South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032
Overview
The South Korea Medical Device Regulatory Affairs Outsourcing Market is expected to reach a 507.07 USD Billion by 2032 and is projected to grow at a CAGR of 23.27% from 2025 to 2032.
Revenue, 2024 (USD Billion)
172.60
Forecast, 2032 (USD Billion)
507.07
CAGR, 2024 - 2032
23.27%
Report Coverage
South Korea
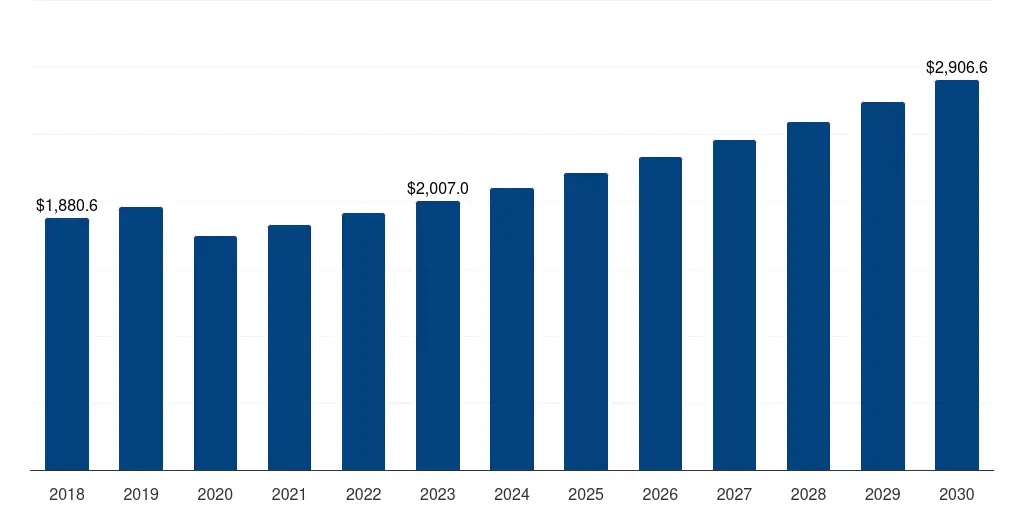
South Korea Medical Device Regulatory Affairs Outsourcing Market 2018-2032 USD Billion
South Korea Medical Device Regulatory Affairs Outsourcing Market, Key Findings (2025-2032)
Market Growth and Projections:
- Market Size (2024): 172.60 USD Billion
- Projected Market Size (2032): 507.07 USD Billion
- CAGR (2025-2032): 23.27%
Key Findings of South Korea Medical Device Regulatory Affairs Outsourcing Market
- The South Korea Medical Device Regulatory Affairs Outsourcing Market was valued at 172.60 USD Billion in 2024.
- The South Korea Medical Device Regulatory Affairs Outsourcing Market is likely to grow at a CAGR of 23.27% during the forecast period of 2024 to 2032.
- In 2024, the Largest segment Regulatory Affairs Services in Services Segment accounted for the largest share of the market with a revenue of 109.75 USD Billion
- The fastest growing segment Cardiology in Application Segment grew Fastest with a CAGR of 30.65% during the forecast period from 2024 to 2032.
South Korea Medical Device Regulatory Affairs Outsourcing Market Scope
South Korea Medical Device Regulatory Affairs Outsourcing Market Segmentation & Scope
Services
- Medical Writing
- Quality Consulting
- Regulatory Affairs Services
Product
- Raw Materials
- Electronics
- Finished Goods
Device Type
- Class III
- Class II
- Class I
Application
- Drug Delivery
- Diabetes Care
- Endoscopy
- Opthalmic
- Dental
- Diagnostic Imaging
- Orthopedic
- Others
- IVD
- General and Plastic Surgery
- Cardiology
End User
- Large Medical Device Company
- Small Medical Device Company
- Medium Medical Device Company
South Korea Medical Device Regulatory Affairs Outsourcing Market Data Coverage Insights
| Study Period | 2024-2032 |
| Base Year | 2020 |
| Unit | Revenue in USD Billion |
| Market Value in 2024 | 172.60 USD Billion |
| Market Value in 2032 | 507.07 USD Billion |
| CAGR (2025-2032) | 23.27% |
| Historic Data | 2016-2023 |
| Market Segments Covered | Services,Product,Device Type,Application,End User |
Regional Insights:
-
Leading Market (2024-2032): South Korea, leading in terms of revenue 172.60 USD Billion in 2024
- Key Country: South Korea, leading in terms of revenue with value of 172.60 USD Billion in 2024.
Segments and Scope
-
South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032, By Services
- Regulatory Affairs Services is the largest segment in South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032 with a revenue of 109.75 USD Billion in the year 2024.
- Regulatory Affairs Services is the Fastest growing segment in South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032 with a Growth rate of 24.74 % in forecast period 2025-2032.
-
South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032, By Product
- Finished Goods is the largest segment in South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032 with a revenue of 83.25 USD Billion in the year 2024.
- Finished Goods is the Fastest growing segment in South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032 with a Growth rate of 26.93 % in forecast period 2025-2032.
-
South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032, By Device Type
- Class I is the largest segment in South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032 with a revenue of 78.01 USD Billion in the year 2024.
- Class I is the Fastest growing segment in South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032 with a Growth rate of 27.43 % in forecast period 2025-2032.
-
South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032, By Application
- Cardiology is the largest segment in South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032 with a revenue of 42.61 USD Billion in the year 2024.
- Cardiology is the Fastest growing segment in South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032 with a Growth rate of 30.65 % in forecast period 2025-2032.
-
South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032, By End User
- Medium Medical Device Company is the largest segment in South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032 with a revenue of 86.35 USD Billion in the year 2024.
- Medium Medical Device Company is the Fastest growing segment in South Korea Medical Device Regulatory Affairs Outsourcing Market to 2032 with a Growth rate of 26.60 % in forecast period 2025-2032.
South Korea Medical Device Regulatory Affairs Outsourcing Market Company Share Analysis
| Company Name |

|
||
| Parexel International Corporation | |||
| North American Science Associates, LLC | |||
| Integer Holdings Corporation | |||
| Labcorp Drug Development | |||
| TÜV SÜD | |||

South Korea Medical Device Regulatory Affairs Outsourcing Market Geographical Sales Distribution, 2018-2032 USD Billion
South Korea Medical Device Regulatory Affairs Outsourcing Market Company Profiling

Industry Related Reports
Frequently Asked Questions
The South Korea Medical Device Regulatory Affairs Outsourcing Market is segmented based on Segmentation Services,Product,Device Type,Application,End User.
South Korea Medical Device Regulatory Affairs Outsourcing Market was valued at USD 172.60(Revenue in USD Billion) in 2020.
South Korea Medical Device Regulatory Affairs Outsourcing Market is projected to grow at a CAGR of 23.27% during the forecast period of 2024 to 2032.
The Regulatory Affairs Services segment is expected to dominate the South Korea Medical Device Regulatory Affairs Outsourcing Market, holding a largest market share of 109.75 USD Billion in 2024
South Korea Medical Device Regulatory Affairs Outsourcing Market Scope
South Korea Medical Device Regulatory Affairs Outsourcing Market Segmentation & Scope
Services
- Medical Writing
- Quality Consulting
- Regulatory Affairs Services
Product
- Raw Materials
- Electronics
- Finished Goods
Device Type
- Class III
- Class II
- Class I
Application
- Drug Delivery
- Diabetes Care
- Endoscopy
- Opthalmic
- Dental
- Diagnostic Imaging
- Orthopedic
- Others
- IVD
- General and Plastic Surgery
- Cardiology
End User
- Large Medical Device Company
- Small Medical Device Company
- Medium Medical Device Company
Frequently Asked Questions
The South Korea Medical Device Regulatory Affairs Outsourcing Market is segmented based on Segmentation Services,Product,Device Type,Application,End User.
South Korea Medical Device Regulatory Affairs Outsourcing Market was valued at USD 172.60(Revenue in USD Billion) in 2020.
South Korea Medical Device Regulatory Affairs Outsourcing Market is projected to grow at a CAGR of 23.27% during the forecast period of 2024 to 2032.
The estimated market value of the South Korea Medical Device Regulatory Affairs Outsourcing Market for final year is USD 507.07 (USD Billion).
South Korea Medical Device Regulatory Affairs Outsourcing Market Company Profiling

Frequently Asked Questions
The South Korea Medical Device Regulatory Affairs Outsourcing Market is segmented based on Segmentation Services,Product,Device Type,Application,End User.
South Korea Medical Device Regulatory Affairs Outsourcing Market was valued at USD 172.60(Revenue in USD Billion) in 2020.
South Korea Medical Device Regulatory Affairs Outsourcing Market is projected to grow at a CAGR of 23.27% during the forecast period of 2024 to 2032.
The estimated market value of the South Korea Medical Device Regulatory Affairs Outsourcing Market for final year is USD 507.07 (USD Billion).
HAVE A QUESTION?
Get expert advice on the right strategy for your business at no cost. We also offer customized subscription plans and affordable discounts for startups and universities.