Global Point-of-Care Testing Market is expected to reach USD 35.3 billion by 2024 from USD 18.6 billion in 2016, growing at a CAGR of 8.4% in the forecast period 2017 to 2024. The new market report contains data for historic years 2015, the base year of calculation is 2016 and the forecast period is 2017 to 2024.
Visit Report @ https://www.databridgemarketresearch.com/press-release/global-point-of-care-testing-market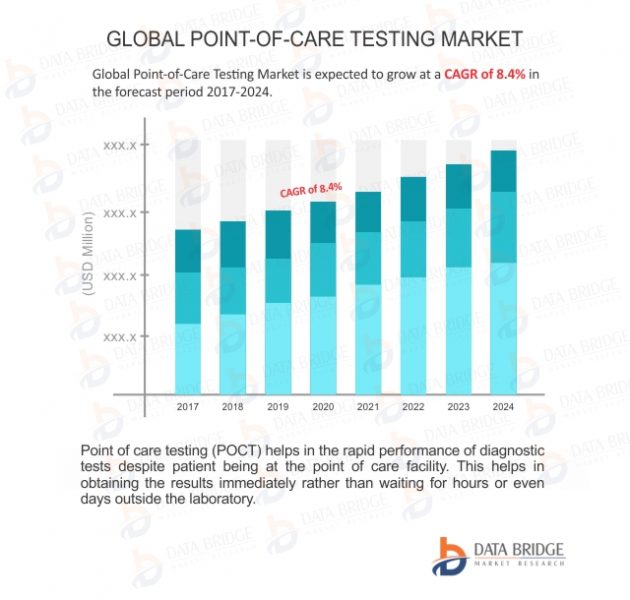 The global point-of-care market is highly concentrated to a few big players and rest to local players who cater to domestic markets only. F. Hoffmann-La Roche Ltd dominated the point-of-care market accounting for a highest market share in 2016, followed by Siemens AG and Abbott. Other players in this market include Beckman Coulter, Inc., Becton, Dickinson and Company, Johnson & Johnson, Alere Inc., Instrumentation Laboratory, PTS Diagnostics, Abaxis, Inc., Medtronic, Accriva Diagnostics, OPTI Medical, Sienco, Inc., Alpha Scientific, AccuBioTech Co., Ltd., ACON Laboratories, Inc., Acrongenomics, Inc, bioMérieux SA, Biomerica, Inc., Atlas Genetics Ltd., Alfa Scientific Designs, Inc., Bio-Rad Laboratories, Inc., Medica Corporation, and Nova Biomedicaamong others holds 53.1% of the global injectable drug delivery market in 2016.
The global point-of-care market is highly concentrated to a few big players and rest to local players who cater to domestic markets only. F. Hoffmann-La Roche Ltd dominated the point-of-care market accounting for a highest market share in 2016, followed by Siemens AG and Abbott. Other players in this market include Beckman Coulter, Inc., Becton, Dickinson and Company, Johnson & Johnson, Alere Inc., Instrumentation Laboratory, PTS Diagnostics, Abaxis, Inc., Medtronic, Accriva Diagnostics, OPTI Medical, Sienco, Inc., Alpha Scientific, AccuBioTech Co., Ltd., ACON Laboratories, Inc., Acrongenomics, Inc, bioMérieux SA, Biomerica, Inc., Atlas Genetics Ltd., Alfa Scientific Designs, Inc., Bio-Rad Laboratories, Inc., Medica Corporation, and Nova Biomedicaamong others holds 53.1% of the global injectable drug delivery market in 2016.
HOFFMANN-LA ROCHE LTD:
Historical Background - Roche was established in 1896
Nature of Business: F. Hoffmann-La Roche Ltd., is one of the largest biotech company with 17 biopharmaceuticals on the market. The company is operating in more than 100 countries around the globe. Some of the subsidiaries are Genetech, Roche Diagnostics, Chugai Pharmaceutical Co., Ventana Medical Systems, InterMune, Inc., Syntex, Anadys Pharmaceuticals, Roche Applied Science, Bioveris, Roche products, Inc., Roche pharma AG, Pt Roche Indonesia, Bina Technologies, Inc., nv Roche sa, Roche SAS, Roche Glycart AG, Roche Co., Ltd.
Main focus: Since the establishment in 1896, the company's main focus is on oncology, immunology, ophthalmology, infectious diseases and neuroscience.
Main Business Areas: Pharmaceuticals and Diagnostics.
Recent Developments:
- In August 2016,Roche Diagnostics Ltd., received FDA approval for cobas Influenza A/B & RSV test for the cobasLiat System that differentiates flu and Respiratory Syncytial Virus (RSV) in 20 minutes
- In May 2015, Roche Diagnostics Ltd., received FDA CLIA Waiver for the cobasLiat PCR System and Strep A assay to test Strep A in throat swab specimen.
- In December 2014, Roche Diagnostics Ltd, received FDA approval for LightMix Ebola Zaire rRT-PCR Test at the time of emergency to treat Ebola Zaire virus infection.
SIEMENS AG:
Historical Background: Siemens AG was established in 1847
Nature of business: Siemens AG is a German leading company specializing in supplier of systems for power generation and transmission, medical diagnosis, infrastructure and industry solutions.
Main focus: Since the establishment in 1847, the company's main focus is on electrification, automation and digitalization
Main Business areas: power and gas, wind power and renewable, energy management, building technologies, mobility, digital factory and process industries.
Subsidiary: Siemens Healthcare operates in more than 200 countries across United States, Europe, Western Hemisphere, Africa, and Asia-Pacific SA/NV, Beersel (Belgium), ETM professional control GmbH, Eisenstadt (Austria), Siemens Healthcare Diagnostics Products GmbH (Marburg), Linacre Investments (Pty) Ltd., Kenilworth (South Africa), Siemens Healthcare FZ LLC (Dubai).
Recent Developments:
- In November 2016, Siemens strengthened its healthcare business through public listing to give more focus and flexibility in pursuing its growth strategy.
- In June 2016, Siemens opened a new headquarter in Munich to enhance its global presence in this market.
ABBOTT:
Historical Background: Abbott was established in 1888.
Nature of business: Abbott is a globally diversified healthcare company focused on development and marketing of diagnostics products, medical devices, nutrition and branded generic pharmaceuticals.
Main focus: Since the establishment in 1888, the company's main focus is on established pharmaceutical, molecular diagnostics, diabetes care and nutritionals.
Main Business areas: Established pharmaceuticals, molecular diagnostics, diabetes care nutritionals, and vascular.
Subsidiary:Abbott operates in more than 150 countries across United States, Europe, Western Hemisphere, Africa, and Asia-Pacific. For instance, Abbott Administration Inc. (Delaware), Abbott Home Infusion Services, Inc. (New York), Abbott Laboratories International Co. (Illinois), Abbott Australasia Pty Ltd.(Australia), Murex Diagnostics International, Inc. (Barbados), Abbott S.A. (Belgium).
Recent Developments:
- In May 2017, Abbott announced CE Mark approval and its use of the new Rx Insertable Cardiac Monitor (ICM), which is the world's first smartphone compatible ICM to identify or detect cardiac arrhythmias, including atrial fibrillation (AF), for therapy.
- In May 2017, Abbott announced the national reimbursement for freestyle Libre in France, providing access to revolutionary technology for people suffering with diabetes. This would help thousands of people to have access to test type 1 and type 2 diabetes.
- In February 2017, FDA granted the use of Abbott Real Time Zika test product to detect Zika virus in whole blood samples for use of in vitro diagnostic tests during emergencies.










