Asia Pacific Hereditary Cancer Testing Market
Market Size in USD Billion
CAGR :
% 
 USD
1.28 Billion
USD
3.65 Billion
2025
2033
USD
1.28 Billion
USD
3.65 Billion
2025
2033
| 2026 –2033 | |
| USD 1.28 Billion | |
| USD 3.65 Billion | |
|
|
|
|
Asia-Pacific Hereditary Cancer Testing Market Size
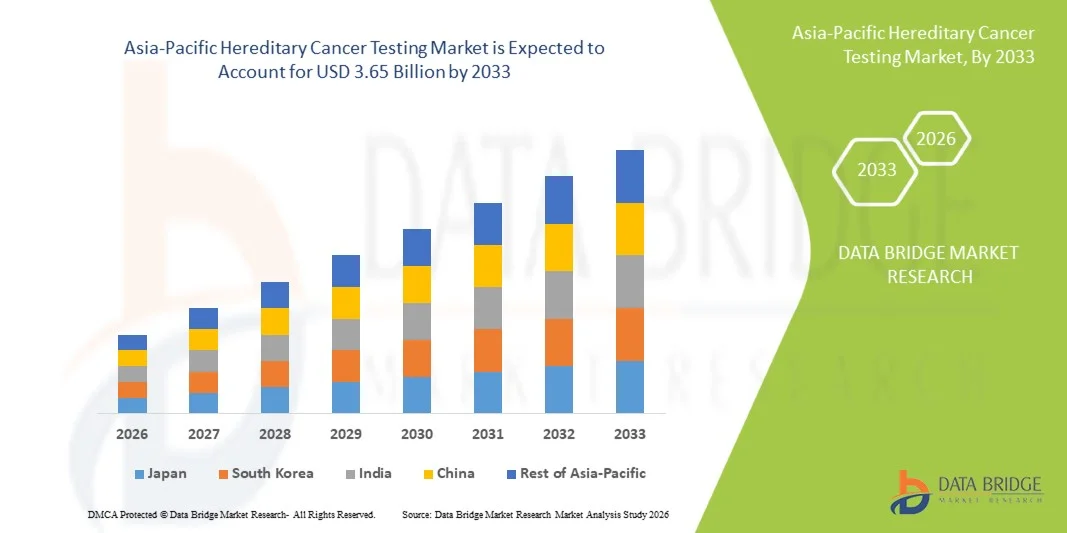
- The Asia-Pacific hereditary cancer testing market size was valued at USD 1.28 billion in 2025 and is expected to reach USD 3.65 billion by 2033, at a CAGR of 14.0% during the forecast period
- The market growth is largely fueled by the increasing prevalence of genetic disorders and rising awareness regarding early cancer detection, leading to greater adoption of advanced genetic testing solutions across the region
- Furthermore, improving healthcare infrastructure, growing investments in precision medicine, and rising demand for personalized treatment approaches are establishing hereditary cancer testing as a critical component of modern oncology care. These converging factors are accelerating the uptake of genetic testing services, thereby significantly boosting the market’s growth
Asia-Pacific Hereditary Cancer Testing Market Analysis
- Hereditary cancer testing, involving genetic screening to identify inherited mutations linked to cancers such as breast, ovarian, and colorectal cancer, is becoming an essential component of modern oncology care across Asia-Pacific due to its role in early detection, risk assessment, and personalized treatment planning
- The escalating demand for hereditary cancer testing is primarily fueled by the rising incidence of cancer, increasing awareness about genetic predisposition, and a growing emphasis on preventive healthcare and precision medicine
- China dominated the hereditary cancer testing market with the largest revenue share of 38.6% in 2025, characterized by rapidly expanding healthcare infrastructure, increasing adoption of advanced genetic testing technologies, and strong government support for cancer screening initiatives, with the country witnessing substantial growth driven by rising healthcare expenditure and the presence of both domestic and international diagnostic companies
- India is expected to be the fastest growing country in the hereditary cancer testing market during the forecast period due to a large patient population, increasing awareness of early cancer detection, and growing investments in genomics and diagnostic services
- Sequencing segment dominated the hereditary cancer testing market with a market share of 41.8% in 2025, driven by its high accuracy, ability to detect a wide range of genetic mutations, and increasing adoption in both clinical diagnostics and research applications
Report Scope and Asia-Pacific Hereditary Cancer Testing Market Segmentation
|
Attributes |
Asia-Pacific Hereditary Cancer Testing Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
Asia-Pacific
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework |
Asia-Pacific Hereditary Cancer Testing Market Trends
“Rising Adoption of AI-Driven Genomic Interpretation”
- A significant and accelerating trend in the Asia-Pacific hereditary cancer testing market is the growing integration of artificial intelligence (AI) and advanced bioinformatics tools with genetic testing platforms to enhance diagnostic accuracy and efficiency. This fusion of technologies is significantly improving the interpretation of complex genetic data and enabling faster clinical decision-making
- For instance, AI-powered genomic platforms are increasingly being utilized to analyze large-scale sequencing data and identify clinically relevant mutations associated with hereditary cancers, thereby supporting oncologists in personalized treatment planning. Similarly, advanced analytics tools are being integrated with sequencing technologies to streamline variant classification and reporting
- AI integration in hereditary cancer testing enables features such as predictive risk modeling, automated variant interpretation, and improved identification of rare mutations. For instance, some diagnostic providers are leveraging machine learning algorithms to continuously refine mutation databases and deliver more precise risk assessments. Furthermore, these capabilities offer healthcare professionals enhanced insights, allowing more accurate and timely intervention strategies
- The seamless integration of genetic testing with digital health platforms and electronic medical records facilitates centralized management of patient data and improves coordination across healthcare systems. Through a unified interface, clinicians can access genetic insights alongside clinical history, enabling a more comprehensive and personalized approach to cancer care
- This trend towards more intelligent, data-driven, and integrated diagnostic solutions is fundamentally reshaping expectations for cancer risk assessment. Consequently, companies are developing advanced hereditary cancer testing solutions with features such as automated reporting, cloud-based analytics, and real-time data sharing capabilities
- The demand for hereditary cancer testing solutions that offer advanced AI-driven insights and seamless digital integration is growing rapidly across hospitals, diagnostic laboratories, and research institutions, as healthcare systems increasingly prioritize precision medicine and early detection strategies
- Rising adoption of direct-to-consumer genetic testing services is further expanding market reach by enabling individuals to access hereditary cancer risk information conveniently, thereby boosting early detection and preventive care initiatives
Asia-Pacific Hereditary Cancer Testing Market Dynamics
Driver
“Growing Demand Due to Rising Cancer Burden and Precision Medicine Adoption”
- The increasing prevalence of hereditary cancers across countries such as China and India, coupled with the expanding adoption of precision medicine, is a significant driver for the heightened demand for hereditary cancer testing
- For instance, in March 2025, several regional healthcare institutions expanded genetic screening programs aimed at early cancer detection, integrating next-generation sequencing technologies into routine diagnostics. Such strategies by key stakeholders are expected to drive the hereditary cancer testing market growth in the forecast period
- As awareness regarding genetic predisposition to cancer increases, patients and healthcare providers are increasingly opting for early screening and preventive diagnostics, offering a compelling shift toward proactive healthcare management
- Furthermore, the growing availability of advanced sequencing technologies and supportive government initiatives promoting cancer screening programs are making hereditary cancer testing more accessible and widely adopted across the region
- The ability to identify high-risk individuals, guide targeted therapies, and support family-based risk assessment are key factors propelling the adoption of hereditary cancer testing in both clinical and research settings. The increasing focus on personalized medicine and expanding insurance coverage for genetic testing further contribute to market growth
- Increasing investments by public and private entities in genomics research and infrastructure development are strengthening testing capabilities and accelerating market expansion across Asia-Pacific
- The rising presence of regional diagnostic laboratories and international market players is enhancing competition and improving service availability, thereby driving wider adoption of hereditary cancer testing
Restraint/Challenge
“High Cost of Testing and Limited Genetic Awareness”
- Concerns surrounding the high cost of advanced genetic testing procedures, including sequencing and multi-gene panel tests, pose a significant challenge to broader market penetration, particularly in price-sensitive regions across Asia-Pacific
- For instance, limited reimbursement policies and uneven access to specialized diagnostic facilities in developing areas have made some patients hesitant to opt for hereditary cancer testing despite potential benefits
- Addressing these cost-related barriers through subsidized programs, public-private partnerships, and technological advancements aimed at reducing testing costs is crucial for expanding adoption. Additionally, limited awareness about genetic testing and hereditary cancer risks among the general population can further restrict market growth
- While awareness initiatives are gradually increasing, misconceptions and lack of education regarding genetic screening still hinder widespread adoption, especially in rural and underserved areas
- Overcoming these challenges through cost optimization, improved healthcare infrastructure, and large-scale awareness campaigns will be vital for sustained market growth
- Regulatory variability across different Asia-Pacific countries can create complexities in test approval, standardization, and commercialization, thereby slowing market expansion
- Data privacy concerns related to genetic information storage and sharing may limit consumer willingness to undergo hereditary cancer testing, posing an additional barrier to adoption
Asia-Pacific Hereditary Cancer Testing Market Scope
The market is segmented on the basis of test type, diagnosis type, technology, disease type, end user, and distribution channel.
- By Test Type
On the basis of test type, the Asia-Pacific hereditary cancer testing market is segmented into multi panel set and single site genetic test. The multi panel set segment dominated the market with the largest market revenue share in 2025, driven by its ability to analyze multiple genes simultaneously, enabling comprehensive risk assessment for various hereditary cancers. Healthcare providers prefer multi panel testing due to its efficiency in identifying a broad spectrum of mutations in a single test, reducing time and cost compared to multiple individual tests. Additionally, advancements in sequencing technologies have improved the accuracy and accessibility of multi gene panels, further supporting their adoption. The growing emphasis on precision medicine and personalized treatment strategies is also contributing to the widespread use of multi panel tests. Increasing awareness among patients regarding early detection and preventive screening is further strengthening the dominance of this segment.
The single site genetic test segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by its cost-effectiveness and targeted approach in identifying specific known mutations within families. These tests are particularly useful for individuals with a confirmed family history of a specific genetic mutation, enabling quick and accurate diagnosis. The rising adoption of targeted genetic screening in developing countries is also supporting segment growth. Furthermore, improvements in diagnostic infrastructure and increased accessibility of genetic counseling services are enhancing the uptake of single site testing. The demand for faster turnaround time and affordability is expected to further drive this segment during the forecast period.
- By Diagnosis Type
On the basis of diagnosis type, the market is segmented into biopsy, imaging, and lab tests. The lab tests segment dominated the market with the largest revenue share in 2025, driven by the central role of genetic and molecular testing in confirming hereditary cancer risks. Lab-based diagnostics, including blood and saliva tests, are widely utilized due to their accuracy, reliability, and non-invasive nature. The increasing integration of advanced molecular diagnostics and sequencing technologies into laboratory workflows is further enhancing their effectiveness. Additionally, the availability of specialized diagnostic laboratories across major Asia-Pacific countries is supporting the widespread adoption of lab tests. Growing investments in healthcare infrastructure and diagnostic capabilities are also contributing to segment dominance. The expanding focus on early detection and preventive healthcare is further reinforcing the importance of lab-based testing.
The imaging segment is expected to witness the fastest growth rate from 2026 to 2033, driven by its increasing role in complementing genetic testing for early cancer detection and monitoring. Advanced imaging technologies are being integrated with genetic insights to provide a more comprehensive diagnostic approach. The rising availability of high-resolution imaging systems and improved accessibility in emerging economies are supporting segment growth. Furthermore, increasing awareness about routine screening and early diagnosis is boosting the adoption of imaging techniques. The combination of imaging with genetic testing enhances clinical decision-making and treatment planning. This integrated approach is expected to drive the growth of the imaging segment in the coming years.
- By Technology
On the basis of technology, the market is segmented into sequencing, polymerase chain reaction (PCR), and microarray. The sequencing segment dominated the market with the largest revenue of 41.8% share in 2025, driven by its high accuracy and ability to analyze multiple genes and mutations simultaneously. Next-generation sequencing technologies have revolutionized hereditary cancer testing by enabling comprehensive genomic analysis at a relatively lower cost. The increasing adoption of sequencing in clinical diagnostics and research applications is further supporting its dominance. Additionally, advancements in bioinformatics tools are enhancing data interpretation and reliability. The growing demand for precision medicine and personalized treatment approaches is also contributing to the widespread use of sequencing technologies. Continuous technological innovations are expected to further strengthen this segment.
The polymerase chain reaction (PCR) segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by its cost-effectiveness, speed, and widespread availability in diagnostic laboratories. PCR-based tests are commonly used for targeted mutation detection and validation, making them an essential tool in hereditary cancer testing. The increasing demand for rapid diagnostic solutions and point-of-care testing is supporting the growth of this segment. Furthermore, advancements in PCR technologies, such as real-time and digital PCR, are improving sensitivity and accuracy. The ease of implementation and lower operational costs make PCR a preferred choice in resource-limited settings. These factors are expected to drive the rapid expansion of the PCR segment.
- By Disease Type
On the basis of disease type, the market is segmented into hereditary breast & ovarian cancer syndrome, Cowden syndrome, Lynch syndrome, hereditary leukemia and hematologic malignancies syndromes, familial adenomatous polyposis (FAP), Li-Fraumeni syndrome, von Hippel-Lindau disease, and multiple endocrine neoplasias (MEN) syndromes. The hereditary breast & ovarian cancer syndrome segment dominated the market with the largest revenue share in 2025, driven by the high prevalence of BRCA1 and BRCA2 gene mutations and increasing awareness about breast and ovarian cancer risks. Screening programs and public health initiatives across Asia-Pacific countries are significantly contributing to the adoption of testing for this condition. Additionally, the availability of targeted therapies and preventive measures is encouraging individuals to undergo genetic testing. The rising focus on women’s health and early cancer detection is further strengthening this segment. Increasing government support and awareness campaigns are also playing a key role in driving demand.
The Lynch syndrome segment is expected to witness the fastest growth rate from 2026 to 2033, fueled by growing awareness regarding hereditary colorectal cancer and advancements in diagnostic technologies. Lynch syndrome is one of the most common hereditary cancer syndromes, and increasing screening initiatives are supporting its early detection. The rising adoption of genetic counseling and family-based testing is also contributing to segment growth. Furthermore, improvements in healthcare infrastructure and diagnostic capabilities are enhancing access to testing services. The integration of genetic testing into routine cancer screening programs is further driving demand. These factors are expected to accelerate the growth of the Lynch syndrome segment.
- By End User
On the basis of end user, the market is segmented into hospitals, clinics, laboratories, radiology centers, diagnostic centers, and others. The hospitals segment dominated the market with the largest revenue share in 2025, driven by the availability of advanced diagnostic facilities and multidisciplinary care teams. Hospitals serve as primary centers for cancer diagnosis, treatment, and genetic counseling, making them key contributors to hereditary cancer testing. The integration of genetic testing into oncology departments is further supporting segment growth. Additionally, increasing patient inflow and access to comprehensive healthcare services are strengthening the dominance of hospitals. The presence of skilled healthcare professionals and advanced technologies also enhances diagnostic accuracy. Growing investments in hospital infrastructure across Asia-Pacific are further contributing to this segment.
The diagnostic centers segment is anticipated to witness the fastest growth rate from 2026 to 2033, fueled by the increasing demand for specialized and cost-effective diagnostic services. Diagnostic centers offer focused genetic testing services with faster turnaround times, making them attractive to patients and healthcare providers. The expansion of private diagnostic chains and increased accessibility in urban and semi-urban areas are supporting segment growth. Furthermore, advancements in diagnostic technologies and automation are improving efficiency and accuracy. The rising trend of outsourcing diagnostic services is also contributing to the growth of this segment. These factors are expected to drive rapid expansion in the coming years.
- By Distribution Channel
On the basis of distribution channel, the market is segmented into direct tender and retail sales. The direct tender segment dominated the market with the largest revenue share in 2025, driven by bulk procurement by hospitals, government organizations, and large diagnostic laboratories. Direct tenders enable cost-effective acquisition of testing kits and services, particularly in public healthcare systems. The increasing involvement of government initiatives in cancer screening programs is further supporting this segment. Additionally, partnerships between diagnostic companies and healthcare institutions are enhancing distribution efficiency. The growing focus on large-scale screening and early detection programs is also contributing to the dominance of direct tenders.
The retail sales segment is expected to witness the fastest growth rate from 2026 to 2033, fueled by the increasing availability of genetic testing kits through pharmacies and online platforms. The rising popularity of direct-to-consumer testing and growing awareness among individuals are driving demand in this segment. Retail channels offer convenience and accessibility, allowing individuals to access testing services without extensive clinical procedures. Furthermore, advancements in at-home testing solutions are enhancing user experience and adoption. The expansion of e-commerce platforms and digital health services is also supporting segment growth. These factors are expected to accelerate the expansion of the retail sales segment.
Asia-Pacific Hereditary Cancer Testing Market Regional Analysis
- China dominated the hereditary cancer testing market with the largest revenue share of 38.6% in 2025, characterized by rapidly expanding healthcare infrastructure, increasing adoption of advanced genetic testing technologies, and strong government support for cancer screening initiatives
- Consumers and healthcare providers in the region highly value the accuracy, early risk identification, and personalized treatment planning offered by hereditary cancer testing, along with its integration with advanced diagnostic technologies such as sequencing and molecular diagnostics
- This widespread adoption is further supported by improving healthcare infrastructure, rising healthcare expenditure, and a growing focus on precision medicine, establishing hereditary cancer testing as a critical solution for early diagnosis and preventive oncology care across both urban and emerging healthcare settings
The China Hereditary Cancer Testing Market Insight
The China hereditary cancer testing market captured the largest revenue share of 38.6% in 2025 within Asia-Pacific, fueled by the rising burden of cancer and expanding adoption of advanced genetic testing technologies. Healthcare providers are increasingly prioritizing early detection and personalized treatment through comprehensive genetic screening solutions. The growing preference for precision medicine, combined with strong government support for cancer screening programs and investments in genomics, further propels the market. Moreover, the increasing integration of sequencing technologies and AI-driven diagnostics is significantly contributing to the market's expansion.
Japan Hereditary Cancer Testing Market Insight
The Japan hereditary cancer testing market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by the country’s advanced healthcare system and strong focus on early cancer diagnosis. Additionally, increasing awareness regarding hereditary cancer risks is encouraging individuals to opt for genetic screening services. Japan’s adoption of advanced sequencing technologies, alongside its well-established healthcare infrastructure, is expected to continue to stimulate market growth. The integration of hereditary cancer testing with precision medicine initiatives is also playing a key role in expanding the market.
South Korea Hereditary Cancer Testing Market Insight
The South Korea hereditary cancer testing market is gaining traction due to the country’s advanced healthcare infrastructure and strong focus on precision medicine and early cancer detection. Increasing government support for genomic research and national screening programs is encouraging the adoption of hereditary cancer testing. The rising awareness among the population regarding genetic risk factors and preventive healthcare is further driving demand. Additionally, the integration of advanced sequencing technologies and digital health platforms is enhancing diagnostic accuracy and accessibility. The presence of leading biotechnology companies and continuous innovation in molecular diagnostics are expected to further accelerate market growth in South Korea.
India Hereditary Cancer Testing Market Insight
The India hereditary cancer testing market accounted for a significant market revenue share in Asia-Pacific in 2025, attributed to the country’s large population base, rising cancer prevalence, and improving access to diagnostic services. India is emerging as a key market for genetic testing, with increasing adoption across hospitals, diagnostic centers, and research institutions. The push towards affordable healthcare solutions and expanding awareness regarding early cancer detection are key factors propelling the market. Additionally, the presence of domestic diagnostic companies and growing investments in genomics are supporting the expansion of hereditary cancer testing in India.
Asia-Pacific Hereditary Cancer Testing Market Share
The Asia-Pacific Hereditary Cancer Testing industry is primarily led by well-established companies, including:
- F. Hoffmann-La Roche Ltd (Switzerland)
- QIAGEN (Netherlands)
- Illumina, Inc. (U.S.)
- Thermo Fisher Scientific Inc. (U.S.)
- Abbott (U.S.)
- Danaher (U.S.)
- Bio-Rad Laboratories, Inc. (U.S.)
- Eurofins Scientific (Luxembourg)
- Myriad Genetics, Inc. (U.S.)
- Invitae Corporation (U.S.)
- Agilent Technologies, Inc. (U.S.)
- Quest Diagnostics Incorporated (U.S.)
- Fulgent Genetics, Inc. (U.S.)
- Natera, Inc. (U.S.)
- Exact Sciences Corporation (U.S.)
- Guardant Health, Inc. (U.S.)
- BGI Genomics Co., Ltd. (China)
- Macrogen, Inc. (South Korea)
- MedGenome Labs Ltd. (India)
- Strand Life Sciences Pvt Ltd (India)
What are the Recent Developments in Asia-Pacific Hereditary Cancer Testing Market?
- In December 2025, Singapore’s national precision health program (PRECISE) revealed plans to expand its genetic testing scope to include hereditary cancers, aiming to integrate genomic screening into population-level healthcare and improve early detection strategies
- In March 2025, Gene Solutions, a leading Asia-based biotech company, announced that its SPOT-MAS multi-cancer blood test successfully completed large-scale clinical validation, becoming one of the first clinically validated multi-cancer early detection tests in Asia. The non-invasive test enables early identification of multiple cancers, including hereditary-linked cancers, marking a major advancement in accessible genomic screening across the region
- In August 2024, Myriad Genetics partnered with Eurobio Scientific to launch the EndoPredict prognostic genetic test, designed to assess breast cancer recurrence risk and support personalized treatment decisions. This development strengthens the adoption of hereditary cancer-related genomic testing in clinical oncology by enabling more precise risk stratification and therapy planning
- In April 2024, a large-scale multi-country study involving Singapore and Malaysia identified improved risk estimates for BRCA1 and BRCA2 mutation carriers in Asian populations, supporting more accurate hereditary breast and ovarian cancer testing and clinical management
- In March 2024, a study published in the Journal of Genetic Counseling highlighted challenges and behavioral patterns in cascade genetic testing among Asian families, emphasizing the need to improve adoption of hereditary cancer screening across the region
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.