Europe Crispr Gene Detection And Diagnostic Market
Market Size in USD million
CAGR :
% 
 USD
700.00 million
USD
2,631.20 million
2022
2030
USD
700.00 million
USD
2,631.20 million
2022
2030
| 2023 –2030 | |
| USD 700.00 million | |
| USD 2,631.20 million | |
|
|
|
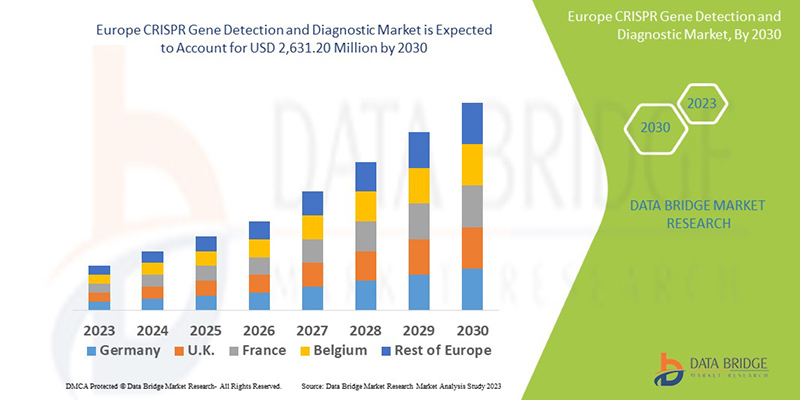
Europe CRISPR Gene Detection and Diagnostic Market Analysis and Size
The demand for CRISPR gene editing will rise because of factors such as cancer, aging, and genetic defects. Several pharmaceutical companies invest heavily in R&D to manufacture new products, with an increase in gene and cell therapy agents. The efficacy of CRISPR-Cas9 for genome editing has led to major investments in genome engineering.
Data Bridge Market Research analyses a growth rate in the CRISPR gene detection and diagnostic market in the forecast period 2023-2030. The expected CAGR of the CRISPR gene detection and diagnostic market is around 18% in the forecast period. The market was valued at USD 700 million in 2022 and will grow to USD 2,631.20 million by 2030. In addition to the market insights such as market value, growth rate, market segments, geographical coverage, market players, and market scenario, the market report curated by the Data Bridge Market Research team also includes in-depth expert analysis, patient epidemiology, pipeline analysis, pricing analysis, and regulatory framework.
Europe CRISPR Gene Detection and Diagnostic Market Scope and Segmentation
|
Report Metric |
Details |
|
Forecast Period |
2023 to 2030 |
|
Base Year |
2022 |
|
Historic Years |
2021 (Customizable to 2015 - 2020) |
|
Quantitative Units |
Revenue in USD Million, Volumes in Units, Pricing in USD |
|
Segments Covered |
Class (Class 1- Multiple Effector Proteins and Class 2 -Single CrRNA-Binding Protein), Products & Services (Products and Services), Application (Biomedical Diagnostics, Genome Engineering, Drug Discovery, Agricultural Applications, and Others), Workflow (Sample Preparation, Pre-Amplification, CrRNA, Cas Enzymes, and Sensing), End User (Hospitals, Diagnostic Centers, Biotechnology Companies, Academic and Research Institutes and Others), Distribution Channel (Direct Tender, Retail Sales) |
|
Countries Covered |
Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe |
|
Market Players Covered |
GenScript (China), OriGene Technologies, Inc (U.S.)., Horizon Discovery Ltd (U.K.), Agilent Technologies Inc. (U.S.)., Merck & Co., Inc. (U.S.), Danaher (U.S.), Thermo Fisher Scientific (U.S.), Takara Bio Inc. (Japan), CRISPR Therapeutics (Switzerland), GeneCopoeia, Inc (U.S.) |
|
Market Opportunities |
|
Market Definition
CRISPR/Cas9 works by precisely cutting DNA and then letting natural DNA repair processes repair. CRISPR-Cas 9 systems have enlarged the scope of diagnostics and services in gene and cell therapies. It is used for several conditions, which include high cholesterol, HIV, and Huntington's disease. It has many applications, including correcting genetic defects and treating and preventing the spread of diseases. CRISPR-based diagnostics have been used for many biomedical applications.
Europe CRISPR Gene Detection and Diagnostic Market Dynamics
Drivers
- Increasing Rate of Chronic Diseases
Chronic diseases are the most prevalent health conditions, where 1 in 3 adults suffer from chronic conditions in Europe. In recent years, CRISPR has become an important tool for gene editing that helps in gene alteration. CRISPR is used in researching and treating different types of chronic diseases. For instance, the data by NORD - National Organization for Rare Disorders, Inc. indicated the diagnosed occurrence of Duchenne Muscular Dystrophy (DMD) in 2021. Duchenne Muscular Dystrophy (DMD) is a frequent genetic condition affecting around 1 in 3,500 male births globally. Many healthcare professionals use CRISPR to treat the conditions. Thus, this increasing prevalence demands more of the use of CRISPR, enhancing market growth.
- Increasing Research Activities
There have been several R&D activities that help in boosting the growth of the market. For instance, an international team of scientists conducted a highly controversial experiment in Italy in 2019. The experiment tends to provide a robust and innovative approach to fighting malaria. Also, CRISPR Therapeutics, a gene-editing firm, launched CTX001, an autologous CRISPR/Cas9 gene-edited hematopoietic stem cell therapy. It is meant to treat patients diagnosed with sickle cell disease and β-thalassemia. Thus, this factor boosts market growth.
Opportunities
- Multiple Strategic Initiatives by Market Players
The major players associated with gene editing are trying to avail specific strategies, such as acquisitions, approvals, product launches, expansions, and partnerships, to ensure long-term market sales growth. For instance, Horizon Discovery Ltd. extended the gene modulation portfolio with the first synthetic single guide RNA and patent pending dcas9 repressor for CRISPR interference in Waltham in 2021. This expansion increased the sales and revenue of the synthetic guide RNA portfolio all over the U.K. region. Thus, this increased market growth.
- Increasing Healthcare Expenditure by Organizations
Several CRISPR gene diagnostics and research are funded by recognized organizations such as the National Institute of Health (NIH) budget. The market players are trying hard to achieve the goal of producing safe and effective treatments for patients. For instance, Synthego had raised USD 200 million as an investment for R&D to increase the development of CRISPR-Based Medicines from early-phase research to the clinic in 2022. Synthego will utilize the investment amount from Series E finance to increase the manufacturing of CRISPR diagnostics and services. Thus, this factor will boost market growth.
Restraints/Challenges
- Increasing Cost of CRISPR Gene Detection and Diagnostics
It has been witnessed that maximum genome editing therapies require adequate development and production time, and therefore the rise in cost occurs. Also, the assay kits and medications associated with CRISPR gene detection and diagnosis are very costly. These costs impact the patients as well. Thus, the high cost is anticipated to hamper the market's growth.
This CRISPR gene detection and diagnostic market report provides details of new recent developments, trade regulations, import-export analysis, production analysis, value chain optimization, market share, impact of domestic and localized market players, analyses opportunities in terms of emerging revenue pockets, changes in market regulations, strategic market growth analysis, market size, category market growths, application niches and dominance, product approvals, product launches, geographic expansions, technological innovations in the market. To gain more info on the CRISPR gene detection and diagnostic market contact Data Bridge Market Research for an Analyst Brief, our team will help you take an informed market decision to achieve market growth.
Recent Development
- In 2020, SHERLOCK BIOSCIENCES collaborated with Dartmouth-Hitchcock Health to conduct the clinical trial of the SHERLOCK Diagnostic Kit for Sars-CoV-2. The kit received emergency approval from the Emergency Use Authorization (EUA) by the USFDA
Europe CRISPR Gene Detection and Diagnostic Market Scope
The CRISPR gene detection and diagnostic market is segmented on the basis of class, products & services, application, workflow, end user, and distribution channel. The growth amongst these segments will help you analyze meagre growth segments in the industries and provide the users with a valuable market overview and market insights to help them make strategic decisions for identifying core market applications.
Class
- Class 1- Multiple Effector Proteins
- Class 2 -Single CrRNA Binding Protein
Products & Services
- Products
- Services
Application
- Biomedical Diagnostics
- Genome Engineering
- Drug Discovery
- Agricultural Applications
- Others
Workflow
- Sample Preparation
- Pre-Amplification
- CrRNA
- Cas Enzymes
- Sensing
End User
- Hospitals
- Diagnostic Centers
- Biotechnology Companies
- Academic and Research Institutes
- Others
Distribution Channel
- Direct Tenders
- Retail Sales
CRISPR Gene Detection and Diagnostic Market Regional Analysis/Insights
The CRISPR gene detection and diagnostic market is analyzed and market size insights and trends are provided by class, products & services, application, workflow, end user, and distribution channel as referenced above.
The major countries covered in the CRISPR gene detection and diagnostic market report are Germany, France, U.K., Netherlands, Switzerland, Belgium, Russia, Italy, Spain, Turkey, Rest of Europe in Europe.
Germany is anticipated to grow with the highest CAGR in the forecast period due to technological advancements in CRISPR-based diagnostics. Also, increased government funding for biotechnology to enhance disease treatment boosts market growth.
The country section of the report also provides individual market impacting factors and changes in regulation in the market domestically that impacts the current and future trends of the market. Also, the presence and availability of Europe brands and their challenges faced due to large or scarce competition from local and domestic brands, impact of domestic tariffs and trade routes are considered while providing forecast analysis of the country data.
Competitive Landscape and Europe CRISPR Gene Detection and Diagnostic Market Share Analysis
The CRISPR gene detection and diagnostic market competitive landscape provides details by competitor. Details included are company overview, company financials, revenue generated, market potential, investment in research and development, new market initiatives, Europe presence, production sites and facilities, production capacities, company strengths and weaknesses, product launch, product width and breadth, application dominance. The above data points provided are only related to the companies' focus related to CRISPR gene detection and diagnostic market
Key players operating in the CRISPR gene detection and diagnostic market include:
- GenScript (China)
- OriGene Technologies, Inc (U.S.)
- Horizon Discovery Ltd (U.K.)
- Agilent Technologies Inc. (U.S.)
- Merck & Co., Inc. (U.S.)
- Danaher (U.S.)
- Thermo Fisher Scientific (U.S.)
- Takara Bio Inc. (Japan)
- CRISPR Therapeutics (Switzerland)
- GeneCopoeia, Inc (U.S.)
SKU-
Get online access to the report on the World's First Market Intelligence Cloud
- Interactive Data Analysis Dashboard
- Company Analysis Dashboard for high growth potential opportunities
- Research Analyst Access for customization & queries
- Competitor Analysis with Interactive dashboard
- Latest News, Updates & Trend analysis
- Harness the Power of Benchmark Analysis for Comprehensive Competitor Tracking
Research Methodology
Data collection and base year analysis are done using data collection modules with large sample sizes. The stage includes obtaining market information or related data through various sources and strategies. It includes examining and planning all the data acquired from the past in advance. It likewise envelops the examination of information inconsistencies seen across different information sources. The market data is analysed and estimated using market statistical and coherent models. Also, market share analysis and key trend analysis are the major success factors in the market report. To know more, please request an analyst call or drop down your inquiry.
The key research methodology used by DBMR research team is data triangulation which involves data mining, analysis of the impact of data variables on the market and primary (industry expert) validation. Data models include Vendor Positioning Grid, Market Time Line Analysis, Market Overview and Guide, Company Positioning Grid, Patent Analysis, Pricing Analysis, Company Market Share Analysis, Standards of Measurement, Global versus Regional and Vendor Share Analysis. To know more about the research methodology, drop in an inquiry to speak to our industry experts.
Customization Available
Data Bridge Market Research is a leader in advanced formative research. We take pride in servicing our existing and new customers with data and analysis that match and suits their goal. The report can be customized to include price trend analysis of target brands understanding the market for additional countries (ask for the list of countries), clinical trial results data, literature review, refurbished market and product base analysis. Market analysis of target competitors can be analyzed from technology-based analysis to market portfolio strategies. We can add as many competitors that you require data about in the format and data style you are looking for. Our team of analysts can also provide you data in crude raw excel files pivot tables (Fact book) or can assist you in creating presentations from the data sets available in the report.