Europe Blood Culture Test Market
Market Size in USD Billion
CAGR :
% 
 USD
3.80 Billion
USD
6.38 Billion
2024
2032
USD
3.80 Billion
USD
6.38 Billion
2024
2032
| 2025 –2032 | |
| USD 3.80 Billion | |
| USD 6.38 Billion | |
|
|
|
|
Blood Culture Test Market Size
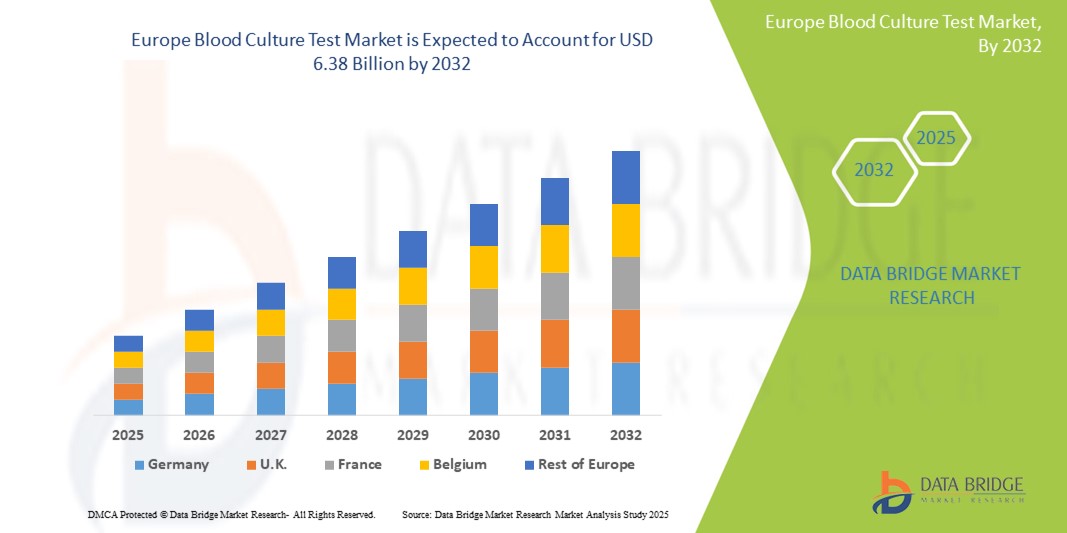
- The Europe Blood Culture Test market size was valued at USD 3.8 billion in 2024 and is projected to reach USD 6.38 billion by 2032, exhibiting a CAGR of 6.7% during the forecast period of 2025-2032.
- Furthermore, the increasing incidence of bloodstream infections and the growing emphasis on early and accurate diagnosis are establishing blood culture tests as essential tools in clinical practice.
- These converging factors are accelerating the adoption of blood culture tests, thereby significantly boosting the industry's growth in the region.
Blood Culture Test Market Analysis
- Blood culture tests, crucial for identifying bloodstream infections and guiding appropriate antimicrobial therapy, are increasingly vital components of modern diagnostic practices in both hospital and laboratory settings due to their ability to improve patient outcomes and reduce mortality.
- The escalating demand for blood culture tests is primarily fueled by the rising prevalence of infectious diseases, the growing awareness of antibiotic resistance necessitating accurate pathogen identification, and technological advancements leading to faster and more sensitive testing methods.
- U.K. dominates the Blood Culture Test market with the largest revenue share of 56.71 % in 2025, due to a well-established healthcare system, high healthcare expenditure, and a large volume of diagnostic testing.
- The Automated blood culture systems segment is expected to dominate the Blood Culture Test market with a market share of 48.34% in 2025, driven by the recurring need for culture media and associated disposables in routine testing procedures.
Report Scope and Blood Culture Test Market Segmentation
|
Attributes |
Blood Culture Test Key Market Insights |
|
Segments Covered |
|
|
Countries Covered |
Europe
|
|
Key Market Players |
|
|
Market Opportunities |
|
|
Value Added Data Infosets |
In addition to the insights on market scenarios such as market value, growth rate, segmentation, geographical coverage, and major players, the market reports curated by the Data Bridge Market Research also include in-depth expert analysis, pricing analysis, brand share analysis, consumer survey, demography analysis, supply chain analysis, value chain analysis, raw material/consumables overview, vendor selection criteria, PESTLE Analysis, Porter Analysis, and regulatory framework. |
Blood Culture Test Market Trends
“Increasing Adoption of Rapid Diagnostic Tests and Automation”
- A significant trend in the Europe blood culture test market is the growing adoption of rapid diagnostic tests (RDTs) and automation technologies. These advancements enable faster turnaround times for results, which is crucial for timely initiation of appropriate antimicrobial therapy and improved patient outcomes.
- For instance, the development of molecular diagnostic techniques that can directly detect pathogens and resistance markers from blood samples is gaining traction, reducing the reliance on traditional culture-based methods that require longer incubation periods.
- Furthermore, the increasing integration of automation in blood culture workflows, from sample loading to result interpretation, is enhancing efficiency, reducing the risk of human error, and improving the overall throughput of diagnostic laboratories.
- The development of user-friendly and compact automated blood culture systems is also facilitating their adoption in smaller hospitals and clinics.
Blood Culture Test Market Dynamics
Driver
“Rising Prevalence of Bloodstream Infections and Sepsis”
- The increasing incidence of bloodstream infections (BSIs) and sepsis across Europe is a significant driver for the heightened demand for blood culture tests.
- For instance, factors such as the aging population, the growing number of patients with compromised immune systems due to chronic diseases or medical treatments, and the rise of antibiotic-resistant bacteria are contributing to a higher burden of BSIs. Sepsis, a life-threatening condition resulting from the body's overwhelming response to an infection, often requires blood culture testing for diagnosis and management.
- As the prevalence of these conditions continues to rise, the demand for rapid and accurate blood culture testing is expected to increase substantially.
- Furthermore, increasing awareness among healthcare professionals about the critical role of early diagnosis in improving sepsis outcomes is driving greater utilization of blood culture tests.
Restraint/Challenge
“Challenges Related to Contamination and False Negatives”
- Despite advancements in blood culture testing, challenges related to contamination and the occurrence of false-negative results can restrain market growth.
- For instance, contamination during blood collection or processing can lead to false-positive results, necessitating additional testing and potentially delaying appropriate treatment.
- Additionally, factors such as prior antibiotic administration or low bacterial load in the bloodstream can result in false-negative results, potentially leading to underdiagnosis and delayed treatment.
- Addressing these challenges through improved blood collection techniques, enhanced quality control measures in laboratories, and the development of more sensitive detection methods will be crucial for maximizing the clinical utility of blood culture tests.
- While ongoing research and technological advancements are focused on minimizing these issues, they remain significant considerations for market growth.
Blood Culture Test Market Scope
The market is segmented on the basis of product type, method, application, and end-user.
By Product Type
On the basis of product type, the blood culture test market can be segmented into automated blood culture systems, culture media, reagents & consumables. The automated blood culture systems segment is anticipated to hold the market share of 48.34% in 2025 due to their efficiency and high throughput capabilities.
The culture media segment is anticipated to hold the market share of 38.45% in 2025 due to the continuous development of specialized media for different types of microorganisms.
By Method
On the basis of method, the market is segmented into conventional/manual blood culture and automated blood culture. The conventional/manual blood culture media segment is anticipated to hold the market share in 2025 due to the advantages of faster turnaround times and reduced contamination risk.
However, the automated blood culture segment is gaining rapid traction due to its ability to deliver faster and more accurate detection of bloodstream infections. The increasing demand for timely diagnosis of sepsis and other critical infections is driving adoption in hospitals and diagnostic laboratories. Technological advancements in automated systems are improving sensitivity, reducing contamination, and streamlining workflow.
By Application
On the basis of application, the market is segmented into bacteremia, septicemia, fungemia, and mycobacterial detection. The bacteremia segment is anticipated to hold the market share in 2025 due to the increasing incidence of severe bloodstream infections.
However, the septicemia segment is gaining rapid traction due to the increasing global incidence of bloodstream infections and sepsis-related complications. The urgent need for early and accurate pathogen detection is driving the use of blood culture tests in septicemia diagnosis. Advancements in culture media and automated systems are enhancing detection speed and reliability.
By End-User
On the basis of end-user, the market is segmented into hospitals, diagnostic laboratories, reference laboratories, and others. The hospitals segment is anticipated to hold the market share in 2025 due to the increasing outsourcing of diagnostic testing.
However, the diagnostic laboratories segment is gaining rapid traction due to their ability to offer high-throughput, accurate, and cost-effective blood culture testing. The growing demand for centralized testing and timely diagnosis of bloodstream infections is driving utilization across healthcare networks.
Blood Culture Test Market Regional Analysis
- The U.K. dominates the Blood Culture Test market with the largest revenue share of 56.71% in 2025, driven by a high prevalence of infectious diseases, advanced healthcare infrastructure, and the presence of major diagnostic companies.
- This significant market share is further supported by favorable reimbursement policies and a high adoption rate of advanced diagnostic technologies.
Germany Blood Culture Test Market Insight
The Germany Blood Culture Test Market is anticipated to grow at a noteworthy CAGR during the forecast period, driven by a well-established healthcare system and increasing awareness of the importance of timely diagnosis of infectious diseases. Government initiatives to improve diagnostic capabilities and rising adoption of advanced technologies are contributing to market expansion.
France Blood Culture Test Market Insight
The France Blood Culture Test Market is expected to expand at a considerable CAGR during the forecast period, due to increasing healthcare expenditure and a growing focus on improving diagnostic infrastructure. The rising incidence of infectious diseases and the need for better diagnostic tools are driving demand for blood culture tests.
Blood Culture Test Market Share
The Blood Culture Test industry is primarily led by well-established companies, including:
- Becton, Dickinson and Company (BD) (U.S.)
- Accelerate Diagnostics (U.S.)
- Biogenix Inc. Pvt. Ltd. (India)
- Copan (Italy)
- bioMérieux SA (France)
- Thermo Fisher Scientific Inc. (U.S.)
- Abbott Laboratories (U.S.)
- Bruker Corporation (U.S.)
- F. Hoffmann-La Roche Ltd. (Switzerland)
- Danaher Corporation (U.S.)
- Luminex Corporation (U.S.)
- T2 Biosystems Inc. (U.S.)
- DiaSorin S.p.A. (Italy)
- Grifols S.A. (Spain)
- Radiometer Medical ApS (Denmark)
- Synlab Group (Germany)
- Cepheid Inc. (U.S.)
- IRIDICA (formerly by Abbott Molecular) (U.S.)
Latest Developments in Europe Blood Culture Test Market
- In April 2025, DiaSorin revealed that it had obtained FDA 510(k) clearance for its Liaison Plex Gram-Negative Blood Culture Assay, the second of three multiplex molecular panels intended for pathogen identification in blood cultures on the Liaison Plex platform. This assay detects 27 pathogens, including 19 gram-negative bacteria and eight key resistance gene targets, such as Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, among others.
- In March 2025, Accelerate Diagnostics submitted its WAVE System and gram-negative test kit for FDA 510(k) clearance. The system delivers rapid antimicrobial susceptibility testing (AST) results in ~4.5 hours directly from blood cultures, enabling same-shift targeted therapy. This addresses sepsis (49 million annual cases, 11 million deaths) and antimicrobial resistance (1.32 million annual deaths), potentially reducing healthcare costs ($62 billion/year U.S. sepsis burden). The WAVE complements existing FDA-cleared solutions like the Pheno system (7-hour AST) and Arc system (automated sample prep), enhancing lab efficiency and patient outcomes.
- In February 2025, Biogenix Inc. Pvt. Ltd. introduced the Bugz32, a fully automated blood culture instrument with a 32-position capacity, aimed at microbiology labs. Designed to deliver fast, accurate, and reliable results, the Bugz32 is part of the company's broader portfolio of diagnostic instruments and kits.
- In November 2024, Copan introduced HEMO, an automated system created to optimize the processing of positive blood cultures by automating sample transfer and tracking.
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.