North America Fertility Testing Devices Market
Market Size in USD Billion
CAGR :
% 
 USD
36.69 Billion
USD
61.87 Billion
2025
2033
USD
36.69 Billion
USD
61.87 Billion
2025
2033
| 2026 –2033 | |
| USD 36.69 Billion | |
| USD 61.87 Billion | |
|
|
|
|
North America Fertility Testing Devices Market Size
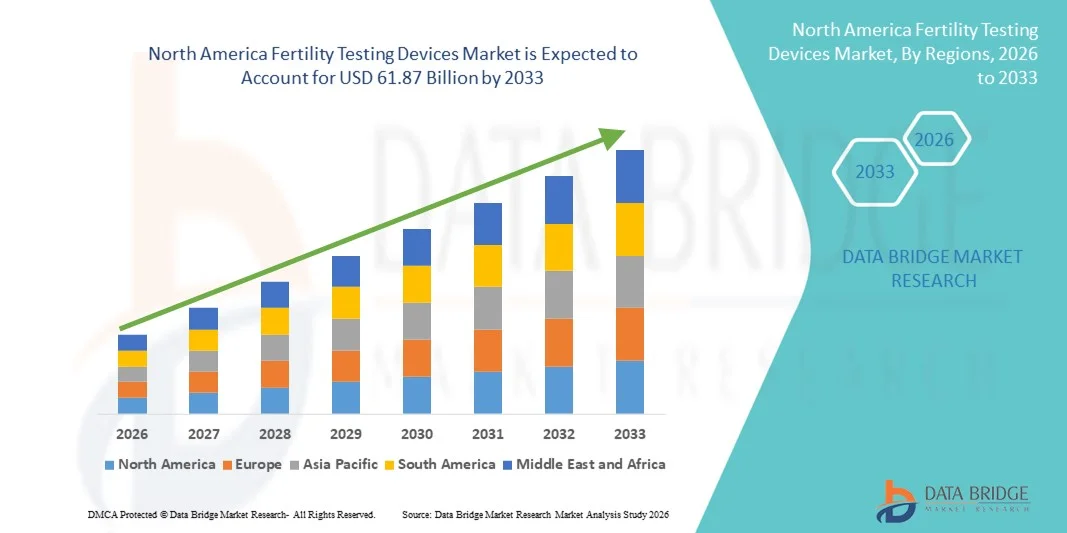
- The North America fertility testing devices market size was valued at USD 36.69 billion in 2025 and is expected to reach USD 61.87 billion by 2033, at a CAGR of6.75% during the forecast period
- The market growth is largely fueled by increasing awareness of reproductive health, rising prevalence of infertility, and advancements in diagnostic technologies, leading to greater adoption of at-home and clinical fertility testing solutions
- Furthermore, growing consumer preference for convenient, accurate, and rapid testing methods, coupled with supportive government initiatives and rising healthcare spending, is establishing fertility testing devices as essential tools for both personal and clinical use. These converging factors are accelerating the uptake of Fertility Testing Devices solutions, thereby significantly boosting the industry's growth
North America Fertility Testing Devices Market Analysis
- Fertility testing devices, offering clinical and at-home diagnostic solutions, are increasingly vital tools in reproductive healthcare due to their enhanced accuracy, ease of use, and integration with digital health platforms
- The escalating demand for fertility testing devices is primarily fueled by rising awareness of reproductive health, growing prevalence of infertility, and the increasing adoption of rapid, convenient, and smartphone-enabled testing solutions
- The U.S. dominated the Fertility Testing Devices market with the largest revenue share of 38.7% in 2025, characterized by advanced healthcare infrastructure, high awareness of reproductive health, and strong presence of key industry players, with substantial growth in digital diagnostics, AI-assisted fertility monitoring, and smartphone-integrated devices
- Canada is expected to be the fastest growing country in the Fertility Testing Devices market during the forecast period, with a projected CAGR of 12.3%, driven by rising fertility awareness, government initiatives promoting reproductive health, and growing adoption of home-based testing kits
- The female fertility testing devices segment dominated the largest market revenue share of 68% in 2025, owing to the widespread use of ovulation prediction kits and hormone-based monitors
Report Scope and Fertility Testing Devices Market Segmentation
|
Attributes |
Fertility Testing Devices Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
North America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework. |
North America Fertility Testing Devices Market Trends
Increasing Adoption of At-Home and Advanced Fertility Monitoring Solutions
- A significant and accelerating trend in the North America Fertility Testing Devices market is the growing adoption of at-home fertility monitoring kits and digital diagnostic solutions. Consumers are increasingly seeking convenient, private, and accurate methods to monitor ovulation, hormone levels, and reproductive health without frequent visits to clinics
- For instance, leading companies such as Clearblue and Everlywell have introduced devices that integrate smartphone connectivity, providing real-time insights, cycle tracking, and predictive analytics for ovulation and fertility windows
- There is also a growing trend toward non-invasive and hormone-based assays that enhance accuracy and reliability of fertility predictions, reducing the chances of false results and improving user confidence
- Wearable fertility monitors, smart basal thermometers, and integrated apps are expanding the functionality of traditional testing kits, enabling users to track multiple parameters such as luteinizing hormone surge, basal body temperature, and cervical mucus changes in a comprehensive manner
- The market is witnessing innovation in multi-function fertility kits, combining ovulation, pregnancy, and hormonal imbalance testing, providing holistic reproductive health insights and reducing the need for multiple individual tests
North America Fertility Testing Devices Market Dynamics
Driver
Rising Infertility Rates and Increased Awareness of Reproductive Health
- The increasing prevalence of infertility and reproductive health challenges among couples in North America is a key driver for the Fertility Testing Devices market. Delayed parenthood, lifestyle factors, and medical conditions such as polycystic ovary syndrome (PCOS) have contributed to heightened demand for early detection and monitoring solutions
- For Instance, Growing awareness about the importance of reproductive health and fertility planning is encouraging individuals and couples to adopt testing solutions at home or in clinics. According to CDC, infertility affects nearly 1 in 8 couples, underscoring the market potential
- Rising demand for personalized fertility management and assisted reproductive technologies, including IVF and ovulation induction, is driving the consumption of accurate and easy-to-use testing devices
- Increasing support from healthcare providers, fertility clinics, and telemedicine platforms promoting self-monitoring solutions is further expanding market adoption
- Convenience, affordability, and time-saving benefits offered by at-home fertility testing kits are major factors influencing the growing consumer preference over traditional clinic-based testing
Restraint/Challenge
High Cost of Advanced Devices and Regulatory Compliance Requirements
- The relatively high cost of advanced fertility testing devices, especially those offering digital tracking, app integration, or multi-parameter monitoring, can limit adoption among price-sensitive consumers.
- Stringent regulatory standards and approval requirements for diagnostic devices in North America may delay market entry for new products or create barriers for smaller manufacturers
- For instance, the FDA requires rigorous validation of accuracy and safety for ovulation and hormone-based testing devices
- Accuracy limitations and user errors in home-based testing can impact consumer confidence, making education and guidance critical for sustained adoption
- Limited awareness in certain demographic segments and the need for healthcare provider consultation to interpret complex hormonal results may restrict widespread acceptance
- Addressing these challenges through cost-effective device innovation, simplified user instructions, and robust regulatory compliance will be essential for long-term growth in the Fertility Testing Devices market
North America Fertility Testing Devices Market Scope
The market is segmented on the basis of mode of purchase, application, product, and end-user.
- By Mode of Purchase
On the basis of mode of purchase, the market is segmented into over-the-counter (OTC) and prescription-based devices. The OTC segment dominated the largest market revenue share of 55% in 2025, driven by the increasing consumer preference for home-based fertility monitoring and ease of access without a physician’s prescription. Growing awareness about reproductive health, rising adoption of self-testing kits, and user-friendly designs support widespread utilization. OTC devices, including ovulation prediction kits and fertility monitors, offer convenience and privacy for users. Marketing campaigns, e-commerce availability, and pharmacy distribution channels enhance reach. Technological advancements improving accuracy further fuel adoption. Social trends encouraging family planning and delayed parenthood are boosting demand. Rising fertility awareness among millennials and Gen Z contributes to sustained growth. Developed regions such as North America and Europe remain key contributors due to higher disposable incomes. Regulatory approvals for safe home use reinforce consumer confidence.
The prescription-based segment is expected to witness the fastest CAGR of 11.5% from 2026 to 2033, driven by the need for clinical-grade fertility testing in complex cases. Hospitals and fertility clinics prefer prescription devices for accurate diagnostics and integration with patient records. Adoption is supported by physician recommendations for high-risk or subfertile patients. Advanced monitoring devices that track hormonal patterns and sperm quality are increasingly used in clinical settings. Technological innovations, including connected monitors and digital data capture, enhance precision and reliability. Regulatory compliance in developed markets ensures patient safety. Insurance coverage and reimbursement policies also promote prescription device uptake. Emerging markets with growing fertility clinics are expanding adoption opportunities. Academic and research centers leverage these devices for studies and clinical trials. Integration with fertility treatment plans, such as IVF, drives usage.
- By Application
On the basis of application, the market is segmented into female fertility testing devices and male fertility testing devices. The female fertility testing devices segment dominated the largest market revenue share of 68% in 2025, owing to the widespread use of ovulation prediction kits and hormone-based monitors. Rising awareness about menstrual health and fertility planning drives adoption. Women increasingly prefer home-based self-testing for convenience and privacy. Hospitals and fertility clinics also utilize advanced devices to support reproductive treatments. Technological advancements, including digital and wearable fertility trackers, enhance accuracy. Social trends of delayed pregnancies and family planning contribute to demand. Marketing campaigns and online retail channels expand reach. Integration with mobile apps provides easy interpretation and tracking. Academic studies supporting the efficacy of female fertility devices encourage adoption. Clinical partnerships with healthcare providers strengthen market penetration.
The male fertility testing devices segment is expected to witness the fastest CAGR of 12% from 2026 to 2033, driven by rising awareness about male infertility and sperm health monitoring. Fertility clinics and hospitals increasingly recommend home-based semen analysis kits. Advancements in microfluidics and smartphone-enabled devices improve convenience and accuracy. Growing awareness campaigns on male reproductive health support uptake. Integration with fertility treatment plans such as IVF encourages clinical adoption. Technological innovations providing rapid results and automated analysis increase usage. Male fertility testing in emerging markets is witnessing adoption due to awareness programs. Insurance policies covering male fertility tests promote growth. The younger population seeking family planning solutions fuels demand. Research initiatives on male fertility trends also drive market growth.
- By Product
On the basis of product, the market is segmented into ovulation prediction kits and fertility monitors. The ovulation prediction kit segment dominated the largest market revenue share of 61% in 2025, driven by their affordability, ease of use, and effectiveness in tracking fertile windows. Home users prefer these kits for planning conception without the need for clinical visits. E-commerce platforms and pharmacy sales enhance accessibility. Rising awareness about menstrual cycles and fertility health fuels adoption. Technological enhancements, including digital readouts and smartphone integration, improve user experience. Clinics and hospitals also use ovulation kits for preliminary assessment. Marketing campaigns targeting reproductive-aged women increase reach. Developed markets with higher disposable income dominate consumption. Growing social trends of planned parenthood and delayed conception encourage usage. Continuous innovation ensures improved accuracy and reliability.
The fertility monitor segment is expected to witness the fastest CAGR of 12.3% from 2026 to 2033, driven by demand for multi-parameter monitoring devices that track hormones, cycle phases, and fertility windows. Advanced monitors offer connectivity with mobile apps, personalized insights, and predictive analytics. Hospitals and fertility clinics adopt monitors for precise ovulation tracking and fertility planning. Integration with AI and cloud-based data storage enhances accuracy and convenience. Awareness about personalized fertility tracking fuels adoption. Premium devices in developed markets attract tech-savvy users. Emerging markets show increasing uptake due to fertility awareness campaigns. Clinical-grade monitors are increasingly incorporated into treatment protocols. Research initiatives supporting monitor efficacy encourage usage. Overall, the segment benefits from digital health trends and consumer demand for reliable, long-term fertility tracking solutions.
- By End-User
On the basis of end-user, the market is segmented into home care settings, hospitals, fertility clinics, and others. The home care settings segment dominated the largest market revenue share of 59% in 2025, driven by the convenience of self-testing and privacy in reproductive health management. Rising digital literacy, availability of user-friendly kits, and online purchase options enhance adoption. Awareness campaigns, social media influence, and telemedicine support usage. Home care adoption reduces clinic visits and empowers users to track fertility independently. Developed countries dominate due to higher disposable income and better access to devices. Integration with mobile apps for cycle tracking and fertility analytics fuels growth. Consumer preference for non-invasive, quick results supports widespread usage. Online education and support services improve trust and utilization. Emerging markets are witnessing gradual adoption due to increasing fertility awareness programs.
The fertility clinics segment is expected to witness the fastest CAGR of 11.8% from 2026 to 2033, driven by rising infertility cases, IVF procedures, and advanced reproductive treatments. Clinics require clinical-grade devices for accurate diagnostics, sperm and ovulation monitoring, and patient treatment plans. Technological advancements, including connected monitors and multi-parameter testing, enhance clinical efficiency. Integration with electronic health records ensures streamlined patient management. Insurance coverage and government support for fertility treatments encourage adoption. Research institutions partner with clinics to evaluate new devices. Emerging regions are seeing new fertility clinics supporting market growth. High demand for precise testing and predictive analytics drives segment expansion.
North America Fertility Testing Devices Market Regional Analysis
- North America dominated the fertility testing devices market with a revenue share in 2025. The region’s growth is driven by advanced healthcare infrastructure, high awareness of reproductive health, and the strong presence of leading industry players
- Rising adoption of both at-home and clinical fertility testing solutions supports market expansion. Innovations in digital diagnostics, AI-assisted analysis, and smartphone-integrated devices are enhancing accessibility and accuracy
- High disposable incomes, supportive reimbursement policies, and technological adoption further encourage uptake. Hospitals, fertility clinics, and home-use platforms are rapidly integrating these solutions. Overall, North America represents a mature, innovation-driven market for fertility testing devices
U.S. Fertility Testing Devices Market Insight
The U.S. fertility testing devices market accounted for the largest revenue share of 38.7% within North America, driven by rising awareness of reproductive health and demand for at-home and clinical fertility testing. The market benefits from advanced healthcare infrastructure, strong industry presence, and innovations such as AI-assisted diagnostics and smartphone-integrated devices. Fertility clinics and hospitals are increasingly adopting digital solutions to improve accuracy and patient convenience. High patient engagement, favorable reimbursement frameworks, and robust research and development further fuel adoption. Government programs, public awareness campaigns, and increasing patient preference for non-invasive testing support growth. Overall, the U.S. market remains the largest and most technologically advanced in North America.
Canada Fertility Testing Devices Market Insight
Canada fertility testing devices market is expected to be the fastest-growing country in the North American Fertility Testing Devices market, with a projected CAGR of 12.3% during the forecast period. Growth is driven by rising healthcare spending, increasing fertility awareness, and government initiatives supporting reproductive health diagnostics. Expanding access to clinics, at-home testing kits, and smartphone-integrated solutions is encouraging adoption. Technological innovations, including AI-assisted fertility analysis and digital diagnostic platforms, enhance patient convenience. Rising demand for minimally invasive and user-friendly testing solutions also supports market expansion. Overall, Canada represents a high-growth opportunity within the North American fertility testing devices market.
North America Fertility Testing Devices Market Share
The Fertility Testing Devices industry is primarily led by well-established companies, including:
- Abbott (U.S.)
- Siemens Healthineers (Germany)
- Roche Diagnostics (Switzerland)
- Clearblue (Switzerland)
- Merck KGaA (Germany)
- Fertility Focus (U.K.)
- Quest Diagnostics (U.S.)
- FertilityFriend (U.K.)
- Life Extension (U.S.)
- Kindara (U.S.)
- Alere Inc. (U.S.)
- CareStart (U.S.)
- Pharmacopeia (U.S.)
- Ferticare Europe (Netherlands)
- CooperSurgical (U.S.)
- Preven Kits (U.S.)
- Obstetrix (U.S.)
Latest Developments in North America Fertility Testing Devices Market
- In July 2023, Quest Diagnostics, a leading clinical laboratory services provider, partnered with Proov to introduce the Proov Confirm PdG home fertility test on QuestHealth.com, marking the commercial launch of an FDA‑cleared home kit designed to confirm successful ovulation by detecting pregnanediol glucuronide (PdG) — a key progesterone metabolite — in urine, expanding consumer access to clinically validated fertility insight tools
- In December 2024, Merck KGaA announced the acquisition of Dexeco, a fertility diagnostics business, as part of its strategy to expand its reproductive health and fertility device portfolio, signaling increasing consolidation and investment in fertility testing technologies
- In February 2025, Clearblue announced a strategic collaboration with Natural Cycles, combining Clearblue’s advanced digital ovulation testing technology with Natural Cycles’ app‑based fertility tracking algorithm to offer an integrated solution that enables users to monitor fertility and reproductive health more comprehensively
- In June 2025, Clearblue launched a next‑generation at‑home fertility testing kit designed to deliver enhanced accuracy, improved ease of use, and greater convenience for consumers seeking reliable insights into fertility windows, supporting the growing demand for user‑friendly home diagnostic tools
- In May 2025, Quest Diagnostics and Fairhaven Health entered into a strategic partnership to expand access to at‑home fertility testing kits through Quest’s broad clinical network, improving the availability of fertility diagnostics at scale across diverse patient populations
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.