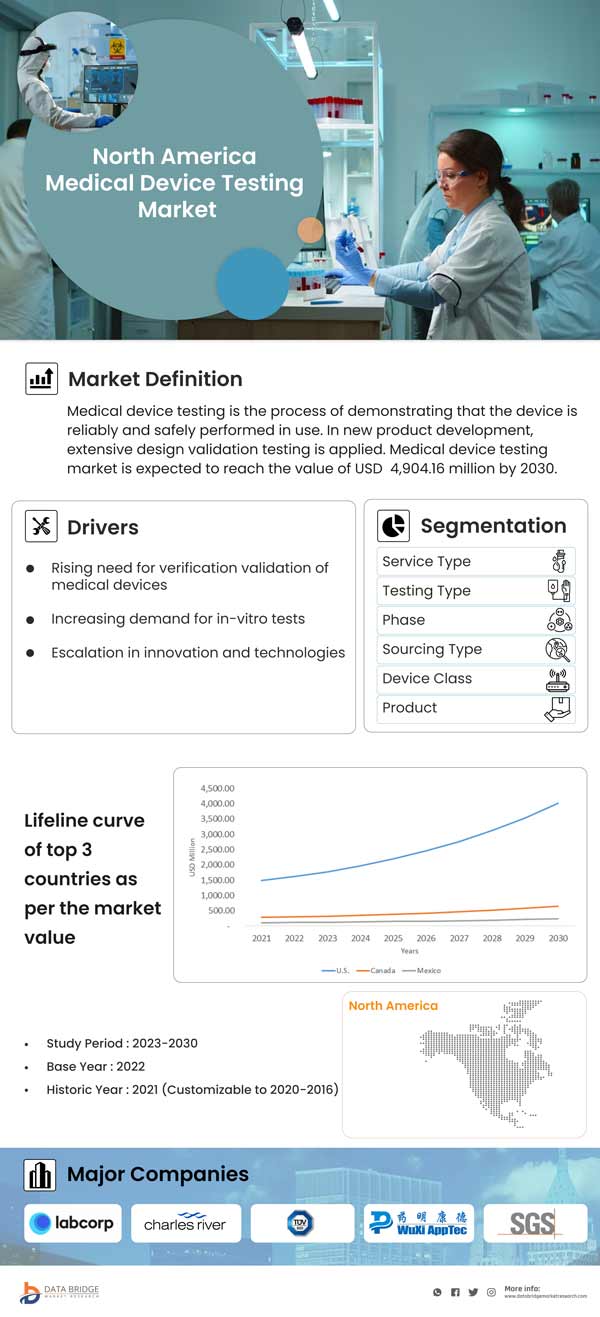
North America Medical Device Testing Market, By Service Type (Testing Services, Inspection Service and Certification Services), Testing Type (Physical Testing, Chemical/Biological Testing, Cybersecurity Testing, Microbiology and Sterility Testing, and Others), Phase (Preclinical and Clinical), Sourcing Type (In-House and Outsourced), Device Class (Class I, Class II and Class III), Product (Active Implant Medical Device, Active Medical Device, Non-active Medical Device, In vitro Diagnostics Medical Device, Opthalmic Medical Device, Orthopedic and Dental Medical Device, Vascular Medical Device, and Others) - Industry Trends and Forecast to 2030.
Access Full Report @ https://www.databridgemarketresearch.com/reports/north-america-medical-device-testing-market
Office Number 402, Amanora Chambers, Magarpatta Road,
Hadapsar Pune – 411028.
Email: [email protected]
Blog @ https://www.databridgemarketresearch.com/news