암은 전 세계적으로 가장 심각한 건강 문제 중 하나입니다. 선진국에서는 주요 사망 원인이고, 개발도상국에서는 두 번째로 큰 사망 원인입니다. 의료 기술에 대한 공학적 응용 분야의 다양한 발전 또한 국가의 경제 성장에 상당한 영향을 미쳤습니다. 폐쇄형 시스템 이송 장치는 처음에는 유해 약물의 조제 및 투여 과정에서 의료 전문가를 노출로부터 보호하기 위한 목적으로만 사용되었지만, 이후 종양학 환경에서 표면 오염을 줄이는 데 도움이 되었습니다.
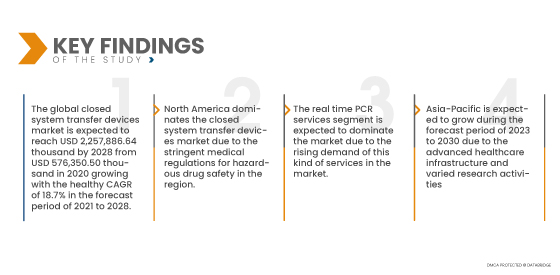
Data Bridge Market Research에 따르면, 글로벌 폐쇄형 시스템 이송 장치 시장은 2020년 5억 7,635만 5,000달러에서 2028년에는 2,257,886.64달러로 성장할 것으로 예상되며, 2021년부터 2028년까지의 예측 기간 동안 18.7%의 건강한 CAGR로 성장할 것으로 전망됩니다. 제약 분야 전반에서 폐쇄형 시스템 이송 장치에 대한 수요가 증가함에 따라 예측 기간 동안 시장 성장에 대한 잠재적 기회가 제공될 것입니다.
기술의 발전이 시장 성장률을 견인할 것으로 예상됩니다.
의료 기술에 대한 공학적 응용 분야의 주요 발전은 경제 성장에 지대한 영향을 미쳤습니다. 폐쇄형 시스템 이송 장치(CSTD)는 약물이 환경으로 누출되는 것을 방지하여 여러 의료 종사자가 취급하는 약물에 노출될 가능성을 차단합니다. 최근 여러 임상 연구에서 의료진의 약물 조제 및 투여 과정에서 CSTD가 안전성 측면에서 향상된 것으로 평가 및 입증되었습니다.
예를 들어,
- BD는 미생물 침투 방지에 도움이 되는 BD PhaSeal 시스템을 출시했으며, 이는 약물 낭비 감소를 위한 약물 바이알 최적화 프로그램의 일부로 사용되었다는 사실이 수많은 저널에 기록되었습니다.
보고서 범위 및 시장 세분화
보고서 메트릭
|
세부
|
예측 기간
|
2021년부터 2028년까지
|
기준 연도
|
2020
|
역사적인 해
|
2019 (2013~2018년으로 맞춤 설정 가능)
|
양적 단위
|
매출(백만 달러), 볼륨(단위), 가격(달러)
|
다루는 세그먼트
|
유형(멤브레인-멤브레인 시스템 및 무침 폐쇄 시스템 전송 장치), 구성 요소(장치 및 액세서리), 폐쇄 메커니즘(푸시-투-턴 시스템, 컬러-투-컬러 정렬 시스템, 루어-락 시스템 및 클릭-투-락 시스템), 기술(다이어프램 기반 장치, 구획화된 장치 및 공기 청정/여과 장치), 최종 사용자(병원, 종양학 센터 및 진료소, 외래 수술 센터, 학술 및 연구 기관), 유통 채널(직접 입찰 및 소매 판매)
|
포함 국가
|
미국, 캐나다 및 멕시코(북미), 독일, 프랑스, 영국, 네덜란드, 스위스, 벨기에, 러시아, 이탈리아, 스페인, 터키, 유럽의 기타 유럽 국가, 중국, 일본, 인도, 한국, 싱가포르, 말레이시아, 호주, 태국, 인도네시아, 필리핀, 아시아 태평양(APAC)의 기타 아시아 태평양 국가(APAC), 사우디 아라비아, UAE, 남아프리카 공화국, 이집트, 이스라엘, 중동 및 아프리카(MEA)의 일부인 기타 중동 및 아프리카(MEA), 브라질, 아르헨티나 및 남미의 일부인 기타 남미
|
시장 참여자 포함
|
B. Braun Medical Inc.(미국), ICU Medical, Inc(미국), BD(미국), Corvida Medical(미국), YUKON MEDICAL(인도), Caragen Ltd.(아일랜드), Baxter(미국), JMS North America Corporation(미국), Vygon(프랑스), Epic Medical Corporation(이탈리아)
|
보고서에서 다루는 데이터 포인트
|
Data Bridge Market Research에서 큐레이팅한 시장 보고서에는 시장 가치, 성장률, 세분화, 지리적 적용 범위, 주요 기업 등 시장 시나리오에 대한 통찰력 외에도 심층적인 전문가 분석, 환자 역학, 파이프라인 분석, 가격 분석, 규제 프레임워크가 포함됩니다.
|
세그먼트 분석:
폐쇄형 시스템 전송 장치 시장은 유형, 구성 요소, 폐쇄 메커니즘, 기술, 최종 사용자 및 유통 채널을 기준으로 6가지 주요 부문으로 구분됩니다.
- 폐쇄형 시스템 전송 장치 시장은 유형을 기준으로 멤브레인 대 멤브레인 시스템과 바늘 없는 폐쇄형 시스템 전송 장치로 구분됩니다.
멤브레인-멤브레인 세그먼트 유형은 폐쇄형 시스템 전송 장치 시장을 지배할 것으로 예상됩니다.
멤브레인 대 멤브레인 부문은 2021년부터 2028년까지의 예측 기간 동안 18.2%의 CAGR로 글로벌 폐쇄 시스템 전송 장치를 주도할 것으로 예상됩니다. 이는 오염에 덜 취약하고 바늘이 없는 폐쇄 시스템 전송 장치와 달리 기존 장비와 함께 사용할 수 있기 때문입니다.
- 폐쇄형 시스템 이송 장치 시장은 구성 요소를 기준으로 장치와 액세서리로 세분화됩니다. 장치는 바이알 접근 장치, 주사기 안전 장치, 백/라인 접근 장치, 벤트형 백 스파이크 등으로 더 세분화됩니다.
구성 요소 유형의 장치 세그먼트는 폐쇄형 시스템 전송 장치 시장을 지배할 것으로 예상됩니다.
장치 부문은 액세서리에 비해 사용량이 많고 가격이 비싸기 때문에 2021년부터 2028년까지 예측 기간 동안 19.0%의 CAGR로 글로벌 폐쇄 시스템 전송 장치를 지배할 것으로 예상됩니다.
- 폐쇄형 시스템 이송 장치 시장은 폐쇄 메커니즘을 기준으로 푸시-투-턴 시스템, 컬러-투-컬러 정렬 시스템, 루어-락 시스템, 클릭-투-락 시스템으로 구분됩니다. 클릭-투-락 시스템은 부품 조립이 필요 없는 직관적인 시스템 설계로 인해 2021년부터 2028년까지 연평균 성장률 19.5%로 세계 폐쇄형 시스템 이송 장치 시장을 주도할 것으로 예상됩니다.
- 기술 측면에서 폐쇄형 시스템 이송 장치 시장은 격막 기반, 구획형, 그리고 공기 청정/여과 장치로 구분됩니다. 격막 기반 장치 부문은 작동 편의성과 안전성 향상으로 인해 2021년부터 2028년까지 연평균 성장률 19.3%로 전 세계 폐쇄형 시스템 이송 장치 시장을 주도할 것으로 예상됩니다.
- 최종 사용자 기준으로 폐쇄형 시스템 이송 장치 시장은 병원, 종양학 센터 및 진료소, 외래 수술 센터, 학술 및 연구 기관 등으로 세분화됩니다. 병원 분야는 조작의 용이성과 안전성으로 인해 2021년부터 2028년까지 연평균 성장률 19.4%로 전 세계 폐쇄형 시스템 이송 장치 시장을 주도할 것으로 예상됩니다.
- 유통 채널을 기준으로 폐쇄형 시스템 이송 장치 시장은 직접 입찰과 소매 판매로 구분됩니다. 소매 부문은 2021년부터 2028년까지 예측 기간 동안 연평균 성장률 18.9%로 전 세계 폐쇄형 시스템 이송 장치 시장을 주도할 것으로 예상됩니다. 이는 병원이 위험 요인으로 더 많이 노출될 가능성이 높고, 폐쇄형 시스템 이송 장치를 통해 의료 전문가가 유해 약물 노출을 예방할 수 있기 때문입니다.
주요 플레이어
Data Bridge Market Research에서는 폐쇄형 시스템 이송 장치 시장의 주요 참여자로 다음과 같은 회사를 인식하고 있습니다. B. Braun Medical Inc.(미국), ICU Medical, Inc(미국), BD(미국), Corvida Medical(미국), YUKON MEDICAL(인도), Caragen Ltd.(아일랜드), Baxter(미국), JMS North America Corporation(미국), Vygon(프랑스), Epic Medical Corporation(이탈리아).
시장 개발
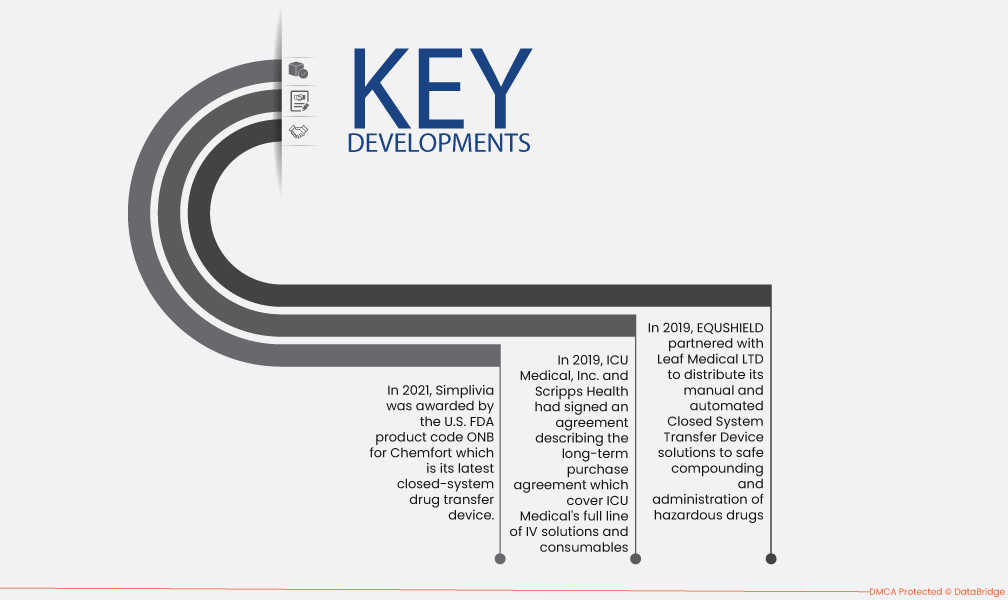
- 2021년, 심플리비아는 최신 폐쇄형 약물 전달 장치인 켐포트(Chemfort)에 대해 미국 FDA 제품 코드 ONB를 획득했습니다. 켐포트는 체액, 비말, 에어로졸의 누출을 방지하여 의료진의 항암제 노출을 효과적으로 줄이는 데 도움을 줄 것입니다.
- 2019년에 ICU Medical, Inc.와 Scripps Health는 ICU Medical의 IV 솔루션 및 소모품 전체 라인과 ICU Medical MedNet IV 약물 안전 소프트웨어가 포함된 Plum 360 주입 시스템을 포괄하는 장기 구매 계약을 체결했습니다.
- 2019년 EQUSHIELD는 Leaf Medical LTD와 협력하여 위험한 약물의 안전한 조제 및 투여를 위한 수동 및 자동 폐쇄형 시스템 전송 장치 솔루션을 배포했습니다.
지역 분석
지리적으로 폐쇄 시스템 전송 장치 시장 보고서에 포함된 국가는 북미의 미국, 캐나다 및 멕시코, 독일, 프랑스, 영국, 네덜란드, 스위스, 벨기에, 러시아, 이탈리아, 스페인, 터키, 유럽의 기타 유럽, 중국, 일본, 인도, 한국, 싱가포르, 말레이시아, 호주, 태국, 인도네시아, 필리핀, 아시아 태평양(APAC)의 기타 아시아 태평양(APAC), 사우디 아라비아, UAE, 남아프리카, 이집트, 이스라엘, 중동 및 아프리카(MEA)의 일부인 기타 중동 및 아프리카(MEA), 브라질, 아르헨티나 및 남미의 일부인 기타 남미입니다.
Data Bridge Market Research 분석에 따르면:
북미는 2023년부터 2030년까지의 예측 기간 동안 폐쇄 시스템 전송 장치 시장에서 지배적인 지역입니다.
북미는 폐쇄형 시스템 전달 장치 시장의 선두 주자입니다. 이 장치는 백신 투여를 더욱 빠르고 효율적으로 수행할 수 있는 방법을 제공하기 때문에 이러한 유형의 사용이 증가하고 있습니다. 또한, 미국은 북미 지역의 유해 약물 안전에 대한 엄격한 의료 규정으로 인해 북미 지역을 장악하고 있습니다.
아시아 태평양 지역은 2023년부터 2030년까지 폐쇄 시스템 전송 장치 시장에서 가장 빠르게 성장하는 지역으로 추정됩니다.
아시아 태평양 지역은 국가의 선진화된 의료 인프라와 종양학 치료 관련 연구 활동으로 인해 2023년부터 2030년까지 성장할 것으로 예상됩니다. 중국은 의료 연구 및 기술 발전으로 아시아 태평양 지역을 선도하고 있습니다.
폐쇄 시스템 전송 장치 시장 보고서 에 대한 자세한 내용은 여기를 클릭하세요 - https://www.databridgemarketresearch.com/reports/global-closed-system-transfer-devices-market