Emeu Pharmaceutical Market
Market Size in USD Billion
CAGR :
% 
 USD
94.49 Billion
USD
173.27 Billion
2025
2033
USD
94.49 Billion
USD
173.27 Billion
2025
2033
| 2026 - 2033 | |
| USD 94.49 Billion | |
| USD 173.27 Billion | |
|
|
|
|
EMEU Pharmaceutical Market Size
- The EMEU pharmaceutical market size was valued at USD 173.27 billion by 2033 from USD 94.49 billion in 2026, growing with a CAGR of 7.9% during the forecast period
- The Europe pharmaceutical market is experiencing steady growth, driven by rising demand for innovative medicines, biologics, and personalized therapies, supported by strong healthcare infrastructure and regulatory frameworks led by the European Medicines Agency. Increasing prevalence of chronic diseases and an aging population are further accelerating pharmaceutical production and consumption across the region.
- Expanding cross-border trade of pharmaceutical products within the European Union, along with growing export activities to Europe markets, is strengthening manufacturing capabilities and supply chain networks. This is boosting demand for high-quality, compliant production standards and regulatory approvals across Europe.
- Advancements in pharmaceutical manufacturing technologies, digital health integration, cold-chain logistics, and stringent quality control systems are enhancing operational efficiency and product safety. Investments in research and development, biologics manufacturing, and specialized production facilities are further supporting large-scale growth and innovation in the Europe pharmaceutical market.
EMEU Pharmaceutical Market Analysis
- The EMEU pharmaceutical market is experiencing steady growth, driven by rising demand for innovative medicines, biosimilars, vaccines, and specialty drugs, supported by strong healthcare systems and regulatory oversight from the European Medicines Agency. Increasing prevalence of chronic diseases and expanding access to advanced therapies are further fueling production and consumption across the region.
- Germany dominated the Europe pharmaceutical market, accounting for approximately 22.13% share in 2025, supported by its robust manufacturing base, advanced research infrastructure, and strong export presence within and beyond the European Union.
- Italy is the fastest-growing country, registering a robust CAGR of 8.5%, driven by increasing investments in generic drug manufacturing, expanding biopharmaceutical capabilities, and rising export activities. The country benefits from strengthening domestic production and growing participation in international supply chains.
- In 2026, the Complex Generics & Injectables segment leads the Europe pharmaceutical market, holding a 32.47% share, owing to increasing demand for cost-effective alternatives to branded drugs, rising prevalence of chronic conditions, and growing hospital and specialty care requirements across the region.
- Increasing adoption of advanced manufacturing technologies, digital quality management systems, cold-chain logistics, and compliance-driven production standards is accelerating pharmaceutical output and supply efficiency. Growing emphasis on sustainability, regulatory compliance, biosimilar development, and value-added formulations is further strengthening long-term growth prospects for the Europe pharmaceutical market.
Report Scope and EMEU Pharmaceutical Market Segmentation
|
Attributes |
EMEU Pharmaceutical Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
Europe
Emerging Market
South East Asia
Latin America
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
EMEU Pharmaceutical Market Trends
“Expansion of Biologics & Advanced Therapy Integration”
- Biopharmaceutical innovations, including monoclonal antibodies, cell and gene therapies, and mRNA-based treatments, are increasingly integrated into Europe’s pharmaceutical pipelines, enabling targeted and personalized treatment approaches for complex and rare diseases.
- Advanced therapy medicinal products (ATMPs) are gaining regulatory support and faster approval pathways under frameworks guided by the European Medicines Agency, accelerating commercialization across the region.
- Investment in specialized biologics manufacturing facilities, single-use technologies, and high-containment production systems is improving scalability, product safety, and compliance with stringent quality standards.
- Growing collaboration between pharmaceutical companies, biotech firms, and research institutions is strengthening innovation ecosystems and enhancing clinical development capabilities.
- Digitalization of clinical trials, real-world evidence integration, and precision medicine strategies are further optimizing treatment outcomes, reinforcing biologics and advanced therapies as a key growth trend in the Europe pharmaceutical market.
EMEU Pharmaceutical Market Dynamics
Driver
“Rising Prevalence of Chronic & Age-Related Diseases”
- The increasing incidence of chronic conditions such as cardiovascular diseases, diabetes, cancer, and respiratory disorders across Europe is significantly driving demand for long-term pharmacological treatments and specialty medicines.
- A rapidly aging population, is expanding the patient pool requiring continuous medication, biologics, and advanced therapeutic solutions.
- Government healthcare expenditure and reimbursement frameworks supported by the European Union are improving access to essential and innovative medicines, further stimulating pharmaceutical production and distribution.
- Increased awareness, early diagnosis initiatives, and improved healthcare infrastructure are accelerating prescription rates and treatment adherence across the region.
- As a result, sustained demand for chronic disease management therapies is acting as a major growth driver for the Europe pharmaceutical market.
Restraint/Challenge
“Stringent Regulatory Compliance & Pricing Pressures”
- The Europe pharmaceutical market operates under strict regulatory frameworks governed by the European Medicines Agency, requiring extensive clinical trials, rigorous quality assessments, and comprehensive pharmacovigilance systems, which increase time-to-market and development costs.
- Complex approval procedures and evolving compliance standards across the European Union create operational challenges for manufacturers seeking multi-country product launches.
- Drug pricing regulations, reference pricing systems, and government-led cost-containment measures are exerting significant pressure on profit margins, particularly for generic and biosimilar manufacturers.
- Lengthy reimbursement negotiations and health technology assessments (HTA) in countries such as France and Italy further delay commercialization timelines.
- Collectively, stringent regulatory requirements and pricing constraints act as a major challenge, limiting revenue growth and increasing compliance burdens in the Europe pharmaceutical market.
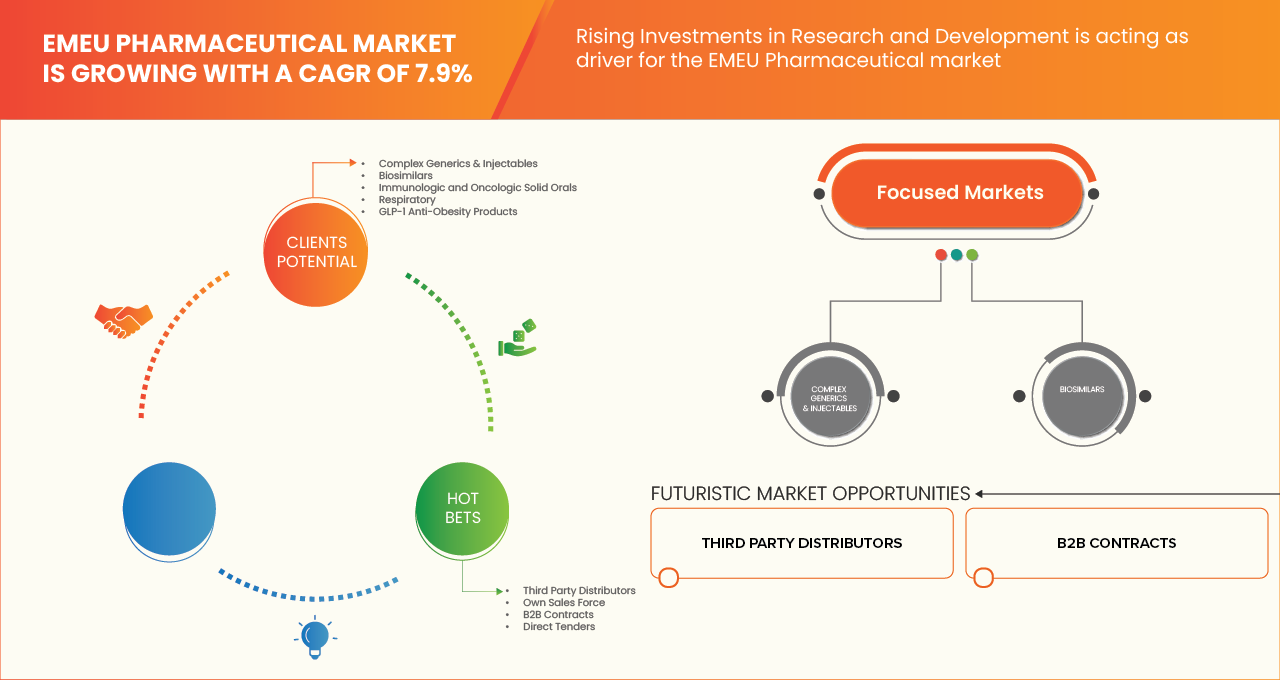
EMEU Pharmaceutical Market Scope
The Europe pharmaceutical market is segmented into two notable segments based on the Product portfolio and distribution channel .
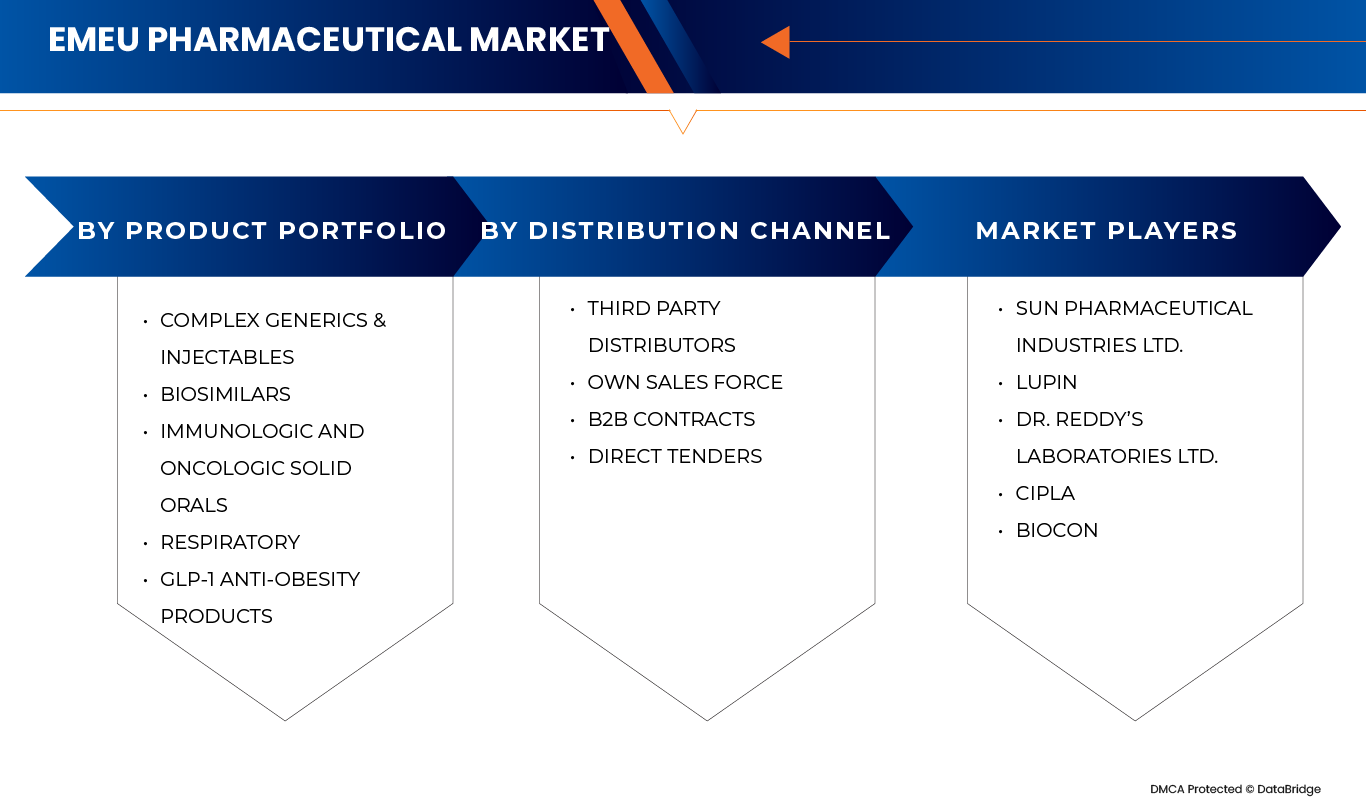
By Product portfolio
On the basis of Product portfolio, the Europe pharmaceutical market is segmented into Complex Generics & Injectables, Biosimilars, Immunologic and Oncologic Solid Orals, Respiratory, and GLP‑1 Anti‑Obesity Products. Complex Generics & Injectables segment is further sub segmented into Anti-Infectives, Cardiac, Metabolic, CNS, Gastro, Ophthalmology, Gynecology, and Others. Biosimilars segment is further sub segmented into Immunologic Biosimilars, and Oncology Biosimilars. Immunologic and Oncologic Solid Orals segment is further sub segmented into Jak-1 Inhibitors, Imatinib, and Gefitinib. Respiratory segment is further sub segmented into Inhalers, and Nebulizers. GLP‑1 Anti‑Obesity Products segment is further sub segmented into Semaglutide, Liraglutide. In 2026, the Complex Generics & Injectables is expected to dominate with 32.47% due to its extensive utilization across high-burden therapeutic areas, strong hospital and institutional demand, and sustained preference for cost-effective alternatives to branded drugs. This segment addresses core disease categories such as anti-infectives, cardiovascular, metabolic disorders, CNS conditions, and gastroenterology, which collectively account for a significant share of pharmaceutical consumption across Europe and emerging markets including Canada, Japan, Southeast Asia, and Latin America. Additionally, complex generics and injectables often involve advanced manufacturing processes, sterile production requirements, and stringent regulatory approvals, limiting the number of market entrants and supporting stable demand
By Distribution Channel
On the basis of Distribution channel, the Europe pharmaceutical market is segmented into On the basis of distribution channel, the EMEU pharmaceutical market is segmented into Third Party Distributors, Own Sales Force, B2B Contracts, Direct Tenders. The Third-Party Distributors segment is further sub segmented into Complex Generics & Injectables, Respiratory, Biosimilars, Immunologic and Oncologic Solid Orals, GLP‑1 Anti‑Obesity Products. The Own Sales Force segment is further sub segmented into Immunologic and Oncologic Solid Orals, Biosimilars, GLP‑1 Anti‑Obesity Products, Complex Generics & Injectables, Respiratory. The B2B Contracts segment is further sub segmented into Biosimilars, Complex Generics & Injectables, Immunologic and Oncologic Solid Orals, Respiratory, GLP‑1 Anti‑Obesity Products. The Direct Tenders segment is further sub segmented into Complex Generics & Injectables, Biosimilars, Immunologic and Oncologic Solid Orals, Respiratory, GLP‑1 Anti‑Obesity Products. In 2026, the Third-Party Distributors segment is expected to dominate with 52.16% market share due to their extensive geographic reach, established logistics infrastructure, and ability to efficiently manage multi-country distribution across both developed European markets and emerging regions. Third-party distributors play a critical role in ensuring timely availability of complex generics & injectables, respiratory products, biosimilars, immunologic and oncologic solid orals, and GLP-1 anti-obesity products, particularly in markets characterized by fragmented healthcare systems and diverse regulatory requirements. Their scale enables cost-efficient warehousing, cold-chain management, inventory optimization, and last-mile delivery, which is especially important for injectables, biosimilars, and temperature-sensitive therapies.
EMEU Pharmaceutical Market Regional Analysis.
- Germany dominates the Europe pharmaceutical market, accounting for approximately 21.79% share in 2026. Its leadership position is supported by a strong domestic manufacturing base, advanced R&D capabilities, and the presence of major pharmaceutical companies. Germany benefits from high healthcare expenditure, robust regulatory compliance standards, and strong export networks across Europe and globally. The country’s well-established healthcare infrastructure and continuous innovation in biologics and specialty medicines further reinforce its dominant position in the regional pharmaceutical landscape.
- Italy is the fastest-growing pharmaceutical market in Europe, registering a CAGR of 8.5% during 2026–2033. Growth is driven by expanding generic drug production, rising biopharmaceutical investments, and increasing export activities. Italy’s strengthening contract manufacturing capabilities and government support for pharmaceutical innovation are enhancing its competitive position. Additionally, increasing demand for complex injectables and specialty drugs is accelerating market expansion. The country’s strategic location and strong integration within European supply chains further contribute to its rapid pharmaceutical sector growth.
France EMEU Pharmaceutical Market Insight
France represents one of the key pharmaceutical markets in Europe, holding a 13.50% share in 2026. The market is projected to grow from USD 6,686.13 million in 2025 to USD 11,570.78 million by 2033, at a CAGR of 7.2%. Growth is supported by strong government healthcare spending, advanced research institutions, and increasing adoption of innovative therapies. France has a well-developed reimbursement framework and strong domestic manufacturing capabilities. Rising demand for biologics, oncology drugs, and chronic disease treatments is further driving market expansion. Continuous investments in R&D and digital health technologies are strengthening France’s position in the regional pharmaceutical ecosystem.
U.K. EMEU Pharmaceutical Market Insight
The United Kingdom holds a 14.95% market share in 2026 and is projected to grow from USD 7,449.99 million in 2025 to USD 12,271.26 million by 2033, registering a CAGR of 6.5%. The market benefits from strong clinical research capabilities, innovation-driven pharmaceutical development, and a well-established regulatory framework. Increasing demand for advanced therapies and specialty medicines is supporting growth. The U.K.’s focus on life sciences innovation, biopharmaceutical research, and global trade partnerships continues to attract investments. Despite regulatory adjustments post-Brexit, the country maintains a competitive pharmaceutical manufacturing and export environment..
Spain EMEU Pharmaceutical Market Insight
Spain accounts for 7.71% of the European pharmaceutical market in 2026 and is expected to grow from USD 3,797.64 million in 2025 to USD 6,855.27 million by 2033, at a CAGR of 7.7%. Growth is driven by increasing healthcare access, rising pharmaceutical exports, and expanding domestic manufacturing capacity. Spain is witnessing higher demand for generic medicines and biosimilars, supported by cost-containment policies and public healthcare expansion. Investments in production facilities and supply chain modernization are improving efficiency and output. The country’s growing role in clinical trials and specialty drug manufacturing further strengthens its position in Europe’s pharmaceutical market.
Japan EMEU Pharmaceutical Market Insight
Japan dominates the developed markets within the emerging pharmaceutical segment, holding a substantial 60.74% market share in 2026. The market is expected to grow from USD 16,100.48 million in 2025 to USD 29,719.03 million by 2033, registering a CAGR of 8.0%. This growth is driven by Japan’s advanced pharmaceutical manufacturing base, strong R&D infrastructure, and robust healthcare system. Aging population and high prevalence of chronic diseases increase demand for innovative and specialty medicines. Japan’s well-established regulatory environment and global export capabilities further strengthen its leadership position in the region.
Canada EMEU Pharmaceutical Market Insight
Canada is the fastest-growing market among the developed countries within the emerging pharmaceutical sector, with a CAGR of 9.2% projected from 2026 to 2033. The market is forecasted to rise from USD 10,297.09 million in 2025 to USD 20,709.97 million by 2033, holding 39.26% share in 2026. Growth is fueled by increasing investments in generic drug manufacturing, expansion of biopharmaceutical capabilities, and rising pharmaceutical exports. Enhanced domestic production capacity and integration into global supply chains are key factors supporting Canada’s rapid pharmaceutical market expansion
EMEU Pharmaceutical Market Share
The pharmaceutical industry is primarily led by well-established companies, including:
- Sun Pharmaceutical Industries Ltd. (India)
- Lupin Limited (India)
- Dr. Reddy’s Laboratories Ltd. (India)
- Cipla Limited (India)
- Biocon Limited (India)
- Aurobindo Pharma Limited (India)
- Teva Pharmaceutical Industries Ltd. (Israel)
- Sandoz Group AG (Switzerland)
- Viatris Inc. (U.S.)
- Zentiva Group, a.s. (Czech Republic)
Latest Developments in EMEU Pharmaceutical Market
- In August 2025, Sandoz has launched a generic version of Rivaroxaban in Germany. This provides an affordable, high-quality treatment option for preventing blood clots. The launch expands patient access to this critical antithrombotic medicine.
- In August 2025, Teva has received U.S. FDA approval for and launched the first generic version of Saxenda (liraglutide) injection. This generic GLP-1 receptor agonist is approved as a weight management tool for adults and pediatric patients, providing a more accessible option for chronic weight management.
- In August 2025, Viatris has received U.S. FDA approval for the first generic version of Iron Sucrose Injection, a medication used to treat iron deficiency anemia. This development provides a more affordable treatment option for patients and healthcare providers in the United States. The approval strengthens Viatris's portfolio of generic injectable medicines and enhances patient access to this critical therapy..
- In May 2025, Aurobindo Pharma, through its subsidiary Curateq, has received approval from the U.S. for its Biologics License Application (BLA) for a biosimilar product. This approval marks Aurobindo's entry into the U.S. biosimilars market, representing a significant milestone in the company's expansion into complex biologic therapeutics and enhancing patient access to more affordable treatment options.
- In July 2025, Sun Pharmaceutical has launched LEQSELVI (selpercatinib) in US for the treatment of specific RET-altered cancers. This provides a new, targeted therapy option for patients with lung and thyroid cancers. The launch enhances Sun Pharma's portfolio of specialized oncology medicines in the American market.
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Table of Content
1 INTRODUCTION
1.1 OBJECTIVES OF THE STUDY
1.2 MARKET DEFINITION
1.3 OVERVIEW OF EMEU PHARMACEUTICAL MARKET
1.4 SCOPE DEFINITION (EMEU VS INDIA & US EXCLUSION)
1.5 RATIONALE FOR FOCUSING ON EMEU
1.6 INDIAN PHARMA OPPORTUNITY IN EMEU
1.6.1 ESTABLISHED GLOBAL MANUFACTURING AND QUALITY CREDENTIALS
1.6.2 LEADERSHIP POSITION IN GENERICS SUPPLY
1.6.3 STRONG PRESENCE IN LATIN AMERICAN AND SOUTHEAST ASIAN MARKETS
1.6.4 DIVERSIFYING REGULATORY FOOTPRINT BEYOND TRADITIONAL MARKETS
1.6.5 COMPETITIVE ADVANTAGE THROUGH COST-EFFICIENT PRODUCTION MODELS
1.6.6 GROWTH OPPORTUNITIES IN BIOSIMILARS AND SPECIALTY PRODUCTS
1.6.7 TRADE AGREEMENTS AND MARKET ACCESS FACILITATION
1.6.8 ESTABLISHED GLOBAL LOGISTICS AND EXPORT ECOSYSTEM
1.7 LIMITATIONS
1.8 MARKETS COVERED
2 MARKET SEGMENTATION
2.1 MARKETS COVERED
2.2 GEOGRAPHICAL SCOPE
2.3 YEARS CONSIDERED FOR THE STUDY
2.4 CURRENCY AND PRICING
2.5 DBMR TRIPOD DATA VALIDATION MODEL
2.6 MULTIVARIATE MODELLING
2.7 PRODUCT PORTFOLIO LIFELINE CURVE
2.8 PRIMARY INTERVIEWS WITH KEY OPINION LEADERS
2.9 DBMR MARKET POSITION GRID
2.1 VENDOR SHARE ANALYSIS
2.11 SECONDARY SOURCES
2.12 ASSUMPTIONS
3 EXECUTIVE SUMMARY
4 PREMIUM INSIGHTS
5 INDUSTRY INSIGHTS
5.1 MICRO AND MACRO ECONOMIC FACTORS
5.2 PENETRATION AND GROWTH PROSPECT MAPPING
5.3 KEY PRICING STRATEGIES
5.4 INTERVIEW WITH SPECIALISTS
5.5 ANALYSIS AND RECOMMENDATION
6 REGULATORY FRAMEWORK
6.1 REGULATORY APPROVAL PROCESS
6.2 GEOGRAPHIES’ EASE OF REGULATORY APPROVAL
6.3 REGULATORY APPROVAL PATHWAYS
6.4 LICENSING AND REGISTRATION
6.5 POST-MARKETING SURVEILLANCE
6.6 GOOD MANUFACTURING PRACTICES (GMPS) GUIDELINE
7 EMEU PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO
7.1 OVERVIEW
7.2 COMPLEX GENERICS & INJECTABLES
7.2.1 EMEU COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
7.2.1.1 ANTI-INFECTIVES
7.2.1.2 CARDIAC
7.2.1.3 METABOLIC
7.2.1.4 CNS
7.2.1.5 GASTRO
7.2.1.6 OPHTHALMOLOGY
7.2.1.7 GYNECOLOGY
7.2.1.8 OTHERS
7.2.2 EMEU COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
7.2.2.1 EUROPE
7.2.2.2 EMERGING MARKETS
7.3 BIOSIMILARS
7.3.1 EMEU BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
7.3.1.1 IMMUNOLOGIC BIOSIMILARS
7.3.1.2 ONCOLOGY BIOSIMILARS
7.3.2 EMEU BIOSIMILARS IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
7.3.2.1 EUROPE
7.3.2.2 EMERGING MARKETS
7.4 IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS
7.4.1 EMEU IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
7.4.1.1 JAK-1 INHIBITORS
7.4.1.2 IMATINIB
7.4.1.3 GEFITINIB
7.4.2 EMEU IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
7.4.2.1 EUROPE
7.4.2.2 EMERGING MARKETS
7.5 RESPIRATORY
7.5.1 EMEU RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
7.5.1.1 INHALERS
7.5.1.2 NEBULIZERS
7.5.2 EMEU RESPIRATORY IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
7.5.2.1 EUROPE
7.5.2.2 EMERGING MARKETS
7.6 GLP‑1 ANTI‑OBESITY PRODUCTS
7.6.1 EMEU GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
7.6.1.1 SEMAGLUTIDE
7.6.1.2 LIRAGLUTIDE
7.6.2 EMEU GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
7.6.2.1 EUROPE
7.6.2.2 EMERGING MARKETS
8 EMEU PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL
8.1 OVERVIEW
8.2 THIRD PARTY DISTRIBUTORS
8.2.1 EMEU THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
8.2.1.1 COMPLEX GENERICS & INJECTABLES
8.2.1.2 RESPIRATORY
8.2.1.3 BIOSIMILARS
8.2.1.4 IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS
8.2.1.5 GLP‑1 ANTI‑OBESITY PRODUCTS
8.2.2 EMEU THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
8.2.2.1 EUROPE
8.2.2.2 EMERGING MARKETS
8.3 OWN SALES FORCE
8.3.1 EMEU OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
8.3.1.1 IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS
8.3.1.2 BIOSIMILARS
8.3.1.3 GLP‑1 ANTI‑OBESITY PRODUCTS
8.3.1.4 COMPLEX GENERICS & INJECTABLES
8.3.1.5 RESPIRATORY
8.3.2 EMEU OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
8.3.2.1 EUROPE
8.3.2.2 EMERGING MARKETS
8.4 B2B CONTRACTS
8.4.1 EMEU B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
8.4.1.1 BIOSIMILARS
8.4.1.2 COMPLEX GENERICS & INJECTABLES
8.4.1.3 IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS
8.4.1.4 RESPIRATORY
8.4.1.5 GLP‑1 ANTI‑OBESITY PRODUCTS
8.4.2 EMEU B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
8.4.2.1 EUROPE
8.4.2.2 EMERGING MARKETS
8.5 DIRECT TENDERS
8.5.1 EMEU DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
8.5.1.1 COMPLEX GENERICS & INJECTABLES
8.5.1.2 BIOSIMILARS
8.5.1.3 IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS
8.5.1.4 RESPIRATORY
8.5.1.5 GLP‑1 ANTI‑OBESITY PRODUCTS
8.5.2 EMEU DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
8.5.2.1 EUROPE
8.5.2.2 EMERGING MARKETS
9 EMEU PHARMACEUTICAL MARKET
9.1 EUROPE
9.1.1 GERMANY
9.1.2 U.K.
9.1.3 FRANCE
9.1.4 ITALY
9.1.5 SPAIN
9.1.6 SWITZERLAND
9.1.7 BELGIUM
9.1.8 SWEDEN
9.1.9 DENMARK
9.1.10 NORWAY
9.1.11 FINLAND
9.1.12 REST OF EUROPE
9.2 EMERGING MARKETS
9.2.1 DEVELOPED MARKETS INCLUDED UNDER EMERGING
9.2.1.1 JAPAN
9.2.1.2 CANADA
9.2.2 LATIN AMERICA
9.2.2.1 BRAZIL
9.2.2.2 MEXICO
9.2.2.3 ARGENTINA
9.2.2.4 COLOMBIA
9.2.2.5 CHILE
9.2.2.6 PERU
9.2.2.7 REST OF LATIN AMERICA
9.2.3 SOUTHEAST ASIA
9.2.3.1 INDONESIA
9.2.3.2 THAILAND
9.2.3.3 VIETNAM
9.2.3.4 PHILIPPINES
9.2.3.5 SINGAPORE
9.2.3.6 MALAYSIA
9.2.3.7 REST OF SOUTHEAST ASIA
10 COMPETITIVE LANDSCAPE
10.1 EMEU PHARMACEUTICAL MARKET: COMPANY LANDSCAPE
10.2 EUROPE PHARMACEUTICAL MARKET: COMPANY LANDSCAPE
10.3 EMERGING MARKETS PHARMACEUTICAL MARKET: COMPANY LANDSCAPE
11 COMPANY SHARE ANALYSIS
11.1 SUN PHARMACEUTICAL INDUSTRIES LTD: BY REGION
11.2 DR. REDDY’S LABORATORIES LTD: BY REGION
11.3 CIPLA: BY REGION
11.4 LUPIN: BY REGION
11.5 BIOCON: BY REGION
12 COMPANY PROFILES
12.1 SANDOZ GROUP AG
12.1.1 COMPANY SNAPSHOT
12.1.2 REVENUE ANALYSIS
12.1.3 GEOGRAPHICAL FINANCIALS
12.1.4 PRODUCT PORTFOLIO
12.1.5 RECENT DEVELOPMENT/ NEWS
12.2 TEVA PHARMACEUTICAL INDUSTRIES LTD.
12.2.1 COMPANY SNAPSHOT
12.2.2 REVENUE ANALYSIS
12.2.3 GEOGRAPHICAL FINANCIALS
12.2.4 PRODUCT PORTFOLIO
12.2.5 RECENT DEVELOPMENT/ NEWS
12.3 VIATRIS INC.
12.3.1 COMPANY SNAPSHOT
12.3.2 REVENUE ANALYSIS
12.3.3 GEOGRAPHICAL FINANCIALS
12.3.4 PRODUCT PORTFOLIO
12.3.5 RECENT DEVELOPMENT/ NEWS
12.4 AUROBINDO PHARMA LIMITED
12.4.1 COMPANY SNAPSHOT
12.4.2 REVENUE ANALYSIS
12.4.3 GEOGRAPHICAL FINANCIALS
12.4.4 PRODUCT PORTFOLIO
12.4.5 RECENT DEVELOPMENT/ NEWS
12.5 SUN PHARMACEUTICAL INDUSTRIES LTD.
12.5.1 COMPANY SNAPSHOT
12.5.2 REVENUE ANALYSIS
12.5.3 GEOGRAPHICAL FINANCIALS
12.5.4 PRODUCT PORTFOLIO
12.5.5 RECENT DEVELOPMENT/ NEWS
12.6 BIOCON.
12.6.1 COMPANY SNAPSHOT
12.6.2 REVENUE ANALYSIS
12.6.3 GEOGRAPHICAL FINANCIALS
12.6.4 PRODUCT PORTFOLIO
12.6.5 RECENT DEVELOPMENT/ NEWS
12.7 CIPLA
12.7.1 COMPANY SNAPSHOT
12.7.2 REVENUE ANALYSIS
12.7.3 GEOGRAPHICAL FINANCIALS
12.7.4 PRODUCT PORTFOLIO
12.7.5 RECENT DEVELOPMENT/ NEWS
12.8 MPANY P DR. REDDY’S LABORATORIES LTD.
12.8.1 COMPANY SNAPSHOT
12.8.2 REVENUE ANALYSIS
12.8.3 GEOGRAPHICAL FINANCIALS
12.8.4 PRODUCT PORTFOLIO
12.8.5 RECENT DEVELOPMENT/ NEWS
12.9 LUPIN
12.9.1 COMPANY SNAPSHOT
12.9.2 REVENUE ANALYSIS
12.9.3 GEOGRAPHICAL FINANCIALS
12.9.4 PRODUCT PORTFOLIO
12.9.5 RECENT DEVELOPMENT/ NEWS
12.1 ZENTIVA GROUP, A.S.
12.10.1 COMPANY SNAPSHOT
12.10.2 SERVICE PORTFOLIO
12.10.3 RECENT DEVELOPMENT
13 QUESTIONNAIRE
14 RELATED REPORTS
List of Table
TABLE 1 EMEU PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 2 EMEU COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 3 EMEU COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
TABLE 4 EMEU BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 5 EMEU BIOSIMILARS IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
TABLE 6 EMEU IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 7 EMEU IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
TABLE 8 EMEU RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 9 EMEU RESPIRATORY IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
TABLE 10 EMEU GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 11 EMEU GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
TABLE 12 EMEU PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 13 EMEU THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 14 EMEU THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
TABLE 15 EMEU OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 16 EMEU OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
TABLE 17 EMEU B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 18 EMEU B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
TABLE 19 EMEU DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 20 EMEU DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
TABLE 21 EUROPE PHARMACEUTICAL MARKET, BY COUNTRY, 2018-2033 (USD MILLION)
TABLE 22 EUROPE PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 23 EUROPE COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 24 EUROPE BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 25 EUROPE IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 26 EUROPE RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 27 EUROPE GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 28 EUROPE PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 29 EUROPE THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 30 EUROPE OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 31 EUROPE B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 32 EUROPE DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 33 GERMANY PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 34 GERMANY COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 35 GERMANY BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 36 GERMANY IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 37 GERMANY RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 38 GERMANY GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 39 GERMANY PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 40 GERMANY THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 41 GERMANY OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 42 GERMANY B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 43 GERMANY DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 44 U.K. PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 45 U.K. COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 46 U.K. BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 47 U.K. IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 48 U.K. RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 49 U.K. GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 50 U.K. PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 51 U.K. THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 52 U.K. OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 53 U.K. B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 54 U.K. DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 55 FRANCE PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 56 FRANCE COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 57 FRANCE BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 58 FRANCE IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 59 FRANCE RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 60 FRANCE GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 61 FRANCE PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 62 FRANCE THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 63 FRANCE OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 64 FRANCE B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 65 FRANCE DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 66 ITALY PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 67 ITALY COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 68 ITALY BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 69 ITALY IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 70 ITALY RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 71 ITALY GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 72 ITALY PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 73 ITALY THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 74 ITALY OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 75 ITALY B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 76 ITALY DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 77 SPAIN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 78 SPAIN COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 79 SPAIN BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 80 SPAIN IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 81 SPAIN RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 82 SPAIN GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 83 SPAIN PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 84 SPAIN THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 85 SPAIN OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 86 SPAIN B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 87 SPAIN DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 88 SWITZERLAND PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 89 SWITZERLAND COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 90 SWITZERLAND BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 91 SWITZERLAND IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 92 SWITZERLAND RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 93 SWITZERLAND GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 94 SWITZERLAND PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 95 SWITZERLAND THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 96 SWITZERLAND OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 97 SWITZERLAND B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 98 SWITZERLAND DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 99 NETHERLAND PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 100 NETHERLAND COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 101 NETHERLAND BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 102 NETHERLAND IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 103 NETHERLAND RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 104 NETHERLAND GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 105 NETHERLAND PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 106 NETHERLAND THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 107 NETHERLAND OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 108 NETHERLAND B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 109 NETHERLAND DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 110 BELGIUM PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 111 BELGIUM COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 112 BELGIUM BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 113 BELGIUM IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 114 BELGIUM RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 115 BELGIUM GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 116 BELGIUM PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 117 BELGIUM THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 118 BELGIUM OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 119 BELGIUM B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 120 BELGIUM DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 121 SWEDEN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 122 SWEDEN COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 123 SWEDEN BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 124 SWEDEN IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 125 SWEDEN RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 126 SWEDEN GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 127 SWEDEN PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 128 SWEDEN THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 129 SWEDEN OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 130 SWEDEN B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 131 SWEDEN DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 132 DENMARK PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 133 DENMARK COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 134 DENMARK BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 135 DENMARK IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 136 DENMARK RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 137 DENMARK GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 138 DENMARK PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 139 DENMARK THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 140 DENMARK OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 141 DENMARK B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 142 DENMARK DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 143 NORWAY PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 144 NORWAY COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 145 NORWAY BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 146 NORWAY IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 147 NORWAY RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 148 NORWAY GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 149 NORWAY PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 150 NORWAY THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 151 NORWAY OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 152 NORWAY B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 153 NORWAY DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 154 FINLAND PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 155 FINLAND COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 156 FINLAND BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 157 FINLAND IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 158 FINLAND RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 159 FINLAND GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 160 FINLAND PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 161 FINLAND THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 162 FINLAND OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 163 FINLAND B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 164 FINLAND DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 165 REST OF EUROPE PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 166 REST OF EUROPE COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 167 REST OF EUROPE BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 168 REST OF EUROPE IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 169 REST OF EUROPE RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 170 REST OF EUROPE GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 171 REST OF EUROPE PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 172 REST OF EUROPE THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 173 REST OF EUROPE OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 174 REST OF EUROPE B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 175 REST OF EUROPE DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 176 EMERGING MARKETS PHARMACEUTICAL MARKET, BY REGION, 2018-2033 (USD MILLION)
TABLE 177 EMERGING MARKETS PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 178 EMERGING MARKETS COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 179 EMERGING MARKETS BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 180 EMERGING MARKETS IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 181 EMERGING MARKETS RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 182 EMERGING MARKETS GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 183 EMERGING MARKETS PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 184 EMERGING MARKETS THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 185 EMERGING MARKETS OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 186 EMERGING MARKETS B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 187 EMERGING MARKETS DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 188 DEVELOPED MARKETS INCLUDED UNDER EMERGING PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 189 DEVELOPED MARKETS INCLUDED UNDER EMERGING COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 190 DEVELOPED MARKETS INCLUDED UNDER EMERGING BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 191 DEVELOPED MARKETS INCLUDED UNDER EMERGING IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 192 DEVELOPED MARKETS INCLUDED UNDER EMERGING RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 193 DEVELOPED MARKETS INCLUDED UNDER EMERGING GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 194 DEVELOPED MARKETS INCLUDED UNDER EMERGING PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 195 DEVELOPED MARKETS INCLUDED UNDER EMERGING THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 196 DEVELOPED MARKETS INCLUDED UNDER EMERGING OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 197 DEVELOPED MARKETS INCLUDED UNDER EMERGING B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 198 DEVELOPED MARKETS INCLUDED UNDER EMERGING DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 199 DEVELOPED MARKETS INCLUDED UNDER EMERGING PHARMACEUTICAL MARKET, BY COUNTRY, 2018-2033 (USD MILLION)
TABLE 200 JAPAN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 201 JAPAN COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 202 JAPAN BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 203 JAPAN IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 204 JAPAN RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 205 JAPAN GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 206 JAPAN PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 207 JAPAN THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 208 JAPAN OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 209 JAPAN B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 210 JAPAN DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 211 CANADA PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 212 CANADA COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 213 CANADA BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 214 CANADA IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 215 CANADA RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 216 CANADA GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 217 CANADA PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 218 CANADA THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 219 CANADA OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 220 CANADA B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 221 CANADA DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 222 LATIN AMERICA PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 223 LATIN AMERICA COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 224 LATIN AMERICA BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 225 LATIN AMERICA IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 226 LATIN AMERICA RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 227 LATIN AMERICA GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 228 LATIN AMERICA PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 229 LATIN AMERICA THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 230 LATIN AMERICA OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 231 LATIN AMERICA B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 232 LATIN AMERICA DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 233 LATIN AMERICA PHARMACEUTICAL MARKET, BY COUNTRY, 2018-2033 (USD MILLION)
TABLE 234 BRAZIL PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 235 BRAZIL COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 236 BRAZIL BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 237 BRAZIL IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 238 BRAZIL RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 239 BRAZIL GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 240 BRAZIL PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 241 BRAZIL THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 242 BRAZIL OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 243 BRAZIL B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 244 BRAZIL DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 245 MEXICO PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 246 MEXICO COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 247 MEXICO BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 248 MEXICO IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 249 MEXICO RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 250 MEXICO GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 251 MEXICO PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 252 MEXICO THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 253 MEXICO OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 254 MEXICO B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 255 MEXICO DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 256 ARGENTINA PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 257 ARGENTINA COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 258 ARGENTINA BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 259 ARGENTINA IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 260 ARGENTINA RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 261 ARGENTINA GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 262 ARGENTINA PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 263 ARGENTINA THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 264 ARGENTINA OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 265 ARGENTINA B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 266 ARGENTINA DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 267 COLOMBIA PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 268 COLOMBIA COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 269 COLOMBIA BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 270 COLOMBIA IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 271 COLOMBIA RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 272 COLOMBIA GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 273 COLOMBIA PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 274 COLOMBIA THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 275 COLOMBIA OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 276 COLOMBIA B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 277 COLOMBIA DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 278 CHILE PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 279 CHILE COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 280 CHILE BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 281 CHILE IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 282 CHILE RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 283 CHILE GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 284 CHILE PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 285 CHILE THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 286 CHILE OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 287 CHILE B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 288 CHILE DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 289 PERU PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 290 PERU COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 291 PERU BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 292 PERU IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 293 PERU RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 294 PERU GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 295 PERU PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 296 PERU THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 297 PERU OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 298 PERU B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 299 PERU DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 300 REST OF LATIN AMERICA PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 301 REST OF LATIN AMERICA COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 302 REST OF LATIN AMERICA BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 303 REST OF LATIN AMERICA IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 304 REST OF LATIN AMERICA RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 305 REST OF LATIN AMERICA GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 306 REST OF LATIN AMERICA PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 307 REST OF LATIN AMERICA THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 308 REST OF LATIN AMERICA OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 309 REST OF LATIN AMERICA B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 310 REST OF LATIN AMERICA DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 311 SOUTHEAST ASIA PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 312 SOUTHEAST ASIA COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 313 SOUTHEAST ASIA BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 314 SOUTHEAST ASIA IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 315 SOUTHEAST ASIA RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 316 SOUTHEAST ASIA GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 317 SOUTHEAST ASIA PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 318 SOUTHEAST ASIA THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 319 SOUTHEAST ASIA OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 320 SOUTHEAST ASIA B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 321 SOUTHEAST ASIA DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 322 SOUTHEAST ASIA PHARMACEUTICAL MARKET, BY COUNTRY, 2018-2033 (USD MILLION)
TABLE 323 INDONESIA PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 324 INDONESIA COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 325 INDONESIA BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 326 INDONESIA IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 327 INDONESIA RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 328 INDONESIA GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 329 INDONESIA PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 330 INDONESIA THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 331 INDONESIA OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 332 INDONESIA B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 333 INDONESIA DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 334 THAILAND PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 335 THAILAND COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 336 THAILAND BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 337 THAILAND IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 338 THAILAND RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 339 THAILAND GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 340 THAILAND PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 341 THAILAND THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 342 THAILAND OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 343 THAILAND B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 344 THAILAND DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 345 VIETNAM PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 346 VIETNAM COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 347 VIETNAM BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 348 VIETNAM IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 349 VIETNAM RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 350 VIETNAM GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 351 VIETNAM PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 352 VIETNAM THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 353 VIETNAM OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 354 VIETNAM B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 355 VIETNAM DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 356 PHILIPPINES PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 357 PHILIPPINES COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 358 PHILIPPINES BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 359 PHILIPPINES IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 360 PHILIPPINES RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 361 PHILIPPINES GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 362 PHILIPPINES PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 363 PHILIPPINES THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 364 PHILIPPINES OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 365 PHILIPPINES B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 366 PHILIPPINES DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 367 SINGAPORE PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 368 SINGAPORE COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 369 SINGAPORE BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 370 SINGAPORE IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 371 SINGAPORE RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 372 SINGAPORE GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 373 SINGAPORE PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 374 SINGAPORE OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 375 SINGAPORE B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 376 SINGAPORE THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 377 SINGAPORE DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 378 MALAYSIA PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 379 MALAYSIA COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 380 MALAYSIA BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 381 MALAYSIA IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 382 MALAYSIA RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 383 MALAYSIA GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 384 MALAYSIA PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 385 MALAYSIA THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 386 MALAYSIA OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 387 MALAYSIA B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 388 MALAYSIA DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 389 REST OF SOUTHEAST ASIA PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 390 REST OF SOUTHEAST ASIA COMPLEX GENERICS & INJECTABLES IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 391 REST OF SOUTHEAST ASIA BIOSIMILARS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 392 REST OF SOUTHEAST ASIA IMMUNOLOGIC AND ONCOLOGIC SOLID ORALS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 393 REST OF SOUTHEAST ASIA RESPIRATORY IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 394 REST OF SOUTHEAST ASIA GLP-1 ANTI-OBESITY PRODUCTS IN PHARMACEUTICAL MARKET, BY TYPE, 2018-2033 (USD MILLION)
TABLE 395 REST OF SOUTHEAST ASIA PHARMACEUTICAL MARKET, BY DISTRIBUTION CHANNEL, 2018-2033 (USD MILLION)
TABLE 396 REST OF SOUTHEAST ASIA THIRD PARTY DISTRIBUTORS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 397 REST OF SOUTHEAST ASIA OWN SALES FORCE IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 398 REST OF SOUTHEAST ASIA B2B CONTRACTS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
TABLE 399 REST OF SOUTHEAST ASIA DIRECT TENDERS IN PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO, 2018-2033 (USD MILLION)
List of Figure
FIGURE 1 EMEU PHARMACEUTICAL MARKET: SEGMENTATION
FIGURE 2 EMEU PHARMACEUTICAL MARKET: DATA TRIANGULATION
FIGURE 3 EMEU PHARMACEUTICAL MARKET: DROC ANALYSIS
FIGURE 4 EMEU PHARMACEUTICAL MARKET: REGIONAL ANALYSIS
FIGURE 5 EMEU PHARMACEUTICAL MARKET: COMPANY RESEARCH ANALYSIS
FIGURE 6 EMEU PHARMACEUTICAL MARKET: INTERVIEW DEMOGRAPHICS
FIGURE 7 EMEU PHARMACEUTICAL MARKET: DBMR MARKET POSITION GRID
FIGURE 8 EMEU PHARMACEUTICAL MARKET: VENDOR SHARE ANALYSIS
FIGURE 9 EXECUTIVE SUMMARY
FIGURE 10 EMEU PHARMACEUTICAL MARKET: SEGMENTATION
FIGURE 11 STRATEGIC DECISIONS
FIGURE 12 FIVE SEGMENTS COMPRISE THE EMEU PHARMACEUTICAL MARKET, BY PRODUCT PORTFOLIO
FIGURE 13 RISING ELDERLY POPULATIONS AND INCREASED CHRONIC CONDITIONS IS EXPECTED TO DRIVE THE EMEU PHARMACEUTICAL MARKET IN THE FORECAST PERIOD OF 2026 TO 2033
FIGURE 14 PRODUCT PORTFOLIO SEGMENT IS EXPECTED TO ACCOUNT FOR THE LARGEST SHARE OF THE EMEU PHARMACEUTICAL MARKET IN 2026 & 2033
FIGURE 15 EMEU PHARMACEUTICAL MARKET: BY PRODUCT PORTFOLIO, 2025
FIGURE 16 EMEU PHARMACEUTICAL MARKET: BY PRODUCT PORTFOLIO, 2026 TO 2033 (USD MILLION)
FIGURE 17 EMEU PHARMACEUTICAL MARKET: BY PRODUCT PORTFOLIO, CAGR (2026- 2033)
FIGURE 18 EMEU PHARMACEUTICAL MARKET: BY PRODUCT PORTFOLIO, LIFELINE CURVE
FIGURE 19 EMEU PHARMACEUTICAL MARKET: BY DISTRIBUTION CHANNEL, 2025
FIGURE 20 EMEU PHARMACEUTICAL MARKET: BY DISTRIBUTION CHANNEL, 2026 TO 2033 (USD MILLION)
FIGURE 21 EMEU PHARMACEUTICAL MARKET: BY DISTRIBUTION CHANNEL, CAGR (2026- 2033)
FIGURE 22 EMEU PHARMACEUTICAL MARKET: BY DISTRIBUTION CHANNEL, LIFELINE CURVE
FIGURE 23 EUROPE PHARMACEUTICAL MARKET: SNAPSHOT (2025)
FIGURE 24 EMEU PHARMACEUTICAL MARKET: COMPANY SHARE 2025 (%)
FIGURE 25 EUROPE PHARMACEUTICAL MARKET: COMPANY SHARE 2025 (%)
FIGURE 26 EMERGING MARKETS PHARMACEUTICAL MARKET: COMPANY SHARE 2025 (%)

Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.