O mercado de randomização e gestão de fornecimento de testes (RTSM) conta com características como a alocação de medicamentos em tempo real, a randomização de doentes e a gestão de stocks. O segmento dominante é a indústria farmacêutica, onde o RTSM simplifica ensaios clínicos complexos. Garante a alocação adequada de doentes, fornecimento de medicamentos e precisão dos dados. O RTSM permite também projetos de testes adaptativos, reduzindo custos e prazos. Com o crescimento da investigação farmacêutica, a importância do mercado de RTSM continua a aumentar, melhorando a eficiência e a precisão dos ensaios clínicos para o desenvolvimento de medicamentos.
Aceda ao relatório completo em https://www.databridgemarketresearch.com/reports/asia-pacific-randomization-and-trial-supply-management-rtsm-market
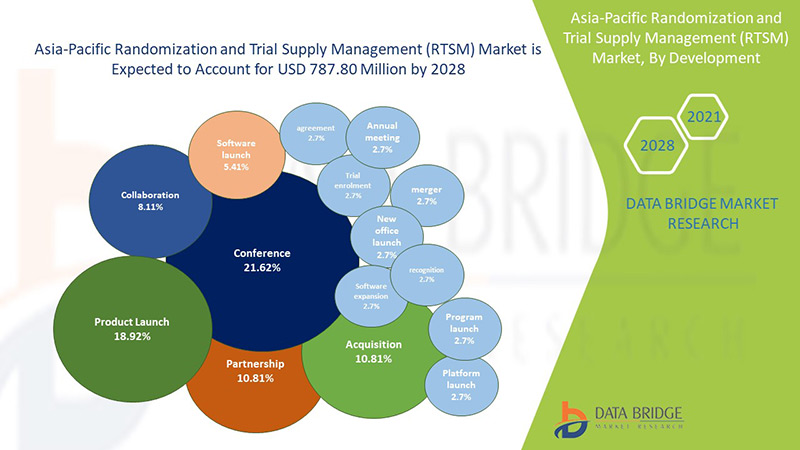
A Data Bridge Market Research analisa que o mercado de randomização e gestão de fornecimento de ensaios clínicos (RTSM) da Ásia-Pacífico , que era de 362,08 milhões de dólares em 2022, deverá atingir 1.084,71 milhões de dólares até 2030 e deverá passar por um CAGR de 14,7% durante o período previsto de 2023 a 2030. Os projetos de ensaios clínicos adaptativos, facilitados pela randomização e gestão de fornecimento de ensaios clínicos (RTSM), permitem ajustes em tempo real em ensaios clínicos com base em dados emergentes, otimizando a alocação de recursos e melhorando a adaptabilidade necessária para abordar a natureza dinâmica do desenvolvimento de medicamentos na indústria farmacêutica.
Principais conclusões do estudo
Espera-se que a crescente complexidade dos ensaios clínicos impulsione a taxa de crescimento do mercado
Com a crescente complexidade dos ensaios clínicos, que frequentemente envolvem um grande número de participantes e projetos de estudo adaptativos, a procura por sistemas eficientes de randomização e gestão de fornecimento de ensaios (RTSM) está a aumentar. Estes sistemas garantem a randomização precisa dos doentes e a gestão simplificada do fornecimento de medicamentos, reduzindo os erros e os desafios logísticos. Numa era de desenvolvimento de medicamentos em evolução, o RTSM desempenha um papel fundamental para garantir que os testes decorrem sem problemas, mantêm a integridade dos dados e se adaptam ao cenário mutável da investigação farmacêutica.
Âmbito do Relatório e Segmentação de Mercado
Métrica de Reporte
|
Detalhes
|
Período de previsão
|
2023 a 2030
|
Ano base
|
2022
|
Anos Históricos
|
2021 (personalizável para 2015-2020)
|
Unidades quantitativas
|
Receita em milhões de dólares americanos, volumes em unidades, preços em dólares americanos
|
Segmentos abrangidos
|
Componente (Software, Serviços), Modo de Entrega (Empresa Licenciada (On-Premise), Baseado na Nuvem (SaaS), Baseado na Web (On Demand), Aplicação (Planeamento e Previsão de Cenários, Agregação/Planeamento de Lotes, Construção de Estudos, Randomização, Dispensa de Medicamentos, Reprevisão e Otimização, Destruição e Reconciliação, Bloqueio de Base de Dados, Gestão de Stocks, Outros), Tipo de Ensaio Clínico (Ensaios de Tratamento, Ensaios de Prevenção, Ensaios de Rastreio, Ensaios de Cuidados de Suporte), Fase do Ensaio Clínico (Fase Inicial, Ensaios Clínicos de Fase I, Ensaios Clínicos de Fase II, Ensaios Clínicos de Fase III, Ensaios Clínicos de Fase IV e Peri/Pós-Aprovação), Área Terapêutica (Oncologia, Doenças Cardiovasculares e Doenças Circulatórias, Doenças Infecciosas , Doenças Digestivas, Doenças Musculoesqueléticas, Doenças do Sistema Nervoso, Endocrinologia e Doenças Metabólicas, Saúde Mental e Perturbações do Comportamento, Doenças do Sangue, Doenças Respiratórias, Outras), Utilizador final (empresas farmacêuticas e biofarmacêuticas, organizações de investigação por contrato, fabricantes de dispositivos médicos, hospitais, institutos de investigação académica, organismos governamentais, outros), canal de distribuição (venda direta, distribuidor externo)
|
Países abrangidos
|
China, Japão, Índia, Coreia do Sul, Singapura, Malásia, Austrália, Tailândia, Indonésia, Filipinas, Resto da Ásia-Pacífico (APAC) na Ásia-Pacífico (APAC).
|
Participantes do mercado abrangidos
|
MEDICAL Information Technology Inc. (EUA), SAP (Alemanha), CPSI (EUA), Meta (EUA), Elinext (EUA), EPIC Systems Corporation (EUA), INFOR (EUA), Cognizant (EUA), Oracle (EUA), Jag products LLC (EUA), Allscripts Healthcare LLC (EUA), Optum Inc. (EUA), Cerner Corporation (EUA), Change Healthcare (EUA), Koninklijke Philips NV (Holanda), athenahealth (EUA), eClinicalWorks (EUA)
|
Pontos de dados abordados no relatório
|
Para além dos insights sobre os cenários de mercado, tais como o valor de mercado, a taxa de crescimento, a segmentação, a cobertura geográfica e os principais participantes, os relatórios de mercado selecionados pela Data Bridge Market Research incluem também análises aprofundadas de especialistas, epidemiologia dos doentes, análise de pipeline, análise de preços e estrutura regulamentar.
|
Análise de Segmentos:
O mercado de randomização e gestão de fornecimento de ensaios clínicos (RTSM) da Ásia-Pacífico está segmentado com base no componente, modo de administração, aplicação, tipo de ensaio clínico, fase do ensaio clínico, área terapêutica, utilizadores finais e canal de distribuição.
- Com base no componente, o mercado de randomização e gestão de fornecimento de testes (RTSM) da Ásia-Pacífico está segmentado em software e serviços.
- Com base no modo de entrega, o mercado de gestão de fornecimento de testes e randomização (RTSM) da Ásia-Pacífico está segmentado em empresa licenciada (no local), baseado na nuvem (SaaS) e baseado na web (on-demand).
- Com base na aplicação, o mercado de randomização e gestão de fornecimento de testes (RTSM) da Ásia-Pacífico está segmentado em planeamento e previsão de cenários, agregação/planeamento de lotes, criação de estudos, randomização, dispensa de medicamentos, reprevisão e otimização, destruição e reconciliação, bloqueio de bases de dados, gestão de stocks e outros.
- Com base no tipo de ensaio clínico, o mercado de randomização e gestão de fornecimento de ensaios (RTSM) da Ásia-Pacífico está segmentado em ensaios de tratamento, ensaios de prevenção, ensaios de triagem e ensaios de cuidados de suporte.
- Com base na fase do ensaio clínico, o mercado de randomização e gestão de fornecimento de ensaios (RTSM) da Ásia-Pacífico está segmentado na fase inicial, ensaios clínicos de fase I, ensaios clínicos de fase II, ensaios clínicos de fase III, ensaios clínicos de fase IV e pré/pós-aprovação.
- Com base na área terapêutica, o mercado de randomização e gestão de fornecimento de testes (RTSM) da Ásia-Pacífico está segmentado em oncologia, doenças cardiovasculares e circulatórias, doenças infeciosas, doenças digestivas, doenças musculoesqueléticas, doenças do sistema nervoso, endocrinologia e doenças metabólicas, saúde mental e distúrbios comportamentais, doenças do sangue, doenças respiratórias e outras.
- Com base nos utilizadores finais, o mercado de randomização e gestão de fornecimento de testes (RTSM) da Ásia-Pacífico está segmentado em empresas farmacêuticas e biofarmacêuticas, organizações de investigação por contrato, fabricantes de dispositivos médicos, hospitais, institutos de investigação académica, organismos governamentais e outros.
- Com base no canal de distribuição, o mercado de randomização e gestão de fornecimento de testes (RTSM) da Ásia-Pacífico está segmentado em vendas diretas e distribuidor terceirizado.
Principais jogadores
A Data Bridge Market Research reconhece as seguintes empresas como participantes do mercado de randomização e gestão de fornecimento de testes (RTSM) na Ásia-Pacífico: MEDICAL Information Technology Inc. (EUA), SAP (Alemanha), CPSI (EUA), Meta (EUA), Elinext (EUA), EPIC Systems Corporation (EUA), INFOR (EUA), Cognizant (EUA), Oracle (EUA).
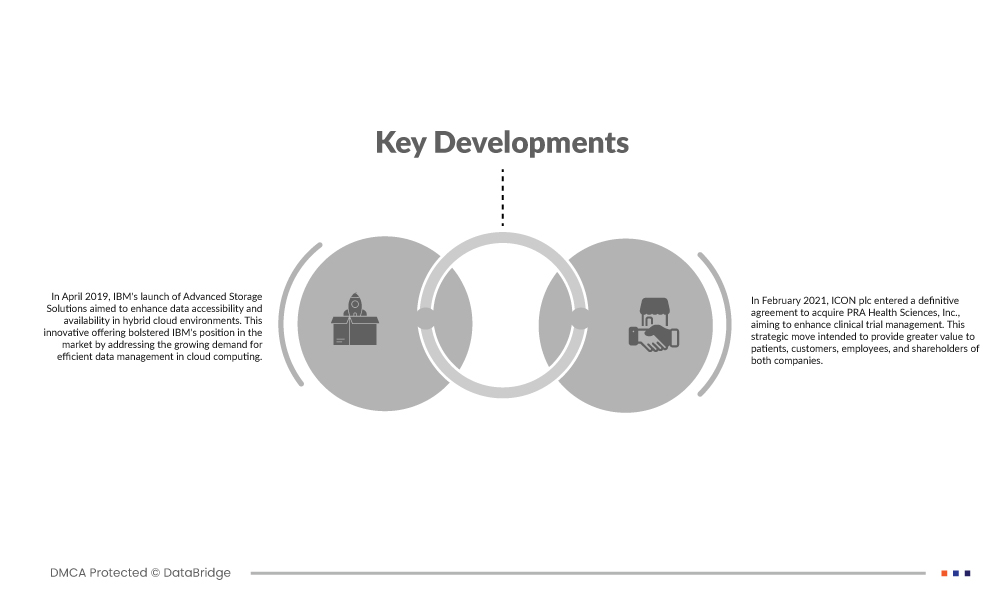
Desenvolvimentos de mercado
- Em abril de 2019, o lançamento do Advanced Storage Solutions pela IBM teve como objetivo melhorar a acessibilidade e a disponibilidade de dados em ambientes de cloud híbrida. Esta oferta inovadora reforçou a posição da IBM no mercado ao satisfazer a crescente procura por uma gestão eficiente de dados na computação em nuvem. Como resultado, atraiu novos clientes e expandiu a quota de mercado da empresa, contribuindo para o aumento das receitas. Esta mudança está alinhada com a estratégia da IBM de fornecer soluções de ponta para o panorama de TI em evolução.
- Em fevereiro de 2021, a ICON plc assinou um acordo definitivo para adquirir a PRA Health Sciences, Inc., com o objetivo de melhorar a gestão dos ensaios clínicos. Esta mudança estratégica teve como objetivo proporcionar maior valor aos doentes, clientes, colaboradores e acionistas de ambas as empresas. A aquisição permitiu a consolidação de recursos, experiência e capacidades no setor da investigação clínica, possibilitando soluções mais eficientes e abrangentes para apoiar o desenvolvimento de terapias e tratamentos inovadores para a saúde.
Análise Regional
Geograficamente, os países abrangidos pelo relatório de mercado de randomização e gestão de fornecimento de testes (RTSM) da Ásia-Pacífico são a China, Japão, Índia, Coreia do Sul, Singapura, Malásia, Austrália, Tailândia, Indonésia, Filipinas e Resto da Ásia-Pacífico (APAC).
De acordo com a análise de pesquisa de mercado da Data Bridge:
Espera-se que o Japão domine o mercado de randomização e gestão de fornecimento de ensaios clínicos (RTSM) da Ásia-Pacífico no período previsto de 2023-2030
O Japão está pronto para liderar o mercado de randomização e gestão de fornecimento de ensaios clínicos (RTSM) na região da Ásia-Pacífico (APAC) devido aos avanços na área da saúde. O compromisso do país com a investigação médica e os ensaios clínicos de ponta, aliado a uma infraestrutura de saúde robusta, posiciona-o como um player dominante na adoção da tecnologia RTSM. O foco do Japão na medicina de precisão e o seu papel como centro de inovação farmacêutica fazem dele um importante impulsionador do crescimento no mercado de RTSM da APAC.
Para obter informações mais detalhadas sobre o relatório de mercado de randomização e gestão de fornecimento de testes (RTSM) da Ásia-Pacífico, clique aqui – https://www.databridgemarketresearch.com/reports/asia-pacific-randomization-and-trial-supply-management-rtsm-market