アジア太平洋地域の CRISPR 遺伝子検出および診断市場、クラス別 (クラス 1 - 複数のエフェクタータンパク質、クラス 2 - 単一の CrRNA 結合タンパク質)、製品とサービス (製品とサービス)、アプリケーション (バイオメディカル診断、ゲノムエンジニアリング、創薬、農業アプリケーションなど)、ワークフロー (サンプル調製、前増幅、CrRNA、Cas 酵素およびセンシング)、エンドユーザー (病院、診断センター、バイオテクノロジー企業、学術研究機関など)、流通チャネル (直接入札、小売販売) 業界動向と 2029 年までの予測
市場の定義と洞察
CRISPR は、規則的に間隔を空けた短い回文反復配列が密集したゲノム編集ツールであり、研究者が DNA 配列を変更し、遺伝子機能を簡単に修正できるようにします。遺伝子欠陥の修正、病気の治療と蔓延の防止など、多くの潜在的な用途があります。CRISPR ベースの診断は、感染性および非感染性疾患の核酸ベースのバイオマーカーの感知や遺伝性疾患の検出など、多くの生物医学用途に使用されています。CRISPR のアッセイ キットは、Cas9 と呼ばれるタンパク質と、特定の遺伝コードを持つ核酸分子の文字列であるガイド RNA の 2 つのコンポーネントで構成されています。
この CRISPR-Cas9 システムは、哺乳類細胞での使用向けに改良されています。非相同末端結合 (NHEJ) によるフレームシフト変異を導入することで、目的の遺伝子に固有のガイド配列 (sgRNA) を導入して特定の遺伝子をノックアウトするか、ノックイン変異を生成することができます。
CRISPR-Cas 9 システムにより、遺伝子および細胞治療における診断とサービスの範囲が拡大しました。製薬会社は新製品の開発に多額の投資を行っており、遺伝子および細胞治療薬が開発初期段階に急増しています。市場プレーヤーが投資することで、深刻な治療を必要とする患者に安全で効果的な治療を提供できるようになります。
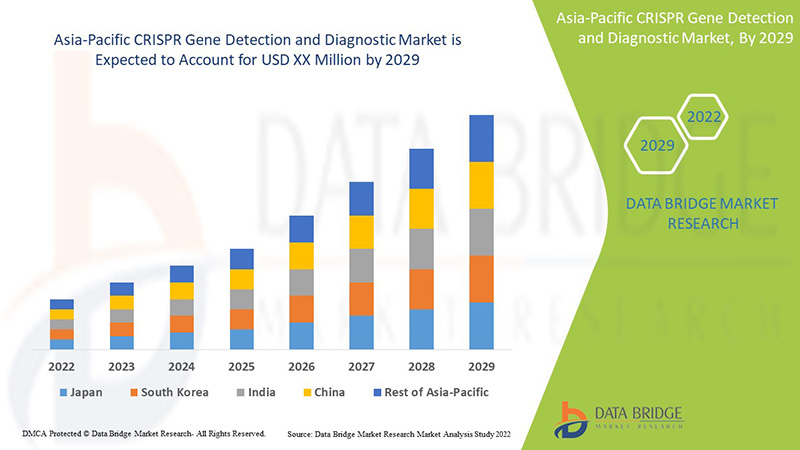
アジア太平洋地域の CRISPR 遺伝子検出および診断は支援的であり、症状の重症度を軽減することを目指しています。Data Bridge Market Research は、CRISPR 遺伝子検出および診断市場は 2022 年から 2029 年の予測期間中に 21.6% の CAGR で成長すると分析しています。
|
レポートメトリック |
詳細 |
|
予測期間 |
2022年から2029年 |
|
基準年 |
2021 |
|
歴史的な年 |
2020 (2019 - 2014 にカスタマイズ可能) |
|
定量単位 |
売上高は百万米ドル、価格は米ドル |
|
対象セグメント |
クラス別(クラス 1 - 複数のエフェクタータンパク質とクラス 2 - 単一の CrRNA 結合タンパク質)、製品とサービス(製品とサービス)、アプリケーション(生物医学診断、ゲノム工学、創薬、農業アプリケーションなど)、ワークフロー(サンプル調製、前増幅、CrRNA、Cas 酵素とセンシング)、エンドユーザー(病院、診断センター、バイオテクノロジー企業、学術研究機関など)、流通チャネル(直接入札、小売販売) |
|
対象国 |
中国、日本、韓国、インド、オーストラリア、シンガポール、タイ、マレーシア、インドネシア、フィリピン、その他のアジア太平洋諸国 |
|
対象となる市場プレーヤー |
GenScript、タカラバイオ株式会社、OriGene Technologies、Inc.、Agilent Technologies、Inc.、Synthego、Merck KGaA、Integrated DNA Technologies、Inc.(Danaher の子会社)、Thermo Fisher Scientific Inc. など。 |
アジア太平洋地域の CRISPR 遺伝子検出および診断市場の動向
ドライバー
- 慢性疾患の有病率と発症率の上昇
慢性疾患は一般的な健康状態であり、成人の 3 人に 1 人が慢性疾患に苦しんでいます。慢性疾患は多くの国民の健康と生活の質に影響を及ぼしています。
CRISPR は、クラスター化された規則的に間隔を置いた短い回文反復の略称です。近年、CRISPR は細胞内の DNA の特定の配列を変更するために使用される遺伝子編集の強力なツールになりました。CRISPR は、ハンチントン病、筋ジストロフィー、癌、高コレステロールの研究と治療に重要な役割を果たしています。
例えば、
- 2021年、NORD(国立希少疾患組織)のデータによると、デュシェンヌ型筋ジストロフィー(DMD)の診断発生率は100%でした。デュシェンヌ型筋ジストロフィー(DMD)は、世界中で出生男児3,500人に1人が罹患する遺伝性疾患です。
- 研究開発への投資の増加
CRISPR-Cas 9 システムなどの遺伝子編集技術により、遺伝子および細胞治療における診断とサービスの範囲が拡大しました。製薬会社は新製品の開発に多額の投資を行っており、遺伝子および細胞治療薬が開発初期段階に急増しています。市場プレーヤーが投資することで、深刻な治療を必要とする患者に安全で効果的な治療を提供するという目標を達成できます。
例えば、
- 2022年2月、シンセゴは、CRISPRベースの医薬品の開発を初期段階の研究から臨床まで促進するための研究開発投資として2億ドルを調達しました。シンセゴは、シリーズEファイナンスからの投資額を使用して、CRISPR診断およびサービスの作成を加速します。
CRISPR遺伝子診断のための資金の入手可能性
CRISPR gene diagnostics and research are funded by the National Institute of Health (NIH) budget. The private sector also funds the CRISPR gene detection and research, but such investment generally occurs later, during the testing and development phase, then during initial basic research. With genome editing being such a new field, an unbiased governmental body must supervise them; the FDA is cautious and thorough, but they are endlessly struggling for funding, making a long-term investment that aligns the payment with the potential future beneficiaries., will further enhance the growth of the CRISPR gene detection and diagnostic market.
Furthermore, advancement in CRISPR gene diagnostics, rising initiatives by public and private organizations to spread awareness and growing government funding are the factors that will expand the Asia-Pacific CRISPR gene detection market. Other factors such as increase in the demand for effective therapies and rising awareness about the timely diagnosis and will positively impact the CRISPR gene detection and diagnostic market's growth rate. Additionally, high disposable income, rising number of chronic diseases, changing lifestyle will result in the expansion of the CRISPR gene detection and diagnostic market.
Opportunities
- The rise in healthcare expenditure
Moreover, the rise in the research and development activities and increasing investments by government and private organization will boost new opportunities for the market's growth rate.
- Strategic initiative by market players
The demand for CRISPR gene detection and diagnosis has increased the demand in the U.S. and Asia-Pacific owing to the timely treatment of chronic conditions. These favorable factors enhance the need for medications, and to achieve the market demand, minor and major market players are utilizing various strategies.
The major players are also trying to devise specific strategies, such as product launches, acquisitions, approvals, expansions, and partnerships, to ensure the smooth running of the business, avoid risks, and increase the long-term growth in the sales of the market.
For instance,
- In May 2021, Horizon Discovery Ltd. extended the gene modulation portfolio with the first synthetic single guide RNA and patent pending dcas9 repressor for CRISPR interference in Waltham. The expansion of the portfolio had increased the sales and revenue of the synthetic guide RNA portfolio across the U.S. and the U.K. region and had increased the collaboration with market players
Also, the launch of effective therapies and continuous clinical trials will provide beneficial opportunities for the CRISPR gene detection and diagnostic market in the forecast period of 2022-2029. Also, high unmet need of current and developments in healthcare technology will escalate the growth rate of the CRISPR gene detection and diagnostic market in future.
Restraints/Challenges
However, high cost of CRISPR diagnostics and risks faced while using the CRISPR diagnostics will impede the growth rate of CRISPR gene detection and diagnostic market. Additionally, the risks incurred while using the MRI devices will hinder the CRISPR gene detection and diagnostic market growth. The lack of skilled expertise and regulations will further challenge the market in the forecast period mentioned above.
- Rise in cost of CRISPR based diagnostics
The vast potential of CRISPR based therapeutics comes with a cost tag. Maximum genome editing therapies require an increased amount of time for development and production, and hence the rise in cost occurs. Besides, the assay kits and medications related to CRISPR gene detection and diagnostic are applicable to large section of population. These costs are pushed on patients. Therefore, the present high cost is expected to show a descending trend in the future.
For instance,
- In July 2021, according to Integrated DNA Technologies, Inc., the first commercially available CRISPR-based diagnostic assay for SARS-CoV-2 including reverse transcription LAMP (RT-LAMP) as preamplification is currently available at USD 30.15 per reaction
The CRISPR gene detection and diagnostic market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on CRISPR gene detection and diagnostic market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
Patient Epidemiology Analysis
According to a study by Globocan. In 2020, breast cancer had high incidence of cases, around 11.7%, followed by lung cancer which is 11.40%, colorectum cancer which is 10.00%, and cervix uteri and oesophagus cancer having less number of incident cases.
CRISPR gene detection and diagnostic market also provides you with detailed market analysis for patient analysis, prognosis and cures. Prevalence, incidence, mortality, adherence rates are some of the data variables that are available in the report. Direct or indirect impact analyses of epidemiology to market growth are analysed to create a more robust and cohort multivariate statistical model for forecasting the market in the growth period.
COVID-19 Impact on the CRISPR Gene Detection and Diagnostic Market
The COVID-19 has negatively affected the market. Lockdowns and isolation during pandemics complicate the diagnosis management and treatment. The lack of access to health-care facilities for routine and medication administration will further affect the market. Social isolation increases stress, despair, and social support, all of which may cause a reduction in anticonvulsant medication adherence during the pandemic.
Recent Development
- In August 2020, SHERLOCK BIOSCIENCES announced a collaboration with Dartmouth-Hitchcock Health, to conduct the clinical trial of SHERLOCK Diagnostic Kit for Sars-CoV-2. The kit received emergency approval from the Emergency Use Authorization (EUA) by the U.S. Food and Drug Administration (FDA).
Asia-Pacific CRISPR Gene Detection and Diagnostic Market Scope
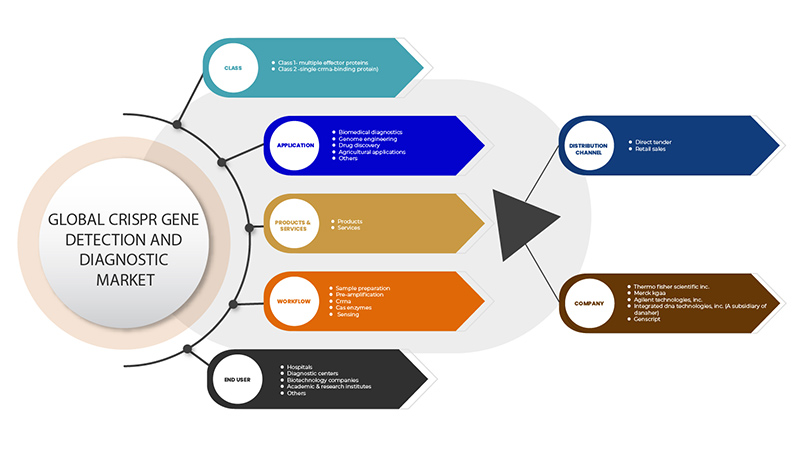
The CRISPR gene detection and diagnostic market is segmented on the basis of six segments: class, products & services, application, workflow, end user, and distribution channel. The growth amongst these segments will help you analyse meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Class
- Class 1- Multiple Effector Proteins
- Class 2 -Single CrRNA Binding Protein
On the basis of class, the Asia-Pacific CRISPR gene detection and diagnostic market is segmented into class 1- multiple effector proteins and class 2 -single CrRNA binding protein.
Products & Services
- Products
- Services
On the basis of products & services, the Asia-Pacific CRISPR gene detection and diagnostic market is segmented into products & services.
Application
- Biomedical Diagnostics
- Genome Engineering
- Drug Discovery
- Agricultural Applications
- Others
On the basis of application, the Asia-Pacific CRISPR gene detection and diagnostic market is segmented into is segmented into biomedical diagnostics, genome engineering, drug discovery, agricultural applications and others.
Workflow
- Sample Preparation
- Pre-Amplification
- CrRNA
- Cas Enzymes
- Sensing
On the basis of workflow, the Asia-Pacific CRISPR gene detection and diagnostic market is segmented into sample preparation, pre-amplification, CrRNA, Cas enzymes and sensing.
End User
- Hospitals
- Diagnostic Centers
- Biotechnology Companies
- Academic and Research Institutes
- Others
On the basis of end user, the Asia-Pacific CRISPR gene detection and diagnostic market is segmented into hospitals, diagnostic centers, biotechnology companies, academic and research institutes and others.
Distribution Channel
- Direct Tenders
- Retail Sales
On the basis of distribution channel, the Asia-Pacific CRISPR gene detection and diagnostic market is segmented into direct tenders and retail sales.
CRISPR Gene Detection and Diagnostic Market Regional Analysis/Insights
アジア太平洋地域の CRISPR 遺伝子検出および診断市場が分析され、上記の地域、クラス、製品とサービス、アプリケーション、ワークフロー、エンドユーザー、流通チャネル別に市場規模の洞察と傾向が提供されます。
CRISPR 遺伝子検出および診断市場レポートで取り上げられている国は、中国、日本、韓国、インド、オーストラリア、シンガポール、タイ、マレーシア、インドネシア、フィリピン、およびその他のアジア太平洋諸国です。
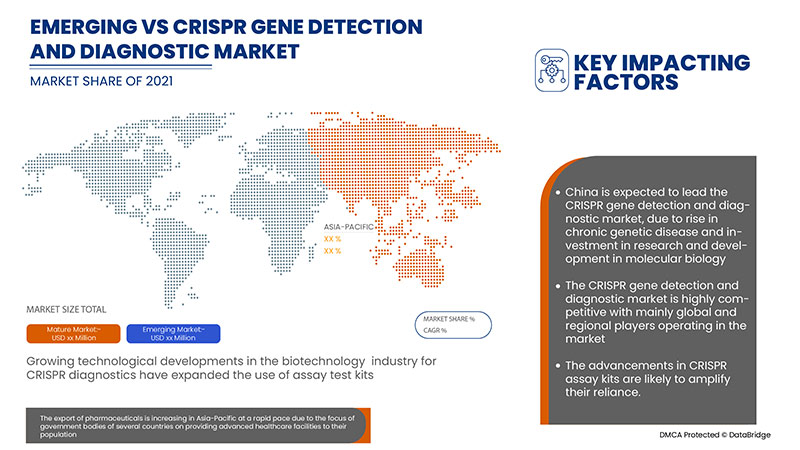
中国は、CRISPR ベースの診断に関する臨床試験の増加により、CRISPR 遺伝子検出および診断市場を支配しています。
レポートの国別セクションでは、市場の現在および将来の動向に影響を与える国内市場における個別の市場影響要因と規制の変更も提供しています。新規販売、交換販売、国の人口統計、疾病疫学、輸出入関税などのデータ ポイントは、各国の市場シナリオを予測するために使用される主要な指標の一部です。また、アジア太平洋ブランドの存在と可用性、および地元および国内ブランドとの競争が激しいか少ないために直面する課題、販売チャネルの影響を考慮しながら、国別データの予測分析を提供します。
競争環境とCRISPR遺伝子検出および診断市場シェア分析
アジア太平洋地域の CRISPR 遺伝子検出および診断市場の競争状況では、競合他社ごとに詳細が提供されます。詳細には、会社概要、会社の財務状況、収益、市場の可能性、研究開発への投資、新しい市場への取り組み、アジア太平洋地域でのプレゼンス、生産拠点と施設、生産能力、会社の強みと弱み、製品の発売、製品の幅と広さ、アプリケーションの優位性が含まれます。提供されている上記のデータ ポイントは、CRISPR 遺伝子検出および診断市場に関連する会社の焦点にのみ関連しています。
CRISPR 遺伝子検出および診断市場で活動している主要企業としては、GenScript、Takara Bio Inc.、OriGene Technologies, Inc.、Agilent Technologies, Inc.、Synthego、Merck KGaA、Integrated DNA Technologies, Inc. (Danaher の子会社)、Thermo Fisher Scientific Inc. などがあります。
SKU-
世界初のマーケットインテリジェンスクラウドに関するレポートにオンラインでアクセスする
- インタラクティブなデータ分析ダッシュボード
- 成長の可能性が高い機会のための企業分析ダッシュボード
- カスタマイズとクエリのためのリサーチアナリストアクセス
- インタラクティブなダッシュボードによる競合分析
- 最新ニュース、更新情報、トレンド分析
- 包括的な競合追跡のためのベンチマーク分析のパワーを活用
目次
1 はじめに
1.1 研究の目的
1.2 市場の定義
1.3 アジア太平洋地域のCRISPR遺伝子検出および診断市場の概要
1.4 通貨と価格
1.5 制限
1.6 対象市場
2 アジア太平洋地域のCRISPR遺伝子検出および診断市場:セグメンテーション
2.1 対象市場
2.2 地理的範囲
研究期間2.3年
2.4 DBMR TRIPODデータ検証モデル
2.5 主要なオピニオンリーダーとの一次インタビュー
2.6 多変量モデリング
2.7 クラスセグメントライフライン曲線
2.8 DBMR市場ポジショングリッド
2.9 ベンダーシェア分析
2.1 市場エンドユーザーカバレッジグリッド
2.11 二次資料
3 概要
4つのプレミアムインサイト
4.1 ペステル
4.2 ポーターの5つの力モデル
5 知的財産ランドスケープ(特許ランドスケープ)
6 疫学
7 アジア太平洋地域のCRISPR遺伝子検出および診断市場:規制シナリオ
8 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるCRISPR診断のパイプライン分析
9 市場概要
9.1 ドライバー
9.1.1 慢性疾患の有病率と発症率の上昇
9.1.2 研究開発への投資の増加
9.1.3 CRISPR遺伝子診断のための資金の入手可能性
9.1.4 CRISPR遺伝子診断のGMP認証承認の増加
9.1.5 CRISPR ベースの診断に関する臨床試験の増加
9.2 拘束
9.2.1 CRISPR ベースの診断コストの上昇
9.2.2 CRISPR診断の使用中に直面するリスク
9.2.3 CRISPR遺伝子検出および診断研究に関連する倫理的懸念
9.2.4 代替手段の利用可能性
9.3 機会
9.3.1 市場参加者による戦略的取り組み
9.3.2 医療費の増加
9.3.3 CRISPR ベースの診断における技術的進歩の出現
9.4 課題
9.4.1 CRISPR診断に必要な熟練した専門家の不足
9.4.2 厳格な規制
10 アジア太平洋地域のCRISPR遺伝子検出および診断市場(クラス別)
10.1 概要
10.2 クラス2単一CRRNA結合タンパク質
10.2.1 バイオメディカル診断
10.2.2 農業への応用
10.2.3 ゲノム工学
10.2.4 創薬
10.2.5 その他
10.3 クラス1マルチエフェクタータンパク質
11 アジア太平洋地域のCRISPR遺伝子検出および診断市場(製品およびサービス別)
11.1 概要
11.2 製品
11.2.1 アッセイキット
11.2.1.1 SGRNAキット
11.2.1.2 ゲノム検出キット
11.2.1.3 その他
11.2.2 タンパク質
11.2.2.1 CAS9
11.2.2.2 CPF1
11.2.2.3 その他
11.2.3 プラスミドとベクター
11.2.4 ライブラリ
11.2.5 コントロールキット
11.2.6 配信システム製品
11.2.7 設計ツール
11.2.8 ゲノムRNA
11.2.9 HDRブロッカー
11.2.9.1 アジドチミジン
11.2.9.2 トリフルオロチミジン
11.2.9.3 その他
11.2.9.4 その他
11.3 サービス
11.3.1 G-RNA設計
11.3.2 細胞株工学
11.3.3 微生物の遺伝子編集
11.3.4 DNA合成
11.3.5 その他
12 アジア太平洋地域のCRISPR遺伝子検出および診断市場(用途別)
12.1 概要
12.2 バイオメディカル診断
12.2.1 がん
12.2.2 血液疾患
12.2.3 遺伝性疾患
12.2.4 筋ジストロフィー
12.2.5 エイズ
12.2.6 神経変性疾患
12.2.7 その他
12.3 農業への応用
12.4 ゲノム工学
12.4.1 細胞株工学
12.4.2 ヒト幹細胞
12.5 創薬
12.6 その他
13 アジア太平洋地域のCRISPR遺伝子検出および診断市場(ワークフロー別)
13.1 概要
13.2 CRRNA
13.3 CAS酵素
13.4 前置増幅
13.4.1 PCR
13.4.2 ランプ
13.4.3 RPA
13.5 サンプルの準備
13.6 センシング
13.6.1 蛍光プローブ
13.6.2 比色分析
14 アジア太平洋地域のCRISPR遺伝子検出および診断市場(エンドユーザー別)
14.1 概要
14.2 バイオテクノロジー企業
14.3 学術研究機関
14.4 診断センター
14.5 病院
14.6 その他
15 アジア太平洋地域のCRISPR遺伝子検出および診断市場(流通チャネル別)
15.1 概要
15.2 直接入札
15.3 小売販売
16 アジア太平洋地域のCRISPR遺伝子検出および診断市場(地域別)
16.1 アジア太平洋
16.1.1 中国
16.1.2 日本
16.1.3 インド
16.1.4 韓国
16.1.5 オーストラリア
16.1.6 シンガポール
16.1.7 タイ
16.1.8 フィリピン
16.1.9 マレーシア
16.1.10 インドネシア
16.1.11 その他のアジア太平洋地域
17 アジア太平洋地域のCRISPR遺伝子検出および診断市場:企業の状況
17.1 企業シェア分析: アジア太平洋
18 SWOT分析
19 会社概要
19.1 サーモフィッシャーサイエンティフィック株式会社
19.1.1 会社概要
19.1.2 収益分析
19.1.3 企業株式分析
19.1.4 製品ポートフォリオ
19.1.5 最近の動向
19.2 メルクKGA
19.2.1 会社のスナップショット
19.2.2 収益分析
19.2.3 企業株式分析
19.2.4 製品ポートフォリオ
19.2.5 最近の動向
19.3 アジレントテクノロジーズ株式会社
19.3.1 会社のスナップショット
19.3.2 収益分析
19.3.3 企業株式分析
19.3.4 製品ポートフォリオ
19.3.5 最近の開発
19.4 インテグレーテッド DNA テクノロジーズ社(ダナハーの子会社)
19.4.1 会社のスナップショット
19.4.2 収益分析
19.4.3 企業株式分析
19.4.4 製品ポートフォリオ
19.4.5 最近の動向
19.5 ジェネスクリプト
19.5.1 会社のスナップショット
19.5.2 収益分析
19.5.3 企業株式分析
19.5.4 製品ポートフォリオ
19.5.5 最近の開発
19.6 10 X ゲノミクス
19.6.1 会社のスナップショット
19.6.2 収益分析
19.6.3 製品ポートフォリオ
19.6.4 最近の動向
19.7 応用幹細胞
19.7.1 会社のスナップショット
19.7.2 製品ポートフォリオ
19.7.3 最近の開発
19.8 アッドジェネ
19.8.1 会社のスナップショット
19.8.2 製品ポートフォリオ
19.8.3 最近の開発
19.9 バイオビジョン株式会社
19.9.1 会社のスナップショット
19.9.2 製品ポートフォリオ
19.9.3 最近の開発
19.1 セルクタ株式会社
19.10.1 会社概要
19.10.2 製品ポートフォリオ
19.10.3 最近の動向
19.11 CAS タグ バイオサイエンス
19.11.1 会社概要
19.11.2 製品ポートフォリオ
19.11.3 最近の開発
19.12 ジェネコポエイア株式会社
19.12.1 会社概要
19.12.2 製品ポートフォリオ
19.12.3 最近の開発
19.13 ホライゾンディスカバリー株式会社
19.13.1 会社概要
19.13.2 製品ポートフォリオ
19.13.3 最近の動向
19.14 ヘラバイオラボ
19.14.1 会社概要
19.14.2 製品ポートフォリオ
19.14.3 最近の開発
19.15 ニューイングランドバイオラボ
19.15.1 会社概要
19.15.2 製品ポートフォリオ
19.15.3 最近の動向
19.16 オリジェンテクノロジーズ株式会社
19.16.1 会社概要
19.16.2 製品ポートフォリオ
19.16.3 最近の開発
19.17 シンセゴ
19.17.1 会社概要
19.17.2 製品ポートフォリオ
19.17.3 最近の動向
19.18 タカラバイオ株式会社
19.18.1 会社概要
19.18.2 収益分析
19.18.3 製品ポートフォリオ
19.18.4 最近の開発
19.19 ツールジェン株式会社
19.19.1 会社概要
19.19.2 製品ポートフォリオ
19.19.3 最近の開発
20 アンケート
関連レポート 21
表のリスト
表1 アジア太平洋地域のCRISPR遺伝子治療薬パイプライン分析
表2 アジア太平洋地域のCRISPR遺伝子検出および診断市場、クラス別、2020年~2029年(百万米ドル)
表3 アジア太平洋地域におけるCRISPR遺伝子検出および診断市場におけるクラス2単一CRRNA結合タンパク質、地域別、2020年~2029年(百万米ドル)
表4 アジア太平洋地域におけるCRISPR遺伝子検出および診断市場におけるクラス2単一CRRNA結合タンパク質、用途別、2020年~2029年(百万米ドル)
表5 アジア太平洋地域におけるCRISPR遺伝子検出および診断市場におけるクラス1多重エフェクタータンパク質、地域別、2020年~2029年(百万米ドル)
表6 アジア太平洋地域のCRISPR遺伝子検出および診断市場、製品およびサービス別、2020年~2029年(百万米ドル)
表7 アジア太平洋地域のCRISPR遺伝子検出および診断市場における製品、地域別、2020年~2029年(百万米ドル)
表8 アジア太平洋地域のCRISPR遺伝子検出および診断市場における製品、タイプ別、2020年~2029年(百万米ドル)
表9 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるアッセイキット、製品タイプ別、2020年~2029年(百万米ドル)
表10 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるタンパク質、製品タイプ別、2020年~2029年(百万米ドル)
表11 アジア太平洋地域におけるCRISPR遺伝子検出および診断市場におけるHDRブロッカー、製品タイプ別、2020年~2029年(百万米ドル)
表12 アジア太平洋地域のCRISPR遺伝子検出および診断サービス市場、地域別、2020年~2029年(百万米ドル)
表13 アジア太平洋地域のCRISPR遺伝子検出および診断サービス市場、タイプ別、2020年~2029年(百万米ドル)
表14 アジア太平洋地域のCRISPR遺伝子検出および診断市場、アプリケーション別、2020年~2029年(百万米ドル)
表15 アジア太平洋地域におけるCRISPR遺伝子検出および診断市場におけるバイオメディカル診断、地域別、2020年~2029年(百万米ドル)
表16 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるバイオメディカル診断、アプリケーション別、2020年~2029年(百万米ドル)
表17 アジア太平洋地域のCRISPR遺伝子検出および診断市場における農業用途、地域別、2020年~2029年(百万米ドル)
表18 アジア太平洋地域におけるゲノムエンジニアリングによるCRISPR遺伝子検出および診断市場、地域別、2020年~2029年(百万米ドル)
表19 アジア太平洋地域におけるゲノムエンジニアリングのCRISPR遺伝子検出および診断市場、アプリケーション別、2020年~2029年(百万米ドル)
表20 アジア太平洋地域における創薬CRISPR遺伝子検出および診断市場、地域別、2020年~2029年(百万米ドル)
表21 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるその他企業、地域別、2020年~2029年(百万米ドル)
表22 アジア太平洋地域のCRISPR遺伝子検出および診断市場、ワークフロー別、2020年~2029年(百万米ドル)
表23 アジア太平洋地域のCRRNA CRISPR遺伝子検出および診断市場、地域別、2020年~2029年(百万米ドル)
表24 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるCAS酵素、地域別、2020年~2029年(百万米ドル)
表25 アジア太平洋地域のCRISPR遺伝子検出および診断市場における前増幅、地域別、2020年~2029年(百万米ドル)
表26 アジア太平洋地域のCRISPR遺伝子検出および診断市場における前増幅、ワークフロー別、2020年~2029年(百万米ドル)
表27 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるサンプル調製、地域別、2020年~2029年(百万米ドル)
表28 アジア太平洋地域のCRISPR遺伝子検出および診断市場、地域別、2020年~2029年(百万米ドル)
表29 アジア太平洋地域のCRISPR遺伝子検出および診断市場、ワークフロー別、2020年~2029年(百万米ドル)
表30 アジア太平洋地域のCRISPR遺伝子検出および診断市場、エンドユーザー別、2020年~2029年(百万米ドル)
表31 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるバイオテクノロジー企業、地域別、2020年~2029年(百万米ドル)
表32 アジア太平洋地域のCRISPR遺伝子検出および診断市場における学術研究機関、地域別、2020年~2029年(百万米ドル)
表33 アジア太平洋地域のCRISPR遺伝子検出および診断市場における診断センター、地域別、2020年~2029年(百万米ドル)
表34 アジア太平洋地域のCRISPR遺伝子検出および診断市場における病院数、地域別、2020年~2029年(百万米ドル)
表35 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるその他企業、地域別、2020年~2029年(百万米ドル)
表36 アジア太平洋地域のCRISPR遺伝子検出および診断市場、流通チャネル別、2020年~2029年(百万米ドル)
表37 アジア太平洋地域におけるCRISPR遺伝子検出および診断市場への直接入札、地域別、2020年~2029年(百万米ドル)
表38 アジア太平洋地域のCRISPR遺伝子検出および診断市場における小売売上高、地域別、2020年~2029年(百万米ドル)
表39 アジア太平洋地域のCRISPR遺伝子検出および診断市場、国別、2020年~2029年(百万米ドル)
表40 アジア太平洋地域のCRISPR遺伝子検出および診断市場、クラス別、2020年~2029年(百万米ドル)
表41 アジア太平洋地域におけるCRISPR遺伝子検出および診断市場におけるクラス2単一CRRNA結合タンパク質、用途別、2020年~2029年(百万米ドル)
表42 アジア太平洋地域のCRISPR遺伝子検出および診断市場、製品およびサービス別、2020年~2029年(百万米ドル)
表43 アジア太平洋地域のCRISPR遺伝子検出および診断市場における製品、タイプ別、2020年~2029年(百万米ドル)
表44 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるアッセイキット、製品タイプ別、2020年~2029年(百万米ドル)
表45 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるHDRブロッカー、製品タイプ別、2020年~2029年(百万米ドル)
表46 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるタンパク質、製品タイプ別、2020年~2029年(百万米ドル)
表47 アジア太平洋地域のCRISPR遺伝子検出および診断サービス市場、タイプ別、2020年~2029年(百万米ドル)
表48 アジア太平洋地域のCRISPR遺伝子検出および診断市場、用途別、2020年~2029年(百万米ドル)
表49 アジア太平洋地域におけるゲノムエンジニアリングのCRISPR遺伝子検出および診断市場、アプリケーション別、2020年~2029年(百万米ドル)
表50 アジア太平洋地域のCRISPR遺伝子検出および診断市場におけるバイオメディカル診断、アプリケーション別、2020年~2029年(百万米ドル)
表51 アジア太平洋地域のCRISPR遺伝子検出および診断市場、ワークフロー別、2020年~2029年(百万米ドル)
表52 アジア太平洋地域の遺伝子検出および診断における前増幅市場、ワークフロー別、2020年~2029年(百万米ドル)
表53 アジア太平洋地域の遺伝子検出および診断市場、ワークフロー別、2020年~2029年(百万米ドル)
表54 アジア太平洋地域のCRISPR遺伝子検出および診断市場、エンドユーザー別、2020年~2029年(百万米ドル)
表55 アジア太平洋地域のCRISPR遺伝子検出および診断市場、流通チャネル別、2020年~2029年(百万米ドル)
表56 中国のCRISPR遺伝子検出および診断市場、クラス別、2020年~2029年(百万米ドル)
表 57 中国クラス 2 単一 CRRNA 結合タンパク質 CRISPR 遺伝子検出および診断市場、用途別、2020-2029 年 (百万米ドル)
表58 中国CRISPR遺伝子検出および診断市場、製品およびサービス別、2020年~2029年(百万米ドル)
表59 CRISPR遺伝子検出および診断市場における中国の製品、タイプ別、2020年~2029年(百万米ドル)
表60 CRISPR遺伝子検出および診断市場における中国のアッセイキット、製品タイプ別、2020年~2029年(百万米ドル)
表61 中国CRISPR遺伝子検出および診断市場におけるHDRブロッカー、製品タイプ別、2020年~2029年(百万米ドル)
表62 中国CRISPR遺伝子検出および診断市場におけるタンパク質、製品タイプ別、2020年~2029年(百万米ドル)
表63 CRISPR遺伝子検出および診断市場における中国のサービス、タイプ別、2020年~2029年(百万米ドル)
表64 中国CRISPR遺伝子検出および診断市場、用途別、2020年~2029年(百万米ドル)
表65 中国ゲノムエンジニアリングCRISPR遺伝子検出および診断市場、アプリケーション別、2020年~2029年(百万米ドル)
表66 CRISPR遺伝子検出および診断市場における中国のバイオメディカル診断、アプリケーション別、2020年~2029年(百万米ドル)
表67 中国CRISPR遺伝子検出および診断市場、ワークフロー別、2020年~2029年(百万米ドル)
表68 中国の遺伝子検出および診断市場における前増幅、ワークフロー別、2020年~2029年(百万米ドル)
表69 中国の遺伝子検出および診断市場におけるセンシング、ワークフロー別、2020年~2029年(百万米ドル)
表70 中国CRISPR遺伝子検出および診断市場、エンドユーザー別、2020年~2029年(百万米ドル)
表71 中国のCRISPR遺伝子検出および診断市場、流通チャネル別、2020年~2029年(百万米ドル)
表72 日本CRISPR遺伝子検出・診断市場、クラス別、2020年~2029年(百万米ドル)
表 73 日本クラス 2 単一 CRRNA 結合タンパク質 CRISPR 遺伝子検出および診断市場、用途別、2020-2029 年 (百万米ドル)
表74 日本CRISPR遺伝子検出・診断市場、製品・サービス別、2020年~2029年(百万米ドル)
表75 CRISPR遺伝子検出および診断市場における日本の製品、タイプ別、2020年~2029年(百万米ドル)
表76 日本におけるCRISPR遺伝子検出・診断市場におけるアッセイキット、製品タイプ別、2020年~2029年(百万米ドル)
表 77 日本 HDR ブロッカーの CRISPR 遺伝子検出および診断市場、製品タイプ別、2020 年 - 2029 年 (百万米ドル)
表78 日本におけるCRISPR遺伝子検出および診断市場におけるタンパク質、製品タイプ別、2020年~2029年(百万米ドル)
表79 CRISPR遺伝子検出および診断市場における日本のサービス、タイプ別、2020年~2029年(百万米ドル)
表80 日本CRISPR遺伝子検出・診断市場、用途別、2020年~2029年(百万米ドル)
表81 日本ゲノムエンジニアリングCRISPR遺伝子検出および診断市場、アプリケーション別、2020年-2029年(百万米ドル)
表82 日本バイオメディカル診断におけるCRISPR遺伝子検出および診断市場、用途別、2020年~2029年(百万米ドル)
表83 日本CRISPR遺伝子検出・診断市場、ワークフロー別、2020年~2029年(百万米ドル)
表84 日本における遺伝子検出および診断用前増幅市場、ワークフロー別、2020年~2029年(百万米ドル)
表85 日本における遺伝子検出・診断市場、ワークフロー別、2020年~2029年(百万米ドル)
表86 日本CRISPR遺伝子検出・診断市場、エンドユーザー別、2020年~2029年(百万米ドル)
表87 日本CRISPR遺伝子検出・診断市場、流通チャネル別、2020年~2029年(百万米ドル)
表88 インドのCRISPR遺伝子検出および診断市場、クラス別、2020年~2029年(百万米ドル)
表 89 インド CRISPR 遺伝子検出および診断市場におけるクラス 2 単一 CRRNA 結合タンパク質、アプリケーション別、2020 年 - 2029 年 (百万米ドル)
表90 インドのCRISPR遺伝子検出および診断市場、製品およびサービス別、2020年~2029年(百万米ドル)
表91 インドのCRISPR遺伝子検出および診断市場における製品、タイプ別、2020年~2029年(百万米ドル)
表92 インドのCRISPR遺伝子検出および診断市場におけるアッセイキット、製品タイプ別、2020年~2029年(百万米ドル)
表93 インドのCRISPR遺伝子検出および診断市場におけるHDRブロッカー、製品タイプ別、2020年~2029年(百万米ドル)
表94 インドのCRISPR遺伝子検出および診断市場におけるタンパク質、製品タイプ別、2020年~2029年(百万米ドル)
表95 インドのCRISPR遺伝子検出および診断市場におけるサービス、タイプ別、2020年~2029年(百万米ドル)
表96 インドのCRISPR遺伝子検出および診断市場、アプリケーション別、2020年~2029年(百万米ドル)
表97 インドのゲノムエンジニアリングCRISPR遺伝子検出および診断市場、アプリケーション別、2020-2029年(百万米ドル)
表98 インドのCRISPR遺伝子検出および診断市場におけるバイオメディカル診断、アプリケーション別、2020年~2029年(百万米ドル)
表99 インドのCRISPR遺伝子検出および診断市場、ワークフロー別、2020年~2029年(百万米ドル)
表 100 インドの遺伝子検出および診断市場における前増幅、ワークフロー別、2020-2029 年 (百万米ドル)
表 101 インドの遺伝子検出および診断市場におけるセンシング、ワークフロー別、2020-2029 年 (百万米ドル)
表 102 インドの CRISPR 遺伝子検出および診断市場、エンドユーザー別、2020-2029 年 (百万米ドル)
表 103 インドの CRISPR 遺伝子検出および診断市場、流通チャネル別、2020-2029 年 (百万米ドル)
表104 韓国のCRISPR遺伝子検出および診断市場、クラス別、2020年~2029年(百万米ドル)
表 105 韓国の CRISPR 遺伝子検出および診断市場におけるクラス 2 単一 CRRNA 結合タンパク質、用途別、2020 年 - 2029 年 (百万米ドル)
表106 韓国のCRISPR遺伝子検出および診断市場、製品およびサービス別、2020年~2029年(百万米ドル)
表107 CRISPR遺伝子検出および診断市場における韓国製品、タイプ別、2020年~2029年(百万米ドル)
表108 韓国のCRISPR遺伝子検出および診断市場におけるアッセイキット、製品タイプ別、2020年~2029年(百万米ドル)
表 109 韓国の CRISPR 遺伝子検出および診断市場における HDR ブロッカー、製品タイプ別、2020 年 - 2029 年 (百万米ドル)
表110 韓国のCRISPR遺伝子検出および診断市場におけるタンパク質、製品タイプ別、2020年~2029年(百万米ドル)
表111 韓国のCRISPR遺伝子検出および診断市場におけるサービス、タイプ別、2020年~2029年(百万米ドル)
表112 韓国のCRISPR遺伝子検出および診断市場、用途別、2020年~2029年(百万米ドル)
表113 韓国のゲノムエンジニアリングCRISPR遺伝子検出および診断市場、アプリケーション別、2020年~2029年(百万米ドル)
表114 韓国のCRISPR遺伝子検出および診断市場におけるバイオメディカル診断、用途別、2020年~2029年(百万米ドル)
表115 韓国のCRISPR遺伝子検出および診断市場、ワークフロー別、2020年~2029年(百万米ドル)
表116 韓国の遺伝子検出および診断における前増幅市場、ワークフロー別、2020年~2029年(百万米ドル)
表 117 韓国の遺伝子検出および診断市場におけるセンシング、ワークフロー別、2020-2029年 (百万米ドル)
表118 韓国のCRISPR遺伝子検出および診断市場、エンドユーザー別、2020年~2029年(百万米ドル)
表119 韓国のCRISPR遺伝子検出および診断市場、流通チャネル別、2020年~2029年(百万米ドル)
表 120 オーストラリアの CRISPR 遺伝子検出および診断市場、クラス別、2020-2029 年 (百万米ドル)
表 121 オーストラリア クラス 2 単一 CRRNA 結合タンパク質 CRISPR 遺伝子検出および診断市場、用途別、2020-2029 年 (百万米ドル)
表 122 オーストラリアの CRISPR 遺伝子検出および診断市場、製品およびサービス別、2020-2029 年 (百万米ドル)
表 123 オーストラリアの CRISPR 遺伝子検出および診断市場における製品、タイプ別、2020-2029 年 (百万米ドル)
表 124 オーストラリアの CRISPR 遺伝子検出および診断市場におけるアッセイキット、製品タイプ別、2020-2029 年 (百万米ドル)
表 125 オーストラリアの CRISPR 遺伝子検出および診断市場における HDR ブロッカー、製品タイプ別、2020 年 - 2029 年 (百万米ドル)
表 126 オーストラリアの CRISPR 遺伝子検出および診断市場におけるタンパク質、製品タイプ別、2020-2029 年 (百万米ドル)
表 127 オーストラリアの CRISPR 遺伝子検出および診断市場におけるサービス、タイプ別、2020-2029 年 (百万米ドル)
表128 オーストラリアのCRISPR遺伝子検出および診断市場、用途別、2020年~2029年(百万米ドル)
表 129 オーストラリアのゲノムエンジニアリング CRISPR 遺伝子検出および診断市場、アプリケーション別、2020-2029 年 (百万米ドル)
表 130 オーストラリアの CRISPR 遺伝子検出および診断市場におけるバイオメディカル診断、アプリケーション別、2020-2029 年 (百万米ドル)
表 131 オーストラリアの CRISPR 遺伝子検出および診断市場、ワークフロー別、2020 年 - 2029 年 (百万米ドル)
表 132 オーストラリアの遺伝子検出および診断市場における前増幅、ワークフロー別、2020-2029 年 (百万米ドル)
表 133 オーストラリアの遺伝子検出および診断市場におけるセンシング、ワークフロー別、2020-2029 年 (百万米ドル)
表 134 オーストラリアの CRISPR 遺伝子検出および診断市場、エンドユーザー別、2020-2029 年 (百万米ドル)
表135 オーストラリアのCRISPR遺伝子検出および診断市場、流通チャネル別、2020年~2029年(百万米ドル)
表136 シンガポールのCRISPR遺伝子検出および診断市場、クラス別、2020年~2029年(百万米ドル)
表 137 シンガポールの CRISPR 遺伝子検出および診断市場におけるクラス 2 単一 CRRNA 結合タンパク質、用途別、2020 年 - 2029 年 (百万米ドル)
表138 シンガポールのCRISPR遺伝子検出および診断市場、製品およびサービス別、2020年~2029年(百万米ドル)
表 139 CRISPR 遺伝子検出および診断市場におけるシンガポール製品、タイプ別、2020-2029 年 (百万米ドル)
表140 シンガポールのCRISPR遺伝子検出および診断市場におけるアッセイキット、製品タイプ別、2020年~2029年(百万米ドル)
表 141 シンガポールの CRISPR 遺伝子検出および診断市場における HDR ブロッカー、製品タイプ別、2020 年 - 2029 年 (百万米ドル)
表 142 シンガポールの CRISPR 遺伝子検出および診断市場におけるタンパク質、製品タイプ別、2020-2029 年 (百万米ドル)
表 143 シンガポールの CRISPR 遺伝子検出および診断市場におけるサービス、タイプ別、2020-2029 年 (百万米ドル)
表144 シンガポールのCRISPR遺伝子検出および診断市場、用途別、2020年~2029年(百万米ドル)
表 145 シンガポールのゲノムエンジニアリング CRISPR 遺伝子検出および診断市場、アプリケーション別、2020-2029 年 (百万米ドル)
表 146 シンガポールの CRISPR 遺伝子検出および診断市場におけるバイオメディカル診断、アプリケーション別、2020 年 - 2029 年 (百万米ドル)
表 147 シンガポールの CRISPR 遺伝子検出および診断市場、ワークフロー別、2020-2029 年 (百万米ドル)
表 148 シンガポールの遺伝子検出および診断市場における前増幅、ワークフロー別、2020-2029 年 (百万米ドル)
表 149 シンガポールの遺伝子検出および診断市場におけるセンシング、ワークフロー別、2020-2029 年 (百万米ドル)
表 150 シンガポールの CRISPR 遺伝子検出および診断市場、エンドユーザー別、2020-2029 年 (百万米ドル)
表151 シンガポールのCRISPR遺伝子検出および診断市場、流通チャネル別、2020年~2029年(百万米ドル)
表 152 タイの CRISPR 遺伝子検出および診断市場、クラス別、2020-2029 年 (百万米ドル)
表 153 タイの CRISPR 遺伝子検出および診断市場におけるクラス 2 単一 CRRNA 結合タンパク質、用途別、2020 年 - 2029 年 (百万米ドル)
表 154 タイの CRISPR 遺伝子検出および診断市場、製品およびサービス別、2020 年 - 2029 年 (百万米ドル)
表 155 タイの CRISPR 遺伝子検出および診断市場における製品、タイプ別、2020-2029 年 (百万米ドル)
表 156 タイの CRISPR 遺伝子検出および診断市場におけるアッセイキット、製品タイプ別、2020-2029 年 (百万米ドル)
表 157 タイの CRISPR 遺伝子検出および診断市場における HDR ブロッカー、製品タイプ別、2020 年 - 2029 年 (百万米ドル)
表 158 タイの CRISPR 遺伝子検出および診断市場におけるタンパク質、製品タイプ別、2020-2029 年 (百万米ドル)
表 159 タイの CRISPR 遺伝子検出および診断市場におけるサービス、タイプ別、2020-2029 年 (百万米ドル)
表 160 タイの CRISPR 遺伝子検出および診断市場、アプリケーション別、2020-2029 年 (百万米ドル)
表 161 タイのゲノムエンジニアリング CRISPR 遺伝子検出および診断市場、アプリケーション別、2020-2029 年 (百万米ドル)
表 162 タイの CRISPR 遺伝子検出および診断市場におけるバイオメディカル診断、アプリケーション別、2020 年 - 2029 年 (百万米ドル)
表 163 タイの CRISPR 遺伝子検出および診断市場、ワークフロー別、2020 年 - 2029 年 (百万米ドル)
表 164 タイの遺伝子検出および診断市場における前増幅、ワークフロー別、2020-2029 年 (百万米ドル)
表 165 タイの遺伝子検出および診断市場におけるセンシング、ワークフロー別、2020-2029 年 (百万米ドル)
表 166 タイの CRISPR 遺伝子検出および診断市場、エンドユーザー別、2020-2029 年 (百万米ドル)
表 167 タイの CRISPR 遺伝子検出および診断市場、流通チャネル別、2020-2029 年 (百万米ドル)
表 168 フィリピンの CRISPR 遺伝子検出および診断市場、クラス別、2020-2029 年 (百万米ドル)
表 169 フィリピンの CRISPR 遺伝子検出および診断市場におけるクラス 2 単一 CRRNA 結合タンパク質、用途別、2020 年 - 2029 年 (百万米ドル)
表 170 フィリピンの CRISPR 遺伝子検出および診断市場、製品およびサービス別、2020-2029 年 (百万米ドル)
表 171 フィリピンの CRISPR 遺伝子検出および診断市場における製品、タイプ別、2020-2029 年 (百万米ドル)
表 172 フィリピンの CRISPR 遺伝子検出および診断市場におけるアッセイキット、製品タイプ別、2020-2029 年 (百万米ドル)
表 173 フィリピンの CRISPR 遺伝子検出および診断市場における HDR ブロッカー、製品タイプ別、2020 年 - 2029 年 (百万米ドル)
表 174 フィリピンの CRISPR 遺伝子検出および診断市場におけるタンパク質、製品タイプ別、2020-2029 年 (百万米ドル)
表 175 フィリピンの CRISPR 遺伝子検出および診断市場におけるサービス、タイプ別、2020-2029 年 (百万米ドル)
表 176 フィリピンの CRISPR 遺伝子検出および診断市場、アプリケーション別、2020-2029 年 (百万米ドル)
表 177 フィリピンのゲノムエンジニアリング CRISPR 遺伝子検出および診断市場、アプリケーション別、2020-2029 年 (百万米ドル)
表 178 フィリピンの CRISPR 遺伝子検出および診断市場におけるバイオメディカル診断、アプリケーション別、2020-2029 年 (百万米ドル)
表 179 フィリピンの CRISPR 遺伝子検出および診断市場、ワークフロー別、2020-2029 年 (百万米ドル)
表 180 フィリピンの遺伝子検出および診断市場における前増幅、ワークフロー別、2020-2029 年 (百万米ドル)
表 181 フィリピンの遺伝子検出および診断市場におけるセンシング、ワークフロー別、2020-2029 年 (百万米ドル)
表 182 フィリピンの CRISPR 遺伝子検出および診断市場、エンドユーザー別、2020-2029 年 (百万米ドル)
表 183 フィリピンの CRISPR 遺伝子検出および診断市場、流通チャネル別、2020-2029 年 (百万米ドル)
表 184 マレーシアの CRISPR 遺伝子検出および診断市場、クラス別、2020-2029 年 (百万米ドル)
表 185 マレーシアの CRISPR 遺伝子検出および診断市場におけるクラス 2 単一 CRRNA 結合タンパク質、用途別、2020 年 - 2029 年 (百万米ドル)
表 186 マレーシアの CRISPR 遺伝子検出および診断市場、製品およびサービス別、2020-2029 年 (百万米ドル)
表 187 マレーシアの CRISPR 遺伝子検出および診断市場における製品、タイプ別、2020-2029 年 (百万米ドル)
表 188 マレーシアの CRISPR 遺伝子検出および診断市場におけるアッセイキット、製品タイプ別、2020-2029 年 (百万米ドル)
表 189 マレーシアの CRISPR 遺伝子検出および診断市場における HDR ブロッカー、製品タイプ別、2020 年 - 2029 年 (百万米ドル)
表 190 マレーシアの CRISPR 遺伝子検出および診断市場におけるタンパク質、製品タイプ別、2020-2029 年 (百万米ドル)
表 191 マレーシアの CRISPR 遺伝子検出および診断市場におけるサービス、タイプ別、2020-2029 年 (百万米ドル)
表 192 マレーシア CRISPR 遺伝子検出および診断市場、アプリケーション別、2020-2029 年 (百万米ドル)
表 193 マレーシアのゲノムエンジニアリング CRISPR 遺伝子検出および診断市場、アプリケーション別、2020-2029 年 (百万米ドル)
表 194 マレーシアの CRISPR 遺伝子検出および診断市場におけるバイオメディカル診断、アプリケーション別、2020-2029 年 (百万米ドル)
表 195 マレーシアの CRISPR 遺伝子検出および診断市場、ワークフロー別、2020-2029 年 (百万米ドル)
表 196 マレーシアの遺伝子検出および診断市場における前増幅、ワークフロー別、2020-2029 年 (百万米ドル)
表 197 マレーシアの遺伝子検出および診断市場におけるセンシング、ワークフロー別、2020-2029 年 (百万米ドル)
表 198 マレーシア CRISPR 遺伝子検出および診断市場、エンドユーザー別、2020-2029 年 (百万米ドル)
表 199 マレーシアの CRISPR 遺伝子検出および診断市場、流通チャネル別、2020-2029 年 (百万米ドル)
表 200 インドネシアの CRISPR 遺伝子検出および診断市場、クラス別、2020-2029 年 (百万米ドル)
表 201 インドネシアの CRISPR 遺伝子検出および診断市場におけるクラス 2 単一 CRRNA 結合タンパク質、用途別、2020 年 - 2029 年 (百万米ドル)
表 202 インドネシア CRISPR 遺伝子検出および診断市場、製品およびサービス別、2020-2029 年 (百万米ドル)
表 203 インドネシアの CRISPR 遺伝子検出および診断市場における製品、タイプ別、2020-2029 年 (百万米ドル)
表 204 インドネシアの CRISPR 遺伝子検出および診断市場におけるアッセイキット、製品タイプ別、2020-2029 年 (百万米ドル)
表 205 インドネシアの CRISPR 遺伝子検出および診断市場における HDR ブロッカー、製品タイプ別、2020 年 - 2029 年 (百万米ドル)
表 206 インドネシアの CRISPR 遺伝子検出および診断市場におけるタンパク質、製品タイプ別、2020-2029 年 (百万米ドル)
表 207 インドネシアの CRISPR 遺伝子検出および診断市場におけるサービス、タイプ別、2020-2029 年 (百万米ドル)
表 208 インドネシア CRISPR 遺伝子検出および診断市場、アプリケーション別、2020-2029 年 (百万米ドル)
表 209 インドネシアのゲノムエンジニアリング CRISPR 遺伝子検出および診断市場、アプリケーション別、2020-2029 年 (百万米ドル)
表 210 インドネシアの CRISPR 遺伝子検出および診断市場におけるバイオメディカル診断、アプリケーション別、2020-2029 年 (百万米ドル)
表 211 インドネシアの CRISPR 遺伝子検出および診断市場、ワークフロー別、2020-2029 年 (百万米ドル)
表 212 インドネシアの遺伝子検出および診断における前増幅市場、ワークフロー別、2020-2029年 (百万米ドル)
表 213 インドネシアの遺伝子検出および診断市場におけるセンシング、ワークフロー別、2020-2029年 (百万米ドル)
表 214 インドネシア CRISPR 遺伝子検出および診断市場、エンドユーザー別、2020-2029 年 (百万米ドル)
表 215 インドネシアの CRISPR 遺伝子検出および診断市場、流通チャネル別、2020-2029 年 (百万米ドル)
表 216 アジア太平洋地域のその他の CRISPR 遺伝子検出および診断市場、クラス別、2020-2029 年 (百万米ドル)
図表一覧
図1 アジア太平洋地域のCRISPR遺伝子検出および診断市場:セグメンテーション
図2 アジア太平洋地域のCRISPR遺伝子検出および診断市場:データの三角測量
図3 アジア太平洋地域のCRISPR遺伝子検出および診断市場:DROC分析
図4 アジア太平洋地域のCRISPR遺伝子検出および診断市場:アジア太平洋地域と地域市場の比較分析
図5 アジア太平洋地域のCRISPR遺伝子検出および診断市場:企業調査分析
図6 アジア太平洋地域のCRISPR遺伝子検出および診断市場:インタビュー人口統計
図7 アジア太平洋地域のCRISPR遺伝子検出および診断市場:DBMRポジショングリッド
図8 アジア太平洋地域のCRISPR遺伝子検出および診断市場:ベンダーシェア分析
図9 アジア太平洋地域のCRISPR遺伝子検出および診断市場:エンドユーザーカバレッジグリッド
図10 アジア太平洋地域のCRISPR遺伝子検出および診断市場:セグメンテーション
図11 北米はアジア太平洋地域のCRISPR遺伝子検出および診断市場を独占すると予想されており、アジア太平洋地域は2022年から2029年の予測期間に最も高いCAGRで成長すると推定されています。
図12 慢性疾患の発生率の増加、CRISPR診断の技術的進歩の高まり、およびCRISPR検出キットの開発に対する政府の資金提供により、2022年から2029年にかけてアジア太平洋地域のCRISPR遺伝子検出および診断市場が拡大すると予想されます。
図13クラスセグメントは、2022年と2029年にアジア太平洋のCRISPR遺伝子検出および診断市場で最大のシェアを占めると予想されています。
図14 アジア太平洋地域のCRISPR遺伝子特許のシナリオ(用途別)
図15 CRISPR特許の状況と2001年から2019年までの全世界での新規特許ファミリーの出願件数
図16 アジア太平洋地域のCRISPR遺伝子検出および診断市場の推進要因、制約要因、機会、課題
図17 2020年の各種がんの発生率
図18 2019年のハンチントン病の有病率
図19 アジア太平洋地域のCRISPR遺伝子検出および診断市場:クラス別、2021年
図20 アジア太平洋地域のCRISPR遺伝子検出および診断市場:クラス別、2022年~2029年(百万米ドル)
図21 アジア太平洋地域のCRISPR遺伝子検出および診断市場:クラス別、CAGR(2022-2029年)
図22 アジア太平洋地域のCRISPR遺伝子検出および診断市場:クラス別、ライフライン曲線
図23 アジア太平洋地域のCRISPR遺伝子検出および診断市場:製品およびサービス別、2021年
図24 アジア太平洋地域のCRISPR遺伝子検出および診断市場:製品およびサービス別、2022年~2029年(百万米ドル)
図25 アジア太平洋地域のCRISPR遺伝子検出および診断市場:製品およびサービス別、CAGR(2022-2029年)
図26 アジア太平洋地域のCRISPR遺伝子検出および診断市場:製品およびサービス別、ライフライン曲線
図27 アジア太平洋地域のCRISPR遺伝子検出および診断市場:アプリケーション別、2021年
図28 アジア太平洋地域のCRISPR遺伝子検出および診断市場:アプリケーション別、2022年~2029年(百万米ドル)
図29 アジア太平洋地域のCRISPR遺伝子検出および診断市場:アプリケーション別、CAGR(2022-2029年)
図30 アジア太平洋地域のCRISPR遺伝子検出および診断市場:アプリケーション別、ライフライン曲線
図31 アジア太平洋地域のCRISPR遺伝子検出および診断市場:ワークフロー別、2021年
図32 アジア太平洋地域のCRISPR遺伝子検出および診断市場:ワークフロー別、2022年~2029年(百万米ドル)
図33 アジア太平洋地域のCRISPR遺伝子検出および診断市場:ワークフロー別、CAGR(2022-2029年)
図34 アジア太平洋地域のCRISPR遺伝子検出および診断市場:ワークフロー、ライフライン曲線別
図35 アジア太平洋地域のCRISPR遺伝子検出および診断市場:エンドユーザー別、2021年
図36 アジア太平洋地域のCRISPR遺伝子検出および診断市場:エンドユーザー別、2022年~2029年(百万米ドル)
図37 アジア太平洋地域のCRISPR遺伝子検出および診断市場:エンドユーザー別、CAGR(2022-2029年)
図38 アジア太平洋地域のCRISPR遺伝子検出および診断市場:エンドユーザー別、ライフライン曲線
図39 アジア太平洋地域のCRISPR遺伝子検出および診断市場:流通チャネル別、2021年
図40 アジア太平洋地域のCRISPR遺伝子検出および診断市場:流通チャネル別、2022年~2029年(百万米ドル)
図41 アジア太平洋地域のCRISPR遺伝子検出および診断市場:流通チャネル別、CAGR(2022-2029年)
図42 アジア太平洋地域のCRISPR遺伝子検出および診断市場:流通チャネル別、ライフライン曲線
図43 アジア太平洋地域のCRISPR遺伝子検出および診断市場:スナップショット(2021年)
図44 アジア太平洋地域のCRISPR遺伝子検出および診断市場:国別(2021年)
図45 アジア太平洋地域のCRISPR遺伝子検出および診断市場:国別(2022年および2029年)
図46 アジア太平洋地域のCRISPR遺伝子検出および診断市場:国別(2021年および2029年)
図47 アジア太平洋地域のCRISPR遺伝子検出および診断市場:クラス別(2022年および2029年)
図48 アジア太平洋地域のCRISPR遺伝子検出および診断市場:企業シェア2021(%)
調査方法
データ収集と基準年分析は、大規模なサンプル サイズのデータ収集モジュールを使用して行われます。この段階では、さまざまなソースと戦略を通じて市場情報または関連データを取得します。過去に取得したすべてのデータを事前に調査および計画することも含まれます。また、さまざまな情報ソース間で見られる情報の不一致の調査も含まれます。市場データは、市場統計モデルと一貫性モデルを使用して分析および推定されます。また、市場シェア分析と主要トレンド分析は、市場レポートの主要な成功要因です。詳細については、アナリストへの電話をリクエストするか、お問い合わせをドロップダウンしてください。
DBMR 調査チームが使用する主要な調査方法は、データ マイニング、データ変数が市場に与える影響の分析、および一次 (業界の専門家) 検証を含むデータ三角測量です。データ モデルには、ベンダー ポジショニング グリッド、市場タイムライン分析、市場概要とガイド、企業ポジショニング グリッド、特許分析、価格分析、企業市場シェア分析、測定基準、グローバルと地域、ベンダー シェア分析が含まれます。調査方法について詳しくは、お問い合わせフォームから当社の業界専門家にご相談ください。
カスタマイズ可能
Data Bridge Market Research は、高度な形成的調査のリーダーです。当社は、既存および新規のお客様に、お客様の目標に合致し、それに適したデータと分析を提供することに誇りを持っています。レポートは、対象ブランドの価格動向分析、追加国の市場理解 (国のリストをお問い合わせください)、臨床試験結果データ、文献レビュー、リファービッシュ市場および製品ベース分析を含めるようにカスタマイズできます。対象競合他社の市場分析は、技術ベースの分析から市場ポートフォリオ戦略まで分析できます。必要な競合他社のデータを、必要な形式とデータ スタイルでいくつでも追加できます。当社のアナリスト チームは、粗い生の Excel ファイル ピボット テーブル (ファクト ブック) でデータを提供したり、レポートで利用可能なデータ セットからプレゼンテーションを作成するお手伝いをしたりすることもできます。