تُحسّن أدوية الورم النقوي المتعدد نتائج المرضى وتُخفف العبء الاقتصادي على أنظمة الرعاية الصحية والمجتمع. فمن خلال الإدارة الفعّالة للمرض من خلال العلاجات الدوائية، تُحسّن هذه الأدوية صحة المرضى، وتُقلّل من معدلات دخولهم إلى المستشفى، وتُطيل متوسط العمر المتوقع. يُساعد نهج الرعاية الصحية الشامل، الذي يشمل التدخلات الطبية والرعاية الداعمة إلى جانب العلاج الدوائي، في الحفاظ على صحة المرضى وجودة حياتهم. ويُعدُّ الأثر الاقتصادي كبيرًا، إذ يُقلّل العلاج الناجح من الحاجة إلى موارد رعاية صحية مكثفة، مما يُقلّل التكاليف المباشرة وغير المباشرة المرتبطة بفترات الإقامة الطويلة في المستشفى وتدابير الرعاية الداعمة.
يمكنك الوصول إلى التقرير الكامل على https://www.databridgemarketresearch.com/reports/us-multiple-myeloma-drugs-market
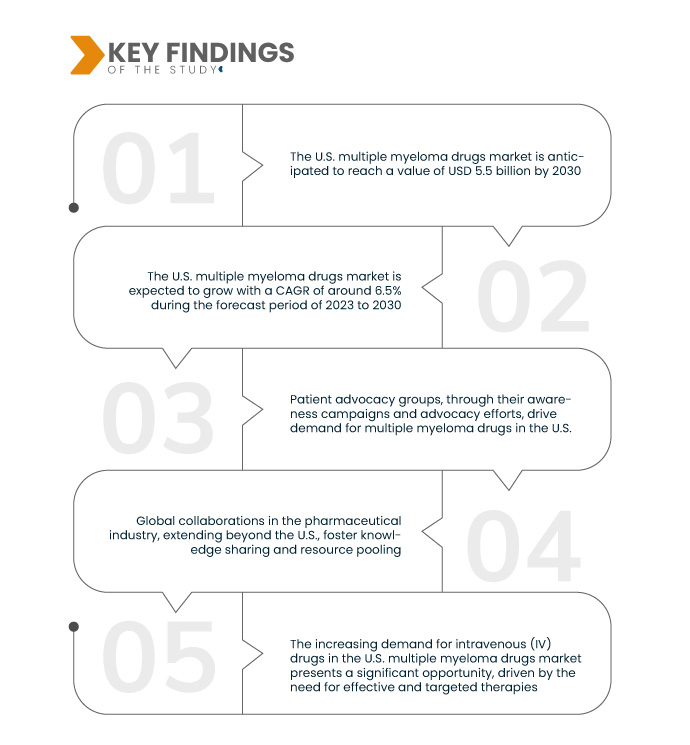
تُحلل شركة داتا بريدج لأبحاث السوق سوق أدوية الورم النقوي المتعدد في الولايات المتحدة ، والذي بلغ 1.79 مليار دولار أمريكي عام 2022، ومن المتوقع أن يصل إلى 5.5 مليار دولار أمريكي بحلول عام 2030، وأن يشهد معدل نمو سنوي مركب بنسبة 15% خلال الفترة المتوقعة من 2023 إلى 2030. تُسهم الاستثمارات في الطب الدقيق وأبحاث المؤشرات الحيوية في تحقيق تقدم في تحديد الأهداف الجزيئية الدقيقة المرتبطة بالورم النقوي المتعدد. وهذا يُعزز تطوير علاجات مُستهدفة، ويعزز فعالية العلاج، ويدعم النهج العلاجية المُخصصة.
النتائج الرئيسية للدراسة
من المتوقع أن يؤدي ارتفاع معدل الإصابة بالورم النقوي المتعدد بسبب شيخوخة السكان إلى دفع معدل نمو السوق
تُعدّ شيخوخة السكان في الولايات المتحدة، إلى جانب عوامل ديموغرافية أخرى، عاملاً رئيسياً في نمو سوق أدوية الورم النقوي المتعدد في الولايات المتحدة. فمع تقدم الأفراد في السن، يميل معدل الإصابة بالورم النقوي المتعدد إلى الارتفاع. وقد أدى هذا التحول الديموغرافي إلى زيادة انتشار المرض. ويتزايد الطلب على الأدوية لإدارة وعلاج الورم النقوي المتعدد بفعالية ضمن هذه الفئة السكانية المتنامية. وتستجيب صناعة الأدوية لهذا الطلب من خلال تطوير وتوفير علاجات مبتكرة، مما يخلق فرصة سوقية كبيرة مدفوعة باحتياجات الرعاية الصحية لفئة سكانية متقدمة في السن ومعرضة للإصابة بالورم النقوي المتعدد.
نطاق التقرير وتقسيم السوق
مقياس التقرير
|
تفاصيل
|
فترة التنبؤ
|
من 2023 إلى 2030
|
سنة الأساس
|
2022
|
السنوات التاريخية
|
2021 (قابلة للتخصيص حتى 2015-2020)
|
الوحدات الكمية
|
الإيرادات بالمليارات من الدولارات الأمريكية، والحجم بالوحدات، والتسعير بالدولار الأمريكي
|
القطاعات المغطاة
|
المرض (ورم النخاع المتعدد الخامل (الخامل)، ورم النخاع المتعدد النشط (المصحوب بأعراض)، ورم البلازما المنفرد للعظم، ورم البلازما خارج النخاع، ورم النخاع الخفيف السلسلة، ورم النخاع غير الإفرازي وأنواع نادرة من ورم النخاع)، العلاج (العلاج المناعي، العلاج الكيميائي، الكورتيكوستيرويدات، وغيرها)، آلية العمل ( مثبط البروتيازوم ، الأجسام المضادة وحيدة النسيلة، العوامل المعدلة للمناعة، مثبطات هيستون دياسيتيل، وغيرها)، العلامات التجارية (دارزالكس، نينلارو، ريفليمود، فيلكيد، إمبليسيتي، فينكليكتا، وغيرها)، نوع الدواء (علامة تجارية، عام)، طريقة الإعطاء (وريدي، فموي، تحت الجلد، عضلي، داخل الشرايين، وغيرها)، المستخدم النهائي (المستشفيات والعيادات المتخصصة والمعاهد الأكاديمية والبحثية و أخرى)، قناة التوزيع (صيدليات التجزئة ومخزن الأدوية، صيدلية المستشفى، الصيدلية عبر الإنترنت، أخرى)
|
الجهات الفاعلة في السوق المغطاة
|
سانوفي (فرنسا)، فايزر (الولايات المتحدة)، جلاكسو سميث كلاين (المملكة المتحدة)، نوفارتس (سويسرا)، آبفي (الولايات المتحدة)، سي آر بارد (الولايات المتحدة)، ستراوب ميديكال (الولايات المتحدة)، فلوجو (الولايات المتحدة)، مختبرات نيوجينوميكس (الولايات المتحدة)، شركة مختبرات هيلينا (الولايات المتحدة)، براكو (إيطاليا)، زيتوفيجن (ألمانيا)، أدابتيف بيوتكنولوجيز (الولايات المتحدة)، بيوفيندور راند (الولايات المتحدة)، تريفيترون هيلثكير (الهند)، يوروفينز ديسكفري (فرنسا)، كويست دراجز (الولايات المتحدة)، تاكيدا للأدوية المحدودة (اليابان).
|
نقاط البيانات التي يغطيها التقرير
|
بالإضافة إلى الرؤى حول سيناريوهات السوق مثل القيمة السوقية ومعدل النمو والتجزئة والتغطية الجغرافية واللاعبين الرئيسيين، فإن تقارير السوق التي تم تنظيمها بواسطة Data Bridge Market Research تتضمن أيضًا تحليلًا متعمقًا من الخبراء وعلم الأوبئة للمرضى وتحليل خطوط الأنابيب وتحليل التسعير والإطار التنظيمي.
|
تحليل القطاعات:
يتم تقسيم سوق أدوية الورم النقوي المتعدد في الولايات المتحدة على أساس المرض والعلاج وآلية العمل والعلامات التجارية ونوع الدواء وطريقة الإدارة والمستخدم النهائي وقناة التوزيع.
- على أساس المرض، يتم تقسيم سوق أدوية المايلوما المتعددة في الولايات المتحدة إلى المايلوما المتعددة الخاملة (الخاملة)، والمايلوما المتعددة النشطة (المصحوبة بأعراض)، والورم البلازمي العظمي الانفرادي، والورم البلازمي خارج النخاع، والمايلوما السلسلة الخفيفة، والمايلوما غير الإفرازية وأنواع نادرة من المايلوما.
- على أساس العلاج، يتم تقسيم سوق أدوية الورم النقوي المتعدد في الولايات المتحدة إلى العلاج المناعي والعلاج الكيميائي والكورتيكوستيرويدات وغيرها
- على أساس آلية العمل، يتم تقسيم سوق أدوية الورم النقوي المتعدد في الولايات المتحدة إلى مثبطات البروتوزوم، والأجسام المضادة وحيدة النسيلة ، وعوامل تعديل المناعة، ومثبطات هيستون ديسيتيلاز، وغيرها.
- على أساس العلامات التجارية، يتم تقسيم سوق أدوية الورم النقوي المتعدد في الولايات المتحدة إلى دارزالكس، ونينلارو، وريفليمود، وفيلكاد، وإمبليسيتي، وفينكليكستا، وغيرها.
- على أساس نوع الدواء، يتم تقسيم سوق أدوية الورم النقوي المتعدد في الولايات المتحدة إلى أدوية ذات علامة تجارية وأدوية عامة
- على أساس طريق الإدارة، يتم تقسيم سوق أدوية الورم النقوي المتعدد في الولايات المتحدة إلى وريدية، وفموية، وتحت الجلد، وعضلية، وداخل الشرايين، وغيرها
- على أساس المستخدم النهائي، يتم تقسيم سوق أدوية الورم النقوي المتعدد في الولايات المتحدة إلى مستشفيات وعيادات متخصصة ومعاهد أكاديمية وبحثية وغيرها
- على أساس قناة التوزيع، يتم تقسيم سوق أدوية الورم النقوي المتعدد في الولايات المتحدة إلى صيدليات التجزئة ومتاجر الأدوية، وصيدليات المستشفيات، والصيدليات عبر الإنترنت، وغيرها
اللاعبون الرئيسيون
تعترف شركة Data Bridge Market Research بالشركات التالية باعتبارها اللاعبين الرئيسيين في سوق أدوية الورم النقوي المتعدد في الولايات المتحدة: Sanofi (فرنسا)، Pfizer Inc. (الولايات المتحدة)، GSK plc (المملكة المتحدة)، Novartis AG (سويسرا)، AbbVie Inc. (الولايات المتحدة)، CR Bard (الولايات المتحدة)، Straub Medical AG (الولايات المتحدة)، FlowJo LLC (الولايات المتحدة)، NeoGenomics Laboratories (الولايات المتحدة)، Helena Laboratories Corporation (الولايات المتحدة).
تطورات السوق
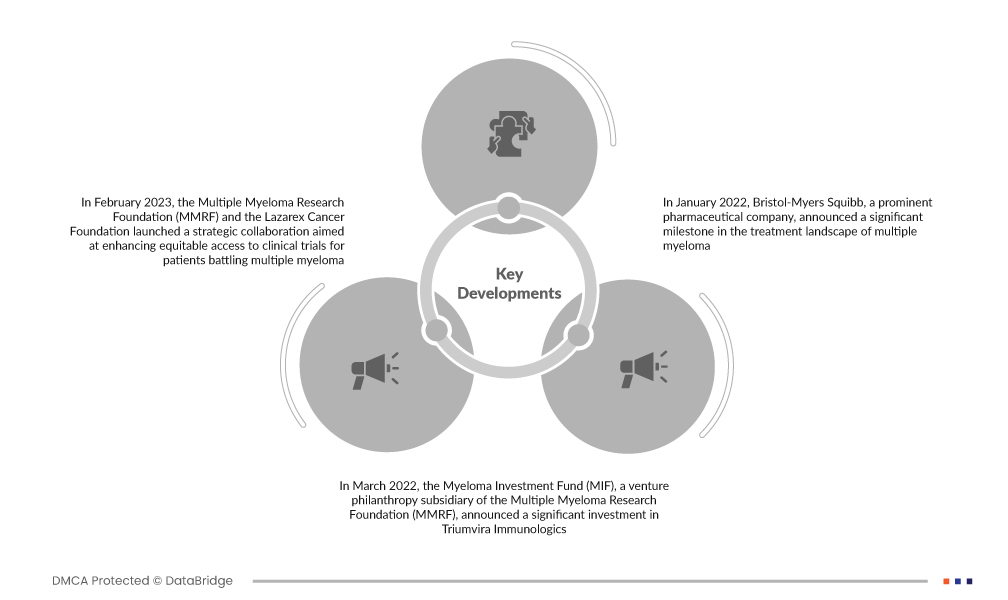
- في فبراير 2023، أعلنت مؤسسة أبحاث الورم النقوي المتعدد (MMRF) ومؤسسة لازاريكس للسرطان عن تعاون استراتيجي لتعزيز الوصول العادل إلى التجارب السريرية لمرضى الورم النقوي المتعدد. وقد تعاونت مؤسسة لازاريكس للسرطان، وهي منظمة غير ربحية تُعنى بتحسين وصول مرضى السرطان إلى التجارب السريرية، مع مؤسسة MMRF لتلبية الحاجة إلى زيادة الشمولية في التجارب السريرية. ومن المتوقع أن يُحدث هذا التعاون تأثيرًا عميقًا على الطلب على أدوية وعلاجات الورم النقوي المتعدد، مما يُحفز نمو السوق.
- في مارس 2022، أعلن صندوق استثمار المايلوما (MIF)، وهو صندوق خيري استثماري تابع لمؤسسة أبحاث المايلوما المتعددة (MMRF)، عن استثمار كبير في شركة تريومفيرا إيمونولوجيكس. تريومفيرا، شركة تكنولوجيا حيوية في المرحلة السريرية، مقرها أوستن، تكساس، متخصصة في تطوير علاجات مناعية جديدة ومستهدفة بالخلايا التائية الذاتية والخيفية. تستفيد هذه العلاجات المبتكرة من الخصائص البيولوجية الطبيعية للخلايا التائية لمكافحة الأورام الصلبة وسرطانات الدم. يؤكد هذا الاستثمار الاستراتيجي من MIF على الاهتمام المتزايد والدعم المالي لتطوير علاجات مناعية رائدة لعلاج المايلوما المتعددة.
- في يناير 2022، أعلنت شركة بريستول-مايرز سكويب، وهي شركة أدوية رائدة، عن إنجازٍ هام في مجال علاج الورم النقوي المتعدد. وافقت وزارة الصحة والعمل والرعاية الاجتماعية اليابانية على دواء أبيكما (idecabtagene vicleucel)، وهو علاج مناعي للخلايا التائية مستقبلات المستضد الكيمري (CAR) موجه لمستضد نضج الخلايا البائية (BCMA). وتتعلق هذه الموافقة تحديدًا بعلاج المرضى البالغين الذين يعانون من الورم النقوي المتعدد المتكرر أو المقاوم للعلاج (R/R). ويؤكد الحصول على الموافقة التنظيمية على أبيكما على التطور المستمر للعلاجات المبتكرة في مواجهة التحديات المعقدة المرتبطة بالورم النقوي المتعدد.
لمزيد من المعلومات التفصيلية حول تقرير سوق أدوية المايلوما المتعددة في الولايات المتحدة، انقر هنا - https://www.databridgemarketresearch.com/reports/us-multiple-myeloma-drugs-market