty balloon technologies, thereby steadily supporting the market’s growth trajectory
North America Angioplasty Balloons Market Analysis
- Angioplasty balloons, used in minimally invasive procedures to dilate narrowed or obstructed blood vessels, are essential components in interventional cardiology and peripheral vascular treatments across hospitals and specialized cardiac centers due to their ability to restore blood flow effectively and improve patient outcomes
- The escalating demand for angioplasty balloons is primarily fueled by the increasing prevalence of cardiovascular diseases, growing number of percutaneous coronary intervention (PCI) procedures, and a rising preference for minimally invasive treatment approaches among both patients and healthcare providers
- United States dominated the angioplasty balloons market in North America with the largest revenue share of 82.5% in 2025, driven by advanced healthcare infrastructure, high adoption of innovative medical technologies, and a strong presence of key industry players, with the country experiencing substantial procedural volumes and product adoption, particularly supported by favorable reimbursement frameworks and continuous technological advancements such as drug-coated and specialty balloons
- Canada is expected to be the fastest growing country in the angioplasty balloons market during the forecast period due to increasing healthcare investments, rising adoption of minimally invasive procedures, and improving access to advanced cardiovascular treatments
- Drug-Coated Balloon (DCB) Angioplasty segment dominated the angioplasty balloons market with a market share of 38.6% in 2025, driven by their superior clinical benefits in reducing restenosis rates and improving long-term vessel patency, making them a preferred choice in complex vascular interventions
Report Scope and North America Angioplasty Balloons Market Segmentation
|
Attributes |
North America Angioplasty Balloons Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
North America Angioplasty Balloons Market Trends
“Advancements in Drug-Coated and Specialty Balloon Technologies”
- A significant and accelerating trend in the North America angioplasty balloons market is the increasing adoption of drug-coated balloons (DCBs) and specialty balloons such as cutting and scoring balloons, which are improving procedural precision and long-term patient outcomes. This advancement is significantly enhancing treatment effectiveness in complex cardiovascular interventions
- For instance, leading medical device manufacturers are introducing drug-coated balloons designed to reduce restenosis rates by delivering antiproliferative drugs directly to the arterial walls. Similarly, specialty balloons such as scoring balloons are gaining traction for their ability to treat calcified and resistant lesions more effectively
- The integration of advanced materials and coating technologies in angioplasty balloons enables better deliverability, controlled drug release, and improved lesion crossing capabilities. For instance, some next-generation balloons are engineered to enhance flexibility and reduce vessel trauma during procedures, thereby improving safety and efficiency. Furthermore, these technological improvements allow physicians to achieve better procedural success rates with fewer complications
- The growing adoption of advanced angioplasty balloons across hospitals and specialized cardiac centers is facilitating improved patient management and optimized treatment protocols. Through enhanced device performance, clinicians can treat a broader range of vascular conditions with greater accuracy, contributing to more consistent and reliable outcomes
- This trend towards more advanced, precise, and clinically effective balloon technologies is fundamentally reshaping interventional cardiology practices. Consequently, companies are developing innovative angioplasty balloon solutions with enhanced drug delivery mechanisms and improved structural designs to address complex cardiovascular conditions
- The demand for technologically advanced angioplasty balloons is growing steadily across healthcare facilities, as providers increasingly prioritize improved clinical outcomes, reduced repeat interventions, and cost-effective treatment solutions
- The integration of imaging-guided procedures and precision-based treatment approaches is enhancing the effectiveness of angioplasty balloons, enabling more accurate lesion targeting and improved procedural outcomes
North America Angioplasty Balloons Market Dynamics
Driver
“Rising Cardiovascular Disease Burden and Increasing Minimally Invasive Procedures”
- The increasing prevalence of cardiovascular diseases across North America, coupled with the growing preference for minimally invasive procedures, is a significant driver for the heightened demand for angioplasty balloons
- For instance, in recent years, healthcare providers have expanded the use of percutaneous coronary intervention (PCI) procedures supported by advanced balloon catheter technologies to improve treatment outcomes. Such strategies by key companies are expected to drive the angioplasty balloons market growth in the forecast period
- As patients and healthcare systems focus on reducing hospital stays and improving recovery times, angioplasty balloons offer effective solutions for restoring blood flow with minimal surgical intervention, providing a strong alternative to traditional surgical methods
- Furthermore, the advancement of healthcare infrastructure and increasing adoption of interventional cardiology procedures are making angioplasty balloons an essential component of cardiovascular treatment protocols, offering improved efficiency and patient care
- The convenience of shorter recovery periods, reduced procedural risks, and enhanced clinical outcomes are key factors propelling the adoption of angioplasty balloons across hospitals and specialty clinics. The increasing availability of technologically advanced balloon products further contributes to market growth
- The rising geriatric population, particularly in United States and Canada, is further increasing the patient pool susceptible to cardiovascular diseases, thereby boosting demand for angioplasty procedures
- Increasing investments by healthcare providers in advanced catheterization laboratories and interventional cardiology infrastructure are supporting higher procedural volumes and technology adoption
Restraint/Challenge
“High Procedure Costs and Product Recall Risks”
- The high cost associated with advanced angioplasty balloon procedures and devices, including drug-coated balloons, poses a significant challenge to broader market adoption, particularly in cost-sensitive healthcare systems
- For instance, pricing pressures and reimbursement limitations in certain healthcare settings can restrict the widespread use of premium angioplasty balloon technologies despite their clinical benefits
- Addressing cost-related challenges through pricing strategies, reimbursement support, and value-based healthcare models is crucial for improving accessibility. Companies are focusing on optimizing production and enhancing product efficiency to justify costs. In addition, the risk of product recalls and stringent regulatory requirements can impact market growth, as device failures or compliance issues may lead to temporary supply disruptions and increased scrutiny
- While technological advancements continue to improve product performance, regulatory approvals and compliance processes can delay product launches and limit rapid adoption, particularly for innovative balloon technologies
- Overcoming these challenges through cost optimization, strong regulatory compliance, and continuous product innovation will be vital for sustained market growth
- The limited availability of skilled interventional cardiologists in certain regions can restrict the adoption of advanced angioplasty procedures, impacting overall market expansion
- Concerns related to procedural complications such as restenosis or vessel damage may influence clinical decision-making and slow the adoption of certain advanced balloon technologies
North America Angioplasty Balloons Market Scope
The market is segmented on the basis of type, material, balloon type, disease indication, and end user.
- By Type
On the basis of type, the North America angioplasty balloons market is segmented into plain old balloon angioplasty, drug-coated balloon (DCB) angioplasty, cutting balloons, scoring balloons, and stent graft balloon catheter. The drug-coated balloon (DCB) angioplasty segment dominated the market with the largest market revenue share 38.6% in 2025, driven by its superior ability to reduce restenosis rates and improve long-term vessel patency without leaving a permanent implant. Healthcare providers increasingly prefer DCBs in complex lesions and peripheral artery disease due to their targeted drug delivery mechanism. The growing clinical evidence supporting improved patient outcomes and reduced repeat interventions further strengthens their adoption. In addition, favorable reimbursement scenarios and rising awareness among clinicians contribute to the segment’s dominance. The continuous innovation in coating technologies and drug formulations also enhances their therapeutic effectiveness and procedural success rates.
The scoring balloons segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by its increasing use in treating calcified and resistant lesions. These balloons provide controlled plaque modification, reducing the risk of vessel damage and improving procedural precision. Their ability to enhance lesion preparation before stent placement makes them highly valuable in complex interventions. Increasing demand for advanced interventional tools and growing procedural volumes further support segment growth. In addition, technological advancements in balloon design are improving flexibility and deliverability. The rising adoption in both coronary and peripheral procedures is accelerating their market expansion.
- By Material
On the basis of material, the market is segmented into nylon, polyurethane, silicone urethane co-polymers, and other materials. The nylon segment dominated the market with the largest market revenue share in 2025, driven by its high tensile strength, durability, and excellent pressure resistance, making it suitable for non-compliant balloon applications. Nylon-based balloons are widely used in high-pressure procedures due to their ability to maintain shape and deliver precise dilation. The material’s reliability and compatibility with advanced catheter technologies further enhance its adoption. In addition, its cost-effectiveness compared to alternative materials supports its widespread use across healthcare facilities. Continuous improvements in polymer processing are also enhancing performance characteristics. These factors collectively contribute to its dominant position in the market.
The silicone urethane co-polymers segment is expected to witness the fastest growth rate during the forecast period, driven by their superior flexibility, biocompatibility, and enhanced deliverability in complex vascular pathways. These materials allow for improved navigation through tortuous vessels, reducing procedural risks. Their growing use in next-generation angioplasty balloons supports advanced treatment approaches. Increasing demand for minimally invasive procedures further accelerates their adoption. In addition, advancements in material science are improving their strength and performance characteristics. This makes them highly suitable for specialized and complex interventions.
- By Balloon Type
On the basis of balloon type, the market is segmented into semi-compliant and non-compliant balloons. The non-compliant segment dominated the market with the largest revenue share in 2025, driven by its ability to maintain consistent diameter under high pressure, ensuring precise lesion dilation. These balloons are widely used in post-dilatation and high-pressure procedures where accuracy is critical. Their effectiveness in treating calcified and rigid lesions further enhances their adoption. The growing demand for precision-based interventional procedures supports their widespread use. In addition, advancements in balloon technology are improving performance and safety. These factors contribute to the strong dominance of non-compliant balloons in the market.
The semi-compliant segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by its flexibility and adaptability to varying vessel sizes. These balloons are particularly useful in initial lesion crossing and pre-dilatation procedures. Their ability to conform to vessel anatomy reduces the risk of complications during interventions. Increasing procedural volumes and the need for versatile treatment options drive their demand. In addition, improvements in material design are enhancing their performance and safety profile. The growing adoption in a wide range of angioplasty procedures supports their rapid growth.
- By Disease Indication
On the basis of disease indication, the market is segmented into coronary angioplasty, venous angioplasty, carotid angioplasty, renal artery angioplasty, and peripheral angioplasty. The coronary angioplasty segment dominated the market with the largest revenue share in 2025, driven by the high prevalence of coronary artery disease and the increasing number of PCI procedures. The segment benefits from strong clinical adoption and well-established treatment protocols. Continuous advancements in interventional cardiology further enhance treatment outcomes. In addition, favorable reimbursement policies support the widespread use of coronary angioplasty procedures. The growing geriatric population also contributes to increased demand. These factors collectively drive the dominance of this segment.
The peripheral angioplasty segment is expected to witness the fastest growth rate during the forecast period, fueled by the rising incidence of peripheral artery disease and increasing awareness regarding its treatment. The growing adoption of minimally invasive procedures in peripheral interventions supports segment expansion. Technological advancements in balloon design are improving treatment effectiveness. In addition, increasing healthcare investments are expanding access to advanced procedures. The rising demand for early diagnosis and treatment further drives growth. These factors contribute to the rapid expansion of the peripheral angioplasty segment.
- By End User
On the basis of end user, the market is segmented into cath labs, hospitals, specialty clinics, ambulatory surgery centers, and diagnostic centers. The hospitals segment dominated the market with the largest revenue share in 2025, driven by the availability of advanced infrastructure and skilled healthcare professionals. Hospitals handle a high volume of complex cardiovascular procedures, supporting the extensive use of angioplasty balloons. The presence of specialized cardiac units further enhances procedural efficiency. In addition, strong reimbursement frameworks contribute to higher patient inflow. Continuous investments in healthcare infrastructure also support segment growth. These factors collectively establish hospitals as the leading end user segment.
The ambulatory surgery centers segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by the increasing shift toward outpatient procedures and cost-effective treatment options. These centers offer shorter hospital stays and faster recovery times, attracting both patients and healthcare providers. The growing preference for minimally invasive procedures supports their expansion. In addition, advancements in medical technology enable complex procedures to be performed in outpatient settings. Increasing healthcare cost pressures further drive the adoption of these centers. These factors contribute to the rapid growth of ambulatory surgery centers in the market.
North America Angioplasty Balloons Market Regional Analysis
- United States dominated the angioplasty balloons market in North America with the largest revenue share of 82.5% in 2025, driven by advanced healthcare infrastructure, high adoption of innovative medical technologies, and a strong presence of key industry players
- Healthcare providers in the region highly prioritize minimally invasive treatment options, clinical efficiency, and improved patient outcomes offered by angioplasty balloons, along with the integration of advanced technologies such as drug-coated and specialty balloons in cardiovascular procedures
- This widespread adoption is further supported by advanced healthcare infrastructure, high healthcare expenditure, and the presence of a technologically advanced medical ecosystem, establishing angioplasty balloons as a critical component in interventional cardiology across both hospitals and specialized cardiac centers
U.S. Angioplasty Balloons Market Insight
The United States angioplasty balloons market captured the largest revenue share of 82.5% in 2025 within North America, fueled by the high prevalence of cardiovascular diseases and the increasing volume of percutaneous coronary intervention (PCI) procedures. Healthcare providers are increasingly prioritizing minimally invasive treatments that offer improved patient outcomes and faster recovery times. The growing adoption of advanced technologies such as drug-coated balloons and specialty balloons further propels the market. Moreover, strong reimbursement frameworks and continuous investments in healthcare infrastructure are significantly contributing to the market's expansion.
Canada Angioplasty Balloons Market Insight
The Canada angioplasty balloons market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by increasing healthcare investments and rising adoption of minimally invasive cardiovascular procedures. In addition, growing awareness regarding early diagnosis and treatment of cardiovascular diseases is encouraging demand for angioplasty balloons. Canada’s well-structured healthcare system and focus on improving patient outcomes are further supporting market growth. The integration of advanced balloon technologies in interventional cardiology practices is expected to continue to stimulate demand.
Mexico Angioplasty Balloons Market Insight
The Mexico angioplasty balloons market is expected to expand at a considerable CAGR during the forecast period, fueled by improving healthcare infrastructure and increasing access to cardiovascular treatments. The rising prevalence of cardiovascular diseases and growing awareness about minimally invasive procedures are driving adoption. Mexico’s expanding private healthcare sector is also contributing to the demand for advanced interventional devices. In addition, ongoing healthcare reforms and investments are supporting the adoption of angioplasty balloon technologies across the country.
North America Angioplasty Balloons Market Share
The North America Angioplasty Balloons industry is primarily led by well-established companies, including:
- Medtronic (Ireland)
- Boston Scientific Corporation (U.S.)
- Abbott (U.S.)
- B. Braun SE (Germany)
- Terumo Corporation (Japan)
- BIOTRONIK SE & Co. KG (Germany)
- Cook (U.S.)
- Cordis (U.S.)
- Koninklijke Philips N.V. (Netherlands)
- BD (U.S.)
- AngioDynamics, Inc. (U.S.)
- Merit Medical Systems, Inc. (U.S.)
- Teleflex Incorporated (U.S.)
- Integer Holdings Corporation (U.S.)
- NATEC Medical Ltd. (Ireland)
- ENDOCOR GmbH (Germany)
- Cardinal Health, Inc. (U.S.)
- Alvimedica Medical Technologies (Turkey)
- Hexacath (France)
- QT Vascular Ltd. (Singapore)
What are the Recent Developments in North America Angioplasty Balloons Market?
- In March 2025, new clinical findings presented from ongoing studies of the AGENT drug-coated balloon demonstrated superior outcomes compared to uncoated balloons, including lower rates of repeat interventions and adverse cardiac events. These results reinforce the long-term safety and effectiveness of drug-coated balloons in treating in-stent restenosis and are expected to further increase physician confidence and clinical adoption across the region
- In October 2024, Medtronic received FDA approval to initiate an Investigational Device Exemption (IDE) trial for its Prevail™ drug-coated balloon, aimed at evaluating its safety and effectiveness in treating coronary artery disease. The study includes head-to-head comparison of drug-coated balloons in the U.S., supporting future regulatory submissions. This development highlights growing innovation and competition in the angioplasty balloon segment, particularly in expanding drug-coated balloon indications
- In March 2024, Boston Scientific announced that it received U.S. FDA approval for its AGENT™ Drug-Coated Balloon, marking the first coronary drug-coated balloon approved in the United States for treating in-stent restenosis. The device delivers paclitaxel directly to the vessel wall to prevent re-narrowing and offers an alternative to repeat stenting. This approval represents a major milestone in interventional cardiology and is expected to significantly accelerate the adoption of drug-coated balloon technologies across North America
- In September 2023, the U.S. FDA approved the SurVeil Drug-Coated Balloon, designed to treat peripheral artery disease in the thigh and knee by delivering paclitaxel to prevent restenosis. Clinical data demonstrated strong efficacy, with over 80% vessel patency at 12 months. This approval strengthened the availability of advanced angioplasty solutions for peripheral interventions and expanded treatment options for patients with vascular diseases in North America
- In March 2022, Philips received U.S. FDA approval for its Stellarex 0.035” over-the-wire drug-coated angioplasty balloon, designed for the treatment of peripheral artery disease (PAD) in the thigh and knee. The device uses a paclitaxel coating to help prevent restenosis by inhibiting cell proliferation after vessel dilation. This approval expanded its indication to include treatment within previously stented vessels, strengthening its clinical utility
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.