This market includes the development, manufacturing, regulatory approval, distribution, and sale of prescription and non-prescription medicines, encompassing small-molecule drugs, biologics, biosimilars, and complex injectables. Within the defined scope, the market is segmented by product portfolio, including complex generics and injectables (cardiac, metabolic, anti-infectives, CNS, ophthalmology, gynecology, gastroenterology, and others), GLP-1 anti-obesity drugs (such as liraglutide and semaglutide), respiratory therapies (inhalers and nebulizers), biosimilars (immunologic and oncology), and immunologic and oncologic solid oral therapies (including gefitinib, imatinib, and JAK-1 inhibitors). Geographically, the market spans highly regulated, mature pharmaceutical systems in Europe, Japan, and Canada, characterized by structured reimbursement mechanisms, stringent regulatory oversight, and significant use of branded, generic, and biosimilar medicines, as well as emerging pharmaceutical markets in Latin America and Southeast Asia, where growth is supported by expanding healthcare access, rising medicine consumption, and increasing adoption of generics and essential drugs. The EMEU pharmaceutical market operates within diverse regulatory, pricing, and distribution frameworks, with medicines supplied through hospital pharmacies, retail pharmacies, and other regulated distribution channels depending on country-specific healthcare systems. Overall, the EMEU Pharmaceutical Market reflects a heterogeneous but interconnected regional landscape, combining established pharmaceutical demand in developed markets with volume-driven expansion in emerging economies, and forms an integral part of the global pharmaceutical industry.
Access Full Report @ https://www.databridgemarketresearch.com/reports/emeu-pharmaceutical-market
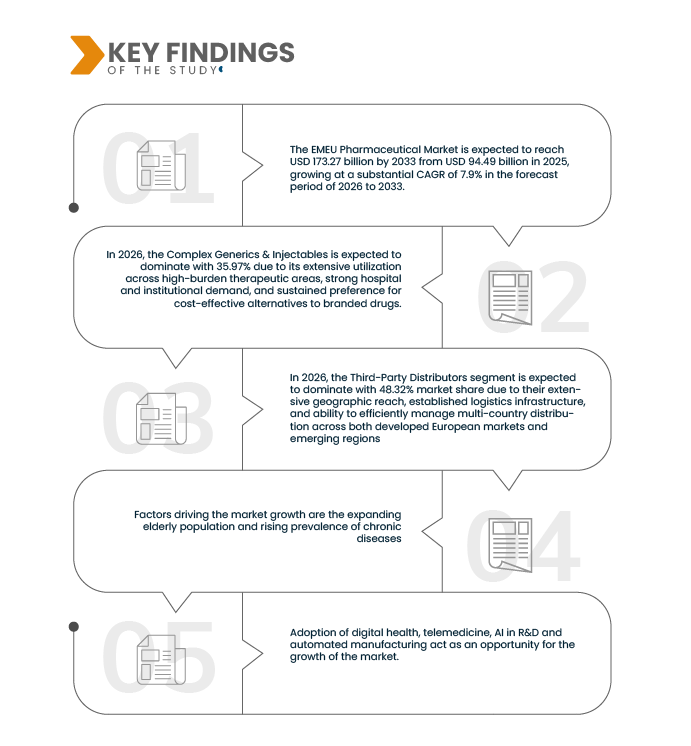
Data Bridge Market Research analyzes that the EMEU Pharmaceutical Market is expected to reach USD 173.27 billion by 2033 from USD 94.49 billion in 2025, growing at a substantial CAGR of 7.9% in the forecast period of 2026 to 2033.
Key Findings of the Study
Rising Elderly Populations and Increased Chronic Conditions
The growing geriatric population coupled with the escalating burden of chronic diseases is a key factor propelling growth in the EMEU (Europe, Middle East, and Europe) pharmaceutical market. Across Europe in particular, demographic aging is accelerating, with a rising proportion of individuals aged 65 years and above. This demographic transition is strongly associated with a higher incidence of chronic conditions such as cardiovascular diseases, diabetes, oncology disorders, respiratory illnesses, and neurodegenerative diseases, all of which require sustained pharmacological treatment and long-term disease management. Meanwhile, several Middle Eastern countries are experiencing an increase in lifestyle-related disorders due to urbanization, reduced physical activity, and dietary changes, further enlarging the patient base. The expanding prevalence of chronic illnesses is driving demand for branded drugs, generics, biologics, and specialty therapies across hospital and retail distribution channels. As a result, demographic and epidemiological shifts are reinforcing steady pharmaceutical consumption and supporting long-term revenue growth across the EMEU region.
Report Scope and Market Segmentation
|
Report Metric
|
Details
|
|
Forecast Period
|
2026 to 2033
|
|
Base Year
|
2025
|
|
Historic Year
|
2024 (Customizable 2018-2024)
|
|
Quantitative Units
|
Revenue in USD million
|
|
Segments Covered
|
Product Portfolio (Complex Generics & Injectables, Biosimilars, Immunologic and Oncologic Solid Orals, Respiratory, GLP‑1 Anti‑Obesity Products), Distribution Channel (Third Party Distributors, Own Sales Force, B2B Contracts, Direct Tenders
|
|
Region Covered
|
Europe, Emerging Markets
|
|
Market Players Covered
|
Sun Pharmaceutical Industries Ltd., Lupin, Dr. Reddy’s Laboratories Ltd., Cipla, Biocon, Aurobindo Pharma Limited, Teva Pharmaceutical Industries Ltd., Sandoz Group AG, Viatris Inc. and Zentiva Group, a.s. and, among others
|
|
Data Points Covered in the Report
|
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
|
Segment Analysis
EMEU Pharmaceutical market is categorized into two notable segments which are based on product portfolio, and distribution channel.
- On the basis of product portfolio, EMEU pharmaceutical market is segmented into Complex Generics & Injectables, Biosimilars, Immunologic and Oncologic Solid Orals, Respiratory, and GLP‑1 Anti‑Obesity Products. Complex Generics & Injectables segment is further sub segmented into Anti-Infectives, Cardiac, Metabolic, CNS, Gastro, Ophthalmology, Gynecology, and Others. Biosimilars segment is further sub segmented into Immunologic Biosimilars, and Oncology Biosimilars. Immunologic and Oncologic Solid Orals segment is further sub segmented into Jak-1 Inhibitors, Imatinib, and Gefitinib. Respiratory segment is further sub segmented into Inhalers, and Nebulizers. GLP‑1 Anti‑Obesity Products segment is further sub segmented into Semaglutide, Liraglutide.
In 2026, the Complex Generics & Injectables segment is expected to dominate the market
In 2026, the Complex Generics & Injectables is expected to dominate with 35.97% due to its extensive utilization across high-burden therapeutic areas, strong hospital and institutional demand, and sustained preference for cost-effective alternatives to branded drugs. This segment addresses core disease categories such as anti-infectives, cardiovascular, metabolic disorders, CNS conditions, and gastroenterology, which collectively account for a significant share of pharmaceutical consumption across Europe and emerging markets including Canada, Japan, Southeast Asia, and Latin America. Additionally, complex generics and injectables often involve advanced manufacturing processes, sterile production requirements, and stringent regulatory approvals, limiting the number of market entrants and supporting stable demand.
- On the basis of distribution channel, the EMEU pharmaceutical market is segmented into Third Party Distributors, Own Sales Force, B2B Contracts, Direct Tenders. The Third-Party Distributors segment is further sub segmented into Complex Generics & Injectables, Respiratory, Biosimilars, Immunologic and Oncologic Solid Orals, GLP‑1 Anti‑Obesity Products. The Own Sales Force segment is further sub segmented into Immunologic and Oncologic Solid Orals, Biosimilars, GLP‑1 Anti‑Obesity Products, Complex Generics & Injectables, Respiratory. The B2B Contracts segment is further sub segmented into Biosimilars, Complex Generics & Injectables, Immunologic and Oncologic Solid Orals, Respiratory, GLP‑1 Anti‑Obesity Products. The Direct Tenders segment is further sub segmented into Complex Generics & Injectables, Biosimilars, Immunologic and Oncologic Solid Orals, Respiratory, GLP‑1 Anti‑Obesity Products.
In 2026, the Third-Party Distributors segment is expected to dominate the market
In 2026, the Third-Party Distributors segment is expected to dominate with 48.32% market share due to their extensive geographic reach, established logistics infrastructure, and ability to efficiently manage multi-country distribution across both developed European markets and emerging regions. Third-party distributors play a critical role in ensuring timely availability of complex generics & injectables, respiratory products, biosimilars, immunologic and oncologic solid orals, and GLP-1 anti-obesity products, particularly in markets characterized by fragmented healthcare systems and diverse regulatory requirements. Their scale enables cost-efficient warehousing, cold-chain management, inventory optimization, and last-mile delivery, which is especially important for injectables, biosimilars, and temperature-sensitive therapies.
Major Players
Data Bridge Market Research analyzes are Sun Pharmaceutical Industries Ltd., Aurobindo Pharma Limited, Teva Pharmaceutical Industries Ltd., Sandoz Group AG, Viatris Inc. as the major market players of the market.
Market Development
- In August 2025, Sandoz has launched a generic version of Rivaroxaban in Germany. This provides an affordable, high-quality treatment option for preventing blood clots. The launch expands patient access to this critical antithrombotic medicine.
- In August 2025, Teva has received U.S. FDA approval for and launched the first generic version of Saxenda (liraglutide) injection. This generic GLP-1 receptor agonist is approved as a weight management tool for adults and pediatric patients, providing a more accessible option for chronic weight management.
- In August 2025, Viatris has received U.S. FDA approval for the first generic version of Iron Sucrose Injection, a medication used to treat iron deficiency anemia. This development provides a more affordable treatment option for patients and healthcare providers in the United States. The approval strengthens Viatris's portfolio of generic injectable medicines and enhances patient access to this critical therapy..
- In May 2025, Aurobindo Pharma, through its subsidiary Curateq, has received approval from the U.S. for its Biologics License Application (BLA) for a biosimilar product. This approval marks Aurobindo's entry into the U.S. biosimilars market, representing a significant milestone in the company's expansion into complex biologic therapeutics and enhancing patient access to more affordable treatment options.
- In July 2025, Sun Pharmaceutical has launched LEQSELVI (selpercatinib) in US for the treatment of specific RET-altered cancers. This provides a new, targeted therapy option for patients with lung and thyroid cancers. The launch enhances Sun Pharma's portfolio of specialized oncology medicines in the American market.
Regional Info
As per Data Bridge Market Research analysis:
Geographically, the countries covered in the EMEU Pharmaceutical Market report are Germany, France, U.K., Italy, Spain, Switzerland, Belgium, Netherlands, Sweden, Denmark, Norway, Finland, Rest of Europe. Canada, Japan, Indonesia, Thailand, Vietnam, Philippines, Malaysia, Singapore, Rest of Southeast Asia, Brazil, Argentina, Chile, Colombia, Peru, Mexico, Rest of Latin America.
Germany is the dominating country in the EMEU Pharmaceutical Market
Germany dominates the EMEU Pharmaceutical Market due to its well-established healthcare infrastructure, strong domestic pharmaceutical manufacturing base, and significant healthcare expenditure. The country hosts leading global pharmaceutical companies, advanced R&D capabilities, and a robust regulatory framework supporting drug innovation and commercialization. High adoption of specialty medicines, biologics, and advanced therapies, combined with comprehensive reimbursement systems and strong hospital networks, further reinforces Germany’s leadership position. Additionally, ongoing investments in biotechnology, clinical trials, and precision medicine continue to drive market expansion.
U.K. is expected to be the fastest growing country in the EMEU Pharmaceutical Market
The U.K. is projected to witness the fastest growth in the EMEU Pharmaceutical Market during the forecast period, supported by increasing clinical research activities, accelerated approval pathways for innovative drugs, and strong government backing for life sciences. Expansion in biologics, oncology therapies, rare disease treatments, and advanced therapeutics is fueling demand. Growing collaboration between academia, biotech firms, and pharmaceutical manufacturers, along with digital health integration and personalized medicine initiatives, is further accelerating pharmaceutical market growth across the country.
For more detailed information about the EMEU Pharmaceutical Market report, click here – https://www.databridgemarketresearch.com/reports/emeu-pharmaceutical-market